One-Stop Management of 230 Consecutive Acute Stroke Patients: Report of Procedural Times and Clinical Outcome
Abstract
1. Introduction
2. Materials and Methods
2.1. Patient Selection
2.2. Image Acquisition and Processing
2.3. Management After Imaging
2.4. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Goyal, M.; Menon, B.K.; van Zwam, W.H.; Dippel, D.W.J.; Mitchell, P.J.; Demchuk, A.M.; Dávalos, A.; Majoie, C.B.L.M.; van der Lugt, A.; de Miquel, M.A.; et al. Endovascular thrombectomy after large-vessel ischaemic stroke: A meta-analysis of individual patient data from five randomised trials. Lancet 2016, 387, 1723–1731. [Google Scholar] [CrossRef]
- Saver, J.L.; Goyal, M.; van der Lugt, A.; Menon, B.K.; Majoie, C.B.; Dippel, D.W.; Campbell, B.C.; Nogueira, R.G.; Demchuk, A.M.; Tomasello, A.; et al. Time to Treatment With Endovascular Thrombectomy and Outcomes From Ischemic Stroke: A Meta-analysis. JAMA 2016, 316, 1279–1288. [Google Scholar] [CrossRef] [PubMed]
- Berkhemer, O.A.; Fransen, P.S.; Beumer, D.; van den Berg, L.A.; Lingsma, H.F.; Yoo, A.J.; Schonewille, W.J.; Vos, J.A.; Nederkoorn, P.J.; Wermer, M.J.; et al. A randomized trial of intraarterial treatment for acute ischemic stroke. N. Engl. J. Med. 2015, 372, 11–20. [Google Scholar] [CrossRef]
- Campbell, B.C.V.; Mitchell, P.J.; Kleinig, T.J.; Dewey, H.M.; Churilov, L.; Yassi, N.; Yan, B.; Dowling, R.J.; Parsons, M.W.; Oxley, T.J.; et al. Endovascular Therapy for Ischemic Stroke with Perfusion-Imaging Selection. N. Engl. J. Med. 2015, 372, 1009–1018. [Google Scholar] [CrossRef] [PubMed]
- Saver, J.L.; Goyal, M.; Bonafe, A.; Diener, H.C.; Levy, E.I.; Pereira, V.M.; Albers, G.W.; Cognard, C.; Cohen, D.J.; Hacke, W.; et al. Stent-retriever thrombectomy after intravenous t-PA vs. t-PA alone in stroke. N. Engl. J. Med. 2015, 372, 2285–2295. [Google Scholar] [CrossRef] [PubMed]
- Goyal, M.; Demchuk, A.M.; Menon, B.K.; Eesa, M.; Rempel, J.L.; Thornton, J.; Roy, D.; Jovin, T.G.; Willinsky, R.A.; Sapkota, B.L.; et al. Randomized Assessment of Rapid Endovascular Treatment of Ischemic Stroke. N. Engl. J. Med. 2015, 372, 1019–1030. [Google Scholar] [CrossRef]
- Jovin, T.G.; Chamorro, A.; Cobo, E.; de Miquel, M.A.; Molina, C.A.; Rovira, A.; San Roman, L.; Serena, J.; Abilleira, S.; Ribo, M.; et al. Thrombectomy within 8 hours after symptom onset in ischemic stroke. N. Engl. J. Med. 2015, 372, 2296–2306. [Google Scholar] [CrossRef]
- Psychogios, M.N.; Behme, D.; Schregel, K.; Tsogkas, I.; Maier, I.L.; Leyhe, J.R.; Zapf, A.; Tran, J.; Bahr, M.; Liman, J.; et al. One-Stop Management of Acute Stroke Patients: Minimizing Door-to-Reperfusion Times. Stroke 2017, 48, 3152–3155. [Google Scholar] [CrossRef]
- Leyhe, J.R.; Tsogkas, I.; Hesse, A.C.; Behme, D.; Schregel, K.; Papageorgiou, I.; Liman, J.; Knauth, M.; Psychogios, M.-N. Latest generation of flat detector CT as a peri-interventional diagnostic tool: A comparative study with multidetector CT. J. Neurointerv. Surg. 2017, 9, 1253–1257. [Google Scholar] [CrossRef]
- Maier, I.L.; Scalzo, F.; Leyhe, J.R.; Schregel, K.; Behme, D.; Tsogkas, I.; Psychogios, M.-N.; Liebeskind, D.S. Validation of collateral scoring on flat-detector multiphase CT angiography in patients with acute ischemic stroke. PLoS ONE 2018, 13, e0202592. [Google Scholar] [CrossRef]
- Schregel, K.; Behme, D.; Tsogkas, I.; Knauth, M.; Maier, I.; Karch, A.; Mikolajczyk, R.; Hinz, J.; Liman, J.; Psychogios, M.N. Effects of Workflow Optimization in Endovascularly Treated Stroke Patients—A Pre-Post Effectiveness Study. PLoS ONE 2016, 11, e0169192. [Google Scholar] [CrossRef] [PubMed]
- Psychogios, M.N.; Bahr, M.; Liman, J.; Knauth, M. One Stop Management in Acute Stroke: First Mothership Patient Transported Directly to the Angiography Suite. Clin. Neuroradiol. 2017, 27, 389–391. [Google Scholar] [CrossRef] [PubMed]
- Holodinsky, J.K.; Kamal, N.; Wilson, A.T.; Hill, M.D.; Goyal, M. Workflow in Acute Stroke: What Is the 90th Percentile? Stroke 2017, 48, 808–812. [Google Scholar] [CrossRef] [PubMed]
- Albers, G.W.; Marks, M.P.; Kemp, S.; Christensen, S.; Tsai, J.P.; Ortega-Gutierrez, S.; McTaggart, R.A.; Torbey, M.T.; Kim-Tenser, M.; Leslie-Mazwi, T.; et al. Thrombectomy for Stroke at 6 to 16 Hours with Selection by Perfusion Imaging. N. Engl. J. Med. 2018, 379, 708–718. [Google Scholar] [CrossRef] [PubMed]
- Nogueira, R.G.; Frei, D.; Kirmani, J.F.; Zaidat, O.; Lopes, D.; Turk, A.S., 3rd; Heck, D.; Mason, B.; Haussen, D.C.; Levy, E.I.; et al. Safety and Efficacy of a 3-Dimensional Stent Retriever With Aspiration-Based Thrombectomy vs Aspiration-Based Thrombectomy Alone in Acute Ischemic Stroke Intervention: A Randomized Clinical Trial. JAMA Neurol. 2018, 75, 304–311. [Google Scholar] [CrossRef] [PubMed]
- Schregel, K.; Behme, D.; Tsogkas, I.; Knauth, M.; Maier, I.; Karch, A.; Mikolajczyk, R.; Bähr, M.; Schäper, J.; Hinz, J.; et al. Optimized Management of Endovascular Treatment for Acute Ischemic Stroke. J. Vis. Exp. 2018, 131, e56397. [Google Scholar] [CrossRef]
- Maus, V.; Behme, D.; Kabbasch, C.; Borggrefe, J.; Tsogkas, I.; Nikoubashman, O.; Wiesmann, M.; Knauth, M.; Mpotsaris, A.; Psychogios, M.N. Maximizing First-Pass Complete Reperfusion with SAVE. Clin. Neuroradiol. 2017, 28, 327–338. [Google Scholar] [CrossRef]
- Bourcier, R.; Goyal, M.; Liebeskind, D.S.; Muir, K.W.; Desal, H.; Siddiqui, A.H.; Dippel, D.W.J.; Majoie, C.B.; van Zwam, W.H.; Jovin, T.G.; et al. Association of Time From Stroke Onset to Groin Puncture With Quality of Reperfusion After Mechanical Thrombectomy: A Meta-analysis of Individual Patient Data From 7 Randomized Clinical Trials. JAMA Neurol 2019, 76, 405–411. [Google Scholar] [CrossRef]
- Strub, W.M.; Leach, J.L.; Tomsick, T.; Vagal, A. Overnight preliminary head CT interpretations provided by residents: Locations of misidentified intracranial hemorrhage. AJNR Am. J. Neuroradiol. 2007, 28, 1679–1682. [Google Scholar] [CrossRef]
- Heldner, M.R.; Hsieh, K.; Broeg-Morvay, A.; Mordasini, P.; Buhlmann, M.; Jung, S.; Arnold, M.; Mattle, H.P.; Gralla, J.; Fischer, U. Clinical prediction of large vessel occlusion in anterior circulation stroke: Mission impossible? J. Neurol. 2016, 263, 1633–1640. [Google Scholar] [CrossRef]
- Pfaff, J.; Schonenberger, S.; Herweh, C.; Pham, M.; Nagel, S.; Ringleb, P.A.; Heiland, S.; Bendszus, M.; Mohlenbruch, M.A. Influence of a combined CT/C-arm system on periprocedural workflow and procedure times in mechanical thrombectomy. Eur. Radiol. 2017, 27, 3966–3972. [Google Scholar] [CrossRef] [PubMed]
- Ribo, M.; Boned, S.; Rubiera, M.; Tomasello, A.; Coscojuela, P.; Hernández, D.; Pagola, J.; Juega, J.; Rodriguez, N.; Muchada, M.; et al. Direct transfer to angiosuite to reduce door-to-puncture time in thrombectomy for acute stroke. J. Neurointerv. Surg. 2018, 10, 221–224. [Google Scholar] [CrossRef] [PubMed]
- Jadhav, A.P.; Kenmuir, C.L.; Aghaebrahim, A.; Limaye, K.; Wechsler, L.R.; Hammer, M.D.; Starr, M.T.; Molyneaux, B.J.; Rocha, M.; Guyette, F.X.; et al. Interfacility Transfer Directly to the Neuroangiography Suite in Acute Ischemic Stroke Patients Undergoing Thrombectomy. Stroke 2017, 48, 1884–1889. [Google Scholar] [CrossRef] [PubMed]
- Mendez, B.; Requena, M.; Aires, A.; Martins, N.; Boned, S.; Rubiera, M.; Tomasello, A.; Coscojuela, P.; Muchada, M.; Rodríguez-Luna, D.; et al. Direct Transfer to Angio-Suite to Reduce Workflow Times and Increase Favorable Clinical Outcome. Stroke 2018, 49, 2723–2727. [Google Scholar] [CrossRef] [PubMed]
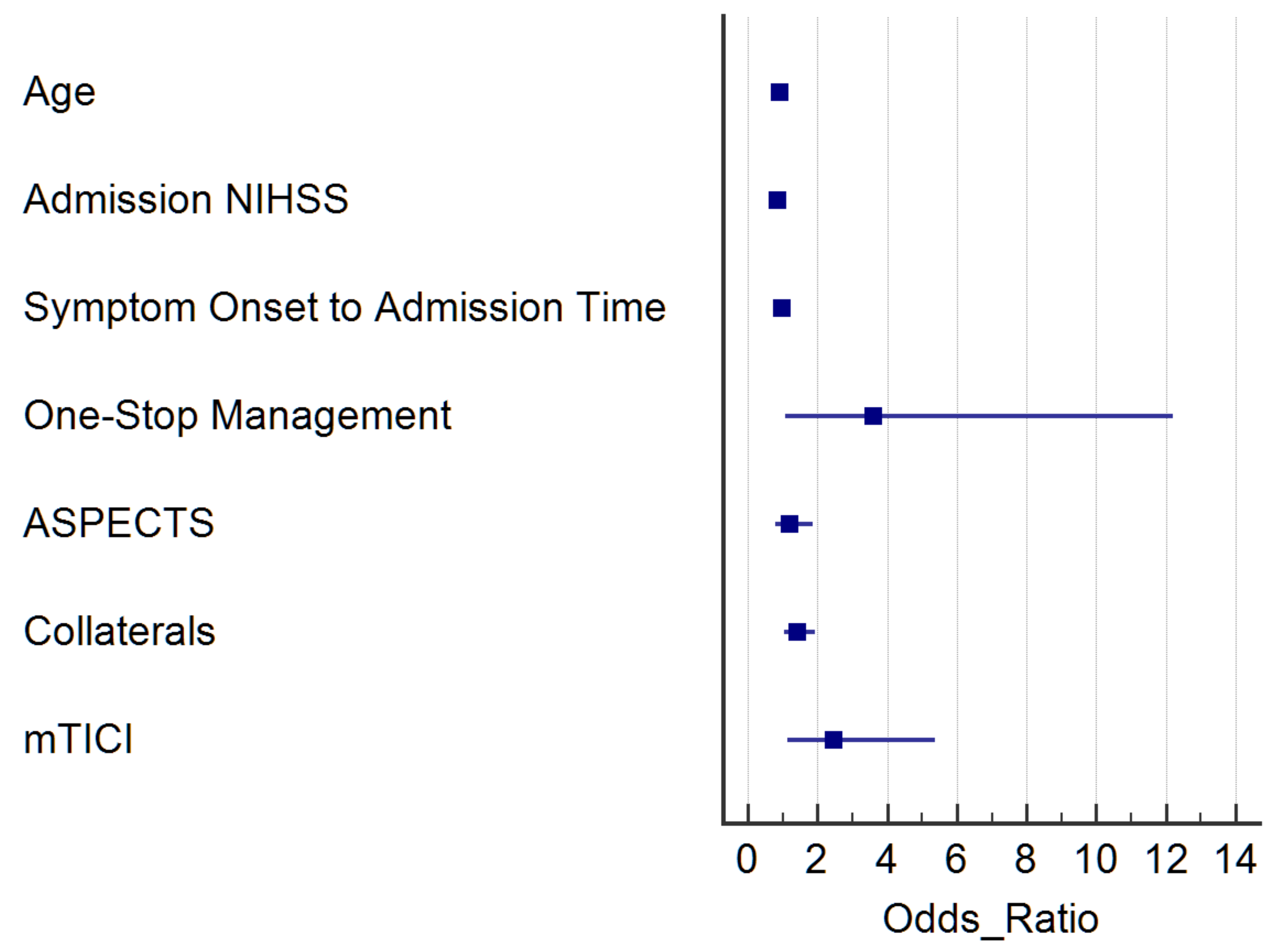

| All, n = 230 | LVO, n = 166 | SVO, n = 25 | ICH, n = 24 | Todd’s, n = 11 | RLVO, n = 4 | |
|---|---|---|---|---|---|---|
| Age, median (IQR) | 78 (69–84) | 77 (68–84) | 79 (71–87) | 78 (75–83) | 79 (73–82) | 81 (77–86) |
| Admission NIHSS | 15 (12–19) | 16 (13–19) | 13 (11–15) | 15 (10–17) | 12 (11–15) | 13 (11–13) |
| Female | 123 (54%) | 90 (54%) | 12 (48%) | 12 (50%) | 5 (46%) | 4 (100%) |
| IV–rtPA | 144 (63%) | 112 (68%) | 23 (92%) | 1 | 4 (36%) | 4 (100%) |
| Hemorrhage on initial FDCT | 25 (11%) | 1 (1%) | 0 (0%) | 24 (100%) | 0 (0%) | 0 (0%) |
| Occlusion site | ||||||
| ICA-T | 41 (25%) | |||||
| M1 | 88 (53%) | |||||
| M2 | 15 (9%) | |||||
| Other | 22 (13%) | |||||
| Tandem occlusion | 34 (21%) | |||||
| Times, min (IQR; 90th Percentile) | ||||||
| Symptom to door | 130 (70–195; 253) | 154 (67–205; 264) | 82 (66–134; 205) | 105 (69–129; 204) | 131(95–146; 186) | 229 (181–259) |
| Door to FDCT | 15 (10–20; 26) | 14 (9–19; 25) | 16 (12–24; 32) | 17 (14–22; 30) | 21 (13–23; 31) | 16 (14–16) |
| Door to IV–rtPA | 22 (20–30; 41) | 22 (20–29; 38) | 26 (20–45; 53) | |||
| Door to treatment startα | 21 (18–33; 34) | 27 (19–32; 37) | ||||
| Door to groin | 29 (22–39; 50) | |||||
| Groin to reperfusion | 40 (28–60; 80) | |||||
| FDCT to reperfusion | 59 (45–82; 101) | |||||
| Door to reperfusion | 72 (58–91; 117) | |||||
| Door to reperfusion M1 | 64 (56–87; 102) | |||||
| extCT to FDCT | 124 (110–155; 218) | |||||
| Direct admission | 127 (55%) | 74 (45%) | 19 (76%) | 23 (96%) | 11 (100%) | 0 (0%) |
| Working hoursβ | 95 (41%) | 68 (41%) | 13 (52%) | 11 (46%) | 2 (18%) | 1 (25%) |
| Reperfusion, mTICI2b-3 | 142 (86%) | |||||
| Any hemorrhage on FU | 49 (21%) | 25 (15%) | 0 (0%) | 24 (100%) | 0 (0%) | |
| PH-2 hematoma on FU | 2 (1%) | |||||
| sICH | 6 (4%) | |||||
| Discharge NIHSS | 5 (2–10) | 5 (2–12) | 7 (4–10) | 5 (1–9) | 4 (1–6) | 6 (4–7) |
| Discharge mRS | 4 (1–5) | 4 (1–5) | 4 (2–5) | 2 (2–6) | 3 (1–4) | 3 (3–4) |
| Mortality | 45/230 (20%) | 36 (22%) | 2 (8%) | 5 (30%) | 2 (18%) | 0 (0%) |
| 90 d mRS | 4 (1–6) | |||||
| 90 d favorable outcome | 54/147 (37%) |
| Direct Admission n = 74 | Min (IQR; 90th Percentile) |
|---|---|
| Door to FDCT | 15 (12–20; 24) |
| Door to IV–rtPA | 22 (20–29; 38) |
| Door to groin | 34 (28–45; 51) |
| Groin to reperfusion | 41 (26–55; 73) |
| FDCT to reperfusion | 61 (47–81; 93) |
| Door to reperfusion | 76 (61–92; 116) |
| Door to reperfusion of M1 | 68 (58–89; 101) |
| Occluded vessel | ICA-T 13 (18%), M1 42 (58%), M2 9 (12%) |
| Tandem occlusions | 15 (20%) |
| Transfer patients n = 92 | |
| extCT to FDCT | 124 (110–155; 218) |
| Door to FDCT | 10 (8–17; 25) |
| Door to groin | 25 (19–33; 41) |
| Groin to reperfusion | 38 (29–65; 87) |
| FDCT to reperfusion | 56 (44–86; 110) |
| Door to reperfusion | 68 (53–90; 126) |
| Door to reperfusion of M1 | 59 (52–84; 118) |
| Occluded vessel | ICA-T 28 (30%), M1 46 (50%), M2 6 (7%) |
| Tandem occlusions | 19 (20%) |
| Working hours n = 68 | |
| Door to FDCT | 12 (7–16; 21) |
| Door to IV–rtPA | 22 (20–26; 34) |
| Door to groin | 25 (19–33; 41) |
| Groin to reperfusion | 38 (25–53; 85) |
| FDCT to reperfusion | 61 (42–69; 101) |
| Door to reperfusion | 66 (52–85; 105) |
| Off-hours n = 98 | |
| Door to FDCT | 15 (10–21; 27) |
| Door to IV–rtPA | 23 (19–29; 38) |
| Door to groin | 33 (25–42; 60) |
| Groin to reperfusion | 38 (25–53; 86) |
| FDCT to reperfusion | 52 (42–69; 101) |
| Door to reperfusion | 66 (52–85; 105) |
| MDCT, n = 43 | FDCT, n = 43 | p-Value | |
|---|---|---|---|
| Age median (IQR) * | 77 (69–81) | 77 (69–82) | 0.962 |
| Admission NIHSS * | 17 (14–20) | 16 (13–20) | 0.796 |
| CT ASPECTS * | 8 (7–9) | 9 (7–10) | 0.138 |
| Onset to door, min (IQR; 90th) * | 129 (76–200; 244) | 160 (74–202; 221) | 0.511 |
| Female | 26 (61%) | 26 (61%) | 1.000 |
| IV-rtPA | 36 (84%) | 30 (70%) | 0.201 |
| Hypertension | 35 (81%) | 33 (77%) | 0.791 |
| Hyperlipidemia | 14 (33%) | 20 (47%) | 0.266 |
| PAD | 2 (5%) | 5 (12%) | 0.433 |
| DM | 11 (26%) | 17 (40%) | 0.249 |
| Collateral grading | 7 (5–8) | 7 (4–8) | 0.699 |
| Direct admissions | 30 (70%) | 18 (42%) | 0.016 |
| Working hours | 22 (51%) | 19 (44%) | 0.666 |
| Door to CT, min (IQR; 90th) | 15 (11–20; 24) | 9 (6–14; 16) | <0.001 |
| Door to IV-rtPA | 27 (22–34; 35) | 19 (12–22; 34) | 0.016 |
| Door to groin | 60 (48–68; 79) | 25 (19–30; 38) | <0.001 |
| Working hours | 60 (42–65; 85) | 21 (17–25; 41) | <0.001 |
| Off-hours | 62 (53–69; 75) | 25 (21–32; 38) | <0.001 |
| Direct admissions | 61 (54–67; 83) | 26 (25–38; 44) | <0.001 |
| Transfer patients | 40 (30–69; 75) | 21 (19–26; 35) | <0.001 |
| Groin to reperfusion | 42 (27–62; 94) | 43 (33–60; 78) | 0.866 |
| CT to reperfusion | 84 (71–99; 144) | 59 (44–75; 96) | <0.001 |
| Door to reperfusion | 102 (85–117; 166) | 68 (53–89; 104) | <0.001 |
| Working hours | 102 (79–145; 191) | 62 (52–81; 104) | 0.006 |
| Off-hours | 103 (93–116; 126) | 74 (55–90; 109) | <0.001 |
| Direct admissions | 103 (85–121; 184) | 72 (58–87; 103) | 0.001 |
| Transfer patients | 102 (68–109; 120) | 64 (51–88; 108) | 0.05 |
| ICA-T | 7 (16%) | 13 (30%) | 0.179 |
| M1 | 26 (61%) | 25 (58%) | 0.888 |
| M2 | 9 (21%) | 3 (7%) | 0.117 |
| Tandem occlusion | 6 (14%) | 7 (16%) | 1 |
| Successful reperfusion (mTICI2b-3) | 31 (72%) | 38 (88%) | 0.102 |
| sICH | 3 (7%) | 2 (5%) | 1 |
| Any hemorrhage | 11 (26%) | 7 (16%) | 0.427 |
| PH–2 hemorrhage | 1 (2%) | 1 (2%) | 1 |
| Discharge mRS | 4 (2–5) | 3 (1–5) | 0.374 |
| 90d mRS | 4 (1–5) | 2 (1–5) | 0.153 |
| 90d mRS of 0–2 | 14 (33%) | 25 (58%) | 0.029 |
| Mortality | 9 (21%) | 10 (23%) | 1 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Psychogios, M.-N.; Maier, I.L.; Tsogkas, I.; Hesse, A.C.; Brehm, A.; Behme, D.; Schnieder, M.; Schregel, K.; Papageorgiou, I.; Liebeskind, D.S.; et al. One-Stop Management of 230 Consecutive Acute Stroke Patients: Report of Procedural Times and Clinical Outcome. J. Clin. Med. 2019, 8, 2185. https://doi.org/10.3390/jcm8122185
Psychogios M-N, Maier IL, Tsogkas I, Hesse AC, Brehm A, Behme D, Schnieder M, Schregel K, Papageorgiou I, Liebeskind DS, et al. One-Stop Management of 230 Consecutive Acute Stroke Patients: Report of Procedural Times and Clinical Outcome. Journal of Clinical Medicine. 2019; 8(12):2185. https://doi.org/10.3390/jcm8122185
Chicago/Turabian StylePsychogios, Marios-Nikos, Ilko L. Maier, Ioannis Tsogkas, Amélie Carolina Hesse, Alex Brehm, Daniel Behme, Marlena Schnieder, Katharina Schregel, Ismini Papageorgiou, David S. Liebeskind, and et al. 2019. "One-Stop Management of 230 Consecutive Acute Stroke Patients: Report of Procedural Times and Clinical Outcome" Journal of Clinical Medicine 8, no. 12: 2185. https://doi.org/10.3390/jcm8122185
APA StylePsychogios, M.-N., Maier, I. L., Tsogkas, I., Hesse, A. C., Brehm, A., Behme, D., Schnieder, M., Schregel, K., Papageorgiou, I., Liebeskind, D. S., Goyal, M., Bähr, M., Knauth, M., & Liman, J. (2019). One-Stop Management of 230 Consecutive Acute Stroke Patients: Report of Procedural Times and Clinical Outcome. Journal of Clinical Medicine, 8(12), 2185. https://doi.org/10.3390/jcm8122185






