The Right Time to Safely Re-Evaluate Empirical Antimicrobial Treatment of Hip or Knee Prosthetic Joint Infections
Abstract
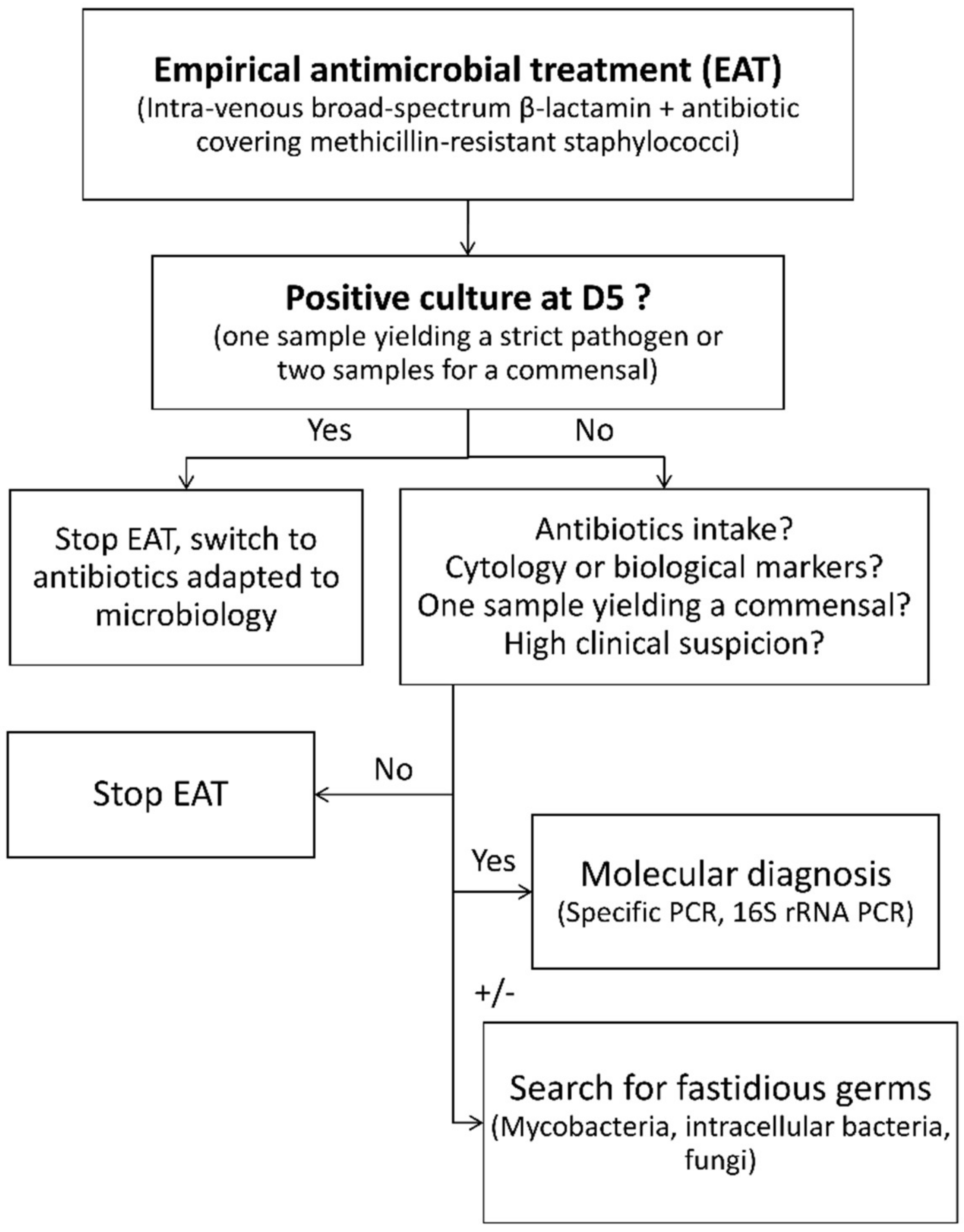
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Definition of PJI
2.3. Laboratory Analysis
2.4. Statistical Analysis
2.5. Ethics
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rodriguez-Merchan, E.C. Preoperative Aspiration Culture (PAC) for the Diagnosis of Infection in a Prosthetic Knee Joint. Arch. Bone Jt. Surg. 2018, 6, 342–345. [Google Scholar] [PubMed]
- Spilf, O. Recommendations for bone and joint prosthetic device infections in clinical practice (prosthesis, implants, osteosynthesis). Med. Mal. Infect. 2010, 40, 185–211. [Google Scholar] [CrossRef] [PubMed]
- Osmon, D.R.; Berbari, E.F.; Berendt, A.R.; Lew, D.; Zimmerli, W.; Steckelberg, J.M.; Rao, N.; Hanssen, A.; Wilson, W.R. Diagnosis and Management of Prosthetic Joint Infection: Clinical Practice Guidelines by the Infectious Diseases Society of America. Clin. Infect. Dis. 2013, 56, e1–e25. [Google Scholar] [CrossRef] [PubMed]
- Esposito, S.; Leone, S.; Bassetti, M.; Borrè, S.; Leoncini, F.; Meani, E.; Venditti, M.; Mazzotta, F.; Bone Joint Infections Committee for the Italian Society of Infectious Tropical Diseases (SIMIT). Italian guidelines for the diagnosis and infectious disease management of osteomyelitis and prosthetic joint infections in adults. Infection 2009, 37, 478–496. [Google Scholar] [CrossRef]
- Ariza, J.; Cobo, J.; Baraia-Etxaburu, J.; Benito, N.; Bori, G.; Cabo, J.; Corona, P.; Esteban, J.; Horcajada, J.P.; Lora-Tamayo, J.; et al. Executive summary of management of prosthetic joint infections. Clinical practice guidelines by the Spanish Society of Infectious Diseases and Clinical Microbiology (SEIMC). Enferm. Infecc. Microbiol. Clin. 2017, 35, 189–195. [Google Scholar] [CrossRef]
- Joseph, C.; Robineau, O.; Titecat, M.; Putman, S.; Blondiaux, N.; Loiez, C.; Valette, M.; Schmit, J.; Beltrand, E.; Dézeque, H.; et al. Daptomycin versus Vancomycin as Post-Operative Empirical Antibiotic Treatment for Prosthetic Joint Infections: A Case-Control Study. J. Bone Jt. Infect. 2019, 4, 72–75. [Google Scholar] [CrossRef][Green Version]
- Takoudju, E.; Bemer, P.; Touchais, S.; Asseray, N.; Corvec, S.; Khatchatourian, L.; Serandour, N.; Boutoille, D.; Nantes Bone and Joint Infections Study Group. Bacteriological relevance of linezolid versus vancomycin in postoperative empirical treatment of osteoarticular infections: A retrospective single-center study. Int. J. Antimicrob. Agents 2018, 52, 663–666. [Google Scholar] [CrossRef]
- Deroche, L.; Plouzeau, C.; Bémer, P.; Tandé, D.; Valentin, A.S.; Jolivet-Gougeon, A.; Lemarié, C.; Bret, L.; Kempf, M.; Héry-Arnaud, G.; et al. Probabilistic chemotherapy in knee and hip replacement infection: The place of linezolid. Eur. J. Clin. Microbiol. Infect. Dis. 2019, 38, 1659–1663. [Google Scholar] [CrossRef]
- Aalirezaie, A.; Bauer, T.W.; Fayaz, H.; Griffin, W.; Higuera, C.A.; Krenn, V.; Krenn, V.; Molano, M.; Moojen, D.-J.; Restrepo, C.; et al. Hip and Knee Section, Diagnosis, Reimplantation: Proceedings of International Consensus on Orthopedic Infections. J. Arthroplast. 2019, 34, S369–S379. [Google Scholar] [CrossRef]
- Rutter, W.C.; Burgess, D.S. Incidence of Acute Kidney Injury among Patients Treated with Piperacillin-Tazobactam or Meropenem in Combination with Vancomycin. Antimicrob. Agents Chemother. 2018, 62. [Google Scholar] [CrossRef]
- Triffault-Fillit, C.; Valour, F.; Guillo, R.; Tod, M.; Goutelle, S.; Lustig, S.; Fessy, M.-H.; Chidiac, C.; Ferry, T.; Lyon BJI Study Group. Prospective Cohort Study of the Tolerability of Prosthetic Joint Infection Empirical Antimicrobial Therapy. Antimicrob. Agents Chemother. 2018, 62, e00163-18. [Google Scholar] [CrossRef] [PubMed]
- Bémer, P.; Plouzeau, C.; Tande, D.; Léger, J.; Giraudeau, B.; Valentin, A.S.; Jolivet-Gougeon, A.; Vincent, P.; Corvec, S.; Gibaud, S.; et al. Evaluation of 16S rRNA Gene PCR Sensitivity and Specificity for Diagnosis of Prosthetic Joint Infection: A Prospective Multicenter Cross-Sectional Study. J. Clin. Microbiol. 2014, 52, 3583–3589. [Google Scholar] [CrossRef] [PubMed]
- Tsukayama, D.T.; Estrada, R.; Gustilo, R.B. Infection after total hip arthroplasty. A study of the treatment of one hundred and six infections. J. Bone Jt. Surg. Am. 1996, 78, 512–523. [Google Scholar] [CrossRef] [PubMed]
- Tsukayama, D.T.; Goldberg, V.M.; Kyle, R. Diagnosis and management of infection after total knee arthroplasty. J. Bone Jt. Surg. Am. 2003, 85, S75–S80. [Google Scholar] [CrossRef] [PubMed]
- Senneville, E.; Joulie, D.; Legout, L.; Valette, M.; Dezèque, H.; Beltrand, E.; Roselé, B.; d’Escrivan, T.; Loïez, C.; Caillaux, M.; et al. Outcome and Predictors of Treatment Failure in Total Hip/Knee Prosthetic Joint Infections Due to Staphylococcus aureus. Clin. Infect. Dis. 2011, 53, 334–340. [Google Scholar] [CrossRef] [PubMed]
- Peel, T.N.; Cheng, A.C.; Choong, P.F.M.; Buising, K.L. Early onset prosthetic hip and knee joint infection: Treatment and outcomes in Victoria, Australia. J. Hosp. Infect. 2012, 82, 248–253. [Google Scholar] [CrossRef]
- Rieber, H.; Frontzek, A.; Jerosch, J.; Alefeld, M.; Strohecker, T.; Ulatowski, M.; Morawietz, T.; Hinsenkamp, S.; Bell, A.; Kücükköylü, D.; et al. Periprosthetic joint infection caused by anaerobes. Retrospective analysis reveals no need for prolonged cultivation time if sensitive supplemented growth media are used. Anaerobe 2018, 50, 12–18. [Google Scholar] [CrossRef]
- Renz, N.; Mudrovcic, S.; Perka, C.; Trampuz, A. Orthopedic implant-associated infections caused by Cutibacterium spp.—A remaining diagnostic challenge. PLoS ONE 2018, 13, e0202639. [Google Scholar] [CrossRef]
- Bossard, D.A.; Ledergerber, B.; Zingg, P.O.; Gerber, C.; Zinkernagel, A.S.; Zbinden, R.; Achermann, Y. Optimal Length of Cultivation Time for Isolation of Propionibacterium acnes in Suspected Bone and Joint Infections Is More than 7 Days. J. Clin. Microbiol. 2016, 54, 3043–3049. [Google Scholar] [CrossRef]
- Holleyman, R.J.; Deehan, D.J.; Charlett, A.; Gould, K.; Baker, P.N. Does pre-operative sampling predict intra-operative cultures and antibiotic sensitivities in knee replacements revised for infection? A study using the NJR dataset. Knee Surg. Sports Traumatol. Arthrosc. 2016, 24, 3056–3063. [Google Scholar] [CrossRef]
- Frangiamore, S.; Saleh, A.; Grosso, M.; Alolabi, B.; Bauer, T.; Iannotti, J.; Ricchetti, E. Early Versus Late Culture Growth of Propionibacterium acnes in Revision Shoulder Arthroplasty. J. Bone Jt. Surg. 2015, 97, 1149–1158. [Google Scholar] [CrossRef] [PubMed]
- Benito, N.; Mur, I.; Ribera, A.; Soriano, A.; Rodríguez-Pardo, D.; Sorlí, L.; Cobo, J.; Fernández-Sampedro, M.; del Toro, M.D.; Guío, L.; et al. The Different Microbial Etiology of Prosthetic Joint Infections according to Route of Acquisition and Time after Prosthesis Implantation, Including the Role of Multidrug-Resistant Organisms. J. Clin. Med. 2019, 8, 673. [Google Scholar] [CrossRef] [PubMed]
- Triffault-Fillit, C.; Ferry, T.; Laurent, F.; Pradat, P.; Dupieux, C.; Conrad, A.; Becker, A.; Lustig, S.; Fessy, M.H.; Chidiac, C.; et al. Microbiologic epidemiology depending on time to occurrence of prosthetic joint infection: A prospective cohort study. Clin. Microbiol. Infect. 2019, 25, 353–358. [Google Scholar] [CrossRef] [PubMed]
- Trampuz, A.; Piper, K.E.; Jacobson, M.J.; Hanssen, A.D.; Unni, K.K.; Osmon, D.R.; Mandrekar, J.N.; Cockerill, F.R.; Steckelberg, J.M.; Greenleaf, J.F.; et al. Sonication of removed hip and knee prostheses for diagnosis of infection. N. Engl. J. Med. 2007, 357, 654–663. [Google Scholar] [CrossRef]
- Peel, T.N.; Dylla, B.L.; Hughes, J.G.; Lynch, D.T.; Greenwood-Quaintance, K.E.; Cheng, A.C.; Mandrekar, J.N.; Patel, R. Improved Diagnosis of Prosthetic Joint Infection by Culturing Periprosthetic Tissue Specimens in Blood Culture Bottles. mBio 2016, 7, e01776-15. [Google Scholar] [CrossRef]
- Bémer, P.; Léger, J.; Milin, S.; Plouzeau, C.; Valentin, A.S.; Stock, N.; Jolivet-Gougeon, A.; Moreau, A.; Corvec, S.; Quintin-Roue, I.; et al. Histopathological Diagnosis of Prosthetic Joint Infection: Does a Threshold of 23 Neutrophils Do Better than Classification of the Periprosthetic Membrane in a Prospective Multicenter Study? J. Clin. Microbiol. 2018, 56, e00536-18. [Google Scholar] [CrossRef]
- Parvizi, J.; Tan, T.L.; Goswami, K.; Higuera, C.; Della Valle, C.; Chen, A.F.; Shohat, N. The 2018 Definition of Periprosthetic Hip and Knee Infection: An Evidence-Based and Validated Criteria. J. Arthroplast. 2018, 33, 1309–1314.e2. [Google Scholar] [CrossRef]
- Parvizi, J.; Erkocak, O.; Valle, C.D. Culture-Negative Periprosthetic Joint Infection. J. Bone Jt. Surg. 2014, 96, 430–436. [Google Scholar] [CrossRef]
- Rieber, H.; Frontzek, A.; Fischer, M. Periprosthetic joint infection associated with Mycoplasma hominis after transurethral instrumentation in an immunocompetent patient. Unusual or underestimated? A case report and review of the literature. Int. J. Infect. Dis. 2019, 82, 86–88. [Google Scholar] [CrossRef]
- Li, H.K.; Rombach, I.; Zambellas, R.; Walker, A.S.; McNally, M.A.; Atkins, B.L.; Lipsky, B.A.; Hughes, H.C.; Bose, D.; Kümin, M.; et al. Oral versus Intravenous Antibiotics for Bone and Joint Infection. N. Engl. J. Med. 2019, 380, 425–436. [Google Scholar] [CrossRef]
- Marks, M.; Bell, L.C.K.; Jones, I.; Rampling, T.; Kranzer, K.; Morris-Jones, S.; Logan, S.; Pollara, G. Clinical and economic impact of implementing OVIVA criteria on patients with bone and joint infections in OPAT. Clin. Infect. Dis. 2019. [Google Scholar] [CrossRef] [PubMed]
- Navalkele, B.; Pogue, J.M.; Karino, S.; Nishan, B.; Salim, M.; Solanki, S.; Pervaiz, A.; Tashtoush, N.; Shaikh, H.; Koppula, S.; et al. Risk of Acute Kidney Injury in Patients on Concomitant Vancomycin and Piperacillin–Tazobactam Compared to Those on Vancomycin and Cefepime. Clin. Infect. Dis. 2017, 64, 116–123. [Google Scholar] [CrossRef] [PubMed]
- Luther, M.K.; Timbrook, T.T.; Caffrey, A.R.; Dosa, D.; Lodise, T.P.; LaPlante, K.L. Vancomycin Plus Piperacillin-Tazobactam and Acute Kidney Injury in Adults: A Systematic Review and Meta-Analysis. Crit. Care Med. 2018, 46, 12–20. [Google Scholar] [CrossRef] [PubMed]
- Schreier, D.J.; Kashani, K.B.; Sakhuja, A.; Mara, K.C.; Tootooni, M.S.; Personett, H.A.; Nelson, S.; Rule, A.D.; Steckelberg, J.M.; Tande, A.J.; et al. Incidence of acute kidney injury among critically ill patients with brief empiric use of anti-pseudomonal beta-lactams with vancomycin. Clin. Infect. Dis. 2018, 68, 1456–1462. [Google Scholar] [CrossRef] [PubMed]
- Weßels, C.; Strommenger, B.; Klare, I.; Bender, J.; Messler, S.; Mattner, F.; Krakau, M.; Werner, G.; Layer, F. Emergence and control of linezolid-resistant Staphylococcus epidermidis in an ICU of a German hospital. J. Antimicrob. Chemother. 2018, 73, 1185–1193. [Google Scholar] [CrossRef] [PubMed]
- Dortet, L.; Glaser, P.; Kassis-Chikhani, N.; Girlich, D.; Ichai, P.; Boudon, M.; Samuel, D.; Creton, E.; Imanci, D.; Bonnin, R.; et al. Long-lasting successful dissemination of resistance to oxazolidinones in MDR Staphylococcus epidermidis clinical isolates in a tertiary care hospital in France. J. Antimicrob. Chemother. 2018, 73, 41–51. [Google Scholar] [CrossRef] [PubMed]
- Beekmann, S.E.; Gilbert, D.N.; Polgreen, P.M. Toxicity of extended courses of linezolid: Results of an Infectious Diseases Society of America Emerging Infections Network survey. Diagn. Microbiol. Infect. Dis. 2008, 62, 407–410. [Google Scholar] [CrossRef]
- Boak, L.M.; Rayner, C.R.; Grayson, M.L.; Paterson, D.L.; Spelman, D.; Khumra, S.; Capitano, B.; Forrest, A.; Li, J.; Nation, R.L.; et al. Clinical Population Pharmacokinetics and Toxicodynamics of Linezolid. Antimicrob. Agents Chemother. 2014, 58, 2334–2343. [Google Scholar] [CrossRef]
| Variable | Monomicrobial Infections (n = 155) | Polymicrobial Infections (n = 28) | All Infections (n = 183) |
|---|---|---|---|
| Mean age (year +/- SD) | 71.4 ± 13.2 | 70.3 ± 11.6 | 71.5 ± 13.3 |
| Male sex (no. [%]) | 85 (54.8) | 12 (42.9) | 97 (53.0) |
| Location of arthroplasty (no. [%]) | |||
| Knee | 53 (34.2) | 6 (21.4) | 59 (32.2) |
| Hip | 102 (65.8) | 22 (78.6) | 124 (67.8) |
| Presentation of infection (no. [%]) | |||
| Acute infection (<1 month) | 33 (21.3) | 10 (35.7) | 43 (23.5) |
| Chronic infection (>1 month) | 122 (78.7) | 18 (64.3) | 140 (76.5) |
| Type of surgery (no. [%]) | |||
| Debridement with retention | 48 (31.0) | 11 (39.3) | 59 (32.2) |
| One-stage technique | 43 (27.7) | 4 (14.3) | 47 (25.7) |
| Two-stage technique | 52 (33.5) | 9 (32.1) | 61 (33.3) |
| Permanent explantation of joint prosthesis | 5 (3.2) | 3 (10.7) | 8 (4.4) |
| Data not available | 7 (4.5) | 1 (3.6) | 8 (4.4) |
| Number of previous joint surgeries (no. [%]) | |||
| 1 | 83 (53.5) | 16 (57.1) | 99 (54.1) |
| 2 | 49 (31.6) | 5 (17.9) | 54 (29.5) |
| ≥3 | 16 (10.3) | 6 (21.4) | 22 (12.0) |
| Data not available | 7 (4.5) | 1 (3.6) | 8 (4.4) |
| Antibiotherapy in the 15 days before surgery (no. [%]) a | 44 (28.4) | 10 (35.7) | 54 (29.5) |
| β-lactams | 21 (47.7) | 5 (50.0) | 26 (48.1) |
| Pristinamycin | 3 (6.8) | 0 | 3 (5.6) |
| Clindamycin | 2 (4.5) | 0 | 2 (3.7) |
| Rifampin | 4 (9.1) | 0 | 4 (7.4) |
| Fluoroquinolones | 8 (18.2) | 1 (10.0) | 9 (16.7) |
| Cotrimoxazole | 5 (11.4) | 2 (20.0) | 7 (13.0) |
| Others | 13 (29.5) | 3 (30.0) | 16 (29.6) |
| S. aureus (n = 75) | CoNS * (n = 58) | Streptococci (n = 32) | Gram-Negative Bacilli (n = 32) | Anaerobes (n = 8) | C. acnes (n = 7) | Others ** (n = 11) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Enterobacterales (n = 26) | P. aeruginosa (n = 6) | |||||||||||||||
| Mono- or Polymicrobial infection | M | P | M | P | M | P | M | P | M | P | M | P | M | P | M | P |
| Day 1 | 54 | 12 | 38 | 10 | 19 | 10 | 10 | 14 | 2 | 2 | 1 | 4 | 3 | |||
| Day 2 | 2 | 1 | 6 | 3 | 3 | 1 | 2 | 1 | 4 | 2 | ||||||
| Day 3 | 1 | 2 | ||||||||||||||
| Day 4 | 1 | 1 | ||||||||||||||
| Day 5 | 2 | 1 | 1 | 1 | 1 | |||||||||||
| Day 7 | 1 | 1 | 1 | 1 | ||||||||||||
| Day 14 | 1 | 1 | ||||||||||||||
 |  |  |  | |||||||||||||
| p-value | 0.2135 | 0.8946 | 0.2332 | 0.2176 | NC | NC | NC | NC | ||||||||
| Monomicrobial (n = 155) | Polymicrobial (n = 28) | Both (n = 183) | ||||
|---|---|---|---|---|---|---|
| n | % | n | % | n | % | |
| Day 1 | 130 | 83.9 | 16 | 57.1 | 146 | 79.8 |
| Day 2 | 146 | 94.2 | 21 | 75.0 | 167 | 91.3 |
| Day 3 | 147 | 94.8 | 22 | 78.6 | 169 | 92.3 |
| Day 4 | 148 | 95.5 | 23 | 82.1 | 171 | 93.4 |
| Day 5 | 151 | 97.4 | 26 | 92.9 | 177 | 96.7 |
| Day 7 | 154 | 99.4 | 27 | 96.4 | 181 | 98.9 |
| Day 14 | 155 | 100.0 | 28 | 100.0 | 183 | 100.0 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Deroche, L.; Bémer, P.; Valentin, A.-S.; Jolivet-Gougeon, A.; Tandé, D.; Héry-Arnaud, G.; Lemarié, C.; Kempf, M.; Bret, L.; Burucoa, C.; et al. The Right Time to Safely Re-Evaluate Empirical Antimicrobial Treatment of Hip or Knee Prosthetic Joint Infections. J. Clin. Med. 2019, 8, 2113. https://doi.org/10.3390/jcm8122113
Deroche L, Bémer P, Valentin A-S, Jolivet-Gougeon A, Tandé D, Héry-Arnaud G, Lemarié C, Kempf M, Bret L, Burucoa C, et al. The Right Time to Safely Re-Evaluate Empirical Antimicrobial Treatment of Hip or Knee Prosthetic Joint Infections. Journal of Clinical Medicine. 2019; 8(12):2113. https://doi.org/10.3390/jcm8122113
Chicago/Turabian StyleDeroche, Luc, Pascale Bémer, Anne-Sophie Valentin, Anne Jolivet-Gougeon, Didier Tandé, Geneviève Héry-Arnaud, Carole Lemarié, Marie Kempf, Laurent Bret, Christophe Burucoa, and et al. 2019. "The Right Time to Safely Re-Evaluate Empirical Antimicrobial Treatment of Hip or Knee Prosthetic Joint Infections" Journal of Clinical Medicine 8, no. 12: 2113. https://doi.org/10.3390/jcm8122113
APA StyleDeroche, L., Bémer, P., Valentin, A.-S., Jolivet-Gougeon, A., Tandé, D., Héry-Arnaud, G., Lemarié, C., Kempf, M., Bret, L., Burucoa, C., Corvec, S., & Plouzeau, C. (2019). The Right Time to Safely Re-Evaluate Empirical Antimicrobial Treatment of Hip or Knee Prosthetic Joint Infections. Journal of Clinical Medicine, 8(12), 2113. https://doi.org/10.3390/jcm8122113






