Influence of Transfusion on the Risk of Acute Kidney Injury: ABO-Compatible versus ABO-Incompatible Liver Transplantation
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Anesthesia, Surgical Technique, and Preoperative Desensitization Preparation
2.3. Data Collection
2.4. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Conflicts of Interest
References
- Hilmi, I.A.; Damian, D.; Al-Khafaji, A.; Planinsic, R.; Boucek, C.; Sakai, T.; Chang, C.C.; Kellum, J.A. Acute kidney injury following orthotopic liver transplantation: Incidence, risk factors, and effects on patient and graft outcomes. Br. J. Anaesth. 2015, 114, 919–926. [Google Scholar] [CrossRef] [PubMed]
- Park, M.H.; Shim, H.S.; Kim, W.H.; Kim, H.J.; Kim, D.J.; Lee, S.H.; Kim, C.S.; Gwak, M.S.; Kim, G.S. Clinical Risk Scoring Models for Prediction of Acute Kidney Injury after Living Donor Liver Transplantation: A Retrospective Observational Study. PLoS ONE 2015, 10, e0136230. [Google Scholar] [CrossRef] [PubMed]
- Utsumi, M.; Umeda, Y.; Sadamori, H.; Nagasaka, T.; Takaki, A.; Matsuda, H.; Shinoura, S.; Yoshida, R.; Nobuoka, D.; Satoh, D.; et al. Risk factors for acute renal injury in living donor liver transplantation: Evaluation of the RIFLE criteria. Transpl. Int. 2013, 26, 842–852. [Google Scholar] [CrossRef] [PubMed]
- Zhu, M.; Li, Y.; Xia, Q.; Wang, S.; Qiu, Y.; Che, M.; Dai, H.; Qian, J.; Ni, Z.; Axelsson, J.; et al. Strong impact of acute kidney injury on survival after liver transplantation. Transplant. Proc. 2010, 42, 3634–3638. [Google Scholar] [CrossRef] [PubMed]
- Inoue, Y.; Soyama, A.; Takatsuki, M.; Hidaka, M.; Muraoka, I.; Kanematsu, T.; Eguchi, S. Acute kidney injury following living donor liver transplantation. Clin. Transplant. 2012, 26, E530–E535. [Google Scholar] [CrossRef]
- Chen, J.; Singhapricha, T.; Hu, K.Q.; Hong, J.C.; Steadman, R.H.; Busuttil, R.W.; Xia, V.W. Postliver transplant acute renal injury and failure by the RIFLE criteria in patients with normal pretransplant serum creatinine concentrations: A matched study. Transplantation 2011, 91, 348–353. [Google Scholar] [CrossRef]
- Lebron Gallardo, M.; Herrera Gutierrez, M.E.; Seller Perez, G.; Curiel Balsera, E.; Fernandez Ortega, J.F.; Quesada Garcia, G. Risk factors for renal dysfunction in the postoperative course of liver transplant. Liver Transpl. 2004, 10, 1379–1385. [Google Scholar] [CrossRef]
- Jun, I.G.; Kwon, H.M.; Jung, K.W.; Moon, Y.J.; Shin, W.J.; Song, J.G.; Hwang, G.S. The Impact of Postreperfusion Syndrome on Acute Kidney Injury in Living Donor Liver Transplantation: A Propensity Score Analysis. Anesth. Analg. 2018, 127, 369–378. [Google Scholar] [CrossRef]
- Jun, I.G.; Lee, B.; Kim, S.O.; Shin, W.J.; Bang, J.Y.; Song, J.G.; Song, G.W.; Lee, S.G.; Hwang, G.S. Comparison of acute kidney injury between ABO-compatible and ABO-incompatible living donor liver transplantation: A propensity matching analysis. Liver Transpl. 2016, 22, 1656–1665. [Google Scholar] [CrossRef]
- Hilmi, I.A.; Damian, D.; Al-Khafaji, A.; Sakai, T.; Donaldson, J.; Winger, D.G.; Kellum, J.A. Acute kidney injury after orthotopic liver transplantation using living donor versus deceased donor grafts: A propensity score-matched analysis. Liver Transpl. 2015, 21, 1179–1185. [Google Scholar] [CrossRef]
- Lee, H.C.; Yoon, S.B.; Yang, S.M.; Kim, W.H.; Ryu, H.G.; Jung, C.W.; Suh, K.S.; Lee, K.H. Prediction of Acute Kidney Injury after Liver Transplantation: Machine Learning Approaches vs. Logistic Regression Model. J. Clin. Med. 2018, 7, 428. [Google Scholar] [CrossRef] [PubMed]
- Thomas, M.E.; Blaine, C.; Dawnay, A.; Devonald, M.A.; Ftouh, S.; Laing, C.; Latchem, S.; Lewington, A.; Milford, D.V.; Ostermann, M. The definition of acute kidney injury and its use in practice. Kidney Int. 2015, 87, 62–73. [Google Scholar] [CrossRef] [PubMed]
- Thongprayoon, C.; Kaewput, W.; Thamcharoen, N.; Bathini, T.; Watthanasuntorn, K.; Lertjitbanjong, P.; Sharma, K.; Salim, S.A.; Ungprasert, P.; Wijarnpreecha, K.; et al. Incidence and Impact of Acute Kidney Injury after Liver Transplantation: A Meta-Analysis. J. Clin. Med. 2019, 8, 372. [Google Scholar] [CrossRef] [PubMed]
- Paramesh, A.S.; Roayaie, S.; Doan, Y.; Schwartz, M.E.; Emre, S.; Fishbein, T.; Florman, S.; Gondolesi, G.E.; Krieger, N.; Ames, S.; et al. Post-liver transplant acute renal failure: Factors predicting development of end-stage renal disease. Clin. Transplant. 2004, 18, 94–99. [Google Scholar] [CrossRef]
- Trinh, E.; Alam, A.; Tchervenkov, J.; Cantarovich, M. Impact of acute kidney injury following liver transplantation on long-term outcomes. Clin. Transplant. 2017, 31. [Google Scholar] [CrossRef]
- Kawagishi, N.; Satomi, S. ABO-incompatible living donor liver transplantation: New insights into clinical relevance. Transplantation 2008, 85, 1523–1525. [Google Scholar] [CrossRef]
- Song, G.W.; Lee, S.G. Living donor liver transplantation. Curr. Opin. Organ Transplant. 2014, 19, 217–222. [Google Scholar] [CrossRef]
- Kim, J.M.; Kwon, C.H.; Joh, J.W.; Kang, E.S.; Park, J.B.; Lee, J.H.; Kim, S.J.; Paik, S.W.; Lee, S.K.; Kim, D.W. ABO-incompatible living donor liver transplantation is suitable in patients without ABO-matched donor. J. Hepatol. 2013, 59, 1215–1222. [Google Scholar] [CrossRef]
- Song, G.W.; Lee, S.G.; Hwang, S.; Kim, K.H.; Ahn, C.S.; Moon, D.B.; Ha, T.Y.; Jung, D.H.; Park, G.C.; Kang, S.H.; et al. Biliary stricture is the only concern in ABO-incompatible adult living donor liver transplantation in the rituximab era. J. Hepatol. 2014, 61, 575–582. [Google Scholar] [CrossRef]
- Ikegami, T.; Taketomi, A.; Soejima, Y.; Yoshizumi, T.; Uchiyama, H.; Harada, N.; Iguchi, T.; Hashimoto, N.; Maehara, Y. Rituximab, IVIG, and plasma exchange without graft local infusion treatment: A new protocol in ABO incompatible living donor liver transplantation. Transplantation 2009, 88, 303–307. [Google Scholar] [CrossRef]
- Kwon, H.M.; Jun, I.G.; Lee, J.; Moon, Y.J.; Jung, K.W.; Jeong, H.W.; Park, Y.S.; Song, J.G.; Hwang, G.S. Prevalent metabolic derangement and severe thrombocytopenia in ABO-incompatible liver recipients with pre-transplant plasma exchange. Sci. Rep. 2018, 8, 6679. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.H.; Lee, H.C.; Lim, L.; Ryu, H.G.; Jung, C.W. Intraoperative Oliguria with Decreased SvO2 Predicts Acute Kidney Injury after Living Donor Liver Transplantation. J. Clin. Med. 2018, 8, 29. [Google Scholar] [CrossRef] [PubMed]
- Song, G.W.; Lee, S.G.; Hwang, S.; Kim, K.H.; Ahn, C.S.; Moon, D.B.; Ha, T.Y.; Jung, D.H.; Park, G.C.; Kim, W.J.; et al. ABO-Incompatible Adult Living Donor Liver Transplantation under the Desensitization Protocol with Rituximab. Am. J. Transplant. 2016, 16, 157–170. [Google Scholar] [CrossRef] [PubMed]
- Durand, F.; Francoz, C.; Asrani, S.K.; Khemichian, S.; Pham, T.A.; Sung, R.S.; Genyk, Y.S.; Nadim, M.K. Acute Kidney Injury after Liver Transplantation. Transplantation 2018, 102, 1636–1649. [Google Scholar] [CrossRef] [PubMed]
- Paugam-Burtz, C.; Kavafyan, J.; Merckx, P.; Dahmani, S.; Sommacale, D.; Ramsay, M.; Belghiti, J.; Mantz, J. Postreperfusion syndrome during liver transplantation for cirrhosis: Outcome and predictors. Liver Transpl. 2009, 15, 522–529. [Google Scholar] [CrossRef]
- Vives, M.; Callejas, R.; Duque, P.; Echarri, G.; Wijeysundera, D.N.; Hernandez, A.; Sabate, A.; Bes-Rastrollo, M.; Monedero, P. Modern hydroxyethyl starch and acute kidney injury after cardiac surgery: A prospective multicentre cohort. Br. J. Anaesth. 2016, 117, 458–463. [Google Scholar] [CrossRef]
- Yang, S.M.; Choi, S.N.; Yu, J.H.; Yoon, H.K.; Kim, W.H.; Jung, C.W.; Suh, K.S.; Lee, K.H. Intraoperative hyponatremia is an independent predictor of one-year mortality after liver transplantation. Sci. Rep. 2018, 8, 18023. [Google Scholar] [CrossRef]
- Selzner, M.; Kashfi, A.; Cattral, M.S.; Selzner, N.; McGilvray, I.D.; Greig, P.D.; Levy, G.A.; Renner, E.L.; Grant, D.R. Live donor liver transplantation in high MELD score recipients. Ann. Surg. 2010, 251, 153–157. [Google Scholar] [CrossRef]
- Shin, S.R.; Kim, W.H.; Kim, D.J.; Shin, I.W.; Sohn, J.T. Prediction and Prevention of Acute Kidney Injury after Cardiac Surgery. Biomed. Res. Int. 2016, 2016, 2985148. [Google Scholar] [CrossRef]
- Sanchez-Urdazpal, L.; Batts, K.P.; Gores, G.J.; Moore, S.B.; Sterioff, S.; Wiesner, R.H.; Krom, R.A. Increased bile duct complications in liver transplantation across the ABO barrier. Ann. Surg. 1993, 218, 152–158. [Google Scholar] [CrossRef]
- Aksu Erdost, H.; Ozkardesler, S.; Ocmen, E.; Avkan-Oguz, V.; Akan, M.; Iyilikci, L.; Unek, T.; Ozbilgin, M.; Meseri Dalak, R.; Astarcioglu, I. Acute Renal Injury Evaluation after Liver Transplantation: With RIFLE Criteria. Transplant. Proc. 2015, 47, 1482–1487. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.H.; Park, M.H.; Kim, H.J.; Lim, H.Y.; Shim, H.S.; Sohn, J.T.; Kim, C.S.; Lee, S.M. Potentially modifiable risk factors for acute kidney injury after surgery on the thoracic aorta: A propensity score matched case-control study. Medicine (Baltimore) 2015, 94, e273. [Google Scholar] [CrossRef] [PubMed]
- Pandey, S.; Vyas, G.N. Adverse effects of plasma transfusion. Transfusion 2012, 52 (Suppl. 1), 65s–79s. [Google Scholar] [CrossRef] [PubMed]
- Sachs, U.J. Non-infectious serious hazards in plasma transfusion. Transfus. Apher. Sci. 2010, 43, 381–386. [Google Scholar] [CrossRef]
- Kim, W.H.; Oh, H.W.; Yang, S.M.; Yu, J.H.; Lee, H.C.; Jung, C.W.; Suh, K.S.; Lee, K.H. Intraoperative Hemodynamic Parameters and Acute Kidney Injury after Living Donor Liver Transplantation. Transplantation 2019, 103, 1877–1886. [Google Scholar] [CrossRef]
- Rohde, J.M.; Dimcheff, D.E.; Blumberg, N.; Saint, S.; Langa, K.M.; Kuhn, L.; Hickner, A.; Rogers, M.A. Health care-associated infection after red blood cell transfusion: A systematic review and meta-analysis. JAMA 2014, 311, 1317–1326. [Google Scholar] [CrossRef]
- Delaney, M.; Wendel, S.; Bercovitz, R.S.; Cid, J.; Cohn, C.; Dunbar, N.M.; Apelseth, T.O.; Popovsky, M.; Stanworth, S.J.; Tinmouth, A.; et al. Transfusion reactions: Prevention, diagnosis, and treatment. Lancet 2016, 388, 2825–2836. [Google Scholar] [CrossRef]
- Kim, W.H.; Lee, H.C.; Ryu, H.G.; Chung, E.J.; Kim, B.; Jung, H.; Jung, C.W. Reliability of Point-of-Care Hematocrit Measurement during Liver Transplantation. Anesth. Analg. 2017, 125, 2038–2044. [Google Scholar] [CrossRef]
- Yoo, Y.C.; Shim, J.K.; Kim, J.C.; Jo, Y.Y.; Lee, J.H.; Kwak, Y.L. Effect of single recombinant human erythropoietin injection on transfusion requirements in preoperatively anemic patients undergoing valvular heart surgery. Anesthesiology 2011, 115, 929–937. [Google Scholar] [CrossRef]
- Wagener, G.; Gubitosa, G.; Renz, J.; Kinkhabwala, M.; Brentjens, T.; Guarrera, J.V.; Emond, J.; Lee, H.T.; Landry, D. Vasopressin decreases portal vein pressure and flow in the native liver during liver transplantation. Liver Transpl. 2008, 14, 1664–1670. [Google Scholar] [CrossRef]
| Before Matching | Standardized Difference | After Matching | Standardized Difference | |||||
|---|---|---|---|---|---|---|---|---|
| Characteristic | ABO-i LT | ABO-c LT | p-Value | ABO-i LT | ABO-c LT | p-Value | ||
| Sample size | 119 | 766 | 111 | 184 | ||||
| Demographic data | ||||||||
| Age, years | 55 (49–60) | 53 (48–60) | 0.054 | 2.41 | 54 (49–61) | 55 (50–60) | 0.691 | 0.73 |
| Female, n | 47 (39.5) | 176 (23.0) | <0.001 | 16.52 | 41 (36.9) | 65 (35.3) | 0.780 | 1.61 |
| Body mass index, kg/m2 | 23.1 (21.6–25.4) | 23.1 (21.1–25.3) | 0.792 | 0.19 | 23.2 (21.5–25.3) | 23.2 (21.6–25.4) | 0.951 | 0.04 |
| Etiology of liver disease | ||||||||
| Alcoholic liver cirrhosis, n | 19 (16.0) | 70 (9.1) | 0.021 | 6.82 | 17 (15.3) | 31(16.8) | 0.374 | 1.50 |
| HBV hepatitis, n | 70 (58.8) | 328 (42.8) | 0.001 | 16.00 | 60 (54.1) | 95 (51.6) | 0.686 | 2.50 |
| HCV hepatitis, n | 8 (6.7) | 58 (7.6) | 0.853 | 0.85 | 8 (7.2) | 12 (6.5) | 0.644 | 0.70 |
| Hepatocellular carcinoma, n | 71 (59.7) | 420 (54.8) | 0.324 | 4.83 | 63 (56.8) | 106 (57.6) | 0.904 | 0.80 |
| Cholestatic disease, n | 13 (10.9) | 16 (2.1) | <0.001 | 8.84 | 12 (10.8) | 10 (6.4) | 0.121 | 6.50 |
| Nonalcoholic steatohepatitis, n | 3 (2.5) | 41 (5.4) | 0.256 | 2.83 | 3 (2.7) | 8 (4.3) | 0.264 | 1.60 |
| Baseline medical status | ||||||||
| Hypertension, n | 17 (14.3) | 77 (10.1) | 0.163 | 4.23 | 16 (14.4) | 26 (14.1) | 0.284 | 0.30 |
| Diabetes mellitus, n | 19 (16.0) | 107 (14.0) | 0.562 | 2.00 | 19 (17.1) | 32 (17.4) | 0.789 | 0.30 |
| Preoperative hemoglobin, g/dL | 10.4 (8.9–12.1) | 10.8 (9.2–12.6) | 0.202 | 0.73 | 10.4 (8.7–12.4) | 11.0 (9.1–12.5) | 0.335 | 0.26 |
| Preoperative serum sodium concentration, mEq/L | 139 (134–141) | 137 (132–140) | 0.050 | 3.82 | 141 (137–144) | 140 (137–142) | 0.180 | 0.32 |
| MELD score | 14.3 (11.1–20.7) | 15.9 (12.0–22.7) | 0.042 | 3.09 | 15.0 (9.9–20.4) | 14.2 (9.9–19.7) | 0.601 | 0.60 |
| Child class, n (A/B/C) | 53 (43.3)/42 (35.3)/ 24 (20.2) | 298 (38.9)/280 (36.6)/188 (24.5) | 0.194 | - | 45 (40.5)/ 42 (37.8)/24 (21.6) | 75 (40.8)/ 71 (38.6)/38 (20.7) | 0.117 | - |
| Preoperative LVEF, % | 66 (62–68) | 65 (62–68) | 0.978 | 0.46 | 66 (61–69) | 65 (61–69) | 0.540 | 0.35 |
| Preoperative beta-blocker, n | 7 (5.9) | 38 (5.0) | 0.670 | 0.92 | 7 (6.3) | 11 (6.0) | 0.273 | 0.30 |
| Preoperative diuretics, n | 6 (17.1) | 29 (3.8) | 0.003 | 13.4 | 5 (12.2) | 18 (9.8) | 0.131 | 2.40 |
| Previous abdominal surgery, n | 2 (1.7) | 24 (3.1) | 0.383 | 1.45 | 2 (1.8) | 5 (2.7) | 0.491 | 0.90 |
| Donor/graft factors | ||||||||
| Age, years | 32 (24–42) | 30 (23–39) | 0.130 | 2.88 | 30 (23–40) | 30 (23–38) | 0.242 | 1.16 |
| Estimated GRWR | 1.24 (1.05–1.46) | 1.20 (1.04–1.41) | 0.393 | 0.02 | 1.24 (1.02–1.45) | 1.21 (1.06–1.47) | 0.455 | 0.00 |
| Operation and anesthesia details | ||||||||
| Operation time, h | 6.4 (5.6–7.8) | 6.9 (5.8–8.0) | 0.074 | 1.55 | 6.5 (5.5–7.5) | 6.6 (5.4–7.6) | 0.093 | 0.38 |
| Cold ischemic time, min | 86 (70–106) | 88 (68–240) | 0.061 | 13.69 | 86 (70–106) | 88 (69–109) | 0.233 | 3.40 |
| Warm ischemic time, min | 28 (26–33) | 30 (28–35) | 0.101 | 4.30 | 28 (25–33) | 30 (26–34) | 0.109 | 1.47 |
| Intraoperative dose of epinephrine bolus, mcg | 0 (0–20) | 0 (0–30) | 0.063 | 3.12 | 0 (0–20) | 0 (0–10) | 0.787 | 0.65 |
| Reperfusion syndrome, n | 9 (7.6) | 79 (10.3) | 0.351 | 2.75 | 9 (8.1) | 12 (6.5) | 0.644 | 1.60 |
| Intraoperative mean blood glucose, mg/dL | 155 (152–161) | 163 (145–180) | <0.001 | 9.37 | 157 (154–164) | 160 (148–170) | 0.092 | 3.61 |
| Crystalloid intake, 100 mL | 33 (22–49) | 36 (25–53) | 0.023 | 0.57 | 34 (23–48) | 34 (23–45) | 0.646 | 0.40 |
| Colloid intake, 100 ml | 0 (0–0) | 0 (0–5) | 0.001 | 2.55 | 0 (0–3) | 0 (0–4) | 0.102 | 0.68 |
| Bleeding and transfusion amount | ||||||||
| pRBC transfusion, units | 5 (2–10) | 6 (2–12) | 0.048 | 1.25 | 5 (0–9) | 5 (0–10) | 0.942 | 0.02 |
| FFP transfusion, units | 5 (1–9) | 6 (1–12) | 0.060 | 3.47 | 3 (0–7) | 3 (0–8) | 0.101 | 0.98 |
| Blood loss per body weight, mL/kg | 42 (22–86) | 47 (24–100) | 0.056 | 22.11 | 36 (19–73) | 36 (18–73) | 0.753 | 9.29 |
| Variable | Odds Ratio | 95% CI | p-Value |
|---|---|---|---|
| Body mass index, recipient | 1.07 | 1.01–1.12 | 0.012 |
| Preoperative beta-blocker administration | 2.00 | 0.86–4.14 | 0.052 |
| Preoperative diuretics administration | 1.89 | 0.86–4.14 | 0.113 |
| ABO-incompatible liver transplantation | 2.54 | 1.20–5.38 | 0.015 |
| MELD score | 1.02 | 1.00–1.04 | 0.049 |
| Preoperative hemoglobin, g/dL | 0.89 | 0.82–0.97 | 0.007 |
| Postreperfusion syndrome | 1.09 | 0.99–1.80 | 0.051 |
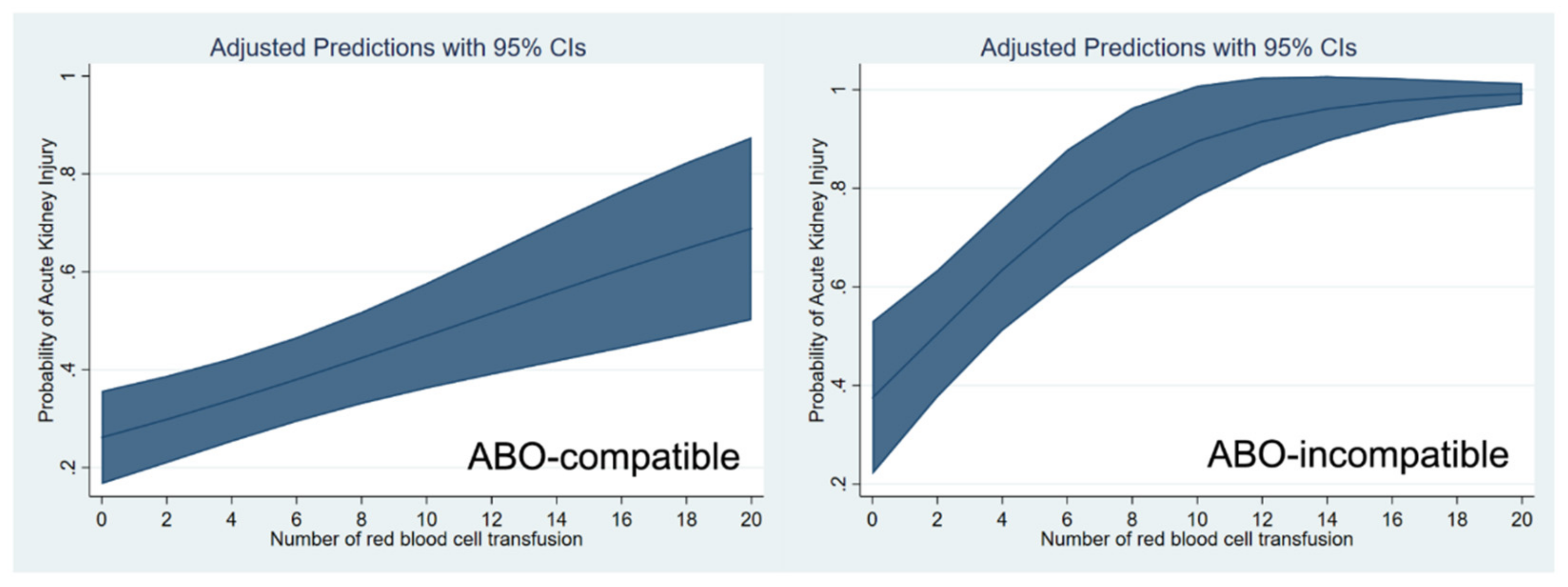
| pRBC transfusion | |||
| As continuous variable, per unit | 1.09 | 1.06–1.11 | <0.001 |
| No red cell transfusion | Reference | ||
| 1–2 units | 1.07 | 0.97–2.04 | 0.050 |
| 3–4 units | 1.92 | 1.11–2.41 | 0.015 |
| 5–9 units | 3.20 | 1.95–5.44 | <0.001 |
| ≥10 units | 4.88 | 3.00–6.84 | <0.001 |
| Variable | Odds Ratio | 95% CI | p-Value |
|---|---|---|---|
| ABO-compatible LT in the matched cohort | |||
| Red cell transfusion as continuous variable | 1.11 | 1.04–1.17 | 0.001 |
| Red cell transfusion as categorized variable | |||
| No transfusion | Reference | ||
| 1–2 units | 1.73 | 0.96–2.35 | 0.068 |
| 3–4 units | 2.01 | 1.04–3.27 | 0.040 |
| 5–9 units | 3.29 | 1.73–5.02 | 0.015 |
| ≥10 units | 4.45 | 2.37–6.61 | <0.001 |
| ABO-incompatible LT in the matched cohorts | |||
| Red cell transfusion as continuous variable | 1.32 | 1.13–1.59 | <0.001 |
| Red cell transfusion as categorized variable | |||
| No transfusion | Reference | ||
| 1–2 units | 1.94 | 1.04–2.64 | 0.010 |
| 3–4 units | 3.05 | 1.43–5.29 | <0.001 |
| 5–9 units | 4.67 | 2.44–6.84 | <0.001 |
| ≥10 units | 5.99 | 3.84–8.01 | <0.001 |
| Variable | Odds Ratio | 95% CI | p-Value |
|---|---|---|---|
| ABO-compatible LT in the matched cohort | |||
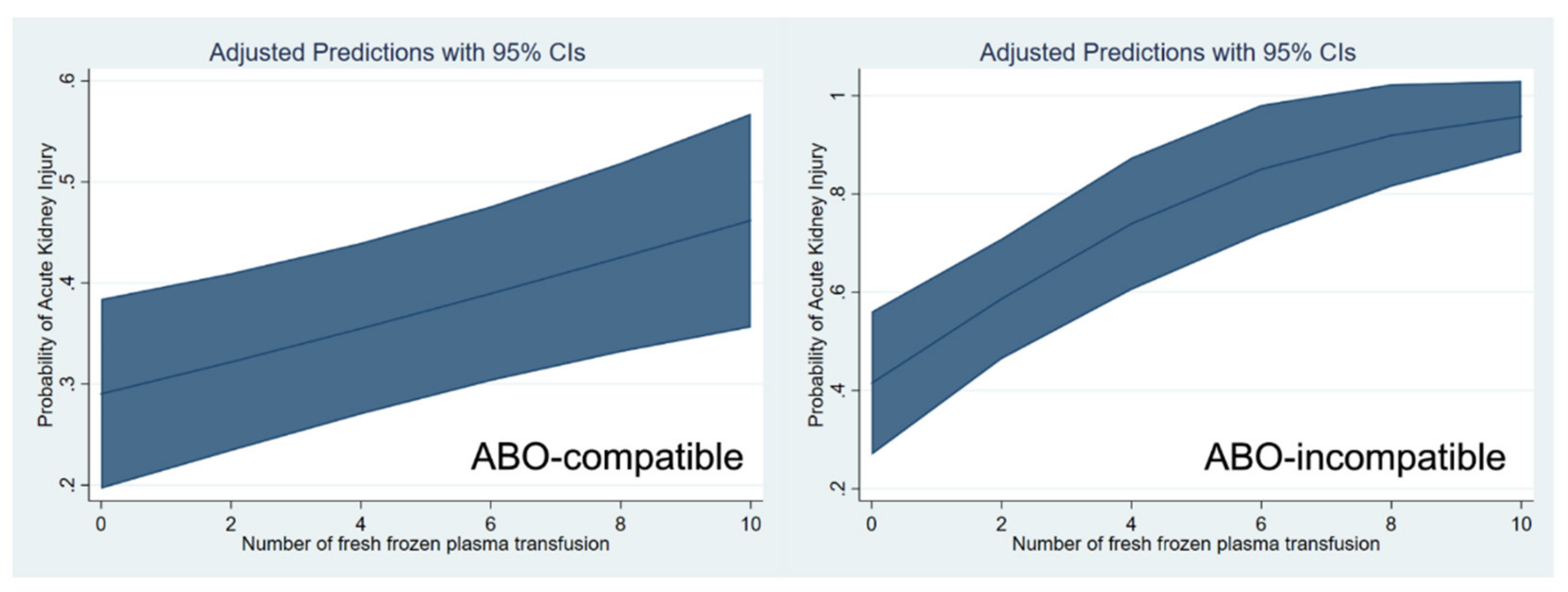
| Fresh frozen plasma as continuous variable | 1.07 | 1.03–1.13 | 0.002 |
| Fresh frozen plasma as categorized variable | |||
| No transfusion | Reference | ||
| 1–5 units | 2.41 | 1.27–4.15 | 0.012 |
| 5–9 units | 3.46 | 1.34–6.19 | 0.004 |
| ≥10 units | 4.68 | 1.81–8.01 | <0.001 |
| ABO-incompatible LT in the matched cohorts | |||
| Fresh frozen plasma as continuous variable | 1.44 | 1.16–1.75 | <0.001 |
| Fresh frozen plasma as categorized variable | |||
| No red cell transfusion | Reference | ||
| 1–5 units | 3.59 | 2.27–5.71 | <0.001 |
| 6–9 units | 4.86 | 2.92–7.22 | <0.001 |
| ≥10 units | 5.98 | 3.02–9.65 | <0.001 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yu, J.H.; Kwon, Y.; Kim, J.; Yang, S.-M.; Kim, W.H.; Jung, C.-W.; Suh, K.-S.; Lee, K.H. Influence of Transfusion on the Risk of Acute Kidney Injury: ABO-Compatible versus ABO-Incompatible Liver Transplantation. J. Clin. Med. 2019, 8, 1785. https://doi.org/10.3390/jcm8111785
Yu JH, Kwon Y, Kim J, Yang S-M, Kim WH, Jung C-W, Suh K-S, Lee KH. Influence of Transfusion on the Risk of Acute Kidney Injury: ABO-Compatible versus ABO-Incompatible Liver Transplantation. Journal of Clinical Medicine. 2019; 8(11):1785. https://doi.org/10.3390/jcm8111785
Chicago/Turabian StyleYu, Je Hyuk, Yongsuk Kwon, Jay Kim, Seong-Mi Yang, Won Ho Kim, Chul-Woo Jung, Kyung-Suk Suh, and Kook Hyun Lee. 2019. "Influence of Transfusion on the Risk of Acute Kidney Injury: ABO-Compatible versus ABO-Incompatible Liver Transplantation" Journal of Clinical Medicine 8, no. 11: 1785. https://doi.org/10.3390/jcm8111785
APA StyleYu, J. H., Kwon, Y., Kim, J., Yang, S.-M., Kim, W. H., Jung, C.-W., Suh, K.-S., & Lee, K. H. (2019). Influence of Transfusion on the Risk of Acute Kidney Injury: ABO-Compatible versus ABO-Incompatible Liver Transplantation. Journal of Clinical Medicine, 8(11), 1785. https://doi.org/10.3390/jcm8111785





