Association of Serum PCSK9 Levels with Antibiotic Resistance and Severity of Disease in Patients with Bacterial Infections Admitted to Intensive Care Units
Abstract
1. Introduction
2. Methods
2.1. Patients
2.2. Blood Sampling and Biochemical Measurements
2.3. Assessment of the Severity of Disease
2.4. Statistical Analyses
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Chrétien, M. My road to Damascus: How I converted to the prohormone theory and the proprotein convertases. Biochem. Cell Biol. 2012, 90, 750–768. [Google Scholar] [CrossRef]
- Le Bras, M.; Roquilly, A.; Deckert, V.; Langhi, C.; Feuillet, F.; Sébille, V.; Mahé, P.-J.; Bach, K.; Masson, D.; Lagrost, L. Plasma PCSK9 is a late biomarker of severity in patients with severe trauma injury. J. Clin. Endocrinol. Metab. 2013, 98, 732–736. [Google Scholar] [CrossRef] [PubMed]
- Nagashima, S.; Morishima, K.; Okamoto, H.; Ishibashi, S. Possible involvement of PCSK9 overproduction in hyperlipoproteinemia associated with hepatocellular carcinoma: A case report. J. Clin. Lipidol. 2016, 10, 1045–1049. [Google Scholar] [CrossRef]
- Momtazi, A.A.; Banach, M.; Pirro, M.; Stein, E.A.; Sahebkar, A. PCSK9 and diabetes: Is there a link? Drug Discov. Today 2017, 22, 883–895. [Google Scholar] [CrossRef] [PubMed]
- Momtazi, A.A.; Banach, M.; Sahebkar, A. PCSK9 inhibitors in sepsis: A new potential indication? Expert Opin. Investig. Drugs 2017, 26, 137–139. [Google Scholar] [CrossRef][Green Version]
- Reiner, Ž. PCSK9 inhibitors–past, present and future. Expert Opin. Drug Metab. Toxicol. 2015, 11, 1517–1521. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Sahebkar, A.; Watts, G.F. New therapies targeting apoB metabolism for high-risk patients with inherited dyslipidaemias: What can the clinician expect? Cardiovasc. Drugs Ther. 2013, 27, 559–567. [Google Scholar] [CrossRef]
- Reiner, Ž. Management of patients with familial hypercholesterolaemia. Nat. Rev. Cardiol. 2015, 12, 565. [Google Scholar] [CrossRef]
- Pećin, I.; Hartgers, M.L.; Hovingh, G.K.; Dent, R.; Reiner, Ž. Prevention of cardiovascular disease in patients with familial hypercholesterolaemia: The role of PCSK9 inhibitors. Eur. J. Prev. Cardiol. 2017, 24, 1383–1401. [Google Scholar] [CrossRef]
- Reiner, Ž. PCSK9 inhibitors in clinical practice: Expectations and reality. Atherosclerosis 2018, 270, 187–188. [Google Scholar] [CrossRef]
- Ding, Z.; Liu, S.; Wang, X.; Deng, X.; Fan, Y.; Shahanawaz, J.; Shmookler Reis, R.J.; Varughese, K.I.; Sawamura, T.; Mehta, J.L. Cross-talk between LOX-1 and PCSK9 in vascular tissues. Cardiovasc. Res. 2015, 107, 556–567. [Google Scholar] [CrossRef] [PubMed]
- Ding, Z.; Liu, S.; Wang, X.; Deng, X.; Fan, Y.; Sun, C.; Wang, Y.; Mehta, J.L. Hemodynamic shear stress via ROS modulates PCSK9 expression in human vascular endothelial and smooth muscle cells and along the mouse aorta. Antioxid. Redox Signal. 2015, 22, 760–771. [Google Scholar] [CrossRef] [PubMed]
- Kohli, P.; Ganz, P.; Ma, Y.; Scherzer, R.; Hur, S.; Weigel, B.; Grunfeld, C.; Deeks, S.; Wasserman, S.; Scott, R. HIV and Hepatitis C–Coinfected Patients Have Lower Low-Density Lipoprotein Cholesterol Despite Higher Proprotein Convertase Subtilisin Kexin 9 (PCSK 9): An Apparent “PCSK 9–Lipid Paradox”. J. Am. Heart Assoc. 2016, 5, e002683. [Google Scholar] [CrossRef] [PubMed]
- Syed, G.H.; Tang, H.; Khan, M.; Hassanein, T.; Liu, J.; Siddiqui, A. Hepatitis C virus stimulates low-density lipoprotein receptor expression to facilitate viral propagation. J. Virol. 2014, 88, 2519–2529. [Google Scholar] [CrossRef]
- Paciullo, F.; Fallarino, F.; Bianconi, V.; Mannarino, M.R.; Sahebkar, A.; Pirro, M. PCSK9 at the crossroad of cholesterol metabolism and immune function during infections. J. Cell Physiol. 2017, 232, 2330–2338. [Google Scholar] [CrossRef]
- Pirro, M.; Bianconi, V.; Francisci, D.; Schiaroli, E.; Bagaglia, F.; Sahebkar, A.; Baldelli, F. Hepatitis C virus and proprotein convertase subtilisin/kexin type 9: A detrimental interaction to increase viral infectivity and disrupt lipid metabolism. J. Cell Mol. Med. 2017, 21, 3150–3161. [Google Scholar] [CrossRef]
- Azzam, K.M.; Fessler, M.B. Crosstalk between reverse cholesterol transport and innate immunity. Trends Endocrinol. Metab. 2012, 23, 169–178. [Google Scholar] [CrossRef]
- Walley, K.R.; Francis, G.A.; Opal, S.M.; Stein, E.A.; Russell, J.A.; Boyd, J.H. The Central Role of Proprotein Convertase Subtilisin/Kexin Type 9 in Septic Pathogen Lipid Transport and Clearance. Am. J. Respir. Crit. Care Med. 2015, 192, 1275–1286. [Google Scholar] [CrossRef]
- Grin, P.M.; Dwivedi, D.J.; Chathely, K.M.; Trigatti, B.L.; Prat, A.; Seidah, N.G.; Liaw, P.C.; Fox-Robichaud, A.E. Low-density lipoprotein (LDL)-dependent uptake of Gram-positive lipoteichoic acid and Gram-negative lipopolysaccharide occurs through LDL receptor. Sci. Rep. 2018, 8, 10496. [Google Scholar] [CrossRef]
- Walley, K.R.; Thain, K.R.; Russell, J.A.; Reilly, M.P.; Meyer, N.J.; Ferguson, J.F.; Christie, J.D.; Nakada, T.A.; Fjell, C.D.; Thair, S.A.; et al. PCSK9 is a critical regulator of the innate immune response and septic shock outcome. Sci. Transl. Med. 2014, 6. [Google Scholar] [CrossRef]
- Genga, K.R.; Lo, C.; Cirstea, M.S.; Leitao Filho, F.S.; Walley, K.R.; Russell, J.A.; Linder, A.; Francis, G.A.; Boyd, J.H. Impact of PCSK9 loss-of-function genotype on 1-year mortality and recurrent infection in sepsis survivors. EBioMedicine 2018, 38, 257–264. [Google Scholar] [CrossRef] [PubMed]
- Feng, Q.; Wei, W.Q.; Chaugai, S.; Carranza Leon, B.G.; Kawai, V.; Carranza Leon, D.A.; Jiang, L.; Zhong, X.; Liu, G.; Ihegword, A.; et al. A Genetic Approach to the Association Between PCSK9 and Sepsis. JAMA Netw. Open 2019, 2, e1911130. [Google Scholar] [CrossRef] [PubMed]
- Boyd, J.H.; Fjell, C.D.; Russell, J.A.; Sirounis, D.; Cirstea, M.S.; Walley, K.R. Increased Plasma PCSK9 Levels Are Associated with Reduced Endotoxin Clearance and the Development of Acute Organ Failures during Sepsis. J. Innate Immun. 2016, 8, 211–220. [Google Scholar] [CrossRef]
- Baruch, A.; Mosesova, S.; Davis, J.D.; Budha, N.; Vilimovskij, A.; Kahn, R.; Peng, K.; Cowan, K.J.; Harris, L.P.; Gelzleichter, T. Effects of RG7652, a monoclonal antibody against PCSK9, on LDL-C, LDL-C subfractions, and inflammatory biomarkers in patients at high risk of or with established coronary heart disease (from the Phase 2 EQUATOR Study). Am. J. Cardiol. 2017, 119, 1576–1583. [Google Scholar] [CrossRef]
- Berger, J.-M.; Valdes, A.L.; Gromada, J.; Anderson, N.; Horton, J.D. Inhibition of PCSK9 does not improve lipopolysaccharide-induced mortality in mice. J. Lipid Res. 2017, 58, 1661–1669. [Google Scholar] [CrossRef] [PubMed]
- Dwivedi, D.J.; Grin, P.M.; Khan, M.; Prat, A.; Zhou, J.; Fox-Robichaud, A.E.; Seidah, N.G.; Liaw, P.C. Differential Expression of PCSK9 Modulates Infection, Inflammation, and Coagulation in a Murine Model of Sepsis. Shock 2016, 46, 672–680. [Google Scholar] [CrossRef]
- Khademi, F.; Momtazi-borojeni, A.A.; Reiner, Ž.; Banach, M.; Al-Rasadi, K.A.; Sahebkar, A. PCSK9 and infection: A potentially useful or dangerous association? J. Cell. Physiol. 2018, 233, 2920–2927. [Google Scholar] [CrossRef]
- Mitchell, K.A.; Moore, J.X.; Rosenson, R.S.; Irvin, R.; Guirgis, F.W.; Shapiro, N.; Safford, M.; Wang, H.E. PCSK9 loss-of-function variants and risk of infection and sepsis in the Reasons for Geographic and Racial Differences in Stroke (REGARDS) cohort. PLoS ONE 2019, 14, e0210808. [Google Scholar] [CrossRef]
- Rannikko, J.; Jacome Sanz, D.; Ortutay, Z.; Seiskari, T.; Aittoniemi, J.; Huttunen, R.; Syrjänen, J.; Pesu, M. Reduced plasma PCSK 9 response in patients with bacteraemia is associated with mortality. J. Innate Immun. 2019. [Google Scholar] [CrossRef]
- Leung, A.K.K.; Genga, K.R.; Topchiy, E.; Cirstea, M.; Shimada, T.; Fjell, C.; Russell, J.A.; Boyd, J.H.; Walley, K.R. Reduced Proprotein convertase subtilisin/kexin 9 (PCSK9) function increases lipoteichoic acid clearance and improves outcomes in Gram positive septic shock patients. Sci. Rep. 2019, 9, 10588. [Google Scholar] [CrossRef]
| All Samples (n = 100) | ||
|---|---|---|
| Age (y) | 67.12 ± 1.34 | |
| Gender (%) | Female | 54 |
| Male | 46 | |
| Weight (kg) | 78.82 ± 1.12 | |
| Height (cm) | 162.32 ± 1.5 | |
| Smoker (%) | 39 | |
| Diabetes (%) | 22 | |
| FBS (mg/dL) | 171.46 ± 6.43 | |
| CRP (mg/dL) | 11.84 ± 0.41 | |
| Lac (mmol/L) | 2 (2–4) | |
| BUN (mg/dL) | 21 (17–31.75) | |
| ALB (g/dL) | 2.9 ± 0.1 | |
| TG (mg/dL) | 291.5 ± 10.14 | |
| TC (mg/dL) | 179.1 ± 5.8 | |
| HDL-C (mg/dL) | 34 (32–45) | |
| LDL-C (mg/dL) | 165.5 (136–192) | |
| PCT (ng/mL) | 1.6 (0.98–2.3) | |
| PT (s) | 16 (13–19) | |
| PTT (s) | 27.3 ± 0.9 | |
| INR (s) | 1.4 (1.2–2) | |
| SGPT (U/L) | 23 (21–32) | |
| SGOT (U/L) | 32 (23–43) | |
| ESR (mm/hr) | 17 (15–24) | |
| K (mEq/L) | 4.31 ± 0.1 | |
| Na (mEq/L) | 138.81 ± 0.82 | |
| PCSK9 (ng/mL) | 98.85 (89.15–123) | |
| APACHE II score | 22 (17–24) | |
| SOFA score | 8.35 ± 0.41 | |
| GCS score | 7 (6–10) | |
| All Samples (n = 100) | ||
|---|---|---|
| Gram-negative bacteria (%) | Klebsiella sp. | 35 |
| Pseudomonas aeruginosa | 23 | |
| Acinetobacter | 35 | |
| Pseudomonas sp. | 1 | |
| Escherichia coli | 3 | |
| Gram-positive bacteria (%) | Staphylococcus aureus | 1 |
| Enterococcus | 2 | |
| Type of antibiotic resistance (%) | Meropenem | 82 |
| Vancomycin | 2 | |
| Amikacin | 74 | |
| Ciprofloxacin | 83 | |
| Ceftriaxone | 86 | |
| Antibiotic resistance (%) | Sensitive | 9 |
| Resistance to one antibiotic | 3 | |
| Resistance to two antibiotics | 9 | |
| Resistance to three antibiotics | 10 | |
| Resistance to four antibiotics | 69 | |
| Outcome Measure | Model | Coefficient | 95% CI | SE | p−Value |
|---|---|---|---|---|---|
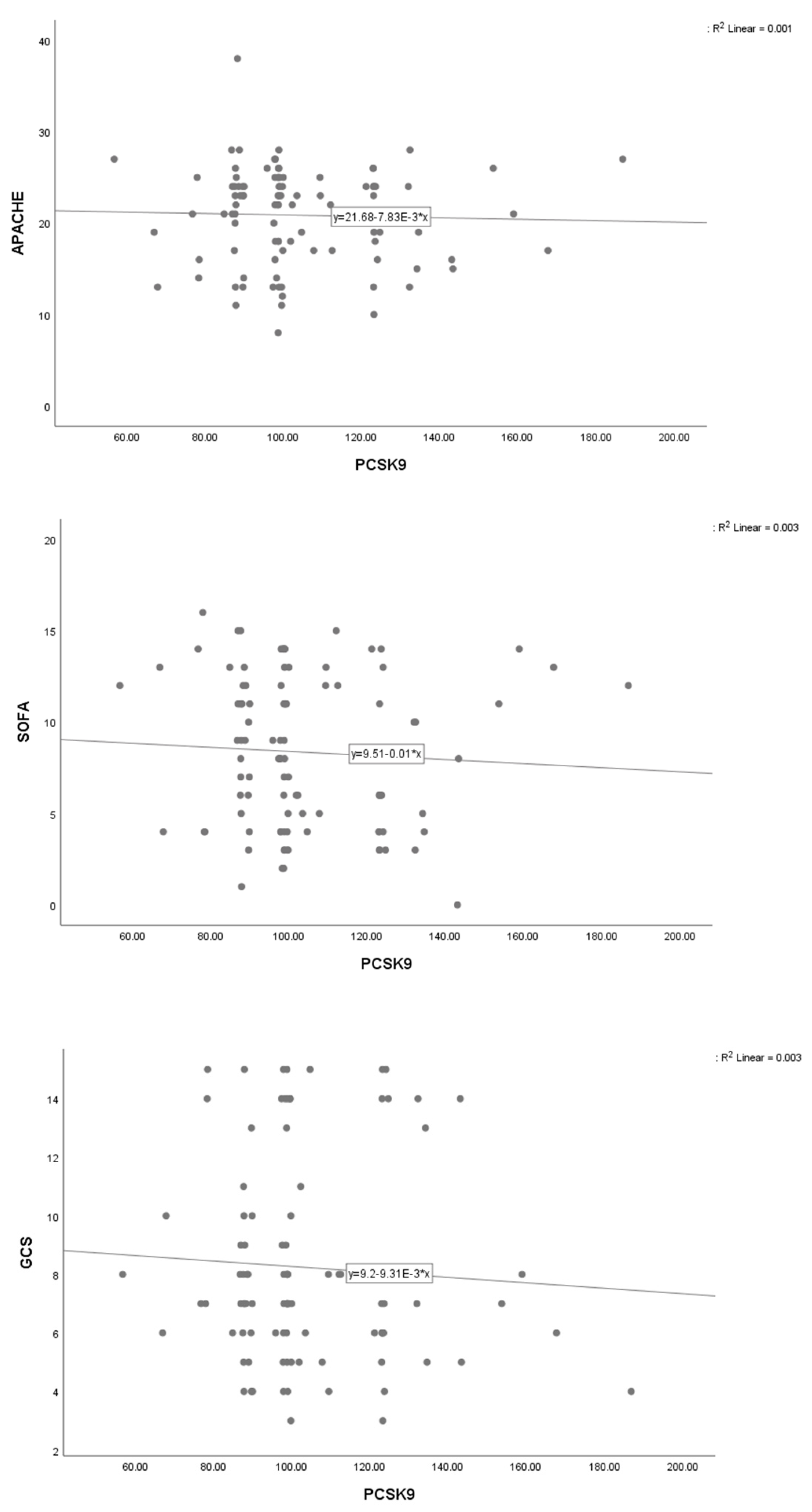
| APACHE II score | Model 1 | −0.04 | −0.12, 0.05 | 0.04 | 0.372 |
| Model 2 | −0.05 | −0.14, 0.04 | 0.04 | 0.261 | |
| Model 3 | −0.08 | −0.20, 0.03 | 0.06 | 0.150 | |
| SOFA score | Model 1 | −0.06 | −0.11, −0.01 | 0.02 | 0.012 |
| Model 2 | −0.07 | −0.12, −0.02 | 0.03 | 0.010 | |
| Model 3 | −0.03 | −0.10, 0.03 | 0.03 | 0.278 | |
| GCS score | Model 1 | 0.01 | −0.04, 0.06 | 0.03 | 0.681 |
| Model 2 | 0.02 | −0.04, 0.07 | 0.03 | 0.492 | |
| Model 3 | 0.01 | −0.05, 0.08 | 0.03 | 0.674 | |
| Antibiotic resistance | Model 1 | 1.03 | 1.00, 1.06 | 0.01 | 0.089 |
| Model 2 | 1.02 | 0.99, 1.06 | 0.02 | 0.162 | |
| Model 3 | 1.03 | 0.96, 1.12 | 0.04 | 0.377 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jamialahmadi, T.; Panahi, Y.; Safarpour, M.A.; Ganjali, S.; Chahabi, M.; Reiner, Z.; Solgi, S.; Vahedian-azimi, A.; Kianpour, P.; Banach, M.; et al. Association of Serum PCSK9 Levels with Antibiotic Resistance and Severity of Disease in Patients with Bacterial Infections Admitted to Intensive Care Units. J. Clin. Med. 2019, 8, 1742. https://doi.org/10.3390/jcm8101742
Jamialahmadi T, Panahi Y, Safarpour MA, Ganjali S, Chahabi M, Reiner Z, Solgi S, Vahedian-azimi A, Kianpour P, Banach M, et al. Association of Serum PCSK9 Levels with Antibiotic Resistance and Severity of Disease in Patients with Bacterial Infections Admitted to Intensive Care Units. Journal of Clinical Medicine. 2019; 8(10):1742. https://doi.org/10.3390/jcm8101742
Chicago/Turabian StyleJamialahmadi, Tannaz, Yunes Panahi, Mohamamd Amin Safarpour, Shiva Ganjali, Mahdi Chahabi, Zeljko Reiner, Saeed Solgi, Amir Vahedian-azimi, Parisa Kianpour, Maciej Banach, and et al. 2019. "Association of Serum PCSK9 Levels with Antibiotic Resistance and Severity of Disease in Patients with Bacterial Infections Admitted to Intensive Care Units" Journal of Clinical Medicine 8, no. 10: 1742. https://doi.org/10.3390/jcm8101742
APA StyleJamialahmadi, T., Panahi, Y., Safarpour, M. A., Ganjali, S., Chahabi, M., Reiner, Z., Solgi, S., Vahedian-azimi, A., Kianpour, P., Banach, M., & Sahebkar, A. (2019). Association of Serum PCSK9 Levels with Antibiotic Resistance and Severity of Disease in Patients with Bacterial Infections Admitted to Intensive Care Units. Journal of Clinical Medicine, 8(10), 1742. https://doi.org/10.3390/jcm8101742







