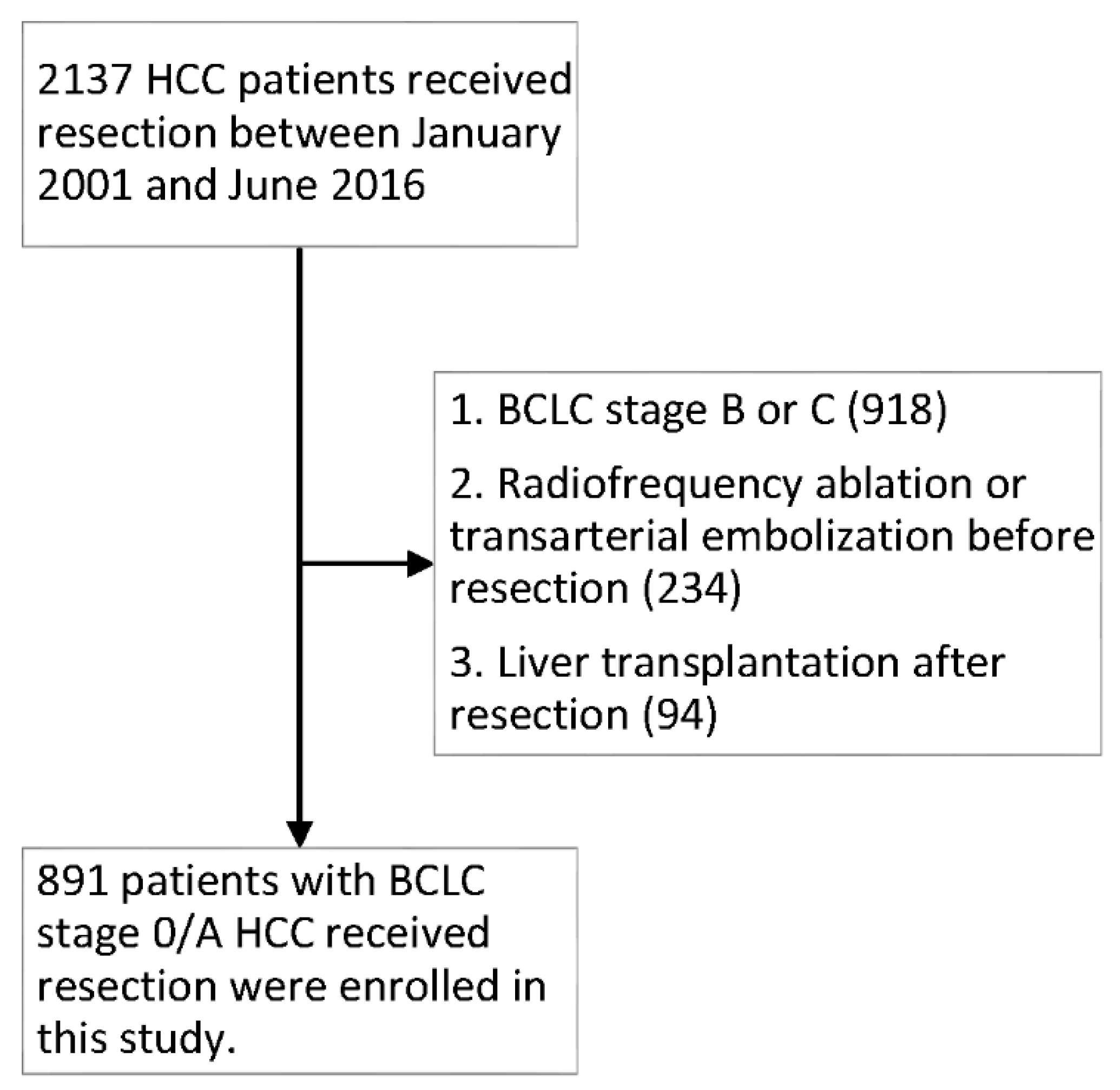
1. Introduction
Liver cancer is the sixth most commonly diagnosed cancer and the fourth leading cause of cancer death worldwide as of 2018. Hepatocellular carcinoma (HCC) comprises 75–85% of primary liver cancer cases [
1]. The management of HCC depends on the tumor stage at diagnosis and liver function. Current management strategies involve resection, liver transplantation (LT), radiofrequency ablation (RFA), transarterial embolization (TAE)/transarterial chemoembolization (TACE), radiation therapy, or systemic therapy [
2,
3]. Patients are considered to have resectable tumors if they have one to three unilobar lesions with an upper limit of 5 cm for a single lesion and 3 cm for more than one lesion; no extrahepatic metastasis or macrovascular invasion; and little or no portal hypertension [
2].
Hepatectomy is a potentially curative treatment for HCC, but the long-term prognosis remains unsatisfactory due to the high incidence of recurrence (50–60%) [
4,
5,
6]. Prognostic factors for recurrence and survival after resection include tumor size, α-fetoprotein level, tumor differentiation, microvascular invasion, cirrhosis, surgical margin, and metabolic syndrome [
6,
7,
8]. Recently, the associations between inflammatory markers and the prognosis of HCC have been actively explored, and systemic inflammation has been thought to be related to poor outcomes. The reason is that inflammatory responses play critical roles in tumorigenesis, including initiation, promotion, angiogenesis, invasion, and metastasis [
9], and an inflammatory microenvironment is an important condition for tumors [
10].
Systemic inflammatory responses have been investigated using markers such as the neutrophil-to-lymphocyte ratio (NLR) [
11,
12,
13,
14], platelet to lymphocyte ratio (PLR) [
15,
16,
17], albumin and lymphocyte counts used in the prognostic nutritional index (PNI) [
18,
19,
20], and neutrophil, lymphocyte, and platelet counts used in the systemic immune-inflammation index (SII) [
21,
22]. To date, most studies have evaluated only one of the inflammatory parameters. Therefore, a comprehensive evaluation of the prognostic values of the inflammatory markers would be valuable for patients with HCC. The aim of this study is to evaluate the effects of inflammatory markers to predict recurrence and survival in HCC patients who have Barcelona Clinic Liver Cancer (BCLC) grades 0–A and are receiving hepatectomy.
4. Discussion
Surgical resection is the mainstay of curative treatment for very-early and early-stage HCC with preserved liver function. Tumor recurrence is still the main concern of resection, and the five-year cumulative HCC recurrent rates are more than 50% [
2,
28]. Many studies have shown the relationships between inflammatory response and the development of tumors, and several scoring systems have been developed to predict the prognosis in cancer patients. NLR, PLR, PNI, and SII have been reported to be useful in predicting OS and recurrence in HCC patients.
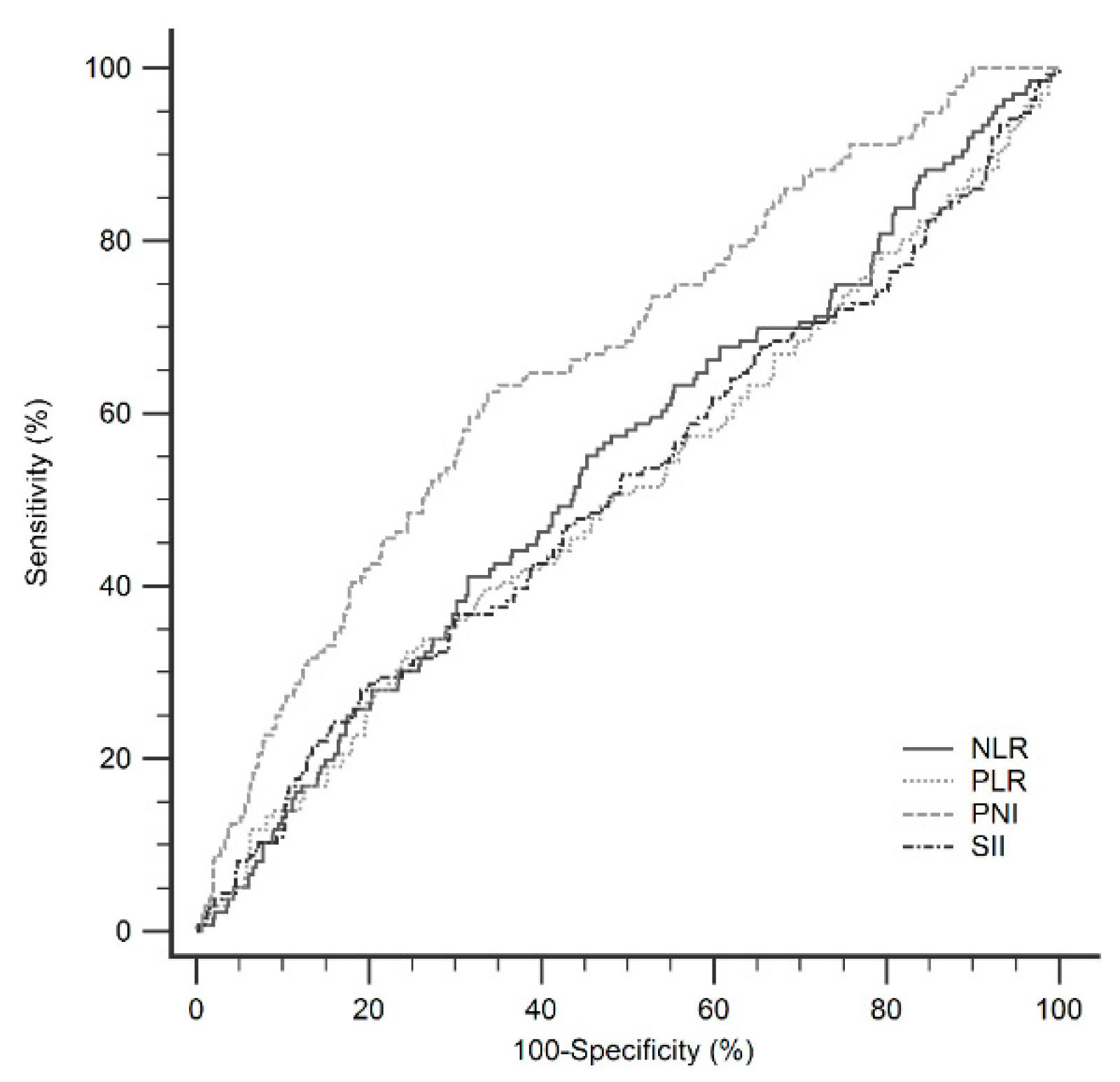
We investigated the clinical and prognostic value of these widely used inflammatory markers in early-stage HCC patients receiving operation and compared their predictive accuracy. Our retrospective cohort of 891 patients revealed that PNI and SII are independent predictors of OS and tumor recurrence. Furthermore, the predictive power of the PNI score outweighs that of other inflammatory markers. PNI and SII are derived from routinely available blood tests, making them easy to apply in routine clinical practice to predict the prognosis in HCC patients.
Studies have shown that the systemic inflammatory response as measured by the NLR, PLR, and SII are good predictors of tumor outcomes. The exact mechanism remains unclear, but several hypotheses have been proposed. Basic studies have revealed that neutrophils can induce tumor proliferation and angiogenesis, as well as enhance the migration and metastasis of cancer cells. In addition, HCC cells induce neutrophils to release hepatocyte growth factor, which makes cancer cells become more aggressive [
29]. Platelets can be activated by immune cells and cancer cells and play an important role in tumor metastasis, tumor growth, and angiogenesis. This occurs by protecting cancer cells from immune surveillance, facilitating cancer cell arrest within the vasculature and subsequent tissue invasion, inducing endothelial cell proliferation and new blood vessel formation, and interacting with cancer cells and stroma in the tumor microenvironment [
30]. However, lymphocytes, including T cells, B cells, and natural killer cells, have antitumor effects either directly or upon the activation of other lymphocytes [
31].
Higher levels of NLR and PLR are associated with poor outcomes in many types of cancer. In a meta-analysis of patients with HCC, Zeng et al. examined 24 studies and concluded that a high NLR predicted a poor OS (HR: 1.54, 95% CI 1.34–1.76,
p < 0.001) and poor RFS (HR: 1.45, 95% CI 1.16–1.82,
p = 0.001), while a high PLR predicted poor OS (HR: 1.63, 95% CI 1.34–1.98,
p < 0.001) and poor RFS (HR: 1.52, 95% CI 1.21–1.91,
p < 0.001) [
32]. In the present study, a high NLR (>1.8) before surgical resection predicted poor OS in HCC patients, but NLR was not an independent factor in predicting OS and RFS in the multivariate analyses. PLR had no significant association with OS or RFS.
Higher SII has been reported to be a poor prognostic factor in multiple cancers. A recent meta-analysis included 22 studies with 7657 patients and showed that a higher level of SII was correlated with poor OS (HR: 1.69, 95% CI 1.42–2.01,
p < 0.001) and poor RFS (HR = 1.66,
p = 0.025) in patients with cancers. Furthermore, a subgroup analysis revealed that higher SII than a cutoff value could predict poor OS in HCC (
p < 0.001) [
33]. However, our study revealed the opposite result: low SII predicted a poor prognosis of OS (HR: 1.674, 95% CI 1.097–2.552,
p = 0.017) and RFS (HR: 1.616, 95% CI: 1.231–2.123,
p = 0.001). Lower SII means lower neutrophil levels, lower platelet levels, and higher lymphocyte levels, as shown in
Table 7. Thrombocytopenia was an independent predictor for survival in patients with compensated cirrhosis and HCC treated with hepatectomy [
34]. Through a meta-analysis, Zhang et al. showed that thrombocytopenia in HCC patients was associated with poor OS (HR: 1.47, 95% CI 1.21–1.78) and poor RFS (HR: 1.41, 95% CI: 1.22–1.62) in HCC patients after hepatic resection [
35]. Platelet count has been used to assess the severity of chronic liver disease and features of portal hypertension [
36]. In the present study, low SII was associated with high APRI and high INR, which are related to significant fibrosis and poor liver function. In the subgroup analysis stratified by platelet count, low SII in the thrombocytopenia group revealed significantly poor OS (
p = 0.019) and RFS (
p = 0.002) compared to those with high SII, but there was no statistical significance in the non-thrombocytopenia group. Therefore, low SII is associated with poor outcome due to advanced liver disease, especially in those with thrombocytopenia.
The PNI was initially designed to assess the immunological and nutritional condition of patients undergoing surgery for digestive diseases [
37]. It was first used to predict HCC outcome as an inflammatory marker by Pinato et al. in 2012 [
18]. Lymphopenia is an unfavorable prognostic factor for OS in several cancers [
38], which may result from an increased rate of turnover of lymphocytes induced by tumor cells [
39] or an altered homeostasis of lymphocytes in patients with tumors [
40]. Albumin synthesis is reduced by the systemic inflammatory response to a tumor [
41], although impaired hepatic synthetic function in advanced liver disease also needs to be considered as an additional cause for reduced serum albumin. Chan et al. reported that the PNI was a significant predictor for OS and RFS in patients with very-early/early-stage HCC receiving curative surgery [
20]. Our data are consistent with previous studies suggesting that low PNI predicts poor OS and RFS in HCC patients receiving hepatectomy. We subsequently compared the predictive power of the PNI with other inflammatory markers, including NLR, PLR, and SII, and the PNI produced superior results.
Nevertheless, there are some limitations to our study. First, it is a retrospective study with patients from a single institution, which could lead to biases. Second, more than half of the patients in the study had HBV infection as the etiology of HCC, whereas chronic HCV infection is the major cause for the development of HCC in Western countries and Japan [
42]. Moreover, laboratory values were only collected before surgery, and a serial follow-up of inflammatory markers after surgery may provide more insights in the prognostic values.
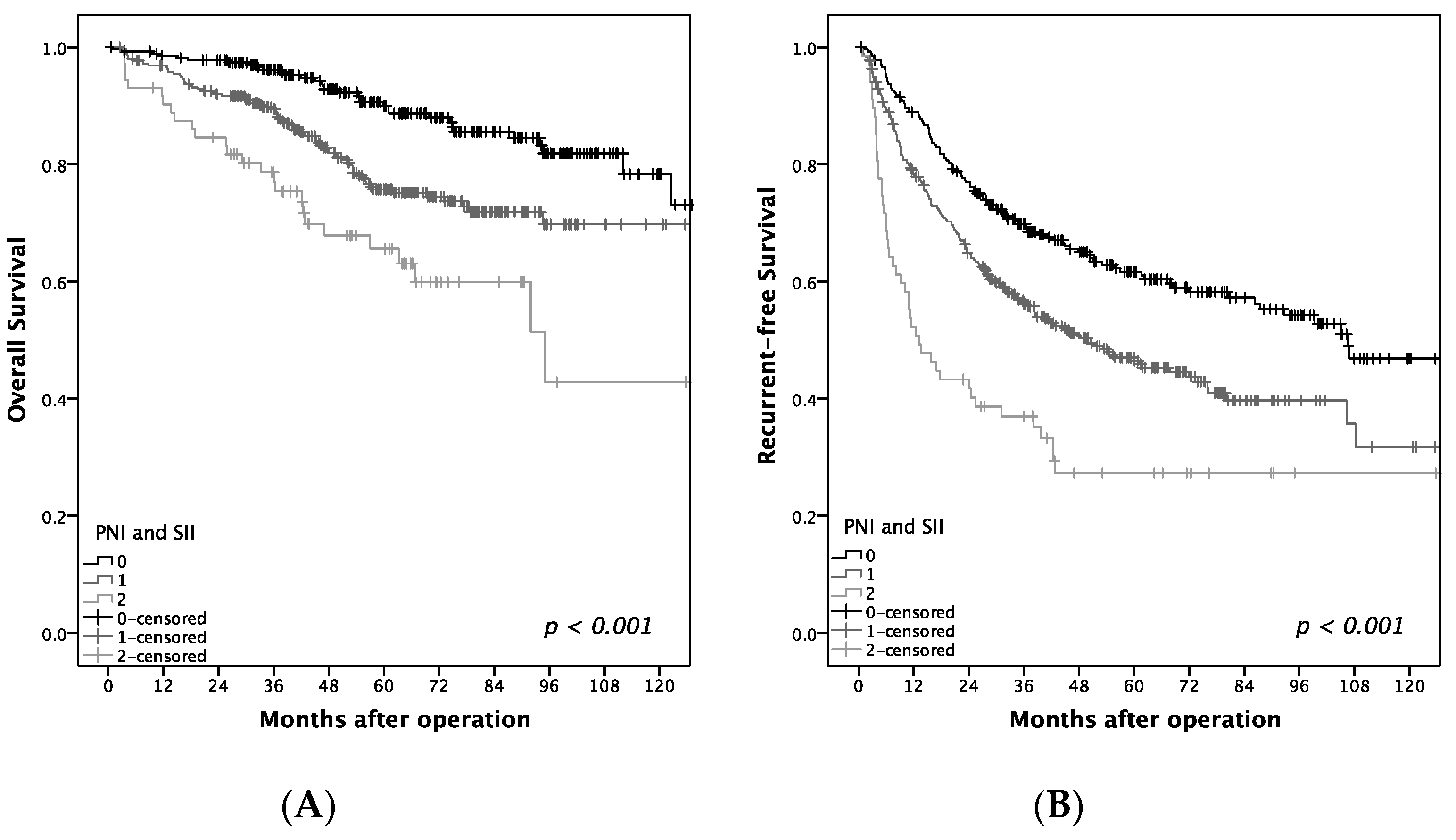
In conclusion, among the evaluated preoperative serum inflammatory markers, low PNI and low SII were independently associated with unfavorable outcomes of OS and RFS in patients with BCLC 0-A hepatocellular carcinoma after hepatectomy. Those observed with low PNI or low SII, especially PNI ≤ 45 and SII ≤ 160 with a high one-year recurrence rate (50%), need a close follow-up, and further adjuvant therapies, such as target or immune-based therapy, might be required. However, the side effects of drugs should be watched for, and additional well-designed studies are needed to confirm this concept. Further evaluation of these markers should be performed on a larger scale and compared between different patient populations.