Non-Vitamin K Antagonist Oral Anticoagulants in Patients with Atrial Fibrillation and Valvular Heart Disease
Abstract
1. Introduction
2. Methods
2.1. Data Source
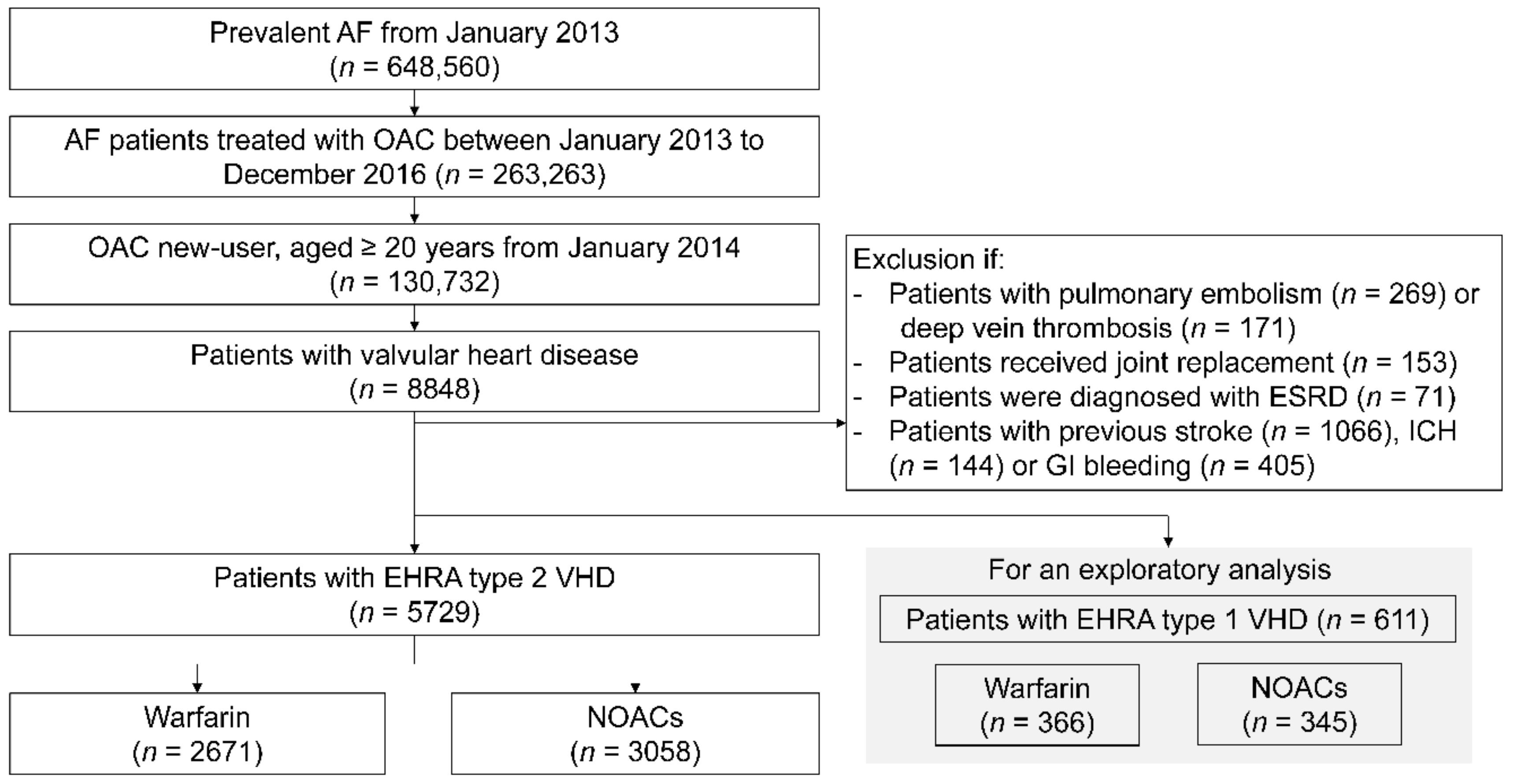
2.2. Study Cohort
2.3. Defining the Comorbidities and Outcomes
2.4. Subgroup Analyses
2.5. Statistical Analysis
2.6. Sensitivity Analyses
3. Results
3.1. Baseline Characteristics of the Study Population
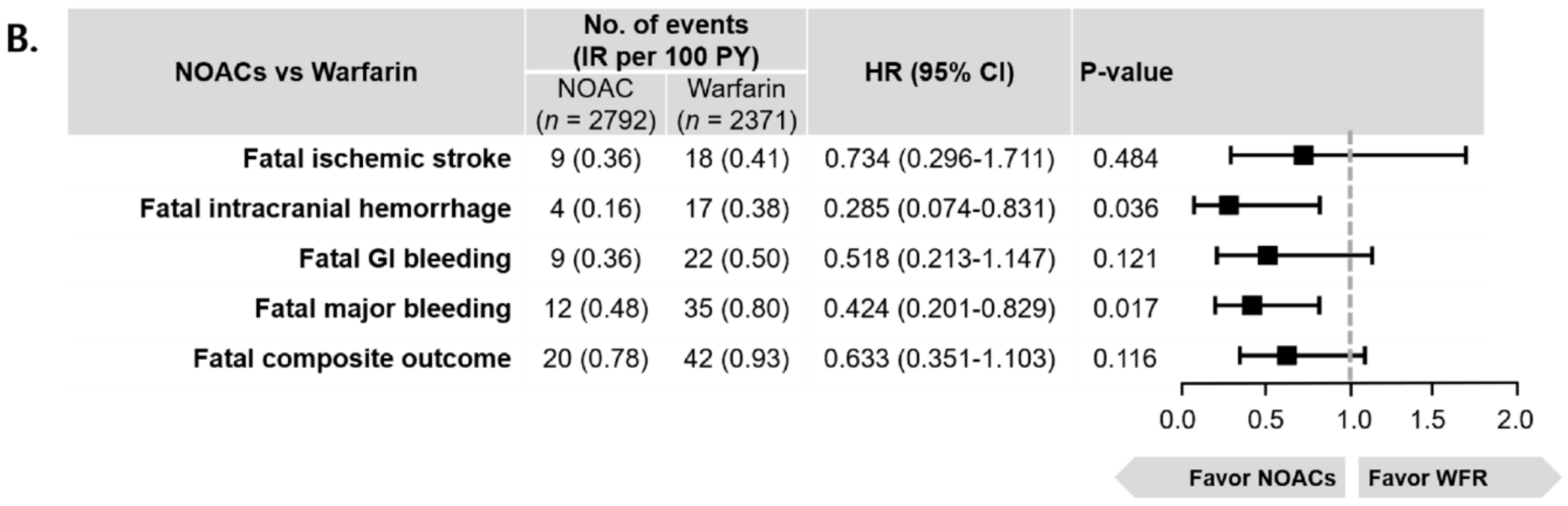
3.2. Clinical Outcomes in Patients with AF and EHRA Type 2 VHDs
3.3. Sensitivity Analysis
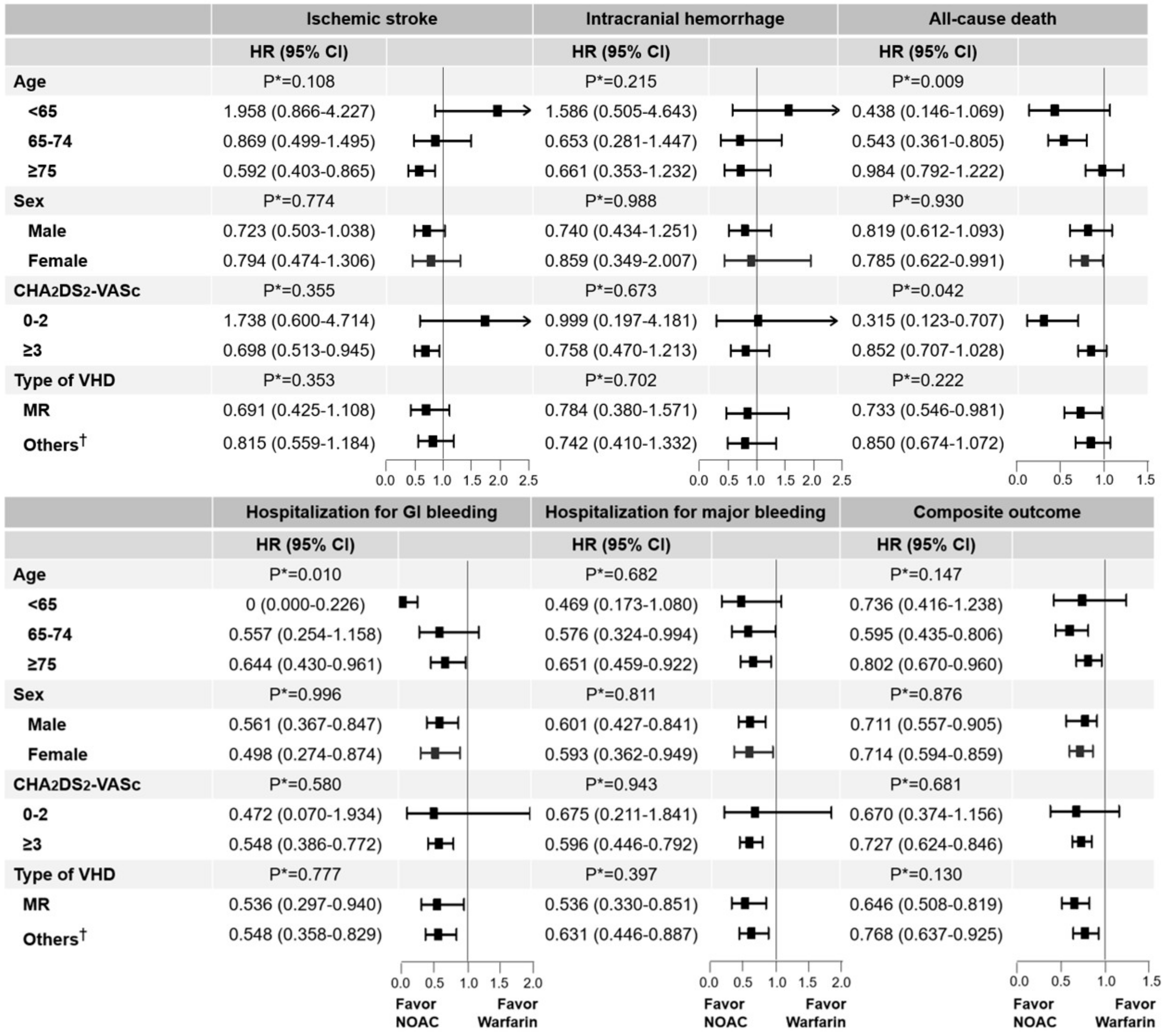
3.4. Subgroup Analyses Stratified by Age, Sex, CHA2DS2-VASc Score, and VHD Types
3.5. Age (<64, 65–74, and ≥75 Years) and Sex
3.6. CHA2DS2-VASc Score (0–2 and ≥3)
3.7. Type of VHD (Mitral Regurgitation and Other VHDs)
3.8. Comparative Effectiveness and Safety of NOAC versus Warfarin in Patients with EHRA Type 1 VHDs: An Exploratory Analysis
4. Discussion
4.1. NOACs in Patients with Non-Valvular AF and EHRA Type 2 VHDs
4.2. NOACs in AF Patients with EHRA Type 1 VHDs (Valvular AF)
4.3. The Clinical Implication with Subgroup Analyses
4.4. Study Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- De Caterina, R.; Camm, A.J. What is ‘valvular’ atrial fibrillation? A reappraisal. Eur. Heart J. 2014, 35, 3328–3335. [Google Scholar] [CrossRef] [PubMed]
- Connolly, S.J.; Ezekowitz, M.D.; Yusuf, S.; Eikelboom, J.; Oldgren, J.; Parekh, A.; Pogue, J.; Reilly, P.A.; Themeles, E.; Varrone, J.; et al. Dabigatran versus warfarin in patients with atrial fibrillation. N. Engl. J. Med. 2009, 361, 1139–1151. [Google Scholar] [CrossRef] [PubMed]
- Granger, C.B.; Alexander, J.H.; McMurray, J.J.; Lopes, R.D.; Hylek, E.M.; Hanna, M.; Al-Khalidi, H.R.; Ansell, J.; Atar, D.; Avezum, A.; et al. Apixaban versus warfarin in patients with atrial fibrillation. N. Engl. J. Med. 2011, 365, 981–992. [Google Scholar] [CrossRef] [PubMed]
- Patel, M.R.; Mahaffey, K.W.; Garg, J.; Pan, G.; Singer, D.E.; Hacke, W.; Breithardt, G.; Halperin, J.L.; Hankey, G.J.; Piccini, J.P.; et al. Rivaroxaban versus warfarin in nonvalvular atrial fibrillation. N Engl. J. Med. 2011, 365, 883–891. [Google Scholar] [CrossRef] [PubMed]
- Giugliano, R.P.; Ruff, C.T.; Braunwald, E.; Murphy, S.A.; Wiviott, S.D.; Halperin, J.L.; Waldo, A.L.; Ezekowitz, M.D.; Weitz, J.I.; Špinar, J.; et al. Edoxaban versus warfarin in patients with atrial fibrillation. N Engl. J. Med. 2013, 369, 2093–2104. [Google Scholar] [CrossRef] [PubMed]
- Lip, G.Y.H.; Collet, J.P.; Caterina, R.; Fauchier, L.; Lane, D.A.; Larsen, T.B.; Marin, F.; Morais, J.; Narasimhan, C.; Olshansky, B.; et al. Antithrombotic therapy in atrial fibrillation associated with valvular heart disease: A joint consensus document from the European Heart Rhythm Association (EHRA) and European Society of Cardiology Working Group on Thrombosis, endorsed by the ESC Working Group on Valvular Heart Disease, Cardiac Arrhythmia Society of Southern Africa (CASSA), Heart Rhythm Society (HRS), Asia Pacific Heart Rhythm Society (APHRS), South African Heart (SA Heart) Association and Sociedad Latinoamericana de Estimulacion Cardiaca y Electrofisiologia (SOLEACE). Europace 2017, 19, 1757–1758. [Google Scholar] [PubMed]
- Ruff, C.T.; Giugliano, R.P.; Braunwald, E.; Hoffman, E.B.; Deenadayalu, N.; Ezekowitz, M.D.; Camm, A.J.; Weitz, J.I.; Lewis, B.S.; Parkhomenko, A.; et al. Comparison of the efficacy and safety of new oral anticoagulants with warfarin in patients with atrial fibrillation: A meta-analysis of randomised trials. Lancet 2014, 383, 955–962. [Google Scholar] [CrossRef]
- Huisman, M.V.; Rothman, K.J.; Paquette, M.; Teutsch, C.; Diener, H.C.; Dubner, S.J.; Halperin, J.L.; Ma, C.S.; Zint, K.; Elsaesser, A.; et al. The Changing Landscape for Stroke Prevention in AF: Findings From the GLORIA-AF Registry Phase 2. J. Am. Coll. Cardiol. 2017, 69, 777–785. [Google Scholar] [CrossRef]
- Lee, S.R.; Choi, E.K.; Han, K.D.; Cha, M.J.; Oh, S.; Lip, G.Y.H. Temporal trends of antithrombotic therapy for stroke prevention in Korean patients with non-valvular atrial fibrillation in the era of non-vitamin K antagonist oral anticoagulants: A nationwide population-based study. PLoS ONE 2017, 12, e0189495. [Google Scholar] [CrossRef]
- Renda, G.; Ricci, F.; Giugliano, R.P.; De Caterina, R. Non-Vitamin K Antagonist Oral Anticoagulants in Patients With Atrial Fibrillation and Valvular Heart Disease. J. Am. Coll. Cardiol. 2017, 69, 1363–1371. [Google Scholar] [CrossRef]
- Noseworthy, P.A.; Yao, X.; Shah, N.D.; Gersh, B.J. Comparative effectiveness and safety of non-vitamin K antagonist oral anticoagulants versus warfarin in patients with atrial fibrillation and valvular heart disease. Int. J. Cardiol. 2016, 209, 181–183. [Google Scholar] [CrossRef] [PubMed]
- Briasoulis, A.; Inampudi, C.; Akintoye, E.; Alvarez, P.; Panaich, S.; Vaughan-Sarrazin, M. Safety and Efficacy of Novel Oral Anticoagulants Versus Warfarin in Medicare Beneficiaries With Atrial Fibrillation and Valvular Heart Disease. J. Am. Hear. Assoc. 2018, 7, e008773. [Google Scholar] [CrossRef] [PubMed]
- Lip, G.Y.; Wang, K.-L.; Chiang, C.-E. Non-vitamin K antagonist oral anticoagulants (NOACs) for stroke prevention in Asian patients with atrial fibrillation: Time for a reappraisal. Int. J. Cardiol. 2015, 180, 246–254. [Google Scholar] [CrossRef] [PubMed]
- Cha, M.-J.; Choi, E.-K.; Han, K.-D.; Lee, S.-R.; Lim, W.-H.; Oh, S.; Lip, G.Y. Effectiveness and Safety of Non-Vitamin K Antagonist Oral Anticoagulants in Asian Patients With Atrial Fibrillation. Stroke 2017, 48, 3040–3048. [Google Scholar] [CrossRef] [PubMed]
- Chan, Y.-H.; Kuo, C.-T.; Yeh, Y.-H.; Chang, S.-H.; Wu, L.-S.; Lee, H.-F.; Tu, H.-T.; See, L.-C. Thromboembolic, Bleeding, and Mortality Risks of Rivaroxaban and Dabigatran in Asians With Nonvalvular Atrial Fibrillation. J. Am. Coll. Cardiol. 2016, 68, 1389–1401. [Google Scholar] [CrossRef]
- Song, S.O.; Jung, C.H.; Song, Y.D.; Park, C.-Y.; Kwon, H.-S.; Cha, B.S.; Park, J.-Y.; Lee, K.-U.; Ko, K.S.; Lee, B.-W. Background and Data Configuration Process of a Nationwide Population-Based Study Using the Korean National Health Insurance System. Diabetes Metab. J. 2014, 38, 395–403. [Google Scholar] [CrossRef] [PubMed]
- Seong, S.C.; Kim, Y.-Y.; Khang, Y.-H.; Park, J.H.; Kang, H.-J.; Lee, H.; Do, C.-H.; Song, J.-S.; Bang, J.H.; Ha, S.; et al. Data Resource Profile: The National Health Information Database of the National Health Insurance Service in South Korea. Int. J. Epidemiol. 2017, 46, 799–800. [Google Scholar]
- Lip, G.Y.H.; Nieuwlaat, R.; Pisters, R.; Lane, D.A.; Crijns, H.J. Refining clinical risk stratification for predicting stroke and thromboembolism in atrial fibrillation using a novel risk factor-based approach: The euro heart survey on atrial fibrillation. Chest 2010, 137, 263–272. [Google Scholar] [CrossRef]
- Lee, S.R.; Choi, E.K.; Han, K.D.; Jung, J.H.; Oh, S.; Lip, G.Y.H. Edoxaban in Asian Patients With Atrial Fibrillation: Effectiveness and Safety. J. Am. Coll. Cardiol. 2018, 72, 838–853. [Google Scholar] [CrossRef]
- Heinze, G.; Jüni, P. An overview of the objectives of and the approaches to propensity score analyses. Eur. Hear. J. 2011, 32, 1704–1708. [Google Scholar] [CrossRef]
- Austin, P.C.; Stuart, E.A. Moving towards best practice when using inverse probability of treatment weighting (IPTW) using the propensity score to estimate causal treatment effects in observational studies. Stat. Med. 2015, 34, 3661–3679. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.R.; Choi, E.K.; Park, C.S.; Han, K.D.; Jung, J.H.; Oh, S.; Lip, G.Y.H. Direct oral anticoagulants in patients with nonvalvular atrial fibrillation and low body weight. J. Am. Coll. Cardiol. 2019, 73, 919–931. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.R.; Choi, E.K.; Han, K.; Cha, M.J.; Oh, S. Prevalence of Non-valvular Atrial Fibrillation Based on Geographical Distribution and Socioeconomic Status in the Entire Korean Population. Korean Circ. J. 2018, 48, 622–634. [Google Scholar] [CrossRef] [PubMed]
- Bisson, A.; Bodin, A.; Clementy, N.; Bernard, A.; Babuty, D.; Lip, G.Y.; Fauchier, L. Stroke, thromboembolism and bleeding in patients with atrial fibrillation according to the EHRA valvular heart disease classification. Int. J. Cardiol. 2018, 260, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Lip, G.Y.H.; Jensen, M.; Melgaard, L.; Skjøth, F.; Nielsen, P.B.; Larsen, T.B. Stroke and bleeding risk scores in patients with atrial fibrillation and valvular heart disease: Evaluating ’valvular heart disease’ in a nationwide cohort study. Europace 2019, 21, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Kamath, S.; Lip, G.Y. Mitral regurgitation and atrial fibrillation: Milder the disease, higher the risk? Int. J. Cardiol. 2000, 72, 235–237. [Google Scholar] [CrossRef]
- Nakagami, H.; Yamamoto, K.; Ikeda, U.; Mitsuhashi, T.; Goto, T.; Shimada, K. Mitral regurgitation reduces the risk of stroke in patients with nonrheumatic atrial fibrillation. Am. Hear. J. 1998, 136, 528–532. [Google Scholar] [CrossRef]
- Sucu, M.; Davutoglu, V.; Sari, I.; Ozer, O.; Aksoy, M. Relationship between platelet indices and aortic valve sclerosis. Clin. Appl. Thromb. Hemost. 2010, 16, 563–567. [Google Scholar] [CrossRef]
- Dimitrow, P.P.; Hlawaty, M.; Undas, A.; Śnieżek-Maciejewska, M.; Sobień, B.; Stepien, E.; Tracz, W. Effect of aortic valve stenosis on haemostasis is independent from vascular atherosclerotic burden. Atherosclerosis 2009, 204, e103–e108. [Google Scholar] [CrossRef]
- Susen, S.; Six, I.; Decoene, C.; Vincentelli, A.; Le Tourneau, T.; Fabre, O.; Juthier, F.; Bauters, A.; Goudemand, J.; Prat, A.; et al. Acquired von Willebrand Syndrome in Aortic Stenosis. N. Engl. J. Med. 2003, 349, 343–349. [Google Scholar]
- Breithardt, G.; Baumgartner, H.; Berkowitz, S.D.; Hellkamp, A.S.; Piccini, J.P.; Lokhnygina, Y.; Halperin, J.L.; Singer, D.E.; Hankey, G.J.; Hacke, W.; et al. Native valve disease in patients with non-valvular atrial fibrillation on warfarin or rivaroxaban. Heart 2016, 102, 1036–1043. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.L.; Lip, G.Y.H.; Lin, S.J.; Chiang, C.E. Non-Vitamin K Antagonist Oral Anticoagulants for Stroke Prevention in Asian Patients With Nonvalvular Atrial Fibrillation: Meta-Analysis. Stroke 2015, 46, 2555–2561. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.-G.; Lee, S.-R.; Choi, E.-K.; Lip, G.Y. Stroke Prevention in Atrial Fibrillation: Focus on Asian Patients. Korean Circ. J. 2018, 48, 665–684. [Google Scholar] [CrossRef]
- Kirchhof, P.; Benussi, S.; Kotecha, D.; Ahlsson, A.; Atar, D.; Casadei, B.; Castellà, M.; Diener, H.-C.; Heidbuchel, H.; Hendriks, J.; et al. 2016 ESC Guidelines for the management of atrial fibrillation developed in collaboration with EACTS. Eur. J. Cardiothorac. Surg. 2016, 74, 1359–1469. [Google Scholar]
- Joung, B.; Lee, J.M.; Lee, K.H.; Kim, T.H.; Choi, E.K.; Lim, W.H.; Kang, K.W.; Shim, J.; Lim, H.E.; Park, J.; et al. 2018 Korean Guideline of Atrial Fibrillation Management. Korean Circ. J. 2018, 48, 1033–1080. [Google Scholar] [CrossRef] [PubMed]
- Singer, D.E.; Hellkamp, A.S.; Piccini, J.P.; Mahaffey, K.W.; Lokhnygina, Y.; Pan, G.; Halperin, J.L.; Becker, R.C.; Breithardt, G.; Hankey, G.J.; et al. Impact of Global Geographic Region on Time in Therapeutic Range on Warfarin Anticoagulant Therapy: Data From the ROCKET AF Clinical Trial. J. Am. Heart Assoc. 2013, 2, e000067. [Google Scholar] [CrossRef] [PubMed]
- Oh, S.; Goto, S.; Accetta, G.; Angchaisuksiri, P.; Camm, A.J.; Cools, F.; Haas, S.; Kayani, G.; Koretsune, Y.; Lim, T.W.; et al. Vitamin K antagonist control in patients with atrial fibrillation in Asia compared with other regions of the world: Real-world data from the GARFIELD-AF registry. Int. J. Cardiol. 2016, 223, 543–547. [Google Scholar] [CrossRef]



| Propensity Score Weighting | ||||||
|---|---|---|---|---|---|---|
| Before | After (with 5% Trimming) | |||||
| Warfarin (n = 2671) | NOACs (n = 3058) | ASD | Warfarin (n = 2371) | NOACs (n = 2792) | ASD | |
| Age, years | 66.5 ± 13.2 | 73.5 ± 9.2 | 0.620 | 71.2 ± 9.9 | 71.2 ± 8.4 | 0.002 |
| <65 | 39.4 | 14.9 | 24.5 | 21.3 | ||
| 65–74 | 30.4 | 34.1 | 33.6 | 38.2 | ||
| ≥75 | 30.2 | 50.9 | 41.8 | 40.5 | ||
| Men | 51.7 | 61.6 | 0.200 | 57.3 | 56.9 | 0.006 |
| CHA2DS2-VASc score | 3.5 ± 1.9 | 4.1 ± 1.6 | 0.340 | 3.95 ± 1.85 | 3.94 ± 1.63 | 0.003 |
| 0–2 | 31.5 | 16.1 | 22.8 | 19.8 | ||
| ≥3 | 68.5 | 83.9 | 77.2 | 80.2 | ||
| Hypertension | 73.9 | 76.4 | 0.057 | 76.7 | 76.5 | 0.005 |
| Diabetes mellitus | 16.5 | 17.9 | 0.036 | 18.4 | 18.5 | 0.000 |
| Dyslipidemia | 36.4 | 40.1 | 0.076 | 40.3 | 39.5 | 0.015 |
| Heart failure | 52.5 | 49.5 | 0.059 | 51.6 | 51.3 | 0.005 |
| Prior MI | 4.3 | 3.5 | 0.043 | 4.2 | 4.2 | 0.002 |
| PAD | 15.5 | 18.5 | 0.081 | 18.3 | 18.0 | 0.006 |
| COPD | 24.6 | 25.2 | 0.012 | 26.2 | 26.2 | 0.000 |
| NOAC dose † | ||||||
| Regular dose | N.A | 36.6 | N.A | N.A | 41.1 | N.A |
| Reduced dose | N.A | 63.4 | N.A | N.A | 58.9 | N.A |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moon, I.; Lee, S.-R.; Choi, E.-K.; Lee, E.; Jung, J.-H.; Han, K.-D.; Cha, M.-J.; Oh, S.; Lip, G.Y.H. Non-Vitamin K Antagonist Oral Anticoagulants in Patients with Atrial Fibrillation and Valvular Heart Disease. J. Clin. Med. 2019, 8, 1624. https://doi.org/10.3390/jcm8101624
Moon I, Lee S-R, Choi E-K, Lee E, Jung J-H, Han K-D, Cha M-J, Oh S, Lip GYH. Non-Vitamin K Antagonist Oral Anticoagulants in Patients with Atrial Fibrillation and Valvular Heart Disease. Journal of Clinical Medicine. 2019; 8(10):1624. https://doi.org/10.3390/jcm8101624
Chicago/Turabian StyleMoon, Inki, So-Ryoung Lee, Eue-Keun Choi, Euijae Lee, Jin-Hyung Jung, Kyung-Do Han, Myung-Jin Cha, Seil Oh, and Gregory Y.H. Lip. 2019. "Non-Vitamin K Antagonist Oral Anticoagulants in Patients with Atrial Fibrillation and Valvular Heart Disease" Journal of Clinical Medicine 8, no. 10: 1624. https://doi.org/10.3390/jcm8101624
APA StyleMoon, I., Lee, S.-R., Choi, E.-K., Lee, E., Jung, J.-H., Han, K.-D., Cha, M.-J., Oh, S., & Lip, G. Y. H. (2019). Non-Vitamin K Antagonist Oral Anticoagulants in Patients with Atrial Fibrillation and Valvular Heart Disease. Journal of Clinical Medicine, 8(10), 1624. https://doi.org/10.3390/jcm8101624






