Long-Term Pancreatic Functional Impairment after Surgery for Neuroendocrine Neoplasms
Abstract
1. Introduction
2. Experimental Section
2.1. Study Design
2.2. Definition of Outcomes
2.3. Data Collection
2.4. Statistical Analysis
3. Results
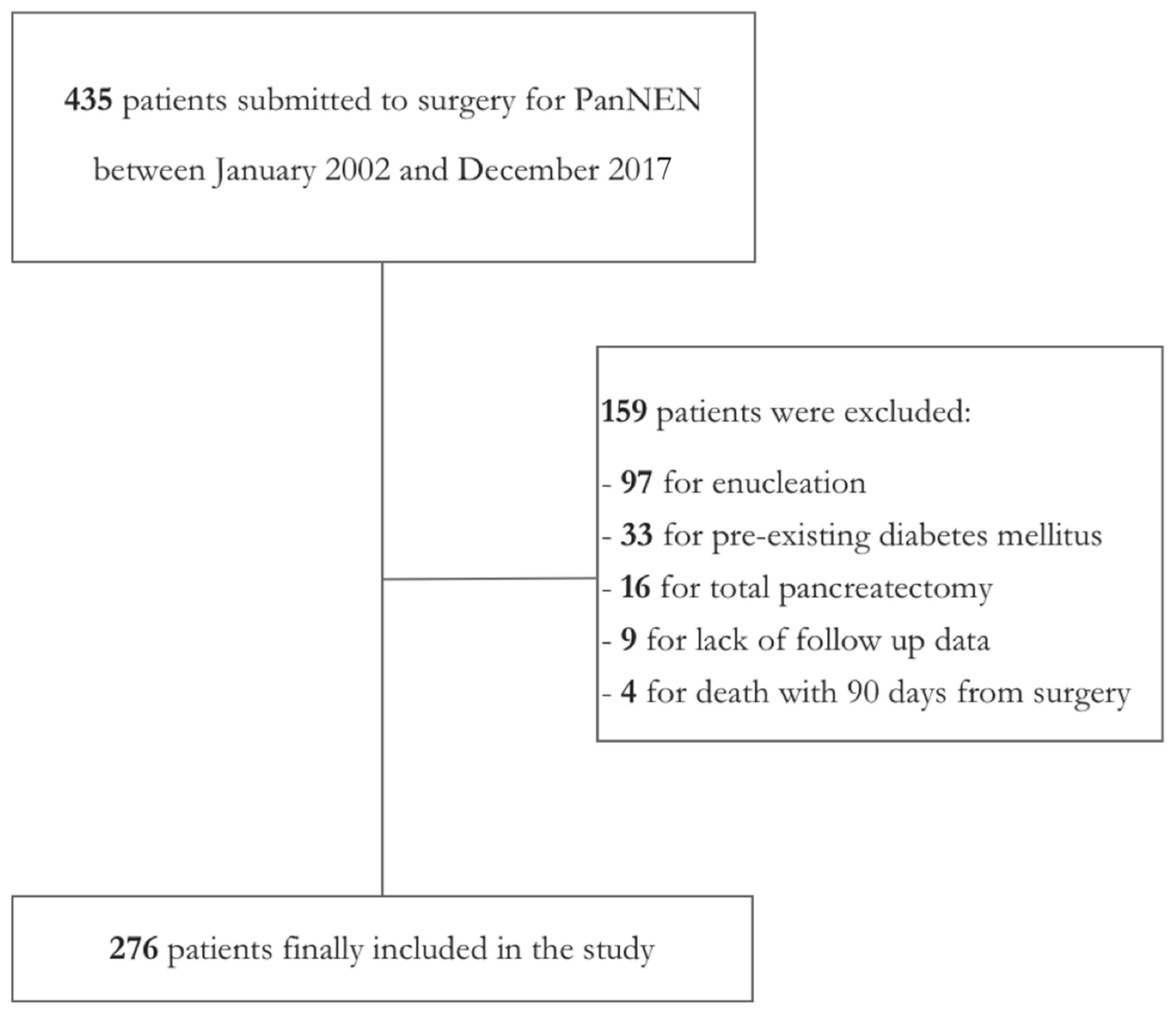
3.1. Study Population
3.2. Postoperative DM
3.3. Postoperative PEI
3.4. Long-Term Oncological Outcomes
4. Discussion
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Dasari, A.; Shen, C.; Halperin, D.; Zhao, B.; Zhou, S.; Xu, Y.; Shih, T.; Yao, J.C. Trends in the Incidence, Prevalence, and Survival Outcomes in Patients With Neuroendocrine Tumors in the United States. JAMA Oncol. 2017, 3, 1335–1342. [Google Scholar] [CrossRef] [PubMed]
- Kuo, E.J.; Salem, R.R. Population-level analysis of pancreatic neuroendocrine tumors 2 cm or less in size. Ann. Surg. Oncol. 2013, 20, 2815–2821. [Google Scholar] [CrossRef] [PubMed]
- Lloyd, R.V.; Osamura, R.Y.; Kloppel, G.; Rosai, J. WHO Classification of Tumours of Endocrine Organs, 4th ed.; Lloyd, R.V., Osamura, R.Y., Kloppel, G., Rosai, J., Eds.; IARC Press: Lyon, France, 2017. [Google Scholar]
- Partelli, S.; Javed, A.A.; Andreasi, V.; He, J.; Muffatti, F.; Weiss, M.J.; Sessa, F.; La Rosa, S.; Doglioni, C.; Zamboni, G.; et al. The number of positive nodes accurately predicts recurrence after pancreaticoduodenectomy for nonfunctioning neuroendocrine neoplasms. Eur. J. Surg. Oncol. 2018, 44, 778–783. [Google Scholar] [CrossRef] [PubMed]
- Genc, C.G.; Jilesen, A.P.; Partelli, S.; Falconi, M.; Muffatti, F.; van Kemenade, F.J.; van Eeden, S.; Verheij, J.; van Dieren, S.; van Eijck, C.H.J.; et al. A New Scoring System to Predict Recurrent Disease in Grade 1 and 2 Nonfunctional Pancreatic Neuroendocrine Tumors. Ann. Surg. 2018, 267, 1148–1154. [Google Scholar] [CrossRef]
- Falconi, M.; Eriksson, B.; Kaltsas, G.; Bartsch, D.K.; Capdevila, J.; Caplin, M.; Kos-Kudla, B.; Kwekkeboom, D.; Rindi, G.; Kloppel, G.; et al. ENETS Consensus Guidelines Update for the Management of Patients with Functional Pancreatic Neuroendocrine Tumors and Non-Functional Pancreatic Neuroendocrine Tumors. Neuroendocrinology 2016, 103, 153–171. [Google Scholar] [CrossRef]
- Phillips, M.E. Pancreatic exocrine insufficiency following pancreatic resection. Pancreatology 2015, 15, 449–455. [Google Scholar] [CrossRef]
- Halloran, C.M.; Cox, T.F.; Chauhan, S.; Raraty, M.G.T.; Sutton, R.; Neoptolemos, J.P.; Ghaneh, P. Partial pancreatic resection for pancreatic malignancy is associated with sustained pancreatic exocrine failure and reduced quality of life: A prospective study. Pancreatology 2011, 11, 535–545. [Google Scholar] [CrossRef]
- Armstrong, T.; Strommer, L.; Ruiz-Jasbon, F.; Shek, F.W.; Harris, S.F.; Permert, J.; Johnson, C.D. Pancreaticoduodenectomy for peri-ampullary neoplasia leads to specific micronutrient deficiencies. Pancreatology 2007, 7, 37–44. [Google Scholar] [CrossRef]
- Winny, M.; Paroglou, V.; Bektas, H.; Kaltenborn, A.; Reichert, B.; Zachau, L.; Kleine, M.; Klempnauer, J.; Schrem, H. Insulin dependence and pancreatic enzyme replacement therapy are independent prognostic factors for long-term survival after operation for chronic pancreatitis. Surg. (U. S.) 2014, 155, 271–279. [Google Scholar] [CrossRef]
- Iacono, C.; Verlato, G.; Ruzzenente, A.; Campagnaro, T.; Bacchelli, C.; Valdegamberi, A.; Bortolasi, L.; Guglielmi, A. Systematic review of central pancreatectomy and meta-analysis of central versus distal pancreatectomy. Br. J. Surg. 2013, 100, 873–885. [Google Scholar] [CrossRef]
- Santangelo, M.; Esposito, A.; Tammaro, V.; Calogero, A.; Criscitiello, C.; Roberti, G.; Candida, M.; Rupealta, N.; Pisani, A.; Carlomagno, N. What indication, morbidity and mortality for central pancreatectomy in oncological surgery? A systematic review. Int. J. Surg. 2016, 28, 172–176. [Google Scholar] [CrossRef] [PubMed]
- Beger, H.G.; Siech, M.; Poch, B.; Mayer, B.; Schoenberg, M.H. Limited surgery for benign tumours of the pancreas: A systematic review. World J. Surg. 2015, 39, 1557–1566. [Google Scholar] [CrossRef] [PubMed]
- Gharios, J.; Hain, E.; Dohan, A.; Prat, F.; Terris, B.; Bertherat, J.; Coriat, R.; Dousset, B.; Gaujoux, S. Pre- and intraoperative diagnostic requirements, benefits and risks of minimally invasive and robotic surgery for neuroendocrine tumors of the pancreas. Best Pract. Res. Clin. Endocrinol. Metab. 2019. [Google Scholar] [CrossRef] [PubMed]
- Crippa, S.; Bassi, C.; Salvia, R.; Falconi, M.; Butturini, G.; Pederzoli, P. Enucleation of pancreatic neoplasms. Br. J. Surg. 2007, 94, 1254–1259. [Google Scholar] [CrossRef] [PubMed]
- Cauley, C.E.; Pitt, H.A.; Ziegler, K.M.; Nakeeb, A.; Schmidt, C.M.; Zyromski, N.J.; House, M.G.; Lillemoe, K.D. Pancreatic Enucleation: Improved Outcomes Compared to Resection. J. Gastrointest. Surg. 2012, 16, 1347–1353. [Google Scholar] [CrossRef]
- Paiella, S.; De Pastena, M.; Faustini, F.; Landoni, L.; Pollini, T.; Bonamini, D.; Giuliani, T.; Bassi, C.; Esposito, A.; Tuveri, M.; et al. Central pancreatectomy for benign or low-grade malignant pancreatic lesions—A single-center retrospective analysis of 116 cases. Eur. J. Surg. Oncol. 2019, 45, 788–792. [Google Scholar] [CrossRef]
- Falconi, M.; Zerbi, A.; Crippa, S.; Balzano, G.; Boninsegna, L.; Capitanio, V.; Bassi, C.; Di Carlo, V.; Pederzoli, P. Parenchyma-Preserving resections for small nonfunctioning pancreatic endocrine tumors. Ann. Surg. Oncol. 2010, 17, 1621–1627. [Google Scholar] [CrossRef]
- Falconi, M.; Mantovani, W.; Crippa, S.; Mascetta, G.; Salvia, R.; Pederzoli, P. Pancreatic insufficiency after different resections for benign tumours. Br. J. Surg. 2008, 95, 85–91. [Google Scholar] [CrossRef]
- Kwon, J.H.; Kim, S.C.; Shim, I.K.; Song, K.B.; Lee, J.H.; Hwang, D.W.; Park, K.M.; Lee, Y.J. Factors affecting the development of diabetes mellitus after pancreatic resection. Pancreas 2015, 44, 1296–1303. [Google Scholar] [CrossRef]
- Malka, D.; Hammel, P.; Sauvanet, A.; Rufat, P. Risk factors for diabetes mellitus in chronic pancreatitis. Gastroenterology 2000, 119, 1324–1332. [Google Scholar] [CrossRef]
- Von Elm, E.; Altman, D.G.; Egger, M.; Pocock, S.J.; Gotzsche, P.C.; Vandenbroucke, J.P. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Statement: Guidelines for reporting observational studies. Int. J. Surg. 2014, 12, 1495–1499. [Google Scholar] [CrossRef] [PubMed]
- American Diabetes Association. 2. Classification and Diagnosis of Diabetes: Standards of Medical Care in Diabetes-2019. Diabetes Care 2019, 42, 13–28. [Google Scholar] [CrossRef] [PubMed]
- Partelli, S.; Boninsegna, L.; Salvia, R.; Bassi, C.; Pederzoli, P.; Falconi, M. Middle-preserving pancreatectomy for multicentric body-sparing lesions of the pancreas. Am. J. Surg. 2009, 198, 49–53. [Google Scholar] [CrossRef]
- Balzano, G.; Maffi, P.; Nano, R.; Mercalli, A.; Melzi, R.; Aleotti, F.; Zerbi, A.; De Cobelli, F.; Gavazzi, F.; Magistretti, P.; et al. Autologous Islet Transplantation in Patients Requiring Pancreatectomy: A Broader Spectrum of Indications Beyond Chronic Pancreatitis. Am. J. Transplant. 2016, 16, 1812–1826. [Google Scholar] [CrossRef] [PubMed]
- Dindo, D.; Demartines, N.; Clavien, P.-A. Classification of surgical complications: A new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann. Surg. 2004, 240, 205–213. [Google Scholar] [CrossRef] [PubMed]
- Bassi, C.; Marchegiani, G.; Dervenis, C.; Sarr, M.; Abu Hilal, M.; Adham, M.; Allen, P.; Andersson, R.; Asbun, H.J.; Besselink, M.G.; et al. The 2016 update of the International Study Group (ISGPS) definition and grading of postoperative pancreatic fistula: 11 Years After. Surgery 2017, 161, 584–591. [Google Scholar] [CrossRef] [PubMed]
- Rindi, G.; Kloppel, G.; Couvelard, A.; Komminoth, P.; Korner, M.; Lopes, J.M.; McNicol, A.-M.; Nilsson, O.; Perren, A.; Scarpa, A.; et al. TNM staging of midgut and hindgut (neuro) endocrine tumors: A consensus proposal including a grading system. Virchows Arch. 2007, 451, 757–762. [Google Scholar] [CrossRef]
- Kusakabe, J.; Anderson, B.; Liu, J.; Williams, G.A.; Chapman, W.C.; Doyle, M.M.B.; Khan, A.S.; Sanford, D.E.; Hammill, C.W.; Strasberg, S.M.; et al. Long-Term Endocrine and Exocrine Insufficiency After Pancreatectomy. J. Gastrointest. Surg. 2019, 23, 1604–1613. [Google Scholar] [CrossRef]
- Shirakawa, S.; Matsumoto, I.; Toyama, H.; Shinzeki, M.; Ajiki, T.; Fukumoto, T.; Ku, Y. Pancreatic Volumetric Assessment as a Predictor of New-Onset Diabetes Following Distal Pancreatectomy. J. Gastrointest. Surg. 2012, 16, 2212–2219. [Google Scholar] [CrossRef]
- You, D.D.; Choi, S.H.; Choi, D.W.; Heo, J.S.; Ho, C.Y.; Kim, W.S. Long-term effects of pancreaticoduodenectomy on glucose metabolism. ANZ J. Surg. 2012, 82, 447–451. [Google Scholar] [CrossRef]
- Neophytou, H.; Wangermez, M.; Gand, E.; Carretier, M.; Danion, J.; Richer, J.-P. Predictive factors of endocrine and exocrine insufficiency after resection of a benign tumour of the pancreas. Ann. Endocrinol. (Paris) 2018, 79, 53–61. [Google Scholar] [CrossRef] [PubMed]
- King, J.; Kazanjian, K.; Matsumoto, J.; Reber, H.A.; Yeh, M.W.; Hines, O.J.; Eibl, G. Distal pancreatectomy: Incidence of postoperative diabetes. J. Gastrointest. Surg. 2008, 12, 1548–1553. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.E.; Jang, J.Y.; Hwang, D.W.; Lee, K.U.; Kim, S.W. Clinical efficacy of organ-preserving pancreatectomy for benign or low-grade malignant potential lesion. J. Korean Med. Sci. 2010, 25, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Shoup, M.; Brennan, M.F.; McWhite, K.; Leung, D.H.Y.; Klimstra, D.; Conlon, K.C. The value of splenic preservation with distal pancreatectomy. Arch. Surg. 2002, 137, 164–168. [Google Scholar] [CrossRef]
- Hirata, K.; Nakata, B.; Amano, R.; Yamazoe, S.; Kimura, K.; Hirakawa, K. Predictive Factors for Change of Diabetes Mellitus Status After Pancreatectomy in Preoperative Diabetic and Nondiabetic Patients. J. Gastrointest. Surg. 2014, 18, 1597–1603. [Google Scholar] [CrossRef]
- Nguyen, N.T.; Nguyen, X.M.T.; Lane, J.; Wang, P. Relationship between obesity and diabetes in a US adult population: Findings from the national health and nutrition examination survey, 1999–2006. Obes. Surg. 2011, 21, 351–355. [Google Scholar] [CrossRef]
- Mann, S.T.W.; Stracke, H.; Lange, U.; Klör, H.U.; Teichmann, J. Vitamin D3 in patients with various grades of chronic pancreatitis, according to morphological and functional criteria of the pancreas. Dig. Dis. Sci. 2003, 48, 533–538. [Google Scholar] [CrossRef]
- Lim, P.W.; Dinh, K.H.; Sullivan, M.; Wassef, W.Y.; Zivny, J.; Whalen, G.F.; LaFemina, J. Thirty-day outcomes underestimate endocrine and exocrine insufficiency after pancreatic resection. HPB 2016, 18, 360–366. [Google Scholar] [CrossRef]
- Matsumoto, J.; Traverso, L.W. Exocrine Function Following the Whipple Operation as Assessed by Stool Elastase. J. Gastrointest. Surg. 2006, 10, 1225–1229. [Google Scholar] [CrossRef]
- Capurso, G.; Traini, M.; Piciucchi, M.; Signoretti, M.; Arcidiacono, P.G. Exocrine pancreatic insufficiency: Prevalence, diagnosis, and management. Clin. Exp. Gastroenterol. 2019, 12, 129–139. [Google Scholar] [CrossRef]
- Kachare, S.D.; Fitzgerald, T.L.; Schuth, O.; Vohra, N.A.; Zervos, E.E. The impact of pancreatic resection on exocrine homeostasis. Am. Surg. 2014, 80, 704–709. [Google Scholar] [PubMed]
- Benini, L.; Gabbrielli, A.; Cristofori, C.; Amodio, A.; Butturini, G.; Cardobi, N.; Sozzi, C.; Frulloni, L.; Mucelli, R.P.; Crinò, S.; et al. Residual pancreatic function after pancreaticoduodenectomy is better preserved with pancreaticojejunostomy than pancreaticogastrostomy: A long-term analysis. Pancreatology 2019, 9, 595–601. [Google Scholar] [CrossRef] [PubMed]
- Tran, T.C.K.; Van Lanschot, J.J.B.; Bruno, M.J.; Van Eijck, C.H.J. Functional changes after pancreatoduodenectomy: Diagnosis and treatment. Pancreatology 2009, 9, 729–737. [Google Scholar] [CrossRef] [PubMed]
- Bettini, R.; Partelli, S.; Boninsegna, L.; Capelli, P.; Crippa, S.; Pederzoli, P.; Scarpa, A.; Falconi, M. Tumor size correlates with malignancy in nonfunctioning pancreatic endocrine tumor. Surgery 2011, 150, 75–82. [Google Scholar] [CrossRef] [PubMed]
- Gaujoux, S.; Partelli, S.; Maire, F.; D’Onofrio, M.; Larroque, B.; Tamburrino, D.; Sauvanet, A.; Falconi, M.; Ruszniewski, P. Observational study of natural history of small sporadic nonfunctioning pancreatic neuroendocrine tumors. J. Clin. Endocrinol. Metab. 2013, 98, 4784–4789. [Google Scholar] [CrossRef] [PubMed]
- Benini, L.; Amodio, A.; Campagnola, P.; Agugiaro, F.; Cristofori, C.; Micciolo, R.; Magro, A.; Gabbrielli, A.; Cabrini, G.; Moser, L.; et al. Fecal elastase-1 is useful in the detection of steatorrhea in patients with pancreatic diseases but not after pancreatic resection. Pancreatology 2013, 13, 38–42. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, H.; Morifuji, M.; Murakami, Y.; Uemura, K.; Ohge, H.; Hayashidani, Y.; Sudo, T.; Sueda, T. Usefulness of a 13C-labeled mixed triglyceride breath test for assessing pancreatic exocrine function after pancreatic surgery. Surgery 2009, 145, 168–175. [Google Scholar] [CrossRef] [PubMed]
- Alfieri, S.; Agnes, A.; Rosa, F.; Di Miceli, D.; Grieco, D.L.; Scaldaferri, F.; Gasbarrini, A.; Doglietto, G.B.; Quero, G. Long-term pancreatic exocrine and endometabolic functionality after pancreaticoduodenectomy. Comparison between pancreaticojejunostomy and pancreatic duct occlusion with fibrin glue. Eur. Rev. Med. Pharmacol. Sci. 2018, 22, 4310–4318. [Google Scholar]
- Lamarca, A.; McCallum, L.; Nuttall, C.; Barriuso, J.; Backen, A.; Frizziero, M.; Leon, R.; Mansoor, W.; McNamara, M.G.; Hubner, R.A.; et al. Somatostatin analogue-induced pancreatic exocrine insufficiency in patients with neuroendocrine tumors: Results of a prospective observational study. Expert Rev. Gastroenterol. Hepatol. 2018, 12, 723–731. [Google Scholar] [CrossRef]
| Variable | n (%) |
|---|---|
| Operative time, min 1 | 240 (180;300) |
| Length of stay, days 1 | 9 (7;11) |
| Readmission | |
| No | 242 (88) |
| Yes | 34 (12) |
| Blood transfusion | |
| No | 229 (83) |
| Yes | 47 (17) |
| Islet autotransplatation | |
| No | 267 (97) |
| Yes | 9 (3) |
| Complications [26] | |
| No complications | 94 (34) |
| I | 56 (20) |
| II | 89 (32) |
| III | 36 (13) |
| IV | 1 (1) |
| POPF [27] | |
| No | 147 (53) |
| Yes | 129 (47) |
| Abdominal Collection | |
| No | 223 (81) |
| Yes | 53 (19) |
| Postoperative Hemorrhage | |
| No | 262 (95) |
| Yes | 14 (5) |
| Variable | Total Population | No Postoperative DM | Postoperative DM | p Value |
|---|---|---|---|---|
| n = 276 | n = 210 | n = 68 | ||
| Age, years | 58 (49;67) | 56 (46;67) | 60 (56;67) | 0.002 |
| Gender | ||||
| Male | 138 (50) | 95 (46) | 43 (63) | |
| Female | 138 (50) | 113 (54) | 25 (37) | 0.017 |
| Preoperative BMI, Kg/m2 | 25 (22;27) | 24 (22;27) | 27 (25;30) | <0.0001 |
| PanNEN functionality | ||||
| Nonfunctioning | 225 (82) | 163 (78) | 62 (91) | |
| Functioning | 51 (18) | 45 (22) | 6 (9) | 0.019 |
| Inherited Syndrome | ||||
| No | 261 (95) | 194 (93) | 67 (99) | |
| Yes | 15 (5) | 14 (7) | 1 (1) | 0.127 |
| Type of Surgery | ||||
| Pancreaticoduodenectomy | 76 (27) | 61 (29) | 15 (22) | |
| Distal Pancreatectomy | 192 (70) | 141 (68) | 51 (75) | |
| Atypical Resection | 8 (3) | 6 (3) | 2 (3) | 0.476 |
| T stage [28] | ||||
| T1–T2 | 180 (65) | 136 (65) | 44 (65) | |
| T3–T4 | 96 (35) | 72 (35) | 24 (35) | 0.919 |
| Tumor grade [3] | ||||
| G1 | 153 (55) | 110 (53) | 43 (63) | |
| G2 | 110 (40) | 85 (41) | 25 (37) | |
| G3 | 13 (5) | 13 (6) | 0 (0) | 0.065 |
| Complications [26] | ||||
| No-I-II | 239 (87) | 179 (85) | 60 (88) | |
| III-IV | 37 (13) | 29 (15) | 8 (12) | 0.647 |
| Variable | OR | 95% C.I. | p |
|---|---|---|---|
| Gender | |||
| Male | 1 | - | |
| Female | 0.481 | 0.178–1.305 | 0.151 |
| Age | |||
| ≤60 years | 1 | - | |
| >60 years | 0.972 | 0.366–2.579 | 0.954 |
| Preoperative BMI | |||
| ≤25 Kg/m2 | 1 | - | |
| >25 Kg/m2 | 4.945 | 1.889–12.943 | 0.001 |
| Type of PanNEN | |||
| Nonfunctioning | 1 | - | |
| Functioning | 0.269 | 0.071–1.022 | 0.054 |
| Variable | Total Population | No Postoperative PEI | Postoperative PEI | p Value |
|---|---|---|---|---|
| n = 276 | n = 158 | n = 118 | ||
| Age, years | 58 (49;67) | 58 (49;65) | 60 (47;68) | 0.556 |
| Gender | ||||
| Male | 138 (50) | 76 (48) | 62 (53) | |
| Female | 138 (50) | 82 (52) | 56 (47) | 0.543 |
| BMI, Kg/m2 | 24.5 (22.5;27) | 25 (23;28) | 24 (22;25) | 0.005 |
| Type of PanNEN | ||||
| Non-functioning | 225 (82) | 123 (78) | 102 (86) | |
| Functioning | 51 (18) | 35 (22) | 16 (14) | 0.085 |
| Inherited Syndrome | ||||
| No | 261 (95) | 150 (95) | 111 (94) | |
| Yes | 15 (5) | 8 (5) | 7 (6) | 0.793 |
| Type of Surgery | ||||
| Pancreaticoduodenectomy | 76 (27) | 8 (5) | 68 (58) | |
| Distal Pancreatectomy | 192 (70) | 144 (91) | 48 (41) | |
| Atypical Resection | 8 (3) | 6 (4) | 2 (1) | <0.0001 |
| T stage [28] | ||||
| T1–T2 | 180 (65) | 116 (73) | 64 (54) | |
| T3–T4 | 96 (35) | 42 (27) | 54 (46) | 0.001 |
| Tumor grade [3] | ||||
| G1 | 153 (55) | 96 (61) | 57 (48) | |
| G2 | 110 (40) | 55 (35) | 55 (47) | |
| G3 | 13 (5) | 7 (4) | 6 (5) | 0.108 |
| Complications [26] | ||||
| No-I-II | 239 (87) | 143 (91) | 96 (81) | |
| III-IV | 37 (13) | 15 (9) | 22 (19) | 0.027 |
| Variable | OR | 95% C.I. | p |
|---|---|---|---|
| BMI | |||
| ≤25 Kg/m2 | 1 | - | |
| >25 Kg/m2 | 0.746 | 0.280–1.989 | 0.558 |
| Type of Surgery | |||
| Distal Pancreatectomy | 1 | - | |
| Pancreaticoduodenectomy | 31.68 | 10.622–94.487 | <0.0001 |
| Atypical resection | 4.8 | 0.626–36.818 | 0.131 |
| T stage [28] | |||
| T1–T2 | 1 | - | |
| T3–T4 | 1.245 | 0.461–3.365 | 0.665 |
| Complications [26] | |||
| No-I–II | 1 | - | |
| III–IV | 1.464 | 0.330–6.486 | 0.616 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Andreasi, V.; Partelli, S.; Capurso, G.; Muffatti, F.; Balzano, G.; Crippa, S.; Falconi, M. Long-Term Pancreatic Functional Impairment after Surgery for Neuroendocrine Neoplasms. J. Clin. Med. 2019, 8, 1611. https://doi.org/10.3390/jcm8101611
Andreasi V, Partelli S, Capurso G, Muffatti F, Balzano G, Crippa S, Falconi M. Long-Term Pancreatic Functional Impairment after Surgery for Neuroendocrine Neoplasms. Journal of Clinical Medicine. 2019; 8(10):1611. https://doi.org/10.3390/jcm8101611
Chicago/Turabian StyleAndreasi, Valentina, Stefano Partelli, Gabriele Capurso, Francesca Muffatti, Gianpaolo Balzano, Stefano Crippa, and Massimo Falconi. 2019. "Long-Term Pancreatic Functional Impairment after Surgery for Neuroendocrine Neoplasms" Journal of Clinical Medicine 8, no. 10: 1611. https://doi.org/10.3390/jcm8101611
APA StyleAndreasi, V., Partelli, S., Capurso, G., Muffatti, F., Balzano, G., Crippa, S., & Falconi, M. (2019). Long-Term Pancreatic Functional Impairment after Surgery for Neuroendocrine Neoplasms. Journal of Clinical Medicine, 8(10), 1611. https://doi.org/10.3390/jcm8101611






