Early Diagnosis and Classification of Cerebral Palsy: An Historical Perspective and Barriers to an Early Diagnosis
Abstract
1. Background
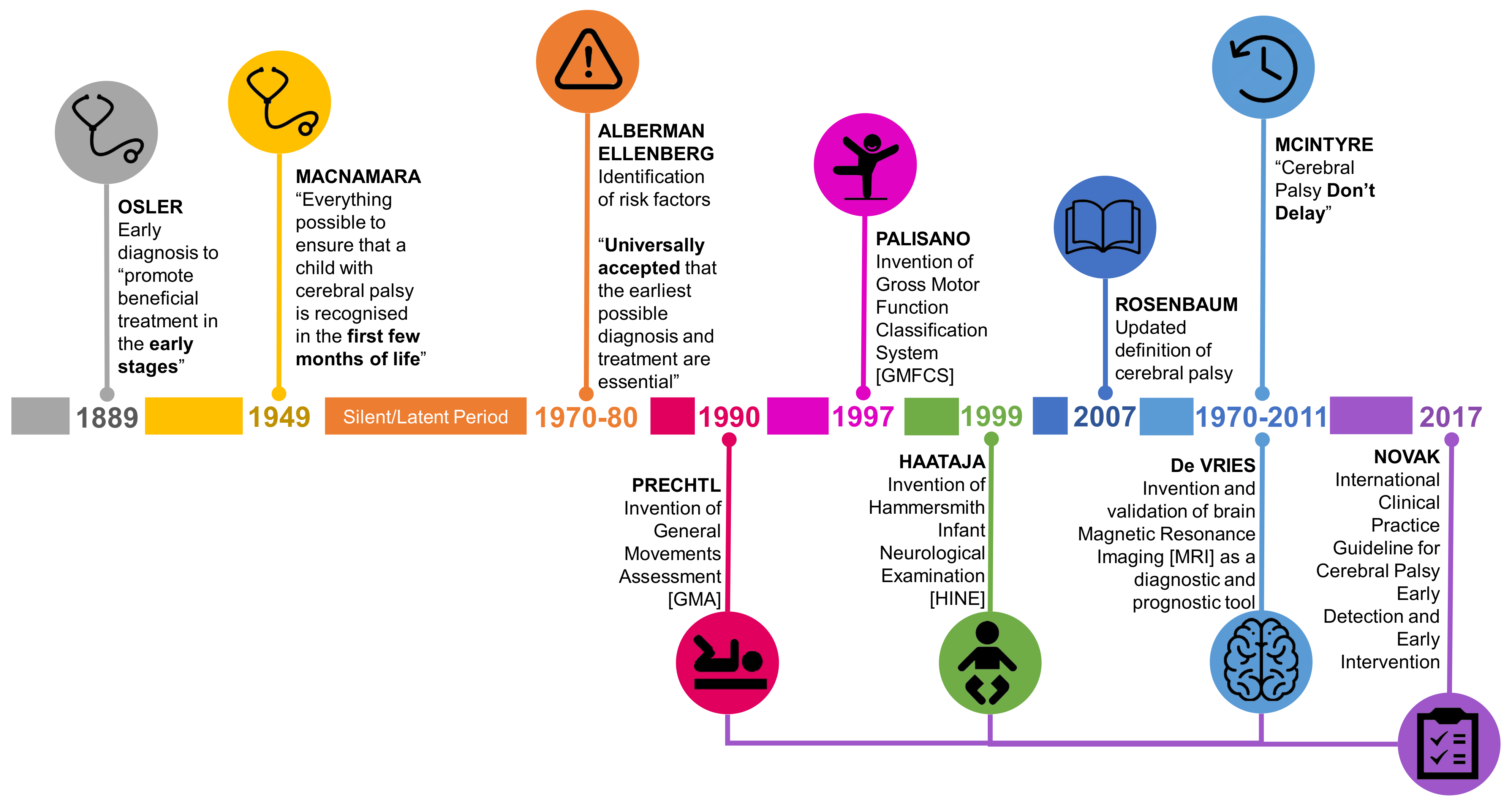
2. The Historical Roundabout Calling for the Earlier Diagnosis of CP
3. Current State of Early Diagnosis of CP Globally
4. Understanding How the Definition of CP Relates to Infants: A Barrier to Early Diagnosis?
4.1. Permanent Disorder of the Development of Movement and Posture
4.2. Activity Limitation
4.3. Non Progressive Brain Lesion
5. Classification of CP
5.1. Is Classification One Type of Pattern Recognition?
5.2. Rethinking Classification Pattern Recognition in Infants With CP—It Will be Different to Older Children
6. Does Diagnosis and Classification of CP Need to Occur Simultaneously in an Infant?
7. Current State of Early Classification of CP under Two Years of Age
7.1. Early Classification of Motor Type
7.2. Early Classification of Topography
7.3. Early Classification of Motor Severity
8. How Can Early Detection of CP be Improved?
9. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Pakula, A.T.; Van Naarden Braun, K.; Yeargin-Allsopp, M. Cerebral palsy: Classification and epidemiology. Phys. Med. Rehabil. Clin. N. Am. 2009, 20, 425–452. [Google Scholar] [CrossRef] [PubMed]
- Shevell, M. Cerebral palsy to cerebral palsy spectrum disorder: Time for a name change? Neurology 2019, 92, 233–235. [Google Scholar] [CrossRef] [PubMed]
- Osler, W. The Cerebral Palsies of Children: A Clinical Study from the Infirmary for Nervous Diseases; H.K. Lewis: London, UK, 1889; p. 103. [Google Scholar]
- Rosenbaum, P.; Paneth, N.; Leviton, A.; Goldstein, M.; Bax, M.; Damiano, D.; Dan, B.; Jacobsson, B. A report: The definition and classification of cerebral palsy April 2006. Dev. Med. Child. Neurol. Suppl. 2007, 109, 8–14. [Google Scholar] [PubMed]
- Little, W.J. On the influence of abnormal parturition, difficult labours, premature birth, and asphyxia neonatorum, on the mental and physical condition of the child, especially in relation to deformities. Dev. Med. Child. Neurol. 1958, 1, 5–34. [Google Scholar] [CrossRef]
- Macnamara, J. Early ascertainment of and prevention of deformity from cerebral palsy. Publ. Health 1949, 62, 121–122. [Google Scholar] [CrossRef]
- Ellenberg, J.H.; Nelson, K.B. Early recognition of infants at high risk for cerebral palsy: Examination at age four months. Dev. Med. Child. Neurol. 1981, 23, 705–716. [Google Scholar] [CrossRef] [PubMed]
- Alberman, E.D.; Goldstein, H. The “at risk” register: A statistical evaluation. Br. J. Prev. Soc. Med. 1970, 24, 129–135. [Google Scholar] [CrossRef] [PubMed]
- Novak, I.; Morgan, C.; Adde, L.; Blackman, J.; Boyd, R.N.; Brunstrom-Hernandez, J.; Cioni, G.; Damiano, D.; Darrah, J.; Eliasson, A.C.; et al. Early, accurate diagnosis and early intervention in cerebral palsy: Advances in diagnosis and treatment. JAMA Pediatr. 2017, 171, 897–907. [Google Scholar] [CrossRef]
- Molnar, G.E. Cerebral palsy: Prognosis and how to judge it. Pediatr. Ann. 1979, 8, 596–605. [Google Scholar] [CrossRef]
- Capute, A.J. Identifying cerebral palsy in infancy through study of primitive-reflex profiles. Pediatr. Ann. 1979, 8, 589–595. [Google Scholar] [CrossRef] [PubMed]
- Pape, K.; Cusick, G.; Blackwell, R.J.; Houang, M.T.W.; Sherwood, A.; Thorburn, R.; Reynolds, E.O.R. Ultrasound detection of brain damage in preterm infants. Lancet 1979, 313, 1261–1264. [Google Scholar] [CrossRef]
- De Vries, L.S.; Dubowitz, V.; Lary, S.; Whitelaw, A.; Dubowitz, L.M.S.; Kaiser, A.; Silverman, M.; Wigglesworth, J.S. Predictive value of cranial ultrasound in the newborn baby: A reappraisal. Lancet 1985, 326, 137–140. [Google Scholar] [CrossRef]
- De Vries, L.S.; Van Haastert, I.C.; Benders, M.J.N.L.; Groenendaal, F. Myth: Cerebral palsy cannot be predicted by neonatal brain imaging. Semin. Fetal. Neonatal. Med. 2011, 16, 279–287. [Google Scholar] [CrossRef] [PubMed]
- Ashwal, S.; Russman, B.S.; Blasco, P.A.; Miller, G.; Sandler, A.; Shevell, M.; Stevenson, R. Practice Parameter: Diagnostic assessment of the child with cerebral palsy: Report of the Quality Standards Subcommittee of the American Academy of Neurology and the Practice Committee of the Child Neurology Society. Neurology 2004, 62, 851–863. [Google Scholar] [CrossRef] [PubMed]
- Prechtl, H.F.R. Qualitative changes of spontaneous movements in fetus and preterm infant are a marker of neurological dysfunction. Early Hum. Dev. 1990, 23, 151–158. [Google Scholar] [CrossRef]
- Prechtl, H.F.R.; Einspieler, C.; Cioni, G.; Bos, A.F.; Ferrari, F.; Sontheimer, D. An early marker for neurological deficits after perinatal brain lesions. Lancet 1997, 349, 1361–1363. [Google Scholar] [CrossRef]
- Einspieler, C.; Prechtl, H.F.R. Prechtl’s Method on the Qualitative Assessment of General Movements in Preterm, Term and Young Infants; Mac Keith Press: London, UK, 2004. [Google Scholar]
- Prechtl, H.F.R.; Fargel, J.W.; Weinmann, H.M.; Bakker, H.H. Postures, motility and respiration of low-risk pre-term infants. Dev. Med. Child. Neurol. 1979, 21, 3–27. [Google Scholar] [CrossRef]
- Haataja, L.; Mercuri, E.; Regev, R.; Cowan, F.; Rutherford, M.; Dubowitz, V.; Dubowitz, L. Optimality score for the neurologic examination of the infant at 12 and 18 months of age. J. Pediatr. 1999, 135, 153–161. [Google Scholar] [CrossRef]
- Guyatt, G.H.; Oxman, A.D.; Vist, G.E.; Kunz, R.; Falck-Ytter, Y.; Alonso-Coello, P.; Schunemann, H.J. GRADE: An emerging consensus on rating quality of evidence and strength of recommendations. BMJ 2008, 336, 924–926. [Google Scholar] [CrossRef]
- McIntyre, S.; Morgan, C.; Walker, K.; Novak, I. Cerebral palsy—Don’t delay. Dev. Disabil. Res. Rev. 2011, 17, 114–129. [Google Scholar] [CrossRef]
- Australian Cerebral Palsy Register Report 2018. Available online: https://cpregister.com/wp-content/uploads/2019/02/Report-of-the-Australian-Cerebral-Palsy-Register-Birth-Years-1995-2012.pdf (accessed on 4 April 2019).
- Galea, C.; Mcintyre, S.; Smithers-Sheedy, H.; Reid, S.M.; Gibson, C.; Delacy, M.; Watson, L.; Goldsmith, S.; Badawi, N.; Blair, E. Cerebral palsy trends in Australia (1995–2009): A population-based observational study. Dev. Med. Child. Neurol. 2019, 61, 186–193. [Google Scholar] [CrossRef] [PubMed]
- Finch-Edmondson, M.; Morgan, C.; Hunt, R.W.; Novak, I. Emergent prophylactic, reparative and restorative brain interventions for infants born preterm with cerebral palsy. Front. Physiol. 2019, 10. [Google Scholar] [CrossRef] [PubMed]
- Baird, G.; McConachie, H.; Scrutton, D. Parents’ perceptions of disclosure of the diagnosis of cerebral palsy. Arch. Dis. Child. 2000, 83, 475–480. [Google Scholar] [CrossRef] [PubMed]
- Byrne, R.; Duncan, A.; Pickar, T.; Burkhardt, S.; Boyd, R.N.; Neel, M.L.; Maitre, N.L. Comparing parent and provider priorities in discussions of early detection and intervention for infants with and at risk of cerebral palsy. Child. Care Health Dev. 2019. [Google Scholar] [CrossRef] [PubMed]
- Whittingham, K.; Sanders, M.R.; McKinlay, L.; Boyd, R.N. Parenting intervention combined with Acceptance and Commitment Therapy: A trial with families of children with cerebral palsy. J. Pediatr. Psychol. 2016, 41, 531–542. [Google Scholar] [CrossRef] [PubMed]
- Spittle, A.J.; Anderson, P.J.; Lee, K.J.; Ferretti, C.; Eeles, A.; Orton, J.; Boyd, R.N.; Inder, T.; Doyle, L.W. Preventive care at home for very preterm infants improves infant and caregiver outcomes at 2 years. Pediatrics 2010, 126, e171–e178. [Google Scholar] [CrossRef] [PubMed]
- Bax, M.; Goldstein, M.; Rosenbaum, P.; Leviton, A.; Paneth, N.; Dan, B.; Jacobsson, B.; Damiano, D. Proposed definition and classification of cerebral palsy, April 2005. Dev. Med. Child. Neurol. 2005, 47, 571–576. [Google Scholar] [CrossRef]
- Maclennan, A.H.; Lewis, S.; Moreno-De-Luca, A.; Fahey, M.; Leventer, R.J.; McIntyre, S.; Ben-Pazi, H.; Corbett, M.; Wang, X.; Baynam, G.; et al. Genetic or other causation should not change the clinical diagnosis of cerebral palsy. J. Child. Neurol. 2019, 34, 472–476. [Google Scholar] [CrossRef]
- Bosanquet, M.; Copeland, L.; Ware, R.; Boyd, R. A systematic review of tests to predict cerebral palsy in young children. Dev. Med. Child. Neurol. 2013, 55, 418–426. [Google Scholar] [CrossRef]
- Romeo, D.M.; Cioni, M.; Palermo, F.; Cilauro, S.; Romeo, M.G. Neurological assessment in infants discharged from a neonatal intensive care unit. Eur. J. Paediatr. Neurol. 2013, 17, 192–198. [Google Scholar] [CrossRef]
- Granild-Jensen, J.B.; Rackauskaite, G.; Flachs, E.M.; Uldall, P. Predictors for early diagnosis of cerebral palsy from national registry data. Dev. Med. Child. Neurol. 2015, 57, 931–935. [Google Scholar] [CrossRef] [PubMed]
- Smithers-Sheedy, H.; Badawi, N.; Blair, E.; Cans, C.; Himmelmann, K.; Krageloh-Mann, I.; McIntyre, S.; Slee, J.; Uldall, P.; Watson, L.; et al. What constitutes cerebral palsy in the twenty-first century? Dev. Med. Child. Neurol. 2014, 56, 323–328. [Google Scholar] [CrossRef] [PubMed]
- Nelson, K.B.; Ellenberg, J.H. Children who “outgrew’ cerebral palsy. Pediatrics 1982, 69, 529–536. [Google Scholar] [PubMed]
- Chen, A.; Dyck Holzinger, S.; Oskoui, M.; Shevell, M. Losing a diagnosis of cerebral palsy: A comparison of variables at 2 and 5 years. Dev. Med. Child. Neurol. 2019, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Nelson, K.B. ‘Outgrowing’ a cerebral palsy diagnosis. Dev. Med. Child. Neurol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Khandaker, G.; Muhit, M.; Karim, T.; Smithers-Sheedy, H.; Novak, I.; Jones, C.; Badawi, N. Epidemiology of cerebral palsy in Bangladesh: A population-based surveillance study. Dev. Med. Child. Neurol. 2018, 61, 601–609. [Google Scholar] [CrossRef] [PubMed]
- Byrne, R.; Noritz, G.; Maitre, N.L.; NCH Early Development Group. Implementation of early diagnosis and intervention guidelines for cerebral palsy in a high-risk infant follow-up clinic. Pediatr. Neurol. 2017, 76, 66–71. [Google Scholar] [CrossRef] [PubMed]
- Krumlinde-Sundholm, L.; Ek, L.; Sicola, E.; Sjostrand, L.; Guzzetta, A.; Sgandurra, G.; Cioni, G.; Eliasson, A.C. Development of the Hand Assessment for Infants: Evidence of internal scale validity. Dev. Med. Child. Neurol. 2017, 59, 1276–1283. [Google Scholar] [CrossRef]
- Ek, L.; Eliasson, A.C.; Sicola, E.; Sjostrand, L.; Guzzetta, A.; Sgandurra, G.; Cioni, G.; Krumlinde-Sundholm, L. Hand Assessment for Infants: Normative reference values. Dev. Med. Child. Neurol. 2019. [Google Scholar] [CrossRef]
- Folio, M.; Fewell, R. Peabody Developmental Motor Scales, 3rd ed.; Pro-ed: Austin, TX, USA, 2000. [Google Scholar]
- Bayley, N. Bayley Scales of Infant and Toddler Development, 3rd ed.; Harcourt Assessment: San Antonio, TX, USA, 2005. [Google Scholar] [CrossRef]
- World Health Organization. ICF: International Classification of Functioning, Disability and Health; World Health Organization: Geneva, Switzerland, 2001. [Google Scholar]
- World Health Organisation Multicentre Growth Reference Study Group; Onis, M. WHO Motor Development Study: Windows of achievement for six gross motor development milestones. Acta Pædiatrica 2006, 95, 86–95. [Google Scholar] [CrossRef]
- Einspieler, C.; Marschik, P.B.; Prechtl, H.F.R. Human motor behavior: Prenatal origin and early postnatal development. Z Psychol. 2008, 16, 147–153. [Google Scholar] [CrossRef]
- Einspieler, C.; Marschik, P.B.; Pansy, J.; Scheuchenegger, A.; Krieber, M.; Yang, H.; Kornacka, M.K.; Rowinska, E.; Soloveichick, M.; Bos, A.F. The general movement optimality score: A detailed assessment of general movements during preterm and term age. Dev. Med. Child. Neurol. 2016, 58, 361–368. [Google Scholar] [CrossRef] [PubMed]
- Hofmeyr, G.J.; Novikova, N. Management of reported decreased fetal movements for improving pregnancy outcomes. Cochrane Database Syst. Rev. 2012, 18. [Google Scholar] [CrossRef] [PubMed]
- Johnson, M. Functional brain development in humans. Nat. Rev. Neurosci. 2001, 2, 475–483. [Google Scholar] [CrossRef] [PubMed]
- Kolb, B.; Muhammad, A. Harnessing the power of neuroplasticity for intervention. Front. Hum. Neurosci. 2014, 8, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Bax, M.; Tydeman, C.; Flodmark, O. Clinical and MRI correlates of cerebral palsy: The European Cerebral Palsy Study. JAMA 2006, 296, 1602–1608. [Google Scholar] [CrossRef]
- Reid, S.M.; Dagia, C.D.; Ditchfield, M.R.; Carlin, J.B.; Reddihough, D.S. Population-based studies of brain imaging patterns in cerebral palsy. Dev. Med. Child. Neurol. 2014, 56, 222–232. [Google Scholar] [CrossRef]
- Towsley, K.; Shevell, M.I.; Dagenais, L. Population-based study of neuroimaging findings in children with cerebral palsy. Eur. J. Padediatr. Neurol. 2011, 15, 29–35. [Google Scholar] [CrossRef]
- Cowan, F. Magnetic resonance imaging of the normal infant brain: Term to 2 years. Available online: http://mrineonatalbrain.com/ch02-04.php#cont-3 (accessed on 20 September 2019).
- Seme-Ciglenecki, P. Predictive values of cranial ultrasound and assessment of general movements for neurological development of preterm infants in the Maribor region of Slovenia. Wien. Klin. Wochenschr. 2007, 119, 490–496. [Google Scholar] [CrossRef]
- Cans, C. Surveillance of cerebral palsy in Europe: A collaboration of cerebral palsy surveys and registers. Dev. Med. Child. Neurol. 2000, 42, 816–824. [Google Scholar] [CrossRef]
- Palisano, R.; Rosenbaum, P.; Walter, S.; Russell, D.; Wood, E.; Galuppi, B. Development and reliability of a system to classify gross motor function in children with cerebral palsy. Dev. Med. Child. Neurol. 1997, 39, 214–223. [Google Scholar] [CrossRef] [PubMed]
- Rosenbaum, P.L.; Walter, S.D.; Hanna, S.E.; Palisano, R.J.; Russell, D.J.; Raina, P.; Wood, E.; Bartlett, D.J.; Galuppi, B.E. Prognosis for gross motor function in cerebral palsy: Creation of motor development curves. JAMA 2002, 288, 1357–1363. [Google Scholar] [CrossRef] [PubMed]
- Bailes, A.F.; Gannotti, M.; Bellows, D.M.; Shusterman, M.; Lyman, J.; Horn, S.D. Caregiver knowledge and preferences for gross motor function information in cerebral palsy. Dev. Med. Child. Neurol. 2018, 60, 1264–1270. [Google Scholar] [CrossRef] [PubMed]
- Wood, E.; Rosenbaum, P. The Gross Motor Function Classification System for Cerebral Palsy: A study of reliability and stability over time. Dev. Med. Child. Neurol. 2000, 42, 292–296. [Google Scholar] [CrossRef] [PubMed]
- Morris, C.; Galuppi, B.E.; Rosenbaum, P.L. Reliability of family report for the Gross Motor Function Classification System. Dev. Med. Child. Neurol. 2004, 46, 455–460. [Google Scholar] [CrossRef]
- Heneghan, C.; Glasziou, P.; Thompson, M.; Rose, P.; Balla, J.; Lasserson, D.; Scott, C.; Perera, R. Diagnostic strategies used in primary care. BMJ 2009, 338, b946. [Google Scholar] [CrossRef] [PubMed]
- Einspieler, C.; Marschik, P.B.; Bos, A.F.; Ferrari, F.; Cioni, G.; Prechtl, H.F.R. Early markers for cerebral palsy: Insights from the assessment of general movements. Future Neurol. 2012, 7, 709–717. [Google Scholar] [CrossRef]
- Morgan, C.; Novak, I.; Dale, R.C.; Guzzetta, A.; Badawi, N. GAME (Goals-Activity-Motor Enrichment): Protocol of a single blind randomised controlled trial of motor training, parent education and environmental enrichment for infants at high risk of cerebral palsy. BMC Neurol. 2014, 14, 203. [Google Scholar] [CrossRef]
- Eliasson, A.-C.; Nordstrand, L.; Ek, L.; Lennartsson, F.; Sjöstrand, L.; Tedroff, K.; Krumlinde-Sundholm, L. The effectiveness of Baby-CIMT in infants younger than 12 months with clinical signs of unilateral-cerebral palsy; an explorative study with randomized design. Res. Dev. Disabil. 2018, 72, 191–201. [Google Scholar] [CrossRef]
- Ryll, U.C.; Wagenaar, N.; Verhage, C.H.; Blennow, M.; De Vries, L.S.; Eliasson, A.C. Early prediction of unilateral cerebral palsy in infants with asymmetric perinatal brain injury—Model development and internal validation. Eur. J. Padediatr. Neurol. 2019. [Google Scholar] [CrossRef]
- Minear, W.L. A classification of cerebral palsy. Pediatrics 1956, 18, 841–852. [Google Scholar] [PubMed]
- Boyd, R.N.; Ziviani, J.; Sakzewski, L.; Novak, I.; Badawi, N.; Pannek, K.; Elliott, C.; Greaves, S.; Guzzetta, A.; Whittingham, K.; et al. REACH: Study protocol of a randomised trial of rehabilitation very early in congenital hemiplegia. BMJ Open 2017, 7, e017204. [Google Scholar] [CrossRef] [PubMed]
- Shevell, M. Classifying cerebral palsy subtypes. Future Neurol. 2010, 5, 765–775. [Google Scholar] [CrossRef]
- Einspieler, C.; Cioni, G.; Paolicelli, P.B.; Bos, A.F.; Dressler, A.; Ferrari, F.; Roversi, M.F.; Prechtl, H.F.R. The early markers for later dyskinetic cerebral palsy are different from those for spastic cerebral palsy. Neuropediatrics 2002, 33, 73–78. [Google Scholar] [CrossRef] [PubMed]
- Jethwa, A.; Mink, J.; Macarthur, C.; Knights, S.; Fehlings, T.; Fehlings, D. Development of the Hypertonia Assessment Tool (HAT): A discriminative tool for hypertonia in children. Dev. Med. Child. Neurol. 2010, 52, e83–e87. [Google Scholar] [CrossRef]
- Kwong, A.K.L.; Fitzgerald, T.L.; Doyle, L.W.; Cheong, J.L.Y.; Spittle, A.J. Predictive validity of spontaneous early infant movement for later cerebral palsy: A systematic review. Dev. Med. Child. Neurol. 2018, 60, 480–489. [Google Scholar] [CrossRef] [PubMed]
- Guzzetta, A.; Pizzardi, A.; Belmonti, V.; Boldrini, A.; Carotenuto, M.; D’acunto, G.; Ferrari, F.; Fiori, S.; Gallo, C.; Ghirri, P.; et al. Hand movements at 3 months predict later hemiplegia in term infants with neonatal cerebral infarction. Dev. Med. Child. Neurol. 2010, 52, 767–772. [Google Scholar] [CrossRef]
- Hay, K.; Nelin, M.; Carey, H.; Chorna, O.; Moore-Clingenpeel, M.M.M.; Maitre, N. Hammersmith Infant Neurological Examination asymmetry score detects hemiplegic cerebral palsy from typical development. Pediatr. Neurol. 2018, 87, 70–74. [Google Scholar] [CrossRef]
- Gorter, J.W.; Ketelaar, M.; Rosenbaum, P.; Helders, P.J.; Palisano, R. Use of the GMFCS in infants with CP: The need for reclassification at age 2 years or older. Dev. Med. Child. Neurol. 2009, 51, 46–52. [Google Scholar] [CrossRef]
- Beckung, E.; Carlsson, G.; Carlsdotter, S.; Uvebrant, P. The natural history of gross motor development in children with cerebral palsy aged 1 to 15 years. Dev. Med. Child. Neurol. 2007, 49, 751–756. [Google Scholar] [CrossRef]
- Romeo, D.M.; Ricci, D.; Brogna, C.; Mercuri, E. Use of the Hammersmith Infant Neurological Examination in infants with cerebral palsy: A critical review of the literature. Dev. Med. Child. Neurol. 2016, 58, 240–245. [Google Scholar] [CrossRef] [PubMed]
- Romeo, D.M.; Cioni, M.; Scoto, M.; Pizzardi, A.; Romeo, M.G.; Guzzetta, A. Prognostic value of a scorable neurological examination from 3 to 12 months post-term age in very preterm infants: A longitudinal study. Early Hum. Dev. 2009, 85, 405–408. [Google Scholar] [CrossRef] [PubMed]
- Haataja, L.; Mercuri, E.; Guzzetta, A.; Rutherford, M.; Counsell, S.; Flavia Frisone, M.; Cioni, G.; Cowan, F.; Dubowitz, L. Neurologic examination in infants with hypoxic-ischemic encephalopathy at age 9 to 14 months: Use of optimality scores and correlation with magnetic resonance imaging findings. J. Pediatr. 2001, 138, 332–337. [Google Scholar] [CrossRef] [PubMed]
- Ricci, D.; Cowan, F.; Pane, M.; Gallini, F.; Haataja, L.; Luciano, R.; Cesarini, L.; Leone, D.; Donvito, V.; Baranello, G.; et al. Neurological examination at 6 to 9 months in infants with cystic periventricular leukomalacia. Neuropediatrics 2006, 37, 247–252. [Google Scholar] [CrossRef] [PubMed]
- Romeo, D.M.; Cioni, M.; Scoto, M.; Mazzone, L.; Palermo, F.; Romeo, M.G. Neuromotor development in infants with cerebral palsy investigated by the Hammersmith Infant Neurological Examination during the first year of age. Eur. J. Padediatr. Neurol. 2008, 12, 24–31. [Google Scholar] [CrossRef] [PubMed]
- Nanba, Y.; Matsui, K.; Aida, N.; Sato, Y.; Toyoshima, K.; Kawataki, M.; Hoshino, R.; Ohyama, M.; Itani, Y.; Goto, A.; et al. Magnetic resonance imaging regional T1 abnormalities at term accurately predict motor outcome in preterm infants. Pediatrics 2007, 120, e10–e19. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.W.; Day, S.M.; Strauss, D.J.; Shavelle, R.M. Prognosis for ambulation in cerebral palsy: A population-based study. Pediatrics 2004, 114, 1264–1271. [Google Scholar] [CrossRef] [PubMed]
- Fedrizzi, E.; Facchin, P.; Marzaroli, M.; Pagliano, E.; Botteon, G.; Percivalle, L.; Fazzi, E. Predictors of independent walking in children with spastic diplegia. J. Child. Neurol. 2000, 15, 228–234. [Google Scholar] [CrossRef]
- Campos da Paz, A.; Burnett, S.M.; Braga, L.W. Walking prognosis in cerebral palsy: A 22-year retrospective analysis. Dev. Med. Child. Neurol. 1994, 36, 130–134. [Google Scholar] [CrossRef]
- Maitre, N.L.; Chorna, O.; Romeo, D.M.; Guzzetta, A. Implementation of the Hammersmith Infant Neurological Examination in a high-risk infant follow-up program. Pediatr. Neurol. 2016, 65, 31–38. [Google Scholar] [CrossRef]
- Kwong, A.K.; Eeles, A.L.; Olsen, J.E.; Cheong, J.L.; Doyle, L.W.; Spittle, A.J. The Baby Moves smartphone app for General Movements Assessment: Engagement amongst extremely preterm and term-born infants in a state-wide geographical study. J. Paediatr. Child. Health 2019, 55, 548–554. [Google Scholar] [CrossRef] [PubMed]
- Marcroft, C.; Khan, A.; Embleton, N.D.; Trenell, M.; Plötz, T. Movement recognition technology as a method of assessing spontaneous general movements in high risk infants. Front. Neurol. 2015, 5, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Kanji, A.; Khoza-Shangase, K.; Moroe, N. Newborn hearing screening protocols and their outcomes: A systematic review. Int. J. Pediatr. Otorhinolaryngol. 2018, 115, 104–109. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
te Velde, A.; Morgan, C.; Novak, I.; Tantsis, E.; Badawi, N. Early Diagnosis and Classification of Cerebral Palsy: An Historical Perspective and Barriers to an Early Diagnosis. J. Clin. Med. 2019, 8, 1599. https://doi.org/10.3390/jcm8101599
te Velde A, Morgan C, Novak I, Tantsis E, Badawi N. Early Diagnosis and Classification of Cerebral Palsy: An Historical Perspective and Barriers to an Early Diagnosis. Journal of Clinical Medicine. 2019; 8(10):1599. https://doi.org/10.3390/jcm8101599
Chicago/Turabian Stylete Velde, Anna, Catherine Morgan, Iona Novak, Esther Tantsis, and Nadia Badawi. 2019. "Early Diagnosis and Classification of Cerebral Palsy: An Historical Perspective and Barriers to an Early Diagnosis" Journal of Clinical Medicine 8, no. 10: 1599. https://doi.org/10.3390/jcm8101599
APA Stylete Velde, A., Morgan, C., Novak, I., Tantsis, E., & Badawi, N. (2019). Early Diagnosis and Classification of Cerebral Palsy: An Historical Perspective and Barriers to an Early Diagnosis. Journal of Clinical Medicine, 8(10), 1599. https://doi.org/10.3390/jcm8101599





