Aerobic Capacity Is Not Associated with Most Cognitive Domains in Patients with Multiple Sclerosis—A Cross-Sectional Investigation
Abstract
1. Introduction
2. Materials and Methods
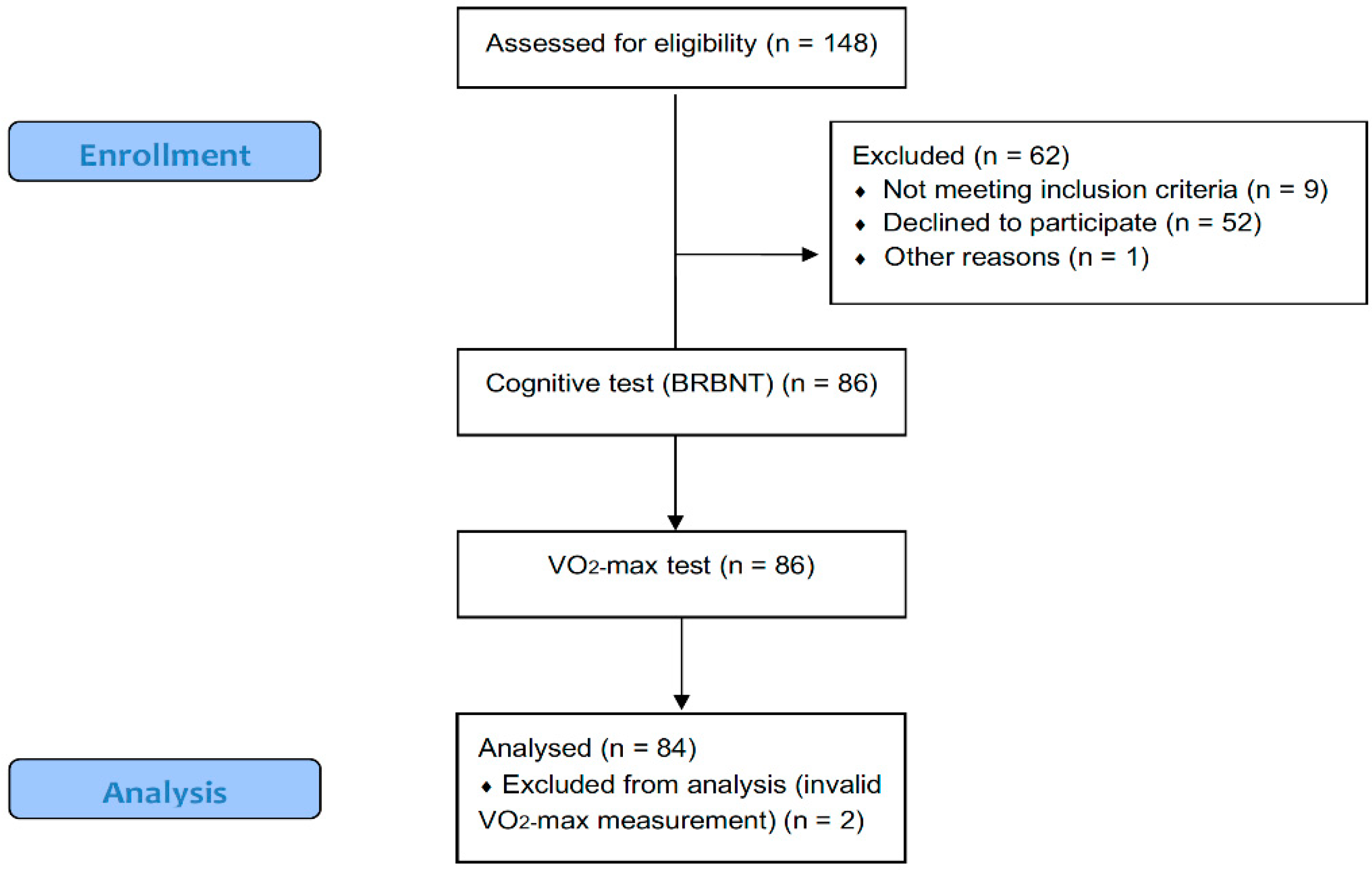
2.1. Subjects
2.2. Cognitive Testing
2.3. Aerobic Capacity
2.4. Statistical Analyses
3. Results
3.1. Subject Characteristics
3.2. Simple Linear Regression Analyses
3.3. Multiple Linear Regression Analyses
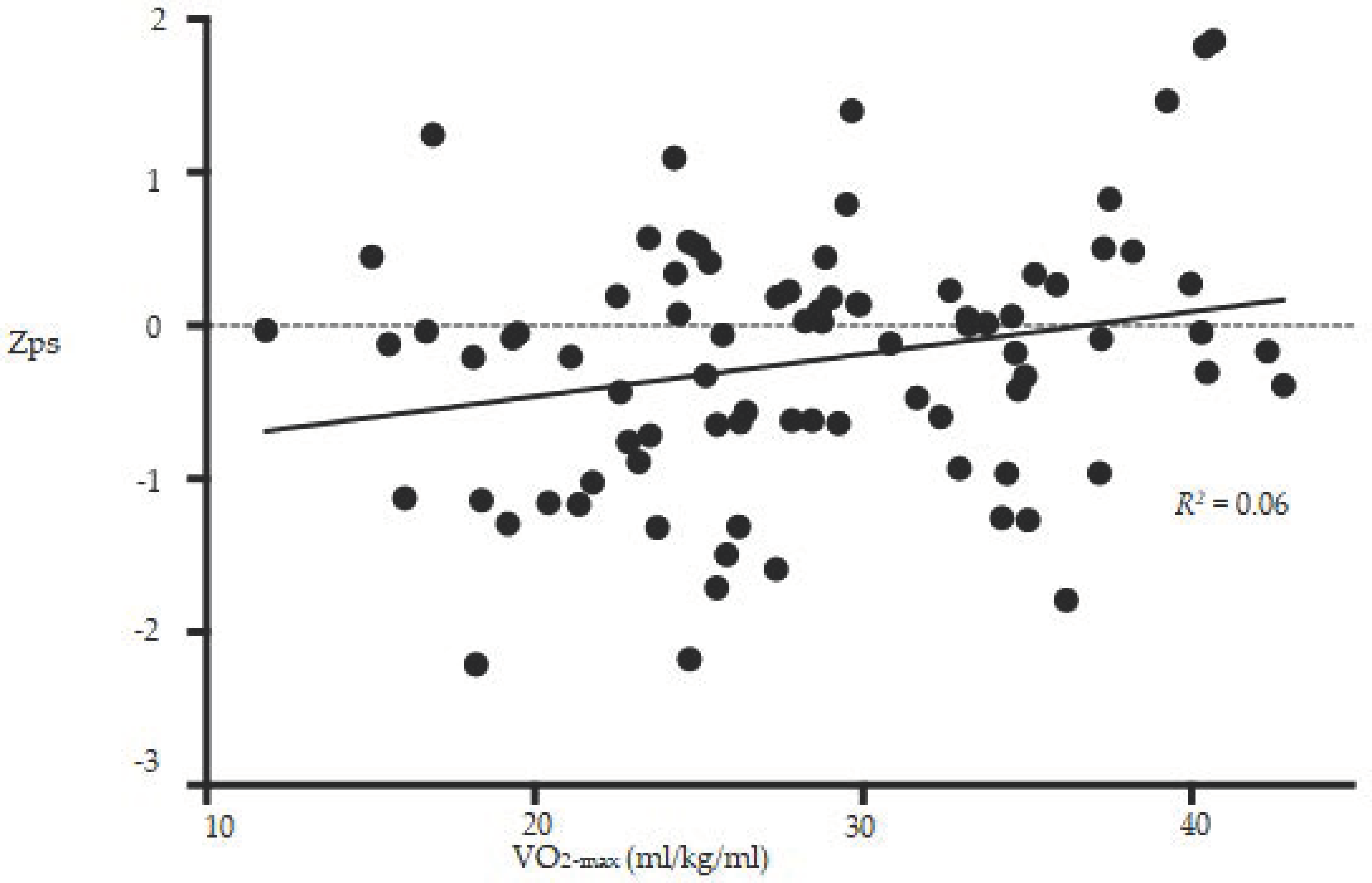
3.4. Z-Scores of the BRBNT
4. Discussion
4.1. Effects on Information Processing
4.2. Effects on Memory and Learning
4.3. Effects on Verbal Fluency
4.4. Cognitively Impaired vs. Nonimpaired
4.5. Direction of Causality
4.6. Potential Underlying Mechanisms
4.7. Limitations
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Study/Domain | Memory and Learning | Information Processing | Executive Function | Verbal Fluency |
|---|---|---|---|---|
| Prakash (2007) n = 24 | SRT: r = −0.22 SPART: r = 0.12 | SDMT: r = 0.17, PASAT: r = 0.42 * | - | WLG: r = 0.30 |
| Prakash (2010) n = 21 | Not shown | Composite Zps score (PASAT, SDMT, WLG): r = 0.46 * | - | Not shown |
| Sandroff (2012) n = 31 | - | Composite Zps score: PASAT, SDMT: r = 0.44 * | - | - |
| Sandroff (2015) n = 28 | - | - | Modified flanker task: Congruent and Incongruent Reaction time: r = −0.54 * r = −0.48 * | - |
| Sandroff (2015) n = 62 | CVLT-2: r = 0.19 BVMT-R: r = 0.18 | SDMT r = 0.41 * | - | - |
| Sandroff (2017) n = 64 | - | SDMT: Not shown Modified flanker task: r = −0.28 * | - | - |
References
- Compston, A.; Coles, A. Multiple sclerosis. Lancet 2008, 372, 1502–1517. [Google Scholar] [CrossRef]
- Benedict, R.H.B.; Zivadinov, R. Risk factors for and management of cognitive dysfunction in multiple sclerosis. Nat. Rev. Neurol. 2011, 7, 332–342. [Google Scholar] [CrossRef] [PubMed]
- Rao, S.M.; Leo, G.J.; Bernardin, L.; Unverzagt, F. Cognitive dysfunction in multiple sclerosis: I. Frequency, patterns, and prediction. Neurology 1991, 41, 685–691. [Google Scholar] [CrossRef] [PubMed]
- Amato, M.P.; Langdon, D.; Montalban, X.; Benedict, R.H.; DeLuca, J.; Krupp, L.B.; Thompson, A.J.; Comi, G. Treatment of cognitive impairment in multiple sclerosis: Position paper. J. Neurol. 2013, 260, 1452–1468. [Google Scholar] [CrossRef] [PubMed]
- Rosti-Otajarvi, E.M.; Hamalainen, P.I. Neuropsychological rehabilitation for multiple sclerosis. Cochrane Database Syst. Rev. 2014. [Google Scholar] [CrossRef] [PubMed]
- Motl, R.W.; Sandroff, B.M.; Benedict, R.H. Cognitive dysfunction and multiple sclerosis: Developing a rationale for considering the efficacy of exercise training. Mult. Scler. 2011, 17, 1034–1040. [Google Scholar] [CrossRef] [PubMed]
- Kramer, A.F.; Colcombe, S. Fitness effects on the cognitive function of older adults: A meta-analytic study—revisited. Perspect. Psychol. Sci. 2018, 13, 213–217. [Google Scholar] [CrossRef] [PubMed]
- Smith, P.J.; Blumenthal, J.A.; Hoffman, B.M.; Cooper, H.; Strauman, T.A.; Welsh-Bohmer, K.; Browndyke, J.N.; Sherwood, A. Aerobic exercise and neurocognitive performance: A meta-analytic review of randomised controlled trials. Psychosom. Med. 2010, 72, 239–252. [Google Scholar] [CrossRef] [PubMed]
- Colcombe, S.; Kramer, A.F. Fitness effects on the cognitive function of older adults: A meta-analytic study. Psychol. Sci. 2003, 14, 125–130. [Google Scholar] [CrossRef] [PubMed]
- Sandroff, B.M.; Pilutti, L.A.; Benedict, R.H.; Motl, R.W. Association between physical fitness and cognitive function in multiple sclerosis: Does disability status matter? Neurorehabil. Neural Repair 2015, 29, 214–223. [Google Scholar] [CrossRef] [PubMed]
- Sandroff, B.M.; Motl, R.W. Fitness and cognitive processing speed in persons with multiple sclerosis: A cross-sectional investigation. J. Clin. Exp. Neuropsychol. 2012, 34, 1041–1052. [Google Scholar] [CrossRef] [PubMed]
- Prakash, R.S.; Snook, E.M.; Erickson, K.I.; Colcombe, S.J.; Voss, M.W.; Motl, R.W.; Kramer, A.F. Cardiorespiratory fitness: A predictor of cortical plasticity in multiple sclerosis. Neuroimage 2007, 34, 1238–1244. [Google Scholar] [CrossRef] [PubMed]
- Sandroff, B.M.; Motl, R.W.; Deluca, J. The influence of cognitive impairment on the fitness-cognition relationship in multiple sclerosis. Med. Sci. Sports Exerc. 2017, 49, 1184–1189. [Google Scholar] [CrossRef] [PubMed]
- Prakash, R.S.; Snook, E.M.; Motl, R.W.; Kramer, A.F. Aerobic fitness is associated with gray matter volume and white matter integrity in multiple sclerosis. Brain Res. 2010, 1341, 41–51. [Google Scholar] [CrossRef] [PubMed]
- Sandroff, B.M.; Hillman, C.H.; Motl, R.W. Aerobic fitness is associated with inhibitory control in persons with multiple sclerosis. Arch. Clin. Neuropsych. 2015, 30, 329–340. [Google Scholar] [CrossRef] [PubMed]
- Rao, S.M. The Cognitive Function Study Group of the National Multiple Sclerosis Society. In A Manual for Brief Repeatable Battery of the Neuropsychological Tests in Multiple Sclerosis; Medical College of Wisconsin: Milwaukee, WI, USA, 1990. [Google Scholar]
- McDonald, W.I.; Compston, A.; Edan, G.; Goodkin, D.; Hartung, H.P.; Lublin, F.D.; McFarland, H.F.; Paty, D.W.; Polman, C.H.; Reingold, S.C.; et al. Recommended diagnostic criteria for multiple sclerosis: Guidelines from the international panel on the diagnosis of multiple sclerosis. Ann. Neurol. 2001, 50, 121–127. [Google Scholar] [CrossRef] [PubMed]
- Boringa, J.B.; Lazeron, R.H.; Reuling, I.E.; Ader, H.J.; Pfennings, L.; Lindeboom, J.; de Sonneville, L.M.; Kalkers, N.F.; Polman, C.H. The brief repeatable battery of neuropsychological tests: Normative values allow application in multiple sclerosis clinical practice. Mult. Scler. 2001, 7, 263–267. [Google Scholar] [CrossRef] [PubMed]
- Schinka, J.A.; Loewenstein, D.A.; Raj, A.; Schoenberg, M.R.; Banko, J.L.; Potter, H.; Duara, R. Defining mild cognitive impairment: Impact of varying decision criteria on neuropsychological diagnostic frequencies and correlates. Am. J. Geriatr. Psychiatry 2010, 18, 684–691. [Google Scholar] [CrossRef] [PubMed]
- Benedict, R.H.; Cookfair, D.; Gavett, R.; Gunther, M.; Munschauer, F.; Garg, N.; Weinstock-Guttman, B. Validity of the minimal assessment of cognitive function in multiple sclerosis (macfims). J. Int. Neuropsychol. Soc. 2006, 12, 549–558. [Google Scholar] [CrossRef] [PubMed]
- Sepulcre, J.; Vanotti, S.; Hernandez, R.; Sandoval, G.; Caceres, F.; Garcea, O.; Villoslada, P. Cognitive impairment in patients with multiple sclerosis using the brief repeatable battery-neuropsychology test. Mult. Scler. 2006, 12, 187–195. [Google Scholar] [CrossRef] [PubMed]
- Buchfuhrer, M.J.; Hansen, J.E.; Robinson, T.E.; Sue, D.Y.; Wasserman, K.; Whipp, B.J. Optimizing the exercise protocol for cardiopulmonary assessment. J. Appl. Physiol. Respir. Environ. Exerc. Physiol. 1983, 55, 1558–1564. [Google Scholar] [CrossRef] [PubMed]
- Borg, G.A. Psychophysical bases of perceived exertion. Med. Sci. Sports Exerc. 1982, 14, 377–381. [Google Scholar] [CrossRef] [PubMed]
- Langeskov-Christensen, M.; Langeskov-Christensen, D.; Overgaard, K.; Moller, A.B.; Dalgas, U. Validity and reliability of vo(2)-max measurements in persons with multiple sclerosis. J. Neurol Sci. 2014, 342, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Hill-Cawthorne, G.A.; Button, T.; Tuohy, O.; Jones, J.L.; May, K.; Somerfield, J.; Green, A.; Giovannoni, G.; Compston, D.A.; Fahey, M.T.; et al. Long term lymphocyte reconstitution after alemtuzumab treatment of multiple sclerosis. J. Neurol. Neurosurg. Psychiatry 2012, 83, 298–304. [Google Scholar] [CrossRef] [PubMed]
- Briken, S.; Gold, S.M.; Patra, S.; Vettorazzi, E.; Harbs, D.; Tallner, A.; Ketels, G.; Schulz, K.H.; Heesen, C. Effects of exercise on fitness and cognition in progressive MS: A randomised, controlled pilot trial. Mult. Scler. 2014, 20, 382–390. [Google Scholar] [CrossRef] [PubMed]
- Carter, A.; Daley, A.; Humphreys, L.; Snowdon, N.; Woodroofe, N.; Petty, J.; Roalfe, A.; Tosh, J.; Sharrack, B.; Saxton, J.M. Pragmatic intervention for increasing self-directed exercise behaviour and improving important health outcomes in people with multiple sclerosis: A randomised controlled trial. Mult. Scler. 2014, 20, 1112–1122. [Google Scholar] [CrossRef] [PubMed]
- Hoang, P.; Schoene, D.; Gandevia, S.; Smith, S.; Lord, S.R. Effects of a home-based step training programme on balance, stepping, cognition and functional performance in people with multiple sclerosis—A randomised controlled trial. Mult. Scler. 2016, 22, 94–103. [Google Scholar] [CrossRef] [PubMed]
- Kucuk, F.; Kara, B.; Poyraz, E.C.; Idiman, E. Improvements in cognition, quality of life, and physical performance with clinical pilates in multiple sclerosis: A randomised controlled trial. J. Phys. Ther. Sci. 2016, 28, 761–768. [Google Scholar] [CrossRef] [PubMed]
- Oken, B.S.; Kishiyama, S.; Zajdel, D.; Bourdette, D.; Carlsen, J.; Haas, M.; Hugos, C.; Kraemer, D.F.; Lawrence, J.; Mass, M. Randomised controlled trial of yoga and exercise in multiple sclerosis. Neurology 2004, 62, 2058–2064. [Google Scholar] [CrossRef] [PubMed]
- Romberg, A.; Virtanen, A.; Ruutiainen, J. Long-term exercise improves functional impairment but not quality of life in multiple sclerosis. J. Neurol. 2005, 252, 839–845. [Google Scholar] [CrossRef] [PubMed]
- Sandroff, B.M.; Balto, J.M.; Klaren, R.E.; Sommer, S.K.; DeLuca, J.; Motl, R.W. Systematically developed pilot randomised controlled trial of exercise and cognition in persons with multiple sclerosis. Neurocase 2016, 22, 443–450. [Google Scholar] [CrossRef] [PubMed]
- Velikonja, O.; Curic, K.; Ozura, A.; Jazbec, S.S. Influence of sports climbing and yoga on spasticity, cognitive function, mood and fatigue in patients with multiple sclerosis. Clin. Neurol. Neurosur. 2010, 112, 597–601. [Google Scholar] [CrossRef] [PubMed]
- Zimmer, P.; Bloch, W.; Schenk, A.; Oberste, M.; Riedel, S.; Kool, J.; Langdon, D.; Dalgas, U.; Kesselring, J.; Bansi, J. High-intensity interval exercise improves cognitive performance and reduces matrix metalloproteinases-2 serum levels in persons with multiple sclerosis: A randomised controlled trial. Mult. Scler. 2017. [Google Scholar] [CrossRef] [PubMed]
- Kierkegaard, M.; Lundberg, I.E.; Olsson, T.; Johansson, S.; Ygberg, S.; Opava, C.; Holmqvist, L.W.; Piehl, F. High-intensity resistance training in multiple sclerosis—An exploratory study of effects on immune markers in blood and cerebrospinal fluid, and on mood, fatigue, health-related quality of life, muscle strength, walking and cognition. J. Neurol. Sci. 2016, 362, 251–257. [Google Scholar] [CrossRef] [PubMed]
- Dalgas, U.; Stenager, E.; Ingemann-Hansen, T. Multiple sclerosis and physical exercise: Recommendations for the application of resistance-, endurance- and combined training. Mult. Scler. 2008, 14, 35–53. [Google Scholar] [CrossRef] [PubMed]
- Roar, M.; Illes, Z.; Sejbaek, T. Practice effect in symbol digit modalities test in multiple sclerosis patients treated with natalizumab. Mult. Scler. Relat. Disord 2016, 10, 116–122. [Google Scholar] [CrossRef] [PubMed]
- Erickson, K.I.; Leckie, R.L.; Weinstein, A.M. Physical activity, fitness, and gray matter volume. Neurobiol. Aging 2014, 35, S20–S28. [Google Scholar] [CrossRef] [PubMed]
- Jonasson, L.S.; Nyberg, L.; Kramer, A.F.; Lundquist, A.; Riklund, K.; Boraxbekk, C.J. Aerobic exercise intervention, cognitive performance, and brain structure: Results from the physical influences on brain in aging (PHIBRA) study. Front. Aging Neurosci. 2016, 8, 336–351. [Google Scholar] [CrossRef] [PubMed]
- Castellano, V.; White, L.J. Serum brain-derived neurotrophic factor response to aerobic exercise in multiple sclerosis. J. Neurol Sci. 2008, 269, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Wens, I.; Keytsman, C.; Deckx, N.; Cools, N.; Dalgas, U.; Eijnde, B.O. Brain derived neurotrophic factor in multiple sclerosis: Effect of 24 weeks endurance and resistance training. Eur. J. Neurol. 2016, 23, 1028–1035. [Google Scholar] [CrossRef] [PubMed]
- White, L.J.; Castellano, V. Exercise and brain health—implications for multiple sclerosis: Part 1—Neuronal growth factors. Sports Med. 2008, 38, 91–100. [Google Scholar] [CrossRef] [PubMed]
- Ainslie, P.N.; Cotter, J.D.; George, K.P.; Lucas, S.; Murrell, C.; Shave, R.; Thomas, K.N.; Williams, M.J.; Atkinson, G. Elevation in cerebral blood flow velocity with aerobic fitness throughout healthy human ageing. J. Physiol. 2008, 586, 4005–4010. [Google Scholar] [CrossRef] [PubMed]
- El-Sayes, J.; Harasym, D.; Turco, C.V.; Locke, M.B.; Nelson, A.J. Exercise-induced neuroplasticity: A mechanistic model and prospects for promoting plasticity. Neuroscientist 2018. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, P.; Brassard, P.; Adser, H.; Pedersen, M.V.; Leick, L.; Hart, E.; Secher, N.H.; Pedersen, B.K.; Pilegaard, H. Evidence for a release of brain-derived neurotrophic factor from the brain during exercise. Exp. Physiol. 2009, 94, 1062–1069. [Google Scholar] [CrossRef] [PubMed]
- Stillman, C.M.; Cohen, J.; Lehman, M.E.; Erickson, K.I. Mediators of physical activity on neurocognitive function: A review at multiple levels of analysis. Front. Hum. Neurosci. 2016, 10. [Google Scholar] [CrossRef] [PubMed]
- Barkhof, F.; Calabresi, P.A.; Miller, D.H.; Reingold, S.C. Imaging outcomes for neuroprotection and repair in multiple sclerosis trials. Nat. Rev. Neurol 2009, 5, 256–266. [Google Scholar] [CrossRef] [PubMed]
- Chiaravalloti, N.D.; DeLuca, J. Cognitive impairment in multiple sclerosis. Lancet Neurol. 2008, 7, 1139–1151. [Google Scholar] [CrossRef]
- Nocentini, U.; Bozzali, M.; Spano, B.; Cercignani, M.; Serra, L.; Basile, B.; Mannu, R.; Caltagirone, C.; De Luca, J. Exploration of the relationships between regional grey matter atrophy and cognition in multiple sclerosis. Brain Imaging Behav. 2014, 8, 378–386. [Google Scholar] [CrossRef] [PubMed]
- Debernard, L.; Melzer, T.R.; Alla, S.; Eagle, J.; Van Stockum, S.; Graham, C.; Osborne, J.R.; Dalrymple-Alford, J.C.; Miller, D.H.; Mason, D.F. Deep grey matter mri abnormalities and cognitive function in relapsing-remitting multiple sclerosis. Psychiat Res. Neuroimag. 2015, 234, 352–361. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.F.; Zhang, F.F.; Huang, D.H.; Wu, L.; Ma, L.; Liu, H.; Zhao, Y.J.; Yu, S.Y.; Shi, J. Contribution of gray and white matter abnormalities to cognitive impairment in multiple sclerosis. Int. J. Mol. Sci. 2017, 18, 46. [Google Scholar] [CrossRef] [PubMed]
- Colcombe, S.J.; Erickson, K.I.; Raz, N.; Webb, A.G.; Cohen, N.J.; McAuley, E.; Kramer, A.F. Aerobic fitness reduces brain tissue loss in aging humans. J. Gerontol. A Biol. Sci. Med. Sci. 2003, 58, 176–180. [Google Scholar] [CrossRef] [PubMed]
- Colcombe, S.J.; Erickson, K.I.; Scalf, P.E.; Kim, J.S.; Prakash, R.; McAuley, E.; Elavsky, S.; Marquez, D.X.; Hu, L.; Kramer, A.F. Aerobic exercise training increases brain volume in aging humans. J. Gerontol. A Biol. Sci. Med. Sci. 2006, 61, 1166–1170. [Google Scholar] [CrossRef] [PubMed]
- Kjolhede, T.; Siemonsen, S.; Wenzel, D.; Stellmann, J.P.; Ringgaard, S.; Pedersen, B.G.; Stenager, E.; Petersen, T.; Vissing, K.; Heesen, C.; et al. Can resistance training impact mri outcomes in relapsing-remitting multiple sclerosis? Mult. Scler. 2017, 24, 1356–1365. [Google Scholar] [CrossRef] [PubMed]
| Cognitive Test | Measures of: | Cognitive Domain |
|---|---|---|
| SRT (including SRT-L, SRT-C & SRT-D) | Verbal learning and memory | Memory and learning |
| SPART (including SPART-D) | Visuospatial learning and delayed recall | Memory and learning |
| SDMT | Sustained attention and information processing speed | Information processing |
| PASAT | Sustained attention and information processing speed | Information processing |
| WLG | Semantic retrieval | Verbal fluency |
| Total Sample | Range | CI Group | CN Group | |
|---|---|---|---|---|
| Sex (men/women) | 35/49 | 15/14 | 20/35 | |
| Age (years) | 44.9 ± 9.4 | (23–64) | 48.8 ± 8.1 | 42.9 ± 9.4 * |
| Height (cm) | 173.9 ± 10.0 | (154–198) | 174.3 ± 10.9 | 173.7 ± 9.6 |
| Weight (kg) | 76.0 ± 16.8 | (46–153) | 74.3 ± 16.2 | 77.0 ± 17.2 |
| Fat percent (%) | 28.4 ± 9.1 | (10–47) | 25.7 ± 8.4 | 29.8 ± 9.3 * |
| BMI (kg/m2) | 25.0 ± 4.2 | (17–39) | 24.2 ± 3.8 | 25.4 ± 4.3 |
| VO2-max (L/min) | 2.13 ± 0.6 | (0.9–3.6) | 1.9 ± 0.7 | 2.2 ± 0.5 * |
| Aerobic Capacity (ml O2/min/kg) | 28.4 ± 7.3 | (11–42) | 25.9 ± 6.7 | 29.7 ± 7.3 * |
| RER (VCO2/VO2) | 1.21 ± 0.08 | (1.01–1.39) | 1.20 ± 0.09 | 1.21 ± 0.08 |
| HRmax (beats/min) | 168 ± 15.8 | (124–200) | 162.7 ± 15.7 | 171.4 ± 15.1 * |
| RPE | 17.2 ± 1.4 | (13–20) | 17.0 ± 1.5 | 17.4 ± 1.3 |
| Education (years) | 16.5 ± 2.8 | (10–25) | 15.1 ± 2.8 | 17.2 ± 2.5 * |
| DMT use (yes/no) | 69/15 | 21/8 | 48/7 | |
| EDSS | 2.67 ± 1.44 | (0–6) | 3.2 ± 1.4 | 2.4 ± 1.4 * |
| MS type (RR, PP, SP) | 73/6/5 | 24/3/2 | 49/3/3 | |
| Disease duration (years) | 9.9 ± 7.1 | (0–38) | 11.2 ± 8.2 | 9.3 ± 6.4 |
| Test (Score Range) | Total Sample | Range | CI Group | CN Group |
|---|---|---|---|---|
| SRT-L (0–72) | 47.2 ± 15.0 | (0–69) | 36.9 ± 17.7 | 52.7 ± 9.8 * |
| SRT-C (0–72) | 37.2 ± 16.8 | (0–69) | 25.6 ± 15.9 | 43.3 ± 13.9 * |
| SRT-D (0–12) | 8.9 ± 2.6 | (3–12) | 7.0 ± 2.6 | 10.0 ± 2.0 * |
| SPART (0–30) | 22.1 ± 5.3 | (11–30) | 18.6 ± 5.2 | 24.0 ± 4.3 * |
| SPART-D (0–10) | 7.6 ± 2.2 | (3–10) | 6.1 ± 2.1 | 8.4 ± 1.8 * |
| SDMT (0–110) | 55.7 ± 14.9 | (33–110) | 47.1 ± 9.4 | 60.2 ± 15.4 * |
| PASAT (0–60) | 43.0 ± 13.0 | (0–60) | 34.2 ± 15.2 | 47.7 ± 8.7 * |
| WLG (0–∞) | 27.5 ± 7.6 | (13–51) | 22.4 ± 7.0 | 30.3 ± 6.5 * |
| Test | Coefficient (SEM) | p-Value | R2 |
|---|---|---|---|
| SRT-L | 0.17 (0.22) | 0.44 | 0.01 |
| SRT-C | 0.28 (0.25) | 0.26 | 0.02 |
| SRT-D | 0.03 (0.04) | 0.47 | 0.01 |
| SPART | 0.05 (0.08) | 0.49 | 0.01 |
| SPART-D | 0.00 (0.03) | 0.95 | 0.00 |
| SDMT | 0.42 (0.22) | 0.06 # | 0.04 |
| PASAT | 0.35 (0.19) | 0.07 # | 0.04 |
| WLG | 0.13 (0.11) | 0.24 | 0.01 |
| Test | Variable | Coefficient (SEM) | p-Value | R2 |
|---|---|---|---|---|
| SRT-L | Aerobic capacity | 0.14 (0.21) | 0.52 | 0.27 ** (p ≤ 0.001) |
| Age | −0.56 (0.16) | 0.001 * | ||
| Sex | 7.08 (3.10) | 0.03 * | ||
| Education years | 1.03 (0.54) | 0.06 | ||
| SRT-C | Aerobic capacity | 0.24 (0.23) | 0.24 | 0.33 ** (p ≤ 0.001) |
| Age | −0.66 (0.17) | <0.001 * | ||
| Sex | 8.60 (3.34) | 0.01 * | ||
| Education years | 1.32 (0.58) | 0.03 * | ||
| SRT-D | Aerobic capacity | 0.02 (0.04) | 0.52 | 0.31 ** (p ≤ 0.001) |
| Age | −0.10 (0.27) | <0.001 * | ||
| Sex | 1.40 (0.53) | 0.01 * | ||
| Education years | 0.17 (0.09) | 0.06 | ||
| SPART | Aerobic capacity | 0.23 (0.08) | 0.77 | 0.19 ** (p = 0.001) |
| Age | −0.16 (0.06) | 0.001 * | ||
| Sex | 1.25 (1.16) | 0.28 | ||
| Education years | 0.43 (0.20) | 0.03 * | ||
| SPART-D | Aerobic capacity | −0.01 (0.03) | 0.70 | 0.16 ** (p = 0.02) |
| Age | −0.06 (0.02) | 0.01 * | ||
| Sex | −0.37 (0.48) | 0.44 | ||
| Education years | 0.17 (0.08) | 0.04 * | ||
| SDMT | Aerobic capacity | 0.33 (0.22) | 0.13 | 0.24 ** (p ≤ 0.001) |
| Age | −0.65 (0.16) | <0.001 * | ||
| Sex | 3.54 (3.16) | 0.27 | ||
| Education years | 0.17 (0.55) | 0.75 | ||
| PASAT | Aerobic capacity | 0.32 (0.20) | 0.12 | 0.10 (p = 0.09) |
| Age | −0.01 (0.15) | 0.92 | ||
| Sex | 0.63 (2.98) | 0.83 | ||
| Education years | 1.11 (0.52) | 0.03 * | ||
| WLG | Aerobic capacity | 0.19 (0.11) | 0.10 | 0.21 ** (p = 0.001) |
| Age | −0.15 (0.08) | 0.08 | ||
| Sex | 5.46 (1.64) | 0.001 * | ||
| Education years | 0.27 (0.28) | 0.34 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Langeskov-Christensen, M.; Eskildsen, S.; Stenager, E.; Boye Jensen, H.; Hvilsted Nielsen, H.; Petersen, T.; Grøndahl Hvid, L.; Hämäläinen, P.; Marstrand, L.; Dalgas, U. Aerobic Capacity Is Not Associated with Most Cognitive Domains in Patients with Multiple Sclerosis—A Cross-Sectional Investigation. J. Clin. Med. 2018, 7, 272. https://doi.org/10.3390/jcm7090272
Langeskov-Christensen M, Eskildsen S, Stenager E, Boye Jensen H, Hvilsted Nielsen H, Petersen T, Grøndahl Hvid L, Hämäläinen P, Marstrand L, Dalgas U. Aerobic Capacity Is Not Associated with Most Cognitive Domains in Patients with Multiple Sclerosis—A Cross-Sectional Investigation. Journal of Clinical Medicine. 2018; 7(9):272. https://doi.org/10.3390/jcm7090272
Chicago/Turabian StyleLangeskov-Christensen, Martin, Søren Eskildsen, Egon Stenager, Henrik Boye Jensen, Helle Hvilsted Nielsen, Thor Petersen, Lars Grøndahl Hvid, Päivi Hämäläinen, Lisbet Marstrand, and Ulrik Dalgas. 2018. "Aerobic Capacity Is Not Associated with Most Cognitive Domains in Patients with Multiple Sclerosis—A Cross-Sectional Investigation" Journal of Clinical Medicine 7, no. 9: 272. https://doi.org/10.3390/jcm7090272
APA StyleLangeskov-Christensen, M., Eskildsen, S., Stenager, E., Boye Jensen, H., Hvilsted Nielsen, H., Petersen, T., Grøndahl Hvid, L., Hämäläinen, P., Marstrand, L., & Dalgas, U. (2018). Aerobic Capacity Is Not Associated with Most Cognitive Domains in Patients with Multiple Sclerosis—A Cross-Sectional Investigation. Journal of Clinical Medicine, 7(9), 272. https://doi.org/10.3390/jcm7090272





