Role of Vitamin A in the Immune System
Abstract
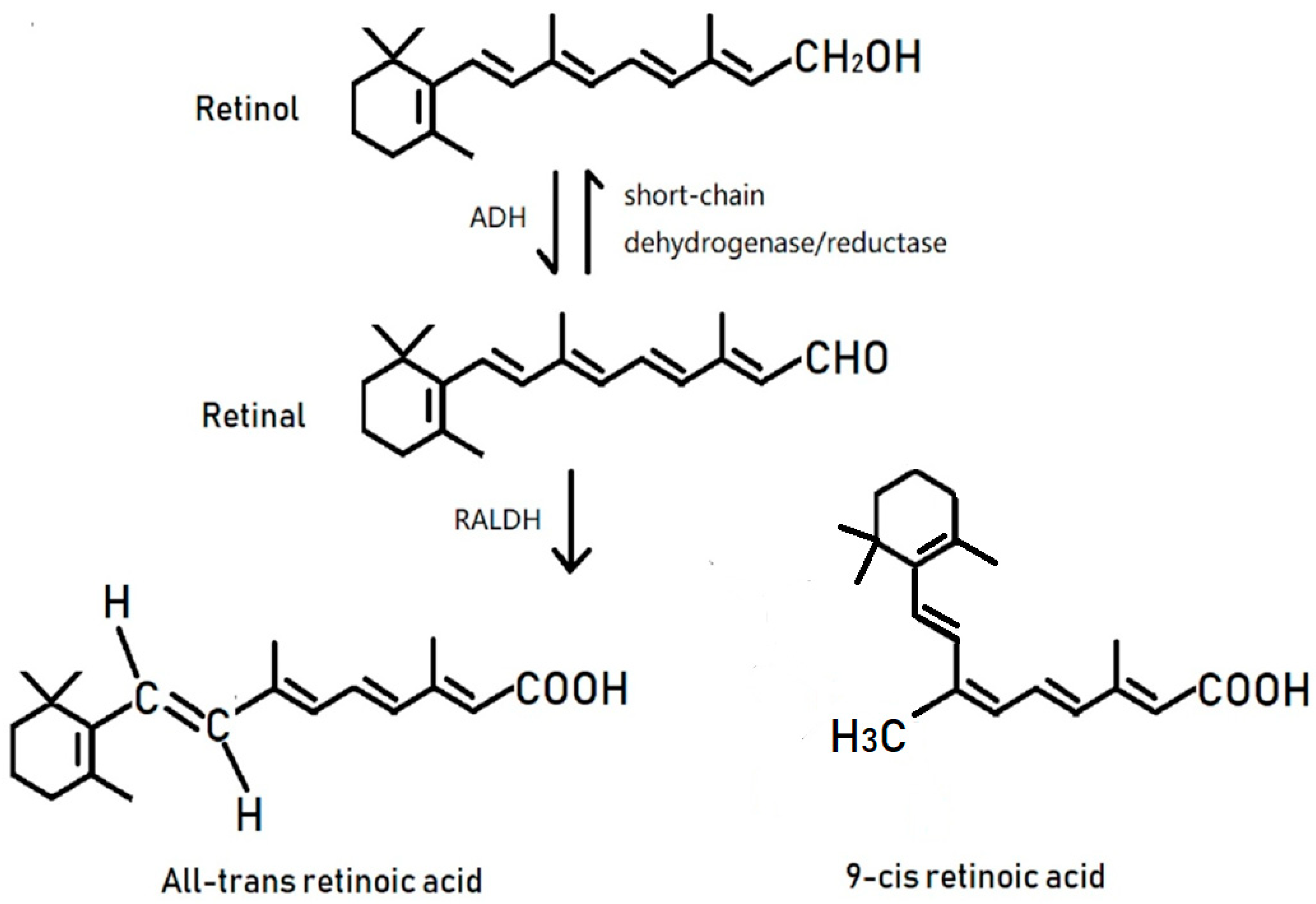
1. Introduction
2. RA Nuclear Receptors
3. VitA Is Involved in the Formation of the Epithelial and Mucous Tissues
4. VitA and Its Impact on the Immune System
5. VitA Affects Cell Differentiation, Maturity, and Immunological Function in Innate Immunity
6. Effects of VitA on T Cells
6.1. RA Induces T Cell Migration
6.2. RA Is a Control Factor for Regulatory T Cells and Maintains Its Homeostasis
6.3. RA May Promote the Ongoing Immune Response
7. Effects of VitA on B Cell Function
7.1. Effects of VitA on Immunoglobulin Production
7.2. VitA Regulation of B Cell Activity
8. Application of VitA in the Treatment of Infectious Diseases
8.1. Tuberculosis
8.2. Acquired Immune Deficiency Syndrome (AIDS)
8.3. Infectious Diseases in Children
9. Summary
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sommer, A. Vitamin a deficiency and clinical disease: An historical overview. J. Nutr. 2008, 138, 1835–1839. [Google Scholar] [CrossRef] [PubMed]
- Mellanby, E.; Green, H.N. Vitamin A as an anti-infective agent. Br. Med. J. 1928, 2, 691–696. [Google Scholar]
- Kolb, E. Recent findings on the importance of vitamin A and its metabolism in man and laboratory animals. Z. Gesamte Inn. Med. 1981, 36, 897–902. [Google Scholar] [PubMed]
- Semba, R.D. Vitamin A, immunity, and infection. Clin. Infect. Dis. 1994, 19, 489–499. [Google Scholar] [CrossRef] [PubMed]
- Muhilal, S.; Permeisih, D.; Idjradinata, Y.R.; Muherdiyantiningsih, D.; Karyadi, D. Vitamin A-fortified monosodium glutamate and health, growth, and survival of children: A controlled field trial. Am. J. Clin. Nutr. 1988, 48, 1271–1276. [Google Scholar] [CrossRef] [PubMed]
- Pino-Lagos, K.; Guo, Y.; Noelle, R.J. Retinoic acid: A key player in immunity. Biofactors 2010, 36, 430–436. [Google Scholar] [CrossRef] [PubMed]
- Ross, S.A.; Mccaffery, P.J.; Drager, U.C.; De Luca, L.M. Retinoids in embryonal development. Physiol. Rev. 2000, 80, 1021–1054. [Google Scholar] [CrossRef] [PubMed]
- Soprano, D.R.; Qin, P.; Soprano, K.J. Retinoic acid receptors and cancers. J. Nutr. 2002, 132, 3809S. [Google Scholar] [CrossRef] [PubMed]
- Rochette-Egly, C.; Germain, P. Dynamic and combinatorial control of gene expression by nuclear retinoic acid receptors (RARs). Nucl. Recept. Signal. 2009, 7, e005. [Google Scholar]
- Repa, J.J.; Hanson, K.K.; Clagett-Dame, M. All-trans-retinol is a ligand for the retinoic acid receptors. Proc. Natl. Acad. Sci. USA 1993, 90, 7293. [Google Scholar] [CrossRef] [PubMed]
- Chambon, P. A decade of molecular biology of retinoic acid receptors. FASEB J. 1996, 10, 940. [Google Scholar] [CrossRef] [PubMed]
- Mangelsdorf, D.; Umesono, K.; Evans, R.M. The Retinoid Receptors. In The Retinoids: Biology, Chemistry and Medicine, 2nd ed.; Sporn, M.B., Roberts, A.B., Goodman, D.S., Eds.; Raven Press: New York, NY, USA, 1994; pp. 319–350. [Google Scholar]
- Glass, C.K.; Rosenfeld, M.G. The coregulator exchange in transcriptional functions of nuclear receptors. Genes Dev. 2000, 14, 121. [Google Scholar] [PubMed]
- Bastien, J.; Rochetteegly, C. Nuclear retinoid receptors and the transcription of retinoid-target genes. Gene 2004, 328, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Taimi, M.; Helvig, C.; Wisniewski, J.; Ramshaw, H.; White, J.; Amad, M.; Korczak, B.; Petkovich, M. A novel human cytochrome P450, CYP26C1, involved in metabolism of 9-cis and all-trans isomers of retinoic acid. Biol. Chem. 2004, 279, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Abu-Abed, S.S.; Beckett, B.R.; Chiba, H.; Chithalen, J.V.; Jones, G.; Metzger, D.; Chambon, P.; Petkovich, M. Mouse P450RAIexpression and retinoic acid-inducible retinoic acid metabolism in F9 cells are regulated by retinoic acid receptor gamma and retinoid X receptor alpha. Biol. Chem. 1998, 273, 2409–2415. [Google Scholar] [CrossRef]
- Ribes, V.; Otto, D.M.; Dickmann, L.; Schmidt, K.; Schuhbaur, B.; Henderson, C.; Blomhoff, R.; Wolf, C.R.; Tickle, C.; Dolle, P. Rescue of cytochrome P450 oxidoreductase (Por) mouse mutants reveals functions in vasculogenesis, brain and limb patterning linked to retinoic acid homeostasis. Dev. Biol. 2007, 303, 66–81. [Google Scholar] [CrossRef] [PubMed]
- Mccullough, F.S.; Northropclewes, C.A.; Thurnham, D.I. The effect of vitamin A on epithelial integrity. Proc. Nutr. Soc. 1999, 58, 289. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.L.; Swartz-Basile, D.A.; Rubin, D.C.; Levin, M.S. Retinoic acid stimulates early cellular proliferation in the adapting remnant rat small intestine after partial resection. J. Nutr. 1997, 127, 1297–1303. [Google Scholar] [CrossRef] [PubMed]
- Amitromach, E.; Uni, Z.; Cheled, S.; Berkovich, Z.; Reifen, R. Bacterial population and innate immunity-related genes in rat gastrointestinal tract are altered by vitamin A-deficient diet. J. Nutr. Biochem. 2009, 20, 70–77. [Google Scholar] [CrossRef] [PubMed]
- Wolbach, S.B.; Howe, P.R. Tissue changes following deprivation of fat-soluble A vitamin. Nutr. Rev. 1925, 42, 753–777. [Google Scholar]
- Wilson, J.R.; Dubois, R.O. Report of a fatal case of keratomalacia in an infant, with postmortem examination. Am. J. Dis. Child. 1923, 26, 431–446. [Google Scholar] [CrossRef]
- Blackfan, K.D.; Wolbach, S.B. Vitamin A deficiency in infants: A clinical and pathological study. J. Pediatr. 1933, 3, 679–706. [Google Scholar] [CrossRef]
- Keenum, D.G.; Semba, R.D.; Wirasasmita, S.; Natadisastra, G.; Muhilal, S.; West, K.P., Jr.; Sommer, A. Assessment of vitamin A status by a disk applicator for conjunctival impression cytology. Arch. Ophthalmol. 1990, 108, 1436–1441. [Google Scholar] [CrossRef] [PubMed]
- Natadisastra, G.; Wittpenn, J.R.; West, K.P., Jr.; Muhilal, S.; Sommer, A. Impression cytology for detection of vitamin A deficiency. Arch. Ophthalmol. 1987, 105, 1224. [Google Scholar] [CrossRef] [PubMed]
- Qi, Y.J.; Niu, Q.L.; Zhu, X.L.; Zhao, X.Z.; Yang, W.W.; Wang, X.J. Relationship between deficiencies in vitamin A and E and occurrence of infectious diseases among children. Eur. Rev. Med. Pharmacol. Sci. 2016, 20, 5009–5012. [Google Scholar] [PubMed]
- Riabroy, N.; Tanumihardjo, S.A. Oral doses of α-retinyl ester track chylomicron uptake and distribution of vitamin A in a male piglet model for newborn infants. J. Nutr. 2014, 144, 1188–1195. [Google Scholar] [CrossRef] [PubMed]
- Kiss, I.; Rühl, R.; Szegezdi, E.; Fritzsche, B.; Toth, B.; Pongrácz, J.; Perlmann, T.; Fésüs, L.; Szondy, Z. Retinoid receptor-activating ligands are produced within the mouse thymus during postnatal development. Eur. J. Immunol. 2008, 38, 147–155. [Google Scholar] [CrossRef] [PubMed]
- Kuwata, T.; Wang, I.M.; Tamura, T.; Ponnamperuma, R.M.; Levine, R.; Holmes, K.L.; Morse, H.C.; De Luca, L.M.; Ozato, K. Vitamin A deficiency in mice causes a systemic expansion of myeloid cells. Blood 2000, 95, 3349. [Google Scholar] [PubMed]
- van Bennekum, A.M.; Wong Yen Kong, L.R.; Gijbels, M.J.; Tielen, F.J.; Roholl, P.J.; Brouwer, A.; Hendriks, H.F. Mitogen response of B cells, but not T cells, is impaired in adult vitamin A-deficient rats. J. Nutr. 1991, 121, 1960–1968. [Google Scholar] [CrossRef] [PubMed]
- Darwiche, N.; Celli, G.; Sly, L.; Lancillotti, F.; De Luca, L.M. Retinoid status controls the appearance of reserve cells and keratin expression in mouse cervical epithelium. Cancer Res. 1993, 53, 2287–2299. [Google Scholar] [PubMed]
- Chang, H.K.; Hou, W.S. Retinoic acid modulates interferon-γ production by hepatic natural killer T cells via phosphatase 2A and the extracellular signal-regulated kinase pathway. J. Interferon Cytokine Res. 2015, 35, 200–212. [Google Scholar] [CrossRef] [PubMed]
- Wynn, T.A.; Vannella, K.M. Macrophages in Tissue Repair, Regeneration, and Fibrosis. Immunity 2016, 44, 450–462. [Google Scholar] [CrossRef] [PubMed]
- Hiemstra, I.H.; Beijer, M.R.; Veninga, H.; Vrijland, K.; Borg, E.G.; Olivier, B.J.; Mebius, R.E.; Kraal, G.; den Haan, J.M. The identification and developmental requirements of colonic CD169+ macrophages. Immunology 2014, 142, 269. [Google Scholar] [CrossRef] [PubMed]
- Pereira, W.F.; Ribeiro-Gomes, F.L.; Guillermo, L.V.; Vellozo, N.S.; Montalvão, F.; Dosreis, G.A.; Lopes, M.F. Myeloid-derived suppressor cells help protective immunity to Leishmania major infection despite suppressed T cell responses. J. Leukoc. Biol. 2011, 90, 1191–1197. [Google Scholar] [CrossRef] [PubMed]
- Vellozo, N.S.; Pereiramarques, S.T.; Cabralpiccin, M.P.; Filardy, A.A.; Ribeirogomes, F.L.; Rigoni, T.S.; Dosreis, G.A.; Lopes, M.F. All-trans retinoic acid promotes an m1- to m2-phenotype shift and inhibits macrophage-mediated immunity to leishmania major. Front. Immunol. 2017, 8, 1560. [Google Scholar] [CrossRef] [PubMed]
- Shrestha, S.; Kim, S.Y.; Yun, Y.J.; Kim, J.K.; Lee, J.M.; Shin, M.; Song, D.K.; Hong, C.W. Retinoic acid induces hypersegmentation and enhances cytotoxicity of neutrophils against cancer cells. Immunol. Lett. 2017, 182, 24–29. [Google Scholar] [CrossRef] [PubMed]
- Worbs, T.; Hammerschmidt, S.I.; Förster, R. Dendritic cell migration in health and disease. Nat. Rev. Immunol. 2017, 17, 30–48. [Google Scholar] [CrossRef] [PubMed]
- Beijer, M.R.; Molenaar, R.; Goverse, G.; Mebius, R.E.; Kraal, G.; den Haan, J.M. A crucial role for retinoic acid in the development of Notch-dependent murine splenic CD8−CD4− and CD4+ dendritic cells. Eur. J. Immunol. 2013, 43, 1608–1616. [Google Scholar] [CrossRef] [PubMed]
- Klebanoff, C.A.; Spencer, S.P.; Torabi-Parizi, P.; Grainger, J.R.; Roychoudhuri, R.; Ji, Y.; Sukumar, M.; Muranski, P.; Scott, C.D.; Hall, J.A.; et al. Retinoic acid controls the homeostasis of pre-cDC-derived splenic and intestinal dendritic cells. J. Exp. Med. 2013, 210, 1961–1976. [Google Scholar] [CrossRef] [PubMed]
- Duriancik, D.M.; Hoag, K.A. Vitamin A deficiency alters splenic dendritic cell subsets and increases CD8+Gr-1+ memory T lymphocytes in C57BL/6J mice. Cell Immunol. 2010, 265, 156–163. [Google Scholar] [CrossRef] [PubMed]
- Zeng, R.; Bscheider, M.; Lahl, K.; Lee, M.; Butcher, E.C. Generation and transcriptional programming of intestinal dendritic cells: Essential role of retinoic acid. Mucosal. Immunol. 2016, 9, 183–193. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.M.; Hall, J.A.; Blank, R.B.; Bouladoux, N.; Oukka, M.; Mora, J.R.; Belkaid, Y. Small intestine lamina propria dendritic cells promote de novo generation of Foxp3 Treg cells via retinoic acid. J. Exp. Med. 2007, 204, 1775–1785. [Google Scholar] [CrossRef] [PubMed]
- Coombes, J.L.; Siddiqui, K.R.; Arancibia-Cárcamo, C.V.; Hall, J.; Sun, C.M.; Belkaid, Y.; Powrie, F. A functionally specialized population of mucosal CD103+ DCs induces Foxp3+ regulatory T cells via a TGF-β– and retinoic acid–dependent mechanism. J. Exp. Med. 2007, 204, 1757–1764. [Google Scholar] [CrossRef] [PubMed]
- Scott, C.L.; Aumeunier, A.M.; Mowat, A.M. Intestinal CD103+, dendritic cells: Master regulators of tolerance? Trends Immunol. 2011, 32, 412–419. [Google Scholar] [CrossRef] [PubMed]
- DePaolo, R.W.; Abadie, V.; Tang, F.; Fehlner-Peach, H.; Hall, J.A.; Wang, W.; Marietta, E.V.; Kasarda, D.D.; Waldmann, T.A.; Murray, J.A.; et al. Co-adjuvant effects of retinoic acid and IL-15 induce inflammatory immunity to dietary antigens. Nature 2011, 471, 220–224. [Google Scholar] [CrossRef] [PubMed]
- Artis, D.; Spits, H. The biology of innate lymphoid cells. Nature 2015, 517, 293–301. [Google Scholar] [CrossRef] [PubMed]
- Sa, V.D.P.; Mebius, R.E. New insights into the development of lymphoid tissues. Nat. Rev. Immunol. 2010, 10, 664–674. [Google Scholar]
- Eberl, G.; Marmon, S.; Sunshine, M.J.; Rennert, P.D.; Choi, Y.; Littman, D.R. An essential function for the nuclear receptor RORgammat in the generation of fetal lymphoid tissue inducer cells. Nat. Immunol. 2004, 5, 64–73. [Google Scholar] [CrossRef] [PubMed]
- Sa, V.D.P.; Ferreira, M.; Domingues, R.G.; Ribeiro, H.; Molenaar, R.; Moreira-Santos, L.; Almeida, F.F.; Ibiza, S.; Barbosa, I.; Goverse, G.; et al. Maternal retinoids control type 3 innate lymphoid cells and set the offspring immunity. Nature 2014, 508, 123–127. [Google Scholar]
- Buonocore, S.; Ahern, P.P.; Uhlig, H.H.; Ivanov, I.I.; Littman, D.R.; Maloy, K.J.; Powrie, F. Innate lymphoid cells drive IL-23 dependent innate intestinal pathology. Nature 2010, 464, 1371–1375. [Google Scholar] [CrossRef] [PubMed]
- Geremia, A.; Arancibia-Cárcamo, C.V.; Fleming, M.P.; Rust, N.; Singh, B.; Mortensen, N.J.; Travis, S.P.; Powrie, F. IL-23-responsive innate lymphoid cells are increased in inflammatory bowel disease. J. Exp. Med. 2011, 208, 1127–1133. [Google Scholar] [CrossRef] [PubMed]
- Spencer, S.P.; Wilhelm, C.; Yang, Q.; Hall, J.A.; Bouladoux, N.; Boyd, A.; Nutman, T.B.; Urban, J.F., Jr.; Wang, J.; Ramalingam, T.R.; et al. Adaptation of innate lymphoid cells to a micronutrient deficiency promotes type 2 barrier immunity. Science 2014, 343, 432–437. [Google Scholar] [CrossRef] [PubMed]
- Mielke, L.A.; Jones, S.A.; Raverdeau, M.; Higgs, R.; Stefanska, A.; Groom, J.R.; Misiak, A.; Dungan, L.S.; Sutton, C.E.; Streubel, G.; et al. Retinoic acid expression associates with enhanced IL-22 production by γδ T cells and innate lymphoid cells and attenuation of intestinal inflammation. J. Exp. Med. 2013, 210, 1117–1124. [Google Scholar] [CrossRef] [PubMed]
- Mora, J.R.; von Andrian, U.H. T-cell homing specificity and plasticity: New concepts and future challenges. Trends Immunol. 2006, 27, 235–243. [Google Scholar] [CrossRef] [PubMed]
- Iwata, M.; Hirakiyama, A.; Eshima, Y.; Kagechika, H.; Kato, C.; Song, S.Y. Retinoic acid imprints gut-homing specificity on T cells. Immunity 2004, 21, 527–538. [Google Scholar] [CrossRef] [PubMed]
- Svensson, M.; Johansson-Lindbom, B.; Zapata, F.; Jaensson, E.; Austenaa, L.M.; Blomhoff, R.; Agace, W.W. Retinoic acid receptor signaling levels and antigen dose regulate gut homing receptor expression on CD8+ T cells. Mucosal. Immunol. 2008, 1, 38–48. [Google Scholar] [CrossRef] [PubMed]
- Hosoe, N.; Miura, S.; Watanabe, C.; Tsuzuki, Y.; Hokari, R.; Oyama, T.; Fujiyama, Y.; Nagata, H.; Ishii, H. Demonstration of functional role of TECK/CCL25 in T lymphocyte-endothelium interaction in inflamed and uninflamed intestinal mucosa. Am. J. Physiol. Gastrointest. Liver Physiol. 2004, 286, 458–466. [Google Scholar] [CrossRef] [PubMed]
- Denucci, C.C.; Pagán, A.J.; Mitchell, J.S.; Shimizu, Y. Control of α4β7 Integrin Expression and CD4 T Cell Homing by the β1 Integrin Subunit. J. Immunol. 2010, 184, 2458–2467. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.G.; Park, J.; Cho, J.Y.; Ulrich, B.; Kim, C.H. Complementary roles of retinoic acid and TGF-β1 in coordinated expression of mucosal integrins by T cells. Mucosal. Immunol. 2011, 4, 66–82. [Google Scholar] [CrossRef] [PubMed]
- Ohoka, Y.; Yokota, A.; Takeuchi, H.; Maeda, N.; Iwata, M. Retinoic acid-induced CCR9 expression requires transient TCR stimulation and cooperativity between NFATc2 and the retinoic acid receptor/retinoid X receptor complex. J. Immunol. 2011, 186, 733–744. [Google Scholar] [CrossRef] [PubMed]
- Tan, X.; Sande, J.L.; Pufnock, J.S.; Blattman, J.N.; Greenberg, P.D. Retinoic acid as a vaccine adjuvant enhances CD8+ T cell response and mucosal protection from viral challenge. J. Virol. 2011, 85, 8316–8327. [Google Scholar] [CrossRef] [PubMed]
- Allie, S.R.; Zhang, W.; Tsai, C.Y.; Noelle, R.J.; Usherwood, E.J. Critical role for all-trans retinoic acid for optimal effector and effector memory CD8 T cell differentiation. J. Immunol. 2013, 190, 2178–2187. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Pino-Lagos, K.; Ahonen, C.A.; Bennett, K.A.; Wang, J.; Napoli, J.L.; Blomhoff, R.; Sockanathan, S.; Chandraratna, R.A.; Dmitrovsky, E.; et al. A retinoic acid--rich tumor microenvironment provides clonal survival cues for tumor-specific CD8+ T cells. Cancer Res. 2012, 72, 5230–5239. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Lee, Y.C.; Brown, C.; Zhang, W.; Usherwood, E.; Noelle, R.J. Dissecting the Role of Retinoic Acid Receptor Isoforms in the CD8 Response to Infection. J. Immunol. 2014, 192, 3336–3344. [Google Scholar] [CrossRef] [PubMed]
- Pino-Lagos, K.; Guo, Y.; Brown, C.; Alexander, M.P.; Elgueta, R.; Bennett, K.A.; De Vries, V.; Nowak, E.; Blomhoff, R.; Sockanathan, S.; et al. A retinoic acid–dependent checkpoint in the development of CD4+ T cell–mediated immunity. J. Exp. Med. 2011, 208, 1767–1775. [Google Scholar] [CrossRef] [PubMed]
- Tao, X.J.; Ma, J.L.; Zhang, Y.H.; Yu, J.N.; Cai, L.; Wang, J.H.; Zheng, S.G. Neutralization of IL-4 and IFN-γ Facilitates inducing TGF-β-induced CD4+Foxp3+ Regulatory Cells. Int. J. Biomed. Sci. 2008, 4, 52–57. [Google Scholar] [PubMed]
- Zheng, S.G.; Gray, J.D.; Ohtsuka, K.; Yamagiwa, S.; Horwitz, D.A. Generation ex vivo of TGF-beta-producing regulatory T cells from CD4+CD25− precursors. J. Immunol. 2002, 169, 4183–4189. [Google Scholar] [CrossRef] [PubMed]
- Zheng, S.G.; Wang, J.H.; Gray, J.D.; Soucier, H.; Horwitz, D.A. Natural and induced CD4+CD25+ cells educate CD4+CD25− cells to develop suppressive activity: The role of IL-2, TGF-beta, and IL-10. J. Immunol. 2004, 172, 5213–5221. [Google Scholar] [CrossRef] [PubMed]
- Zheng, S.G.; Wang, J.; Wang, P.; Gray, J.D.; Horwitz, D.A. IL-2 is essential for TGF-beta to convert naive CD4+CD25− cells to CD25+Foxp3+ regulatory T cells and for expansion of these cells. J. Immunol. 2007, 178, 2018–2027. [Google Scholar] [CrossRef] [PubMed]
- Hori, S.; Nomura, T.; Sakaguchi, S. Control of Regulatory T Cell Development by the Transcription Factor Foxp3. Science 2003, 299, 1057–1061. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Liu, Y.; Li, Y.; Gu, J.; Liu, J.; Tang, J.; Wang, J.; Ryffel, B.; Shen, Y.; Brand, D.; et al. Differential role of all-trans retinoic acid in promoting the development of CD4+ and CD8+ regulatory T cells. J. Leukoc. Biol. 2014, 95, 275–283. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.M.; Wang, K.P.; Ma, J.; Zheng, S.G. The role of all-trans retinoic acid in the biology of Foxp3+ regulatory T cells. Cell Mol. Immunol. 2015, 12, 553–557. [Google Scholar] [CrossRef] [PubMed]
- Mucida, D.; Park, Y.; Kim, G.; Turovskaya, O.; Scott, I.; Kronenberg, M.; Cheroutre, H. Reciprocal th17 and regulatory t cell differentiation mediated by retinoic acid. Science 2007, 317, 256–260. [Google Scholar] [CrossRef] [PubMed]
- Schambach, F.; Schupp, M.; Lazar, M.A.; Reiner, S.L. Activation of retinoic acid receptor-α favours regulatory T cell induction at the expense of IL-17-secreting T helper cell differentiation. Eur. J. Immunol. 2007, 37, 2396–2399. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.; Ma, J.; Li, Z.; Lan, Q.; Chen, M.; Liu, Y.; Xia, Z.; Wang, J.; Han, Y.; Shi, W.; et al. All-Trans Retinoic Acid Promotes TGF-β-Induced Tregs via Histone Modification but Not DNA Demethylation on Foxp3 Gene Locus. PLoS ONE 2011, 6, e24590. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Kong, N.; Wang, J.; Fan, H.; Zou, H.; Horwitz, D.; Brand, D.; Liu, Z.; Zheng, S.G. Cutting edge: All-trans retinoic acid sustains the stability and function of natural regulatory T cells in an inflammatory milieu. J. Immunol. 2010, 185, 2675–2679. [Google Scholar] [CrossRef] [PubMed]
- Kwok, S.K.; Park, M.K.; Cho, M.L.; Oh, H.J.; Park, E.M.; Lee, D.G.; Lee, J.; Kim, H.Y.; Park, S.H. Retinoic acid attenuates rheumatoid inflammation in mice. J. Immunol. 2012, 189, 1062–1067. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.; Lan, Q.; Li, Z.; Zhou, X.; Gu, J.; Li, Q.; Wang, J.; Chen, M.; Liu, Y.; Shen, Y.; et al. Critical role of all-trans retinoic acid in stabilizing human natural regulatory T cells under inflammatory conditions. Proc. Natl. Acad. Sci. USA 2014, 111, 3432–3440. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.; Zhou, X.H.; Wang, J.L.; Zheng, S.G. Correction: Characterization of Protective Human CD4+CD25+ FOXP3+ Regulatory T Cells Generated with IL-2, TGF-β and Retinoic Acid. PLoS ONE 2010, 5, e15150. [Google Scholar] [CrossRef] [PubMed]
- Hammerschmidt, S.I.; Ahrendt, M.; Bode, U.; Wahl, B.; Kremmer, E.; Forster, R.; Pabst, O. Stromal mesenteric lymph node cells are essential for the generation of gut-homing T cells in vivo. J. Exp. Med. 2008, 205, 2483–2490. [Google Scholar] [CrossRef] [PubMed]
- Molenaar, R.; Greuter, M.; van der Marel, A.P.; Roozendaal, R.; Martin, S.F.; Edele, F.; Huehn, J.; Forster, R.; O’Toole, T.; Jansen, W.; et al. Lymph node stromal cells support dendritic cell-induced gut-homing of T cells. Immunology 2009, 183, 6395–6402. [Google Scholar] [CrossRef] [PubMed]
- Hall, J.A.; Cannons, J.L.; Grainger, J.R.; Dos Santos, L.M.; Hand, T.W.; Naik, S.; Wohlfert, E.A.; Chou, D.B.; Oldenhove, G.; Robinson, M.; et al. Essential role for retinoic acid in the promotion of CD4+ T cell effector responses via retinoic acid receptor alpha. Immunity 2011, 34, 435–447. [Google Scholar] [CrossRef] [PubMed]
- Xiao, S.; Jin, H.; Korn, T.; Liu, S.M.; Oukka, M.; Lim, B.; Kuchroo, V.K. Retinoic acid increases Foxp3+ regulatory T cells and inhibits development of Th17 cells by enhancing TGF-β-driven Smad3 signaling and inhibiting IL-6 and IL-23 receptor expression. Immunology 2008, 181, 2277–2284. [Google Scholar] [CrossRef]
- Rampal, R.; Awasthi, A.; Ahuja, V. Retinoic acid-primed human dendritic cells inhibit Th9 cells and induce Th1/Th17 cell differentiation. J. Leukoc. Biol. 2016, 100, 111–120. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, H.; Kanno, T.; Nakayamada, S.; Hirahara, K.; Sciume, G.; Muljo, S.A.; Kuchen, S.; Casellas, R.; Wei, L.; Kanno, Y.; et al. TGF-[beta] and retinoic acid induce the microRNA miR-10a, which targets Bcl-6 and constrains the plasticity of helper T cells. Nat. Immunol. 2012, 13, 587–595. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Kang, S.G.; HogenEsch, H.; Love, P.E.; Kim, C.H. Retinoic acid determines the precise tissue tropism of inflammatory Th17 cells in the intestine. J. Immunol. 2010, 184, 5519–5526. [Google Scholar] [CrossRef] [PubMed]
- Cha, H.R.; Chang, S.Y.; Chang, J.H.; Kim, J.O.; Yang, J.Y.; Kim, C.H.; Kweon, M.N. Downregulation of Th17 cells in the small intestine by disruption of gut flora in the absence of retinoic acid. Immunology 2010, 184, 6799–6806. [Google Scholar] [CrossRef] [PubMed]
- Raverdeau, M.; Mills, K.H. Modulation of T cell and innate immune responses by retinoic Acid. J. Immunol. 2014, 192, 2953–2958. [Google Scholar] [CrossRef] [PubMed]
- Ghodratizadeh, S.; Kanbak, G.; Beyramzadeh, M.; Dikmen, Z.G.; Memarzadeh, S.; Habibian, R. Effect of carotenoid β-cryptoxanthin on cellular and humoral immune response in rabbit. Vet. Res. Commun. 2014, 38, 59–62. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Yuan, Y.; Tao, Y.; Wang, W. Effects of vitamin A deficiency on mucosal immunity and response to intestinal infection in rats. Nutrition 2011, 27, 227–232. [Google Scholar] [CrossRef] [PubMed]
- Mora, J.R.; Iwata, M.; Eksteen, B.; Song, S.Y.; Junt, T.; Senman, B.; Otipoby, K.L.; Yokota, A.; Takeuchi, H.; Ricciardi-Castagnoli, P.; et al. Generation of gut-homing IgA-secreting B cells by intestinal dendritic cells. Science 2006, 314, 1157–1160. [Google Scholar] [CrossRef] [PubMed]
- Pantazi, E.; Marks, E.; Stolarczyk, E.; Lycke, N.; Noelle, R.J.; Elgueta, R. Cutting Edge: Retinoic Acid Signaling in B Cells Is Essential for Oral Immunization and Microflora Composition. J. Immunol. 2015, 195, 1368–1371. [Google Scholar] [CrossRef] [PubMed]
- Xu, A.; Liu, Y.; Chen, W.; Wang, J.; Xue, Y.; Huang, F.; Rong, L.; Lin, J.; Liu, D.; Yan, M.; et al. TGF-β-induced regulatory T cells directly suppress B cell responses through a non-cytotoxic mechanism. J. Immunol. 2016, 196, 3631–3641. [Google Scholar] [CrossRef] [PubMed]
- Heine, G.; Hollstein, T.; Treptow, S.; Radbruch, A.; Worm, M. 9-cis retinoic acid modulates the type I allergic immune response. J. Allergy. Clin. Immunol. 2018, 141, 650–658. [Google Scholar] [CrossRef] [PubMed]
- Seo, G.Y.; Lee, J.M.; Jang, Y.S.; Kang, S.G.; Yoon, S.I.; Ko, H.J.; Lee, G.S.; Park, S.R.; Nagler, C.R.; Kim, P.H. Mechanism underlying the suppressor activity of retinoic acid on IL4-induced IgE synthesis and its physiological implication. Cell Immunol. 2017, 322, 49–55. [Google Scholar] [CrossRef] [PubMed]
- Dilillo, D.J.; Matsushita, T.; Tedder, T.F. B10 cells and regulatory B cells balance immune responses during inflammation, autoimmunity, and cancer. Ann. N. Y. Acad. Sci. 2010, 1183, 38–57. [Google Scholar] [CrossRef] [PubMed]
- Yanaba, K.; Bouaziz, J.D.; Haas, K.M.; Poe, J.C.; Fujimoto, M.; Tedder, T.F. A regulatory B cell subset with a unique CD1dhi CD5+ phenotype controls T cell-dependent inflammatory responses. Immunity 2008, 28, 639–650. [Google Scholar] [CrossRef] [PubMed]
- Yanaba, K.; Bouaziz, J.D.; Matsushita, T.; Tsubata, T.; Tedder, T.F. The Development and Function of Regulatory B Cells Expressing IL-10 (B10 cells) Requires Antigen Receptor Diversity and TLR Signals. J. Immunol. 2009, 182, 7459–7472. [Google Scholar] [CrossRef] [PubMed]
- Wolf, S.D.; Dittel, B.N.; Hardardottir, F.; Janeway, C.A., Jr. Experimental autoimmune encephalomyelitis induction in genetically B cell-deficient mice. J. Exp. Med. 1996, 184, 2271–2278. [Google Scholar] [CrossRef] [PubMed]
- Fillatreau, S.; Sweenie, C.H.; McGeachy, M.J.; Gray, D.; Anderton, S.M. B cells regulate autoimmunity by provision of IL-10. Nat. Immunol. 2002, 3, 944–950. [Google Scholar] [CrossRef] [PubMed]
- Di Caro, V.; Phillips, B.; Engman, C.; Harnaha, J.; Trucco, M.; Giannoukakis, N. Retinoic acid-producing, ex-vivo-generated human tolerogenic dendritic cells induce the proliferation of immunosuppressive B lymphocytes. Clin. Exp. Immunol. 2013, 174, 302–317. [Google Scholar] [CrossRef] [PubMed]
- Iwata, Y.; Matsushita, T.; Horikawa, M.; Dilillo, D.J.; Yanaba, K.; Venturi, G.M.; Szabolcs, P.M.; Bernstein, S.H.; Magro, C.M.; Williams, A.D.; et al. Characterization of a rare IL-10-competent B-cell subset in humans that parallels mouse regulatory B10 cells. Blood 2011, 117, 530–541. [Google Scholar] [CrossRef] [PubMed]
- Mauri, C.; Gray, D.; Mushtaq, N.; Londei, M. Prevention of arthritis by interleukin 10-producing B cells. J. Exp. Med. 2003, 197, 489–501. [Google Scholar] [CrossRef] [PubMed]
- Blair, P.A.; Noreña, L.Y.; Flores-Borja, F.; Rawlings, D.J.; Isenberg, D.A.; Ehrenstein, M.R.; Mauri, C. CD19+ CD24hi CD38hi B cells exhibit regulatory capacity in healthy individuals but are functionally impaired in systemic Lupus Erythematosus patients. Immunity 2010, 32, 129–140. [Google Scholar] [CrossRef] [PubMed]
- Ramachandran, G.; Santha, T.; Garg, R.; Baskaran, D.; Iliayas, S.A.; Venkatesan, P.; Fathima, R.; Narayanan, P.R. Vitamin A levels in sputum-positive pulmonary tuberculosis patients in comparison with household contacts and healthy ‘normals’. Int. J. Tuberc. Lung Dis. 2004, 8, 1130–1133. [Google Scholar] [PubMed]
- Mugusi, F.M.; Rusizoka, O.; Habib, N.; Fawzi, W. Vitamin A status of patients presenting with pulmonary tuberculosis and asymptomatic HIV-infected individuals, Dar es Salaam, Tanzania. Int. J. Tuberc. Lung Dis. 2003, 7, 804–807. [Google Scholar] [PubMed]
- Qrafli, M.; El Kari, K.; Aguenaou, H.; Bourkadi, J.E.; Sadki, K.; El Mzibri, M. Low plasma vitamin A concentration is associated with tuberculosis in Moroccan population: A preliminary case control study. BMC Res. Notes 2017, 10, 421. [Google Scholar] [CrossRef] [PubMed]
- Aibana, O.; Franke, M.F.; Huang, C.C.; Galea, J.T.; Calderon, R.; Zhang, Z.; Becerra, M.C.; Smith, E.R.; Ronnenberg, A.G.; Contreras, C.; et al. Impact of Vitamin A and Carotenoids on the Risk of Tuberculosis Progression. Clin. Infect. Dis. 2017, 65, 900–909. [Google Scholar] [CrossRef] [PubMed]
- Anand, P.K.; Kaul, D.; Sharma, M. Synergistic action of vitamin D and retinoic acid restricts invasion of macrophages by pathogenic mycobacteria. J. Microbiol. Immunol. Infect. 2008, 41, 17–25. [Google Scholar] [PubMed]
- Wheelwright, M.; Kim, E.W.; Inkeles, M.S.; De Leon, A.; Pellegrini, M.; Krutzik, S.R.; Liu, P.T. All-trans retinoic acid triggered antimicrobial activity against Mycobacterium tuberculosis is dependent on NPC2. J. Immunol. 2014, 192, 2280–2290. [Google Scholar] [CrossRef] [PubMed]
- Mourik, B.C.; Leenen, P.J.; de Knegt, G.J.; Huizinga, R.; van der Eerden, B.C.; Wang, J.; Krois, C.R.; Napoli, J.L.; Bakker-Woudenberg, I.A.; de Steenwinkel, J.E. Immunotherapy added to antibiotic treatment reduces relapse of disease in a mouse model of tuberculosis. Am. J. Respir. Cell Mol. Biol. 2017, 56, 233–241. [Google Scholar] [CrossRef] [PubMed]
- Drain, P.K.; Kupka, R.; Mugusi, F.; Fawzi, W.W. Micronutrients in HIV-positive persons receiving highly active antiretroviral therapy. Am. J. Clin. Nutr. 2007, 85, 333–345. [Google Scholar] [CrossRef] [PubMed]
- Chin, J. The AIDS Pandemic: The collision of epidemiology with political correctness. Drugs Alcohol. Today 2007, 7, 46–47. [Google Scholar] [CrossRef]
- Schreck, R.; Rieber, P.; Baeuerle, P.A. Reactive oxygen intermediates as apparently widely used messengers in the activation of the NF-kappa B transcription factor and HIV-1. EMBO J. 1991, 10, 2247–2258. [Google Scholar] [PubMed]
- Makinde, O.; Rotimi, K.; Ikumawoyi, V.; Adeyemo, T.; Olayemi, S. Effect of vitamin A and vitamin C supplementation on oxidative stress in HIV and HIV-TB co-infection at Lagos University Teaching Hospital (LUTH) Nigeria. Afr. Health Sci. 2017, 17, 308–314. [Google Scholar] [CrossRef] [PubMed]
- Visser, M.E.; Durao, S.; Sinclair, D.; Irlam, J.H.; Siegfried, N. Micronutrient supplementation in adults with HIV infection. Cochrane Database Syst. Rev. 2017, 5, CD003650. [Google Scholar] [CrossRef] [PubMed]
- Wiysonge, C.S.; Ndze, V.N.; Kongnyuy, E.J.; Shey, M.S. Vitamin A supplements for reducing mother-to-child HIV transmission. Cochrane Database Syst. Rev. 2017, 9, CD003648. [Google Scholar] [CrossRef] [PubMed]
- Campa, A.; Baum, M.K.; Bussmann, H.; Martinez, S.S.; Farahani, M.; van Widenfelt, E.; Moyo, S.; Makhema, J.; Essex, M.; Marlink, R. The effect of micronutrient supplementation on active TB incidence early in HIV infection in Botswana. Nutr. Diet. Suppl. 2017, 2017, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Fawzi, W.W.; Msamanga, G.I.; Kupka, R.; Spiegelman, D.; Villamor, E.; Mugusi, F.; Wei, R.; Hunter, D. Multivitamin supplementation improves hematologic status in HIV-infected women and their children in Tanzania. Am. J. Clin. Nutr. 2007, 85, 1335–1343. [Google Scholar] [CrossRef] [PubMed]
- Shivakoti, R.; Christian, P.; Yang, W.T.; Gupte, N.; Mwelase, N.; Kanyama, C.; Pillay, S.; Samaneka, W.; Santos, B.; Poongulali, S.; et al. Prevalence and risk factors of micronutrient deficiencies pre- and post-antiretroviral therapy (ART) among a diverse multicountry cohort of HIV-infected adults. Clin. Nutr. 2016, 35, 183–189. [Google Scholar] [CrossRef] [PubMed]
- Rytter, M.J.; Kolte, L.; Briend, A.; Friis, H.; Christensen, V.B. The Immune System in Children with Malnutrition—A Systematic Review. PLoS ONE 2014, 9, e105017. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; Zhang, X.; Li, T.Y.; Chen, L.; Qu, P.; Liu, Y.X. Co-assessment of iron, vitamin A and growth status to investigate anemia in preschool children in suburb Chongqing, China. World J. Pediatr. 2009, 5, 275–281. [Google Scholar] [CrossRef] [PubMed]
- Semba, R.D.; de Pee, S.; Sun, K.; Campbell, A.A.; Bloem, M.W.; Raju, V.K. Low intake of vitamin A-rich foods among children, aged 12–35 months, in India: Association with malnutrition, anemia, and missed child survival interventions. Nutrition 2010, 26, 958–962. [Google Scholar] [CrossRef] [PubMed]
- Sivakumar, B.; Reddy, V. Absorption of labelled vitamin A in children during infection. Br. J. Nutr. 1972, 27, 299–304. [Google Scholar] [CrossRef] [PubMed]
- Sivakumar, B.; Reddy, V. Absorption of vitamin A in children with ascariasis. J. Trop. Med. Hyg. 1975, 78, 114–115. [Google Scholar] [PubMed]
- Stephensen, C.B. Vitamin A, infection, and immune function. Annu. Rev. Nutr. 2001, 21, 167–192. [Google Scholar] [CrossRef] [PubMed]
- Nan, H.U.; Qu-Bei, L.I.; Zou, S.Y. Effect of vitamin A as an adjuvant therapy for pneumonia in children: A meta analysis. Chin. J. Contemp. Pediatr. 2018, 20, 146–153. [Google Scholar]
- Mayo-Wilson, E.; Imdad, A.; Herzer, K.; Yakoob, M.Y.; Bhutta, Z.A. Vitamin A supplements for preventing mortality, illness, and blindness in children aged under 5: Systematic review and meta-analysis. BMJ 2011, 343, d5094. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Yang, Y.; Yan, X.; Chen, J.; Yu, H.; Wang, W. Influence of vitamin A status on the antiviral immunity of children with hand, foot and mouth disease. Clin. Nutr. 2012, 31, 543–548. [Google Scholar] [CrossRef] [PubMed]
- Fisker, A.B.; Bale, C.; Rodrigues, A.; Balde, I.; Fernandes, M.; Jørgensen, M.J.; Danneskiold-Samsøe, N.; Hornshøj, L.; Rasmussen, J.; Christensen, E.D.; et al. High-dose Vitamin A with Vaccination after 6 Months of Age: A Randomized Trial. Pediatrics 2014, 134, 739–748. [Google Scholar] [CrossRef] [PubMed]
- Beare-Rogers, J.L. Recommended Nutrient Intakes for Canadians. Dept. Natl. Health Welf. 1983, 48–50. [Google Scholar] [CrossRef]
- Oliver, T.K. Chronic vitamin A intoxication. Am. J. Dis. Child. 1953, 95, 57–68. [Google Scholar] [CrossRef]
- Bush, M.E.; Dahms, B.B. Fatal hypervitaminosis A in a neonate. Arch. Pathol. Lab Med. 1984, 108, 838–842. [Google Scholar] [PubMed]
- James, M.B.; Leonard, J.C.; Fraser, J.J., Jr.; Stuemky, J.H. Hypervitaminosis A: A case report. Pediatrics 1982, 69, 112–115. [Google Scholar] [PubMed]
- Lippe, B.; Hensen, L.; Mendoza, G.; Finerman, M.; Welch, M. Chronic vitamin A intoxication: A multisystem disease that could reach epidemic proportions. Am. J. Dis. Child. 1981, 135, 634–636. [Google Scholar] [CrossRef]
- Rubin, L.P.; Ross, A.C.; Stephensen, C.B.; Bohn, T.; Tanumihardjo, S.A. Metabolic effects of inflammation on vitamin A and carotenoids in humans and animal models. Adv. Nutr. 2017, 8, 197–212. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.M.; Mao, M.; Wan, C. Vitamin A for treating measles in children. Cochrane Database Syst. Rev. 2005, 5, 85–86. [Google Scholar]
- Sudfeld, C.R.; Marie, N.A.; Halsey, N.A. Effectiveness of measles vaccination and vitamin A treatment. Int. J. Epidemiol. 2010, 39, i48–i55. [Google Scholar] [CrossRef] [PubMed]
- Bhandari, N.; Bhan, M.K.; Sazawal, S. Impact of massive dose of vitamin A given to preschool children with acute diarrhoea on subsequent respiratory and diarrhoeal morbidity. BMJ 1994, 309, 1404–1407. [Google Scholar] [CrossRef] [PubMed]
- Cui, D.; Moldoveanu, Z.; Stephensen, C.B. High-level dietary vitamin A enhances T-helper type 2 cytokine production and secretory immunoglobulin A response to influenza A virus infection in BALB/c mice. J. Nutr. 2000, 130, 1132–1139. [Google Scholar] [CrossRef] [PubMed]
- Nikawa, T.; Odahara, K.; Koizumi, H.; Kido, Y.; Teshima, S.; Rokutan, K.; Kishi, K. Vitamin A prevents the decline in immunoglobulin A and Th2 cytokine levels in small intestinal mucosa of protein-malnourished mice. J. Nutr. 1999, 129, 934–941. [Google Scholar] [CrossRef] [PubMed]
- McDaniel, K.L.; Restori, K.H.; Dodds, J.W.; Kennett, M.J.; Ross, A.C.; Cantorna, M.T. Vitamin A-deficient hosts become nonsymptomatic reservoirs of escherichia coli-like enteric infections. Infect. Immun. 2015, 83, 2984–2991. [Google Scholar] [CrossRef] [PubMed]
- Elom, M.O.; Okafor, F.C.; Eyo, J.E. Vitamin A supplementation of malaria-infected pregnant women and infant birth weight outcomes a case study of Ebonyi State, Nigeria. Gastro 2014, 2, 109. [Google Scholar]
- Owusu-Agyei, S.; Newton, S.; Mahama, E.; Febir, L.G.; Ali, M.; Adjei, K.; Tchum, K.; Alhassan, L.; Moleah, T.; Tanumihardjo, S.A. Impact of vitamin A with zinc supplementation on malaria morbidity in Ghana. Nutr. J. 2013, 12, 131. [Google Scholar] [CrossRef] [PubMed]
- Zeba, A.N.; Sorgho, H.; Rouamba, N.; Zongo, I.; Rouamba, J.; Guiguemdé, R.T.; Hamer, D.H.; Mokhtar, N.; Ouedraogo, J.B. Major reduction of malaria morbidity with combined vitamin A and zinc supplementation in young children in Burkina Faso: A randomized double blind trial. Nutr. J. 2008, 7, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Soye, K.J.; Trottier, C.; Di Lenardo, T.Z.; Restori, K.H.; Reichman, L.; Miller, W.H., Jr.; Ward, B.J. In vitro inhibition of mumps virus by retinoids. Virol. J. 2013, 10, 337. [Google Scholar] [CrossRef] [PubMed]
| Diseases | Role of VitA | Method Setting | Model [Reference] |
|---|---|---|---|
| Measles | Reduce mortality | Meta-analysis | Human [138] |
| Measles | Reduce morbidity and mortality | Systematic review and meta-analysis | Human [129] |
| Measles | Reduce mortality | Meta-analysis | Human [139] |
| Measles | Reduce morbidity | Randomized double-blind controlled trial | Human [140] |
| Acute pneumonia | Promoting the production of specific antibodies | Randomized controlled trial | Mice [141] |
| Acute pneumonia | Relieving clinical symptoms and signs | Meta-analysis | Human [128] |
| Infantile diarrhea | Reduce morbidity and mortality | Systematic review and meta-analysis | Human [129] |
| Infantile diarrhea | Promote the production of IgA in the intestinal tract and enhance the mucosal immune function | Randomized controlled trial | Mice [142] |
| Infantile diarrhea | Reduce morbidity | Randomized double-blind controlled trial | Human [140] |
| Enteric infection | Reduce morbidity and mortality | Randomized controlled trial | Mice [143] |
| Malaria | Reduce morbidity | Randomized double-blind controlled trial | Human [144] |
| Malaria | Reduce morbidity | Randomized controlled trial | Human [145] |
| Malaria | Reduce morbidity | Randomized double-blind controlled trial | Human [146] |
| Hand foot and mouth disease | Promote production of immunoglobulin and enhance antiviral function | Cross-sectional observation and study | Human [130] |
| Mumps | Up-regulation of type 1 interferon and inhibition of viral replication | In vitro controlled experiment | Cells [147] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, Z.; Liu, Y.; Qi, G.; Brand, D.; Zheng, S.G. Role of Vitamin A in the Immune System. J. Clin. Med. 2018, 7, 258. https://doi.org/10.3390/jcm7090258
Huang Z, Liu Y, Qi G, Brand D, Zheng SG. Role of Vitamin A in the Immune System. Journal of Clinical Medicine. 2018; 7(9):258. https://doi.org/10.3390/jcm7090258
Chicago/Turabian StyleHuang, Zhiyi, Yu Liu, Guangying Qi, David Brand, and Song Guo Zheng. 2018. "Role of Vitamin A in the Immune System" Journal of Clinical Medicine 7, no. 9: 258. https://doi.org/10.3390/jcm7090258
APA StyleHuang, Z., Liu, Y., Qi, G., Brand, D., & Zheng, S. G. (2018). Role of Vitamin A in the Immune System. Journal of Clinical Medicine, 7(9), 258. https://doi.org/10.3390/jcm7090258





