The Emperor Has No Clothes? Searching for Dysregulation in Sepsis
Abstract
1. Introduction
“We Have Met the Enemy and He Is Us”—Walt Kelly
2. Dysregulation in Sepsis versus Function
3. The Failure of Immunomodulators in Sepsis
3.1. Lipopolysaccharide/TLR4 Pathway
3.2. Recombinant Activated Protein C
3.3. Statins
3.4. Corticosteroids
4. Normalization of Sepsis Physiology
4.1. Fever
4.2. Elevated Blood Sugar
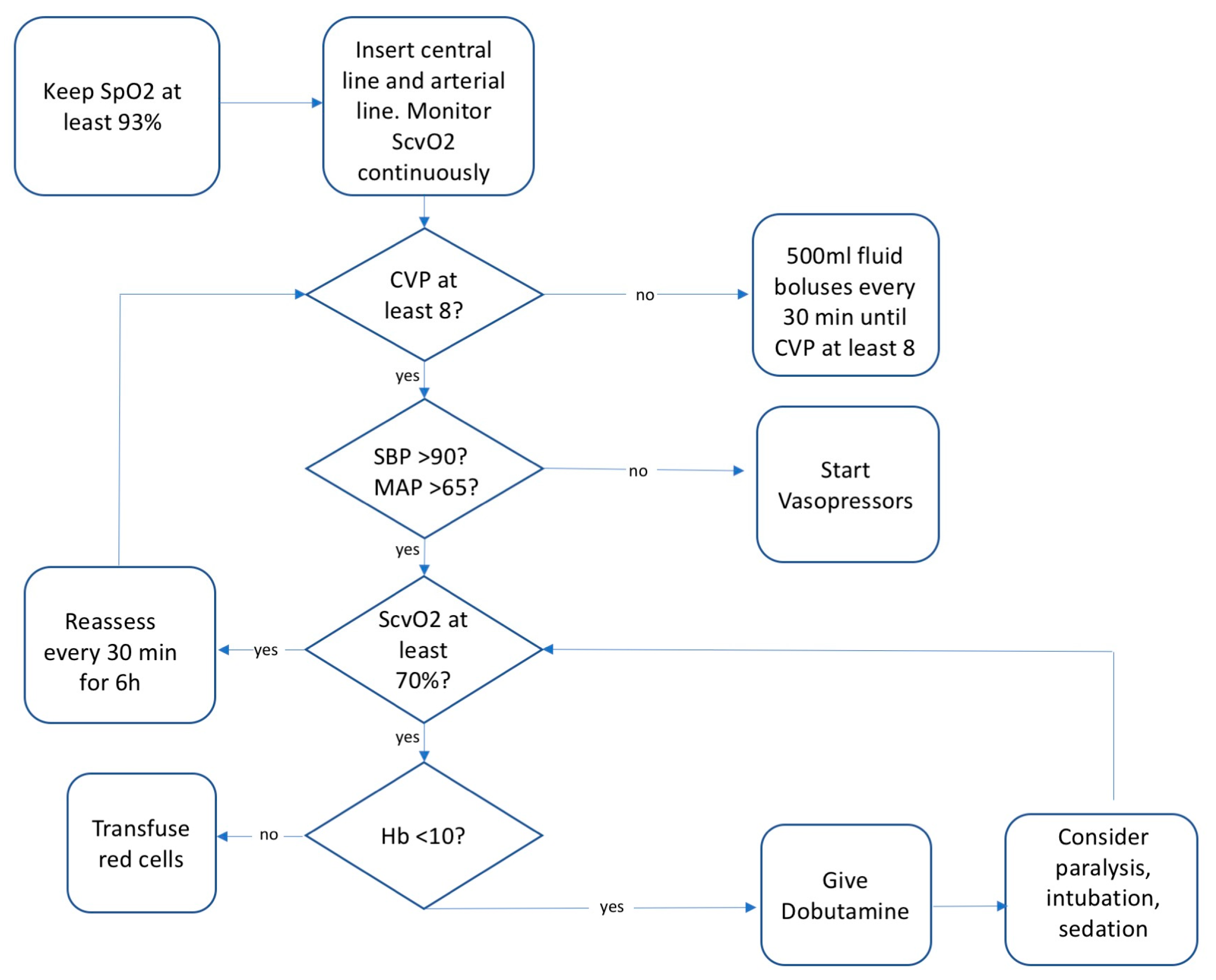
4.3. Sepsis Bundles and Early Goal Directed Therapy
4.4. Fluid Therapy
4.5. Organ Failure
4.6. Interventions with No Verdict Yet
5. Regulation and Adaptation in Sepsis
Funding
Acknowledgments
Conflicts of Interest
References
- Thomas, L. Germs. N. Engl. J. Med. 1972, 287, 553–555. [Google Scholar] [CrossRef] [PubMed]
- Singer, M.; Deutschman, C.S.; Seymour, C.W.; Shankar-Hari, M.; Annane, D.; Bauer, M.; Bellomo, R.; Bernard, G.R.; Chiche, J.-D.; Coopersmith, C.M.; et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA 2016, 315, 801–810. [Google Scholar] [CrossRef] [PubMed]
- Bernard, A.M.; Bernard, G.R. The immune response: Targets for the treatment of severe sepsis. Int. J. Inflamm. 2012, 2012, 697592. [Google Scholar] [CrossRef] [PubMed]
- Alverdy, J.C.; Krezalek, M.A. Collapse of the microbiome, emergence of the pathobiome and the immunopathology of sepsis. Crit. Care Med. 2017, 45, 337. [Google Scholar] [CrossRef] [PubMed]
- Bernard, G.R.; Vincent, J.-L.; Laterre, P.-F.; LaRosa, S.P.; Dhainaut, J.-F.; Lopez-Rodriguez, A.; Steingrub, J.S.; Garber, G.E.; Helterbrand, J.D.; Ely, E.W. Efficacy and safety of recombinant human activated protein C for severe sepsis. N. Engl. J. Med. 2001, 344, 699–709. [Google Scholar] [CrossRef] [PubMed]
- Ranieri, V.M.; Thompson, B.T.; Barie, P.S.; Dhainaut, J.-F.; Douglas, I.S.; Finfer, S.; Gårdlund, B.; Marshall, J.C.; Rhodes, A.; Artigas, A. Drotrecogin alfa (activated) in adults with septic shock. N. Engl. J. Med. 2012, 366, 2055–2064. [Google Scholar] [CrossRef] [PubMed]
- Eichacker, P.Q.; Parent, C.; Kalil, A.; Esposito, C.; Cui, X.; Banks, S.M.; Gerstenberger, E.P.; Fitz, Y.; Danner, R.L.; Natanson, C. Risk and the efficacy of antiinflammatory agents: Retrospective and confirmatory studies of sepsis. Am. J. Respir. Crit. Care Med. 2002, 166, 1197–1205. [Google Scholar] [CrossRef] [PubMed]
- Martin-Loeches, I.; Muriel-Bombín, A.; Ferrer, R.; Artigas, A.; Sole-Violan, J.; Lorente, L.; Andaluz-Ojeda, D.; Prina-Mello, A.; Herrán-Monge, R.; Suberviola, B.; et al. GRECIA group The protective association of endogenous immunoglobulins against sepsis mortality is restricted to patients with moderate organ failure. Ann. Intensive Care 2017, 7, 44. [Google Scholar] [CrossRef] [PubMed]
- Van der Poll, T.; van de Veerdonk, F.L.; Scicluna, B.P.; Netea, M.G. The immunopathology of sepsis and potential therapeutic targets. Nat. Rev. Immunol. 2017, 17, 407–420. [Google Scholar] [CrossRef] [PubMed]
- Seymour, C.W.; Gesten, F.; Prescott, H.C.; Friedrich, M.E.; Iwashyna, T.J.; Phillips, G.S.; Lemeshow, S.; Osborn, T.; Terry, K.M.; Levy, M.M. Time to Treatment and Mortality during Mandated Emergency Care for Sepsis. N. Engl. J. Med. 2017, 376, 2235–2244. [Google Scholar] [CrossRef] [PubMed]
- Hotchkiss, R.S.; Monneret, G.; Payen, D. Sepsis-induced immunosuppression: From cellular dysfunctions to immunotherapy. Nat. Rev. Immunol. 2013, 13, 862–874. [Google Scholar] [CrossRef] [PubMed]
- Feuerecker, M.; Sudhoff, L.; Crucian, B.; Pagel, J.-I.; Sams, C.; Strewe, C.; Guo, A.; Schelling, G.; Briegel, J.; Kaufmann, I.; et al. Early immune anergy towards recall antigens and mitogens in patients at onset of septic shock. Sci. Rep. 2018, 8, 1754. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J.; Vincent, J.-L.; Adhikari, N.K.; Machado, F.R.; Angus, D.C.; Calandra, T.; Jaton, K.; Giulieri, S.; Delaloye, J.; Opal, S. Sepsis: A roadmap for future research. Lancet Infect. Dis. 2015, 15, 581–614. [Google Scholar] [CrossRef]
- Cohen, J. The immunopathogenesis of sepsis. Nature 2002, 420, 885–891. [Google Scholar] [CrossRef] [PubMed]
- Pinsky, M.R. Dysregulation of the immune response in severe sepsis. Am. J. Med. Sci. 2004, 328, 220–229. [Google Scholar] [CrossRef] [PubMed]
- Marshall, J.C. Why have clinical trials in sepsis failed? Trends Mol. Med. 2014, 20, 195–203. [Google Scholar] [CrossRef] [PubMed]
- Prasad, V.; Vandross, A.; Toomey, C.; Cheung, M.; Rho, J.; Quinn, S.; Chacko, S.J.; Borkar, D.; Gall, V.; Selvaraj, S. A decade of reversal: An analysis of 146 contradicted medical practices. Mayo Clin. Proc. 2013, 88, 790–798. [Google Scholar] [CrossRef] [PubMed]
- Rivers, E.; Nguyen, B.; Havstad, S.; Ressler, J.; Muzzin, A.; Knoblich, B.; Peterson, E.; Tomlanovich, M. Early goal-directed therapy in the treatment of severe sepsis and septic shock. N. Engl. J. Med. 2001, 345, 1368–1377. [Google Scholar] [CrossRef] [PubMed]
- Berghe, G.V.D.; Wouters, P.; Weekers, F.; Verwaest, C.; Bruyninckx, F.; Schetz, M.; Vlasselaers, D.; Ferdinande, P.; Lauwers, P.; Bouillon, R. Intensive insulin therapy in critically ill patients. N. Engl. J. Med. 2001, 345, 1359–1367. [Google Scholar] [CrossRef] [PubMed]
- Beutler, B.; Milsark, I.W.; Cerami, A.C. Passive immunization against cachectin/tumor necrosis factor protects mice from lethal effect of endotoxin. Science 1985, 229, 869–871. [Google Scholar] [CrossRef] [PubMed]
- Opal, S.M.; Laterre, P.-F.; Francois, B.; LaRosa, S.P.; Angus, D.C.; Mira, J.-P.; Wittebole, X.; Dugernier, T.; Perrotin, D.; Tidswell, M. Effect of eritoran, an antagonist of MD2-TLR4, on mortality in patients with severe sepsis: The ACCESS randomized trial. JAMA 2013, 309, 1154–1162. [Google Scholar] [CrossRef] [PubMed]
- Fisher, C.J.; Yan, S.B. Protein C levels as a prognostic indicator of outcome in sepsis and related diseases. Crit. Care Med. 2000, 28, S49–S56. [Google Scholar]
- Eichacker, P.Q.; Natanson, C.; Danner, R.L. Surviving sepsis-practice guidelines, marketing campaigns, and Eli Lilly. N. Engl. J. Med. 2006, 355, 1640. [Google Scholar] [CrossRef] [PubMed]
- Abraham, E.; Laterre, P.-F.; Garg, R.; Levy, H.; Talwar, D.; Trzaskoma, B.L.; François, B.; Guy, J.S.; Brückmann, M.; Rea-Neto, A.; et al. Administration of Drotrecogin Alfa (Activated) in Early Stage Severe Sepsis (ADDRESS) Study Group Drotrecogin alfa (activated) for adults with severe sepsis and a low risk of death. N. Engl. J. Med. 2005, 353, 1332–1341. [Google Scholar] [CrossRef] [PubMed]
- Lai, P.S.; Matteau, A.; Iddriss, A.; Hawes, J.C.L.; Ranieri, V.; Thompson, B.T. An updated meta-analysis to understand the variable efficacy of drotrecogin alfa (activated) in severe sepsis and septic shock. Minerva Anestesiol. 2013, 79, 33–43. [Google Scholar] [PubMed]
- Warren, B.L.; Eid, A.; Singer, P.; Pillay, S.S.; Carl, P.; Novak, I.; Chalupa, P.; Atherstone, A.; Pénzes, I.; Kübler, A.; et al. KyberSept Trial Study Group Caring for the critically ill patient. High-dose antithrombin III in severe sepsis: A randomized controlled trial. JAMA 2001, 286, 1869–1878. [Google Scholar] [CrossRef] [PubMed]
- Fiusa, M.M.L.; Carvalho-Filho, M.A.; Annichino-Bizzacchi, J.M.; De Paula, E.V. Causes and consequences of coagulation activation in sepsis: An evolutionary medicine perspective. BMC Med. 2015, 13, 105. [Google Scholar] [CrossRef] [PubMed]
- National Heart, Lung, and Blood Institute ARDS Clinical Trials Network. Rosuvastatin for sepsis-associated acute respiratory distress syndrome. N. Engl. J. Med. 2014, 370, 2191–2200. [Google Scholar] [CrossRef] [PubMed]
- Annane, D.; Renault, A.; Brun-Buisson, C.; Megarbane, B.; Quenot, J.-P.; Siami, S.; Cariou, A.; Forceville, X.; Schwebel, C.; Martin, C.; et al. CRICS-TRIGGERSEP Network Hydrocortisone plus Fludrocortisone for Adults with Septic Shock. N. Engl. J. Med. 2018, 378, 809–818. [Google Scholar] [CrossRef] [PubMed]
- Rygård, S.L.; Butler, E.; Granholm, A.; Møller, M.H.; Cohen, J.; Finfer, S.; Perner, A.; Myburgh, J.; Venkatesh, B.; Delaney, A. Low-dose corticosteroids for adult patients with septic shock: A systematic review with meta-analysis and trial sequential analysis. Intensive Care Med. 2018. [Google Scholar] [CrossRef] [PubMed]
- Vincent, J.L.; Singer, M. Critical care: Advances and future perspectives. Lancet 2010, 376, 1354–1361. [Google Scholar] [CrossRef]
- Sundén-Cullberg, J.; Rylance, R.; Svefors, J.; Norrby-Teglund, A.; Björk, J.; Inghammar, M. Fever in the Emergency Department Predicts Survival of Patients With Severe Sepsis and Septic Shock Admitted to the ICU. Crit. Care Med. 2017, 45, 591–599. [Google Scholar] [CrossRef] [PubMed]
- Young, P.J.; Saxena, M.; Beasley, R.; Bellomo, R.; Bailey, M.; Pilcher, D.; Finfer, S.; Harrison, D.; Myburgh, J.; Rowan, K. Early peak temperature and mortality in critically ill patients with or without infection. Intensive Care Med. 2012. [Google Scholar] [CrossRef] [PubMed]
- Henriksen, D.P.; Havshøj, U.; Pedersen, P.B.; Laursen, C.B.; Jensen, H.K.; Brabrand, M.; Lassen, A.T. Hospitalized acute patients with fever and severe infection have lower mortality than patients with hypo- or normothermia: A follow-up study. QJM Mon. J. Assoc. Phys. 2016, 109, 473–479. [Google Scholar] [CrossRef] [PubMed]
- Filbin, M.R.; Lynch, J.; Gillingham, T.D.; Thorsen, J.E.; Pasakarnis, C.L.; Nepal, S.; Matsushima, M.; Rhee, C.; Heldt, T.; Reisner, A.T. Presenting Symptoms Independently Predict Mortality in Septic Shock: Importance of a Previously Unmeasured Confounder. Crit. Care Med. 2018. [Google Scholar] [CrossRef] [PubMed]
- Itenov, T.S.; Johansen, M.E.; Bestle, M.; Thormar, K.; Hein, L.; Gyldensted, L.; Lindhardt, A.; Christensen, H.; Estrup, S.; Pedersen, H.P.; et al. Cooling and Surviving Septic Shock (CASS) Trial Collaboration Induced hypothermia in patients with septic shock and respiratory failure (CASS): A randomised, controlled, open-label trial. Lancet Respir. Med. 2018, 6, 183–192. [Google Scholar] [CrossRef]
- Bernard, G.R.; Wheeler, A.P.; Russell, J.A.; Schein, R.; Summer, W.R.; Steinberg, K.P.; Fulkerson, W.J.; Wright, P.E.; Christman, B.W.; Dupont, W.D.; et al. The effects of ibuprofen on the physiology and survival of patients with sepsis. The Ibuprofen in Sepsis Study Group. N. Engl. J. Med. 1997, 336, 912–918. [Google Scholar] [CrossRef] [PubMed]
- Young, P.J.; Saxena, M.K.; Bellomo, R.; Freebairn, R.C.; Hammond, N.E.; van Haren, F.M.; Henderson, S.J.; McArthur, C.J.; McGuinness, S.P.; Mackle, D.; et al. The heat trial: A protocol for a multicentre randomised placebo-controlled trial of IV paracetamol in ICU patients with fever and infection. Crit. Care Resusc. 2012, 14, 290. [Google Scholar] [PubMed]
- Agus, M.S.D.; Wypij, D.; Hirshberg, E.L.; Srinivasan, V.; Faustino, E.V.; Luckett, P.M.; Alexander, J.L.; Asaro, L.A.; Curley, M.A.Q.; Steil, G.M.; et al. HALF-PINT Study Investigators and the PALISI Network Tight Glycemic Control in Critically Ill Children. N. Engl. J. Med. 2017, 376, 729–741. [Google Scholar] [CrossRef] [PubMed]
- Investigators, T.P. A Randomized Trial of Protocol-Based Care for Early Septic Shock. N. Engl. J. Med. 2014, 370, 1683–1693. [Google Scholar] [CrossRef] [PubMed]
- ARISE Investigators; ANZICS Clinical Trials Group; Peake, S.L.; Delaney, A.; Bailey, M.; Bellomo, R.; Cameron, P.A.; Cooper, D.J.; Higgins, A.M.; Holdgate, A.; et al. Goal-Directed Resuscitation for Patients with Early Septic Shock. N. Engl. J. Med. 2014, 371, 1496–1506. [Google Scholar] [CrossRef] [PubMed]
- Mouncey, P.R.; Osborn, T.M.; Power, G.S.; Harrison, D.A.; Sadique, M.Z.; Grieve, R.D.; Jahan, R.; Tan, J.C.K.; Harvey, S.E.; Bell, D.; et al. Protocolised Management In Sepsis (ProMISe): A multicentre randomised controlled trial of the clinical effectiveness and cost-effectiveness of early, goal-directed, protocolised resuscitation for emerging septic shock. Health Technol. Assess. Winch. Engl. 2015, 19, 1–150. [Google Scholar] [CrossRef] [PubMed]
- Marik, P.; Bellomo, R. A rational approach to fluid therapy in sepsis. Br. J. Anaesth. 2016, 116, 339–349. [Google Scholar] [CrossRef] [PubMed]
- Farkas, J. Available online: https://emcrit.org/pulmcrit/ssc-petition/ (accessed on 28 August 2018).
- Maitland, K.; Kiguli, S.; Opoka, R.O.; Engoru, C.; Olupot-Olupot, P.; Akech, S.O.; Nyeko, R.; Mtove, G.; Reyburn, H.; Lang, T.; et al. Mortality after Fluid Bolus in African Children with Severe Infection. N. Engl. J. Med. 2011, 364, 2483–2495. [Google Scholar] [CrossRef] [PubMed]
- Andrews, B.; Semler, M.W.; Muchemwa, L.; Kelly, P.; Lakhi, S.; Heimburger, D.C.; Mabula, C.; Bwalya, M.; Bernard, G.R. Effect of an Early Resuscitation Protocol on In-hospital Mortality Among Adults With Sepsis and Hypotension: A Randomized Clinical Trial. JAMA 2017, 318, 1233–1240. [Google Scholar] [CrossRef] [PubMed]
- Bernheim, H.A.; Kluger, M.J. Fever: Effect of drug-induced antipyresis on survival. Science 1976, 193, 237–239. [Google Scholar] [CrossRef] [PubMed]
- Kathryn Maitland: Asking the Difficult Questions—The Lancet Child & Adolescent Health. Available online: https://www.thelancet.com/journals/lanchi/article/PIIS2352-4642(17)30075-5/abstract (accessed on 17 July 2018).
- Mongardon, N.; Dyson, A.; Singer, M. Is MOF an outcome parameter or a transient, adaptive state in critical illness? Curr. Opin. Crit. Care 2009, 15, 431–436. [Google Scholar] [CrossRef] [PubMed]
- Shapiro, N.; Howell, M.D.; Bates, D.W.; Angus, D.C.; Ngo, L.; Talmor, D. The Association of Sepsis Syndrome and Organ Dysfunction with Mortality in Emergency Department Patients with Suspected Infection. Ann. Emerg. Med. 2006, 48, 583–590. [Google Scholar] [CrossRef] [PubMed]
- Hoste, E.A.J.; Bagshaw, S.M.; Bellomo, R.; Cely, C.M.; Colman, R.; Cruz, D.N.; Edipidis, K.; Forni, L.G.; Gomersall, C.D.; Govil, D.; et al. Epidemiology of acute kidney injury in critically ill patients: The multinational AKI-EPI study. Intensive Care Med. 2015, 41, 1411–1423. [Google Scholar] [CrossRef] [PubMed]
- Kosaka, J.; Lankadeva, Y.R.; May, C.N.; Bellomo, R. Histopathology of Septic Acute Kidney Injury: A Systematic Review of Experimental Data. Crit. Care Med. 2016, 44, e897–e903. [Google Scholar] [CrossRef] [PubMed]
- Maiden, M.J.; Otto, S.; Brealey, J.K.; Finnis, M.E.; Chapman, M.J.; Kuchel, T.R.; Nash, C.H.; Edwards, J.; Bellomo, R. Structure and Function of the Kidney in Septic Shock. A Prospective Controlled Experimental Study. Am. J. Respir. Crit. Care Med. 2016, 194, 692–700. [Google Scholar] [CrossRef] [PubMed]
- Karakala, N.; Tolwani, A.J. Timing of Renal Replacement Therapy for Acute Kidney Injury. J. Intensive Care Med. 2018, 885066618774257. [Google Scholar] [CrossRef] [PubMed]
- Marik, P.E.; Khangoora, V.; Rivera, R.; Hooper, M.H.; Catravas, J. Hydrocortisone, Vitamin C, and Thiamine for the Treatment of Severe Sepsis and Septic Shock: A Retrospective Before-After Study. Chest 2017, 151, 1229–1238. [Google Scholar] [CrossRef] [PubMed]
- Oudemans-van Straaten, H.M.; Man, A.M.S.; de Waard, M.C. Vitamin C revisited. Crit. Care 2014, 18, 460. [Google Scholar] [CrossRef] [PubMed]
- Aberegg, S.K. Ionized Calcium in the ICU: Should It Be Measured and Corrected? Chest 2016, 149, 846–855. [Google Scholar] [CrossRef] [PubMed]
- Kallio, J.; Jaakkola, M.; Mäki, M.; Kilpeläinen, P.; Virtanen, V. Vitamin C inhibits staphylococcus aureus growth and enhances the inhibitory effect of quercetin on growth of Escherichia coli in vitro. Planta Med. 2012, 78, 1824–1830. [Google Scholar] [CrossRef] [PubMed]
- Meyer, N.; Harhay, M.O.; Small, D.S.; Prescott, H.C.; Bowles, K.H.; Gaieski, D.F.; Mikkelsen, M.E. Temporal Trends in Incidence, Sepsis-Related Mortality, and Hospital-Based Acute Care After Sepsis. Crit. Care Med. 2018, 46, 354–360. [Google Scholar] [CrossRef] [PubMed]
- Nesse, R.M.; Williams, G.C. Why We Get Sick? The New Science of Darwinian Medicine; Times Books: New York, NY, USA, 1994. [Google Scholar]
- Williams, G.C. Adaptation and Natural Selection: A Critique of Some Current Evolutionary Thought; Princeton University Press: Princeton, NJ, USA, 2008; ISBN 978-1-4008-2010-8. [Google Scholar]
- Ellison, P.T.; Jasienska, G. Constraint, pathology, and adaptation: How can we tell them apart? Am. J. Hum. Biol. 2007, 19, 622–630. [Google Scholar] [CrossRef] [PubMed]
- Barber, M.F.; Elde, N.C. Nutritional immunity. Escape from bacterial iron piracy through rapid evolution of transferrin. Science 2014, 346, 1362–1366. [Google Scholar] [CrossRef] [PubMed]
- LeGrand, E.K.; Alcock, J. Turning up the heat: Immune brinksmanship in the acute-phase response. Q. Rev. Biol. 2012, 87, 3–18. [Google Scholar] [CrossRef] [PubMed]
- Cuenca, A.G.; Delano, M.J.; Kelly-Scumpia, K.M.; Moreno, C.; Scumpia, P.O.; LaFace, D.M.; Heyworth, P.G.; Efron, P.A.; Moldawer, L.L. A Paradoxical Role for Myeloid-Derived Suppressor Cells in Sepsis and Trauma. Mol. Med. 2011, 17, 281–292. [Google Scholar] [CrossRef] [PubMed]
- Francois, B.; Jeannet, R.; Daix, T.; Walton, A.H.; Shotwell, M.S.; Unsinger, J.; Monneret, G.; Rimmelé, T.; Blood, T.; Morre, M.; et al. Interleukin-7 restores lymphocytes in septic shock: The IRIS-7 randomized clinical trial. JCI Insight 2018, 3. [Google Scholar] [CrossRef] [PubMed]
| Dysregulated Pathway | Sepsis Treatment |
|---|---|
| Lipopolysaccharide (LPS; endotoxin) | |
| Anti-LPS human monoclonal antibody HA-1 Anti-LPS E5 murine monoclonal IgM antibody | |
| Enterobacterial common antigen | |
| Toll-like receptor 4 antagonists | |
| Eritoran | |
| TAK-242 (resatorvid) | |
| Anti-CD14 | |
| Taurolidine | |
| Alkaline phosphatase | |
| Polymyxin B | |
| Lipid emulsion | |
| Tumor necrosis factor (TNF) | Monoclonal or polyclonal antibodies |
| Soluble receptor constructs | |
| Interleukin-1 (IL-1) | Recombinant IL-1 receptor antagonist |
| Small-molecule inhibitors | |
| Platelet activating factor (PAF) | PAF acetylhydrolase |
| Ibuprofen | |
| Eicosanoids | Phospholipase A2 inhibitor |
| NO synthase inhibitor l-NNMA | |
| Hypercoagulability | Methylene blue |
| Activated protein C | |
| Tissue factor pathway inhibitor | |
| Antithrombin III | |
| Anti-tissue factor antibody | |
| Thrombomodulin | |
| Immune suppression | Intravenous (IV) immunoglobulin |
| Granulocyte Colony Stimulating Factor and Granulocyte/Macrophage Colony Stimulating Factor | |
| Adrenal insufficiency | Corticosteroids |
| Statins | |
| Organ failure | Extracorporeal hemoperfusion |
| Inadequate perfusion | IV fluid bolus |
| Dobutamine | |
| Insulin resistance, metabolism | Intensive insulin |
© 2018 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alcock, J. The Emperor Has No Clothes? Searching for Dysregulation in Sepsis. J. Clin. Med. 2018, 7, 247. https://doi.org/10.3390/jcm7090247
Alcock J. The Emperor Has No Clothes? Searching for Dysregulation in Sepsis. Journal of Clinical Medicine. 2018; 7(9):247. https://doi.org/10.3390/jcm7090247
Chicago/Turabian StyleAlcock, Joe. 2018. "The Emperor Has No Clothes? Searching for Dysregulation in Sepsis" Journal of Clinical Medicine 7, no. 9: 247. https://doi.org/10.3390/jcm7090247
APA StyleAlcock, J. (2018). The Emperor Has No Clothes? Searching for Dysregulation in Sepsis. Journal of Clinical Medicine, 7(9), 247. https://doi.org/10.3390/jcm7090247





