Diagnostic Criteria and Clinical Outcomes in Sarcopenia Research: A Literature Review
Abstract
1. Introduction
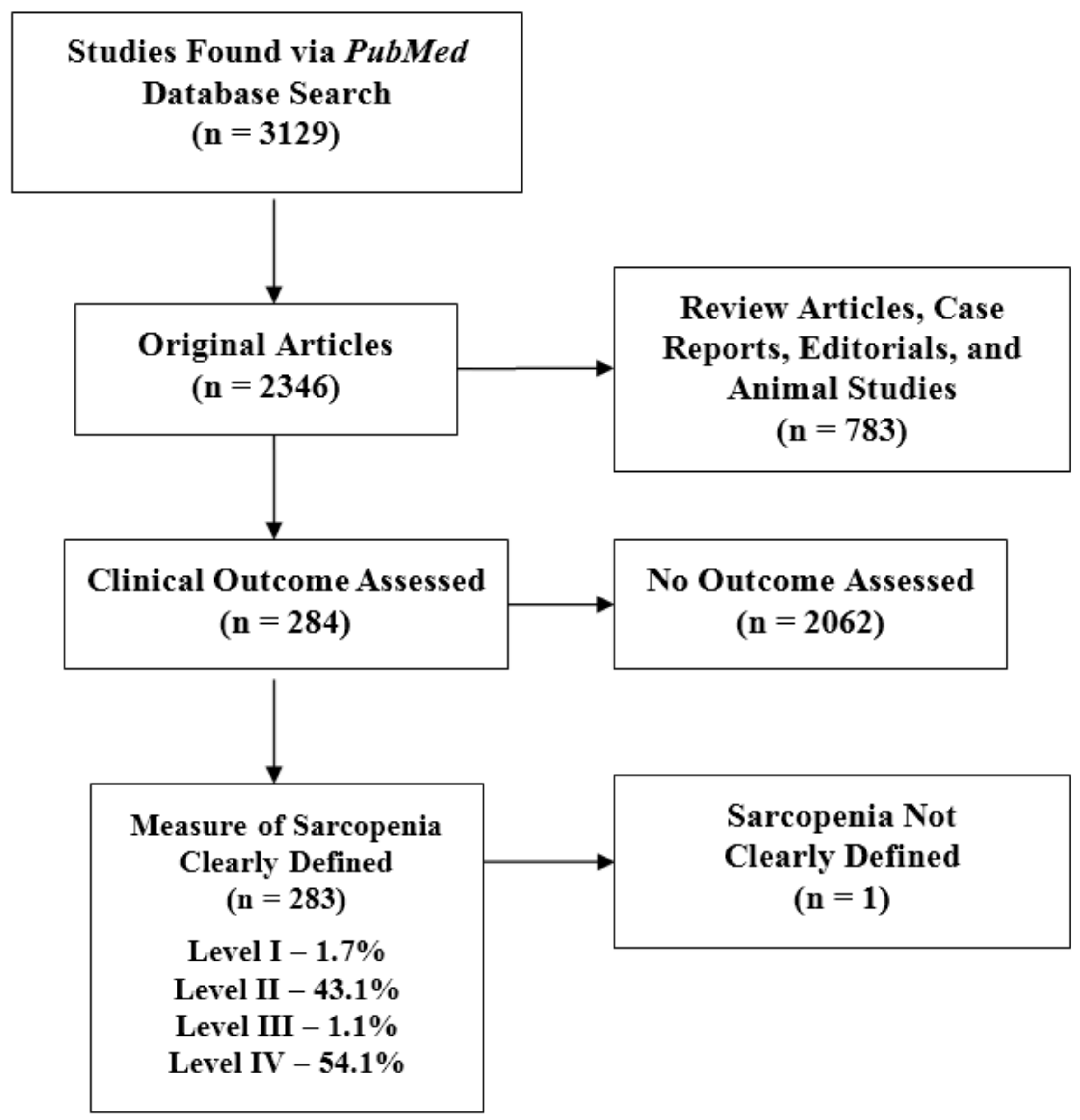
2. Methods
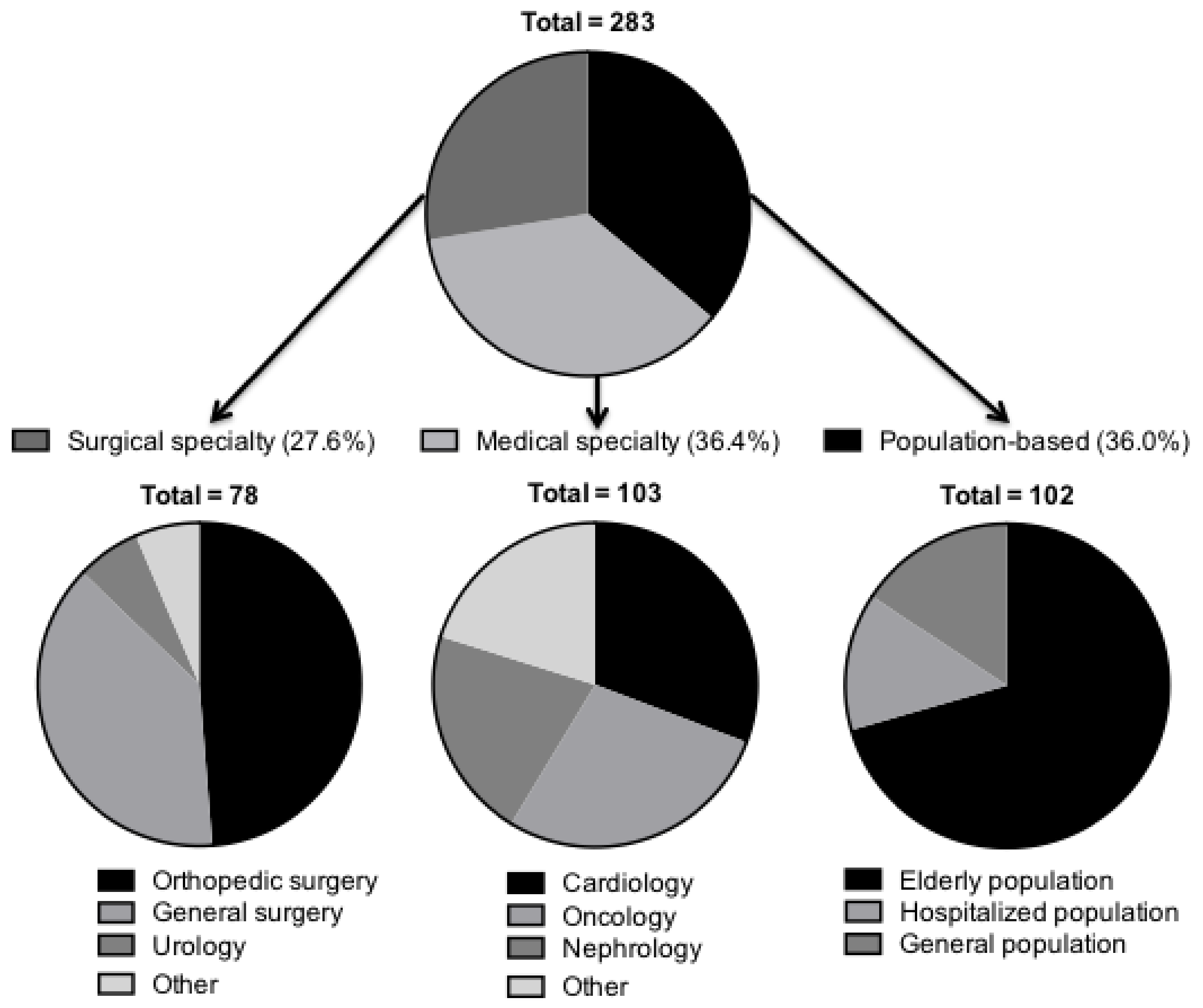
3. Results
4. Sarcopenia in Medicine
4.1. Nephrology
4.2. Hepatobiliary
4.3. Hematology/Oncology
4.4. Cardiology
4.5. Pulmonology
4.6. Population-Based
5. Sarcopenia in Surgery
5.1. General Surgery
5.2. Colorectal Surgery
5.3. Transplant Surgery
5.4. Hepatobiliary Surgery
5.5. Orthopedic Surgery
5.6. Urology
5.7. Gynecologic Oncology
5.8. Ear, Nose, and Throat
6. Discussion
7. Conclusions
Supplementary Materials
Author Contributions
Conflicts of Interest
References
- Cruz-Jentoft, A.J.; Baeyens, J.P.; Bauer, J.M.; Boirie, Y.; Cederholm, T.; Landi, F.; Martin, F.C.; Michel, J.-P.; Rolland, Y.; Schneider, S.M.; et al. Sarcopenia: European consensus on definition and diagnosis: Report of the European Working Group on Sarcopenia in Older People. Age Ageing 2010, 39, 412–423. [Google Scholar] [CrossRef] [PubMed]
- Melton, L.J.; Khosla, S.; Crowson, C.S.; O’Connor, M.K.; O’Fallon, W.M.; Riggs, B.L. Epidemiology of sarcopenia. J. Am. Geriatr. Soc. 2000, 48, 625–630. [Google Scholar] [PubMed]
- Peterson, S.J.; Braunschweig, C.A. Prevalence of sarcopenia and associated outcomes in the clinical setting. Nutr. Clin. Pract. 2016, 31, 40–48. [Google Scholar] [CrossRef] [PubMed]
- Marty, E.; Liu, Y.; Samuel, A.; Or, O.; Lane, J. A review of sarcopenia: Enhancing awareness of an increasingly prevalent disease. Bone 2017, 105, 276–286. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.H.; Park, J.W.; Yoon, K.W.; Do, J.Y. Limb/trunk lean mass ratio as a risk factor for mortality in peritoneal dialysis patients. J. Ren. Nutr. 2013, 23, 315–323. [Google Scholar] [CrossRef] [PubMed]
- Huillard, O.; Mir, O.; Peyromaure, M.; Tlemsani, C.; Giroux, J.; Boudou-Rouquette, P.; Ropert, S.; Delongchamps, N.B.; Zerbib, M.; Goldwasser, F. Sarcopenia and body mass index predict sunitinib-induced early dose-limiting toxicities in renal cancer patients. Br. J. Cancer 2013, 108, 1034–1041. [Google Scholar] [CrossRef] [PubMed]
- Sharma, P.; Zargar-Shoshtari, K.; Caracciolo, J.T.; Fishman, M.; Poch, M.A.; Pow-Sang, J.; Sexton, W.J.; Spiess, P.E. Sarcopenia as a predictor of overall survival after cytoreductive nephrectomy for metastatic renal cell carcinoma. Urol. Oncol. 2015, 33, 339e17–339e23. [Google Scholar] [CrossRef] [PubMed]
- Merli, M.; Giusto, M.; Lucidi, C.; Giannelli, V.; Pentassuglio, I.; Di Gregorio, V.; Lattanzi, B.; Riggio, O. Muscle depletion increases the risk of overt and minimal hepatic encephalopathy: Results of a prospective study. Metab. Brain Dis. 2013, 28, 281–284. [Google Scholar] [CrossRef] [PubMed]
- Mir, O.; Coriat, R.; Blanchet, B.; Durand, J.-P.; Boudou-Rouquette, P.; Michels, J.; Ropert, S.; Vidal, M.; Pol, S.; Chaussade, S.; et al. Sarcopenia predicts early dose-limiting toxicities and pharmacokinetics of sorafenib in patients with hepatocellular carcinoma. PLoS ONE 2012, 7, e37563. [Google Scholar] [CrossRef] [PubMed]
- Cousin, S.; Hollebecque, A.; Koscielny, S.; Mir, O.; Varga, A.; Baracos, V.E.; Soria, J.C.; Antoun, S. Low skeletal muscle is associated with toxicity in patients included in phase I. trials. Investig. New Drugs 2014, 32, 382–387. [Google Scholar] [CrossRef] [PubMed]
- Prado, C.M.M.; Baracos, V.E.; McCargar, L.J.; Reiman, T.; Mourtzakis, M.; Tonkin, K.; Mackey, J.R.; Koski, S.; Pituskin, E.; Sawyer, M.B. Sarcopenia as a determinant of chemotherapy toxicity and time to tumor progression in metastatic breast cancer patients receiving capecitabine treatment. Clin. Cancer Res. 2009, 15, 2920–2926. [Google Scholar] [CrossRef] [PubMed]
- Tan, B.H.L.; Birdsell, L.A.; Martin, L.; Baracos, V.E.; Fearon, K.C.H. Sarcopenia in an overweight or obese patient is an adverse prognostic factor in pancreatic cancer. Clin. Cancer Res. 2009, 15, 6973–6979. [Google Scholar] [CrossRef] [PubMed]
- Han, K.; Park, Y.M.; Kwon, H.S.; Ko, S.H.; Lee, S.H.; Yim, H.W.; Lee, W.C.; Park, Y.G.; Kim, M.K.; Park, Y.M. Sarcopenia as a determinant of blood pressure in older Koreans: Findings from the Korea National Health and Nutrition Examination Surveys (KNHANES) 2008–2010. PLoS ONE 2014, 9, e86902. [Google Scholar] [CrossRef] [PubMed]
- Baek, S.J.; Nam, G.E.; Han, K.D.; Choi, S.W.; Jung, S.W.; Bok, A.R.; Kim, Y.H.; Lee, K.S.; Han, B.D.; Kim, D.H. Sarcopenia and sarcopenic obesity and their association with dyslipidemia in Korean elderly men: The 2008-2010 Korea National Health and Nutrition Examination Survey. J. Endocrinol. Investig. 2014, 37, 247–260. [Google Scholar] [CrossRef] [PubMed]
- Fulster, S.; Tacke, M.; Sandek, A.; Ebner, N.; Tschope, C.; Doehner, W.; Anker, S.D.; von Haehling, S. Muscle wasting in patients with chronic heart failure: Results from the studies investigating co-morbidities aggravating heart failure (SICA-HF). Eur. Heart J. 2013, 34, 512–519. [Google Scholar] [CrossRef] [PubMed]
- Narumi, T.; Watanabe, T.; Kadowaki, S.; Takahashi, T.; Yokoyama, M.; Kinoshita, D.; Honda, Y.; Funayama, A.; Nishiyama, S.; Takahashi, H.; et al. Sarcopenia evaluated by fat-free mass index is an important prognostic factor in patients with chronic heart failure. Eur. J. Intern. Med. 2015, 26, 118–122. [Google Scholar] [CrossRef] [PubMed]
- Sheean, P.M.; Peterson, S.J.; Gomez Perez, S.; Troy, K.L.; Patel, A.; Sclamberg, J.S.; Ajanaku, F.C.; Braunschweig, C.A. The prevalence of sarcopenia in patients with respiratory failure classified as normally nourished using computed tomography and subjective global assessment. JPEN J. Parenter. Enteral Nutr. 2014, 38, 873–879. [Google Scholar] [CrossRef] [PubMed]
- Jeon, Y.K.; Shin, M.J.; Kim, M.H.; Mok, J.H.; Kim, S.S.; Kim, B.H.; Kim, S.-J.; Kim, Y.K.; Chang, J.H.; Shin, Y.B.; et al. Low pulmonary function is related with a high risk of sarcopenia in community-dwelling older adults: The Korea National Health and Nutrition Examination Survey (KNHANES) 2008–2011. Osteoporos. Int. 2015, 26, 2423–2429. [Google Scholar] [CrossRef] [PubMed]
- Cosquéric, G.; Sebag, A.; Ducolombier, C.; Thomas, C.; Piette, F.; Weill-Engerer, S. Sarcopenia is predictive of nosocomial infection in care of the elderly. Br. J. Nutr. 2006, 96, 895–901. [Google Scholar] [CrossRef] [PubMed]
- Batsis, J.A.; Mackenzie, T.A.; Barre, L.K.; Lopez-Jimenez, F.; Bartels, S.J. Sarcopenia, sarcopenic obesity and mortality in older adults: Results from the National Health and Nutrition Examination Survey III. Eur. J. Clin. Nutr. 2014, 68, 1001–1007. [Google Scholar] [CrossRef] [PubMed]
- Chan, O.Y.A.; van Houwelingen, A.H.; Gussekloo, J.; Blom, J.W.; den Elzen, W.P.J. Comparison of quadriceps strength and handgrip strength in their association with health outcomes in older adults in primary care. Age (Dordr) 2014, 36, 9714. [Google Scholar] [CrossRef] [PubMed]
- Janssen, I.; Shepard, D.S.; Katzmarzyk, P.T.; Roubenoff, R. The healthcare costs of sarcopenia in the United States. J. Am. Geriatr. Soc. 2004, 52, 80–85. [Google Scholar] [CrossRef] [PubMed]
- Kirk, P.S.; Friedman, J.F.; Cron, D.C.; Terjimanian, M.N.; Wang, S.C.; Campbell, D.A.; Englesbe, M.J.; Werner, N.L. One-year postoperative resource utilization in sarcopenic patients. J. Surg. Res. 2015, 199, 51–55. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Du, Y.; Karvellas, C.J.; Baracos, V.; Williams, D.C.; Khadaroo, R.G.; Acute Care and Emergency Surgery (ACES) Group. Sarcopenia is a predictor of outcomes in very elderly patients undergoing emergency surgery. Surgery 2014, 156, 521–527. [Google Scholar] [CrossRef] [PubMed]
- Reisinger, K.W.; van Vugt, J.L.A.; Tegels, J.J.W.; Snijders, C.; Hulsewé, K.W.E.; Hoofwijk, A.G.M.; Stoot, J.H.; Von Meyenfeldt, M.F.; Beets, G.L.; Derikx, J.P.M.; et al. Functional compromise reflected by sarcopenia, frailty, and nutritional depletion predicts adverse postoperative outcome after colorectal cancer surgery. Ann. Surg. 2015, 261, 345–352. [Google Scholar] [CrossRef] [PubMed]
- Miyamoto, Y.; Baba, Y.; Sakamoto, Y.; Ohuchi, M.; Tokunaga, R.; Kurashige, J.; Hiyoshi, Y.; Iwagami, S.; Yoshida, N.; Yoshida, M.; et al. Sarcopenia is a negative prognostic factor after curative resection of colorectal cancer. Ann. Surg. Oncol. 2015, 22, 2663–2668. [Google Scholar] [CrossRef] [PubMed]
- Englesbe, M.J.; Patel, S.P.; He, K.; Lynch, R.J.; Schaubel, D.E.; Harbaugh, C.; Holcombe, S.A.; Wang, S.C.; Segev, D.L.; Sonnenday, C.J. Sarcopenia and mortality after liver transplantation. J. Am. Coll. Surg. 2010, 211, 271–278. [Google Scholar] [CrossRef] [PubMed]
- Fujiwara, N.; Nakagawa, H.; Kudo, Y.; Tateishi, R.; Taguri, M.; Watadani, T.; Nakagomi, R.; Kondo, M.; Nakatsuka, T.; Minami, T.; et al. Sarcopenia, intramuscular fat deposition, and visceral adiposity independently predict the outcomes of hepatocellular carcinoma. J. Hepatol. 2015, 63, 131–140. [Google Scholar] [CrossRef] [PubMed]
- Otsuji, H.; Yokoyama, Y.; Ebata, T.; Igami, T.; Sugawara, G.; Mizuno, T.; Nagino, M. Preoperative sarcopenia negatively impacts postoperative outcomes following major hepatectomy with extrahepatic bile duct resection. World J. Surg. 2015, 39, 1494–1500. [Google Scholar] [CrossRef] [PubMed]
- Iolascon, G.; Giamattei, M.T.; Moretti, A.; Di Pietro, G.; Gimigliano, F.; Gimigliano, R. Sarcopenia in women with vertebral fragility fractures. Aging Clin. Exp. Res. 2013, 25 (Suppl. 1), S129–S131. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Kim, T.-N.; Kim, S.-H. Sarcopenic obesity is more closely associated with knee osteoarthritis than is nonsarcopenic obesity: A cross-sectional study. Arthritis Rheum. 2012, 64, 3947–3954. [Google Scholar] [CrossRef] [PubMed]
- Fukushima, H.; Yokoyama, M.; Nakanishi, Y.; Tobisu, K.; Koga, F. Sarcopenia as a prognostic biomarker of advanced urothelial carcinoma. PLoS ONE 2015, 10, e0115895. [Google Scholar] [CrossRef] [PubMed]
- Kuroki, L.M.; Mangano, M.; Allsworth, J.E.; Menias, C.O.; Massad, L.S.; Powell, M.A.; Mutch, D.G.; Thaker, P.H. Pre-operative assessment of muscle mass to predict surgical complications and prognosis in patients with endometrial cancer. Ann. Surg. Oncol. 2015, 22, 972–979. [Google Scholar] [CrossRef] [PubMed]
- Chamchod, S.; Fuller, C.D.; Grossberg, A.J. (P091) sarcopenia/cachexia is associated with reduced survival and locoregional control in head and neck cancer patients receiving radiotherapy: Results from quantitative imaging analysis of lean body mass. Oncology (Williston Park NY) 2015, 29, 205153. [Google Scholar]
- Su, J.; Ekman, C.; Oskolkov, N.; Lahti, L.; Ström, K.; Brazma, A.; Groop, L.; Rung, J.; Hansson, O. A novel atlas of gene expression in human skeletal muscle reveals molecular changes associated with aging. Skelet. Muscle 2015, 5, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Roubenoff, R.; Hughes, V.A. Sarcopenia: Current concepts. J. Gerontol. A Biol. Sci. Med. Sci. 2000, 55, M716–M724. [Google Scholar] [CrossRef] [PubMed]
- Rubbieri, G.; Mossello, E.; Di Bari, M. Techniques for the diagnosis of sarcopenia. Clin. Cases Miner. Bone Metab. 2014, 11, 181–184. [Google Scholar] [CrossRef] [PubMed]
- Ingenbleek, Y.; Bernstein, L.H. Plasma transthyretin as a biomarker of lean body mass and catabolic states. Adv. Nutr. 2015, 6, 572–580. [Google Scholar] [CrossRef] [PubMed]
- Narici, M.V.; Maffulli, N. Sarcopenia: Characteristics, mechanisms and functional significance. Br. Med. Bull. 2010, 95, 139–159. [Google Scholar] [CrossRef] [PubMed]
- Santilli, V.; Bernetti, A.; Mangone, M.; Paoloni, M. Clinical definition of sarcopenia. Clin. Cases Miner. Bone Metab. 2014, 11, 177–180. [Google Scholar] [CrossRef] [PubMed]
- Gori, A.M.; Corsi, A.M.; Fedi, S.; Gazzini, A.; Sofi, F.; Bartali, B.; Bandinelli, S.; Gensini, G.F.; Abbate, R.; Ferrucci, L. A proinflammatory state is associated with hyperhomocysteinemia in the elderly. Am. J. Clin. Nutr. 2005, 82, 335–341. [Google Scholar] [CrossRef] [PubMed]
- Škovierová, H.; Vidomanová, E.; Mahmood, S.; Sopková, J.; Drgová, A.; Červeňová, T.; Halašová, E.; Lehotský, J. The molecular and cellular effect of homocysteine metabolism imbalance on human health. Int. J. Mol. Sci. 2016, 17, 1733. [Google Scholar] [CrossRef] [PubMed]
- Ingenbleek, Y. Lean body mass harbors sensing mechanisms that allow safeguarding of methionine homeostasis. Nutrients 2017, 9, 1035. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Jentoft, A.J.; Landi, F.; Schneider, S.M.; Zuniga, C.; Arai, H.; Boirie, Y.; Chen, L.-K.; Fielding, R.A.; Martin, F.C.; Michel, J.-P.; et al. Prevalence of and interventions for sarcopenia in ageing adults: A systematic review. Report of the International Sarcopenia Initiative (EWGSOP and IWGS). Age Ageing 2014, 43, 748–759. [Google Scholar] [CrossRef] [PubMed]
- Friedman, J.; Lussiez, A.; Sullivan, J.; Wang, S.; Englesbe, M. Implications of sarcopenia in major surgery. Nutr. Clin. Pract. 2015, 30, 175–179. [Google Scholar] [CrossRef] [PubMed]
| Level | Type of Evidence |
|---|---|
| I | Individual randomized controlled trials (RCTs) |
| Systematic review of RCTs | |
| II | Individual cohort studies |
| Systematic review of cohort studies | |
| III | Individual case-control studies |
| Systematic review of case-control studies | |
| IV | Cross-sectional studies |
| Chart review | |
| Case series | |
| V | Expert opinion |
| Clinical observations | |
| Animal/bench research |
| Definition | Percentage of Studies Utilizing | |
|---|---|---|
| Dual Energy X-ray Absorptiometry (DEXA) | Uses X-ray absorption to provide an estimate of appendicular muscle mass | 43.6% |
| Bioelectrical Impedance Analysis (BIA) | Approximates total body muscle and water composition by measuring resistance to electrical flow | 19.3% |
| L3 Total Psoas Index (CT measurement) | Total area of both psoas at L3 vertebrae divided by height squared | 13.6% |
| L4 Total Psoas Area (CT) | Total area of both psoas at L4 vertebrae | 5.3% |
| L3 Psoas Area (CT) | Area of one psoas muscle at L3 vertebrae | 3.5% |
| Total Quadriceps Area (CT) | Cross sectional area of both quadriceps muscles | 1.8% |
| L3 Total Psoas Area (CT) | Total area of both psoas at L3 vertebrae | 1.4% |
| Other | A definition of muscle mass used by less than three studies total | 11.5% |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Han, A.; Bokshan, S.L.; Marcaccio, S.E.; DePasse, J.M.; Daniels, A.H. Diagnostic Criteria and Clinical Outcomes in Sarcopenia Research: A Literature Review. J. Clin. Med. 2018, 7, 70. https://doi.org/10.3390/jcm7040070
Han A, Bokshan SL, Marcaccio SE, DePasse JM, Daniels AH. Diagnostic Criteria and Clinical Outcomes in Sarcopenia Research: A Literature Review. Journal of Clinical Medicine. 2018; 7(4):70. https://doi.org/10.3390/jcm7040070
Chicago/Turabian StyleHan, Alex, Steven L. Bokshan, Stephen E. Marcaccio, J. Mason DePasse, and Alan H. Daniels. 2018. "Diagnostic Criteria and Clinical Outcomes in Sarcopenia Research: A Literature Review" Journal of Clinical Medicine 7, no. 4: 70. https://doi.org/10.3390/jcm7040070
APA StyleHan, A., Bokshan, S. L., Marcaccio, S. E., DePasse, J. M., & Daniels, A. H. (2018). Diagnostic Criteria and Clinical Outcomes in Sarcopenia Research: A Literature Review. Journal of Clinical Medicine, 7(4), 70. https://doi.org/10.3390/jcm7040070





