18F-Fluorodeoxyglucose Positron Emission Tomography/Computed Tomography for Assessing Treatment Response of Pulmonary Multidrug-Resistant Tuberculosis
Abstract
1. Introduction
2. Experimental Section
2.1. Subjects
2.2. 18F-FDG PET/CT
2.3. MDR-TB Treatment and Clinical Follow-Up
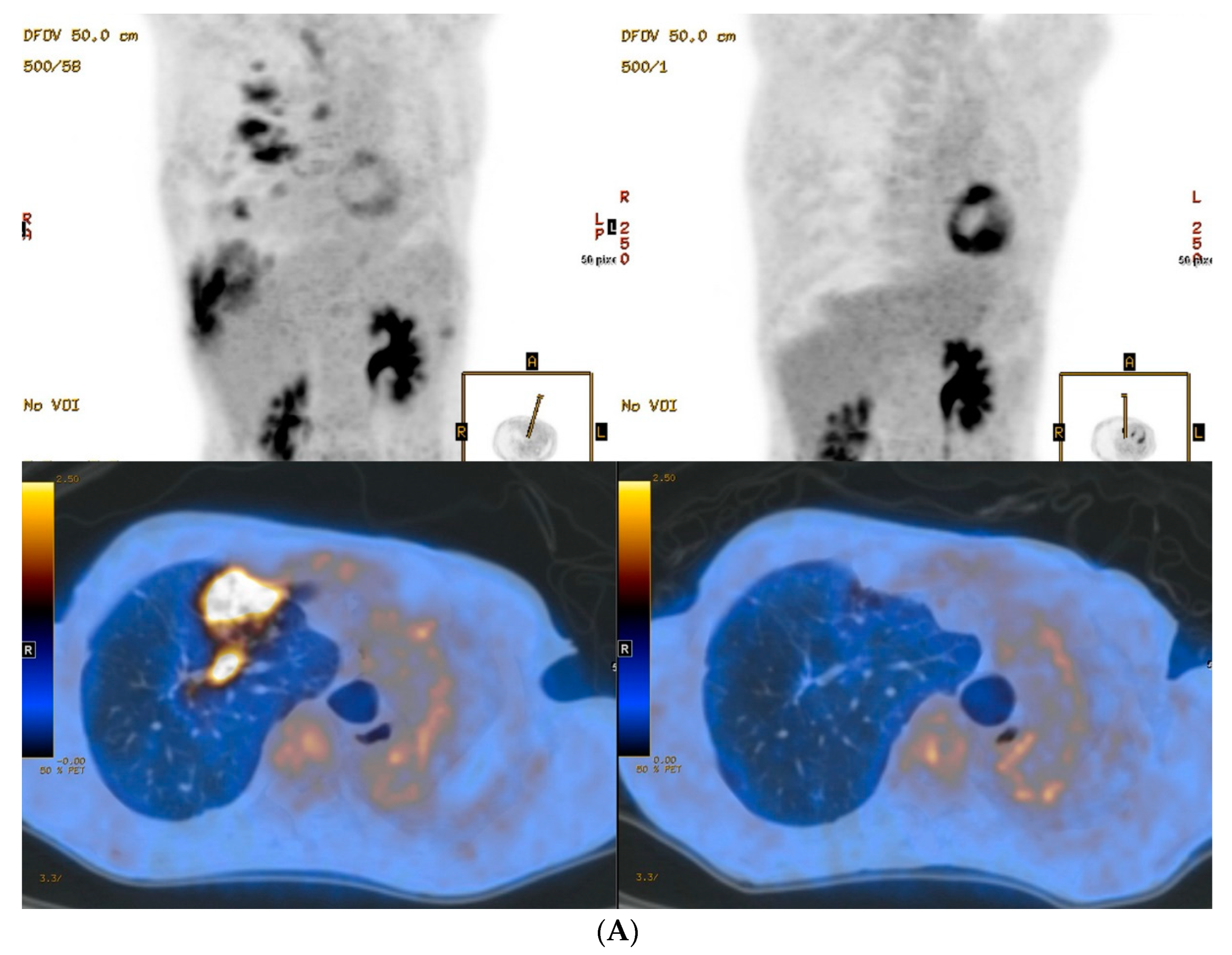
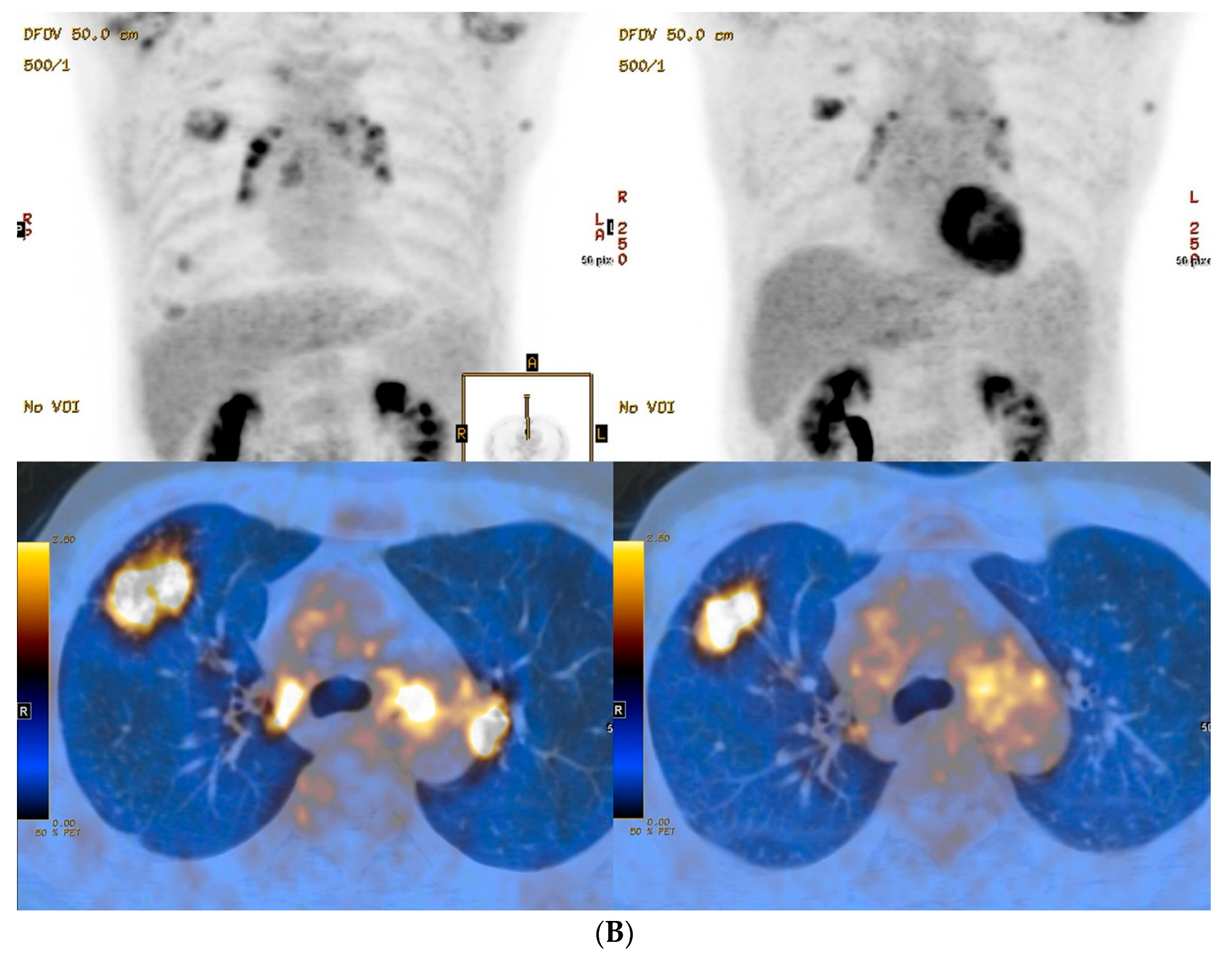
3. Results
4. Discussion
5. Conclusions
6. Ethics Approval and Consent to Participate
Author Contributions
Funding
Conflicts of Interest
References
- Davies, P.D.; Yew, W.W.; Ganguly, D.; Davidow, A.L.; Reichman, L.B.; Dheda, K.; Rook, G.A. Smoking and tuberculosis: The epidemiological association and immunopathogenesis. Trans. R. Soc. Trop. Med. Hyg. 2006, 100, 291–298. [Google Scholar] [CrossRef] [PubMed]
- Ahuja, S.D.; Ashkin, D.; Avendano, M.; Banerjee, R.; Bauer, M.; Bayona, J.N.; Becerra, M.C.; Benedetti, A.; Burgos, M.; Centis, R.; et al. Multidrug resistant pulmonary tuberculosis treatment regimens and patient outcomes: An individual patient data meta-analysis of 9,153 patients. PLoS Med. 2012, 9, e1001300. [Google Scholar] [CrossRef]
- Lange, C.; Abubakar, I.; Alffenaar, J.W.; Bothamley, G.; Caminero, J.A.; Carvalho, A.C.; Chang, K.C.; Codecasa, L.; Correia, A.; Crudu, V.; et al. Management of patients with multidrug-resistant/extensively drug-resistant tuberculosis in Europe: A TBNET consensus statement. Eur. Respir. J. 2014, 44, 23–63. [Google Scholar] [CrossRef] [PubMed]
- Mitnick, C.D.; White, R.A.; Lu, C.; Rodriguez, C.A.; Bayona, J.; Becerra, M.C.; Burgos, M.; Centis, R.; Cohen, T.; Cox, H.; et al. Multidrug-resistant tuberculosis treatment failure detection depends on monitoring interval and microbiological method. Eur. Respir. J. 2016, 48, 1160–1170. [Google Scholar] [CrossRef] [PubMed]
- Horne, D.J.; Royce, S.E.; Gooze, L.; Narita, M.; Hopewell, P.C.; Nahid, P.; Steingart, K.R. Sputum monitoring during tuberculosis treatment for predicting outcome: Systematic review and meta-analysis. Lancet Infect. Dis. 2010, 10, 387–394. [Google Scholar] [CrossRef]
- Holtz, T.H.; Sternberg, M.; Kammerer, S.; Laserson, K.F.; Riekstina, V.; Zarovska, E.; Skripconoka, V.; Wells, C.D.; Leimane, V. Time to sputum culture conversion in multidrug-resistant tuberculosis: Predictors and relationship to treatment outcome. Ann. Intern. Med. 2006, 144, 650–659. [Google Scholar] [CrossRef] [PubMed]
- Capitanio, S.; Nordin, A.J.; Noraini, A.R.; Rossetti, C. PET/CT in nononcological lung diseases: Current applications and future perspectives. Eur. Respir. Rev. 2016, 25, 247–258. [Google Scholar] [CrossRef] [PubMed]
- Ankrah, A.O.; van der Werf, T.S.; de Vries, E.F.; Dierckx, R.A.; Sathekge, M.M.; Glaudemans, A.W. PET/CT imaging of Mycobacterium tuberculosis infection. Clin. Transl. Imaging 2016, 4, 131–144. [Google Scholar] [CrossRef] [PubMed]
- Kubota, R.; Kubota, K.; Yamada, S.; Tada, M.; Ido, T.; Tamahashi, N. Microautoradiographic study for the differentiation of intratumoral macrophages, granulation tissues and cancer cells by the dynamics of fluorine-18-fluorodeoxyglucose uptake. J. Nucl. Med. 1994, 35, 104–112. [Google Scholar] [PubMed]
- Ziai, P.; Hayeri, M.R.; Salei, A.; Salavati, A.; Houshmand, S.; Alavi, A.; Teytelboym, O.M. Role of optimal quantification of FDG PET imaging in the clinical practice of radiology. Radiographics 2016, 36, 481–496. [Google Scholar] [CrossRef] [PubMed]
- Demura, Y.; Tsuchida, T.; Uesaka, D.; Umeda, Y.; Morikawa, M.; Ameshima, S.; Ishizaki, T.; Fujibayashi, Y.; Okazawa, H. Usefulness of 18F-fluorodeoxyglucose positron emission tomography for diagnosing disease activity and monitoring therapeutic response in patients with pulmonary mycobacteriosis. Eur. J. Nucl. Med. Mol. Imaging 2009, 36, 632–639. [Google Scholar] [CrossRef] [PubMed]
- Coleman, M.T.; Chen, R.Y.; Lee, M.; Lin, P.L.; Dodd, L.E.; Maiello, P.; Via, L.E.; Kim, Y.; Marriner, G.; Dartois, V.; et al. PET/CT imaging reveals a therapeutic response to oxazolidinones in macaques and humans with tuberculosis. Sci. Transl. Med. 2014, 6, 265ra167. [Google Scholar] [CrossRef] [PubMed]
- Stelzmueller, I.; Huber, H.; Wunn, R.; Hodolic, M.; Mandl, M.; Lamprecht, B.; Schinko, H.; Fellner, F.; Skanjeti, A.; Giammarile, F.; et al. 18F-FDG PET/CT in the initial assessment and for follow-up in patients with tuberculosis. Clin. Nucl. Med. 2016, 41, e187–e194. [Google Scholar] [CrossRef] [PubMed]
- Malherbe, S.T.; Shenai, S.; Ronacher, K.; Loxton, A.G.; Dolganov, G.; Kriel, M.; Van, T.; Chen, R.Y.; Warwick, J.; Via, L.E.; et al. Persisting positron emission tomography lesion activity and Mycobacterium tuberculosis mRNA after tuberculosis cure. Nat. Med. 2016, 22, 1094–1100. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Companion Handbook to the WHO Guidelines for the Programmatic Management of Drug-Resistant Tuberculosis; World Health Organization: Geneva, Switzerland, 2014. [Google Scholar]
- Chen, R.Y.; Dodd, L.E.; Lee, M.; Paripati, P.; Hammoud, D.A.; Mountz, J.M.; Jeon, D.; Zia, N.; Zahiri, H.; Coleman, M.T.; et al. PET/CT imaging correlates with treatment outcome in patients with multidrug-resistant tuberculosis. Sci. Transl. Med. 2014, 6, 265ra166. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | Patient No. | |||
|---|---|---|---|---|
| 1 | 2 | 3 | 4 | |
| Age, years | 35 | 59 | 42 | 61 |
| Sex | Female | Female | Female | Male |
| AFB smear | 2+ | 2+ | - | - |
| Body mass index, kg/m2 | 20.6 | 15.1 | 24.1 | 25.0 |
| Previous history of TB | TB LN | MDR-TB | - | DS-TB |
| Time from treatment completion of previous TB to inclusion of current study | 13 years | 8 years | - | 30 years |
| Duration of previous TB treatment | 9 months | 23 months | - | NA |
| Regimens of previous TB treatment | 1st line anti-TB drugs | EMB PZA KM MFX PTO CS PAS | - | 1st line anti-TB drugs |
| Previous lung surgery | - | pneumonectomy | - | - |
| Time from starting MDR-TB treatment to PET/CT scan | 0 day | 0 day | 0 day | 0 day |
| Cavity size | 12 mm | 17 mm | 31 mm | - |
| WBC count, 103/uL | 4510 | 7350 | 5720 | 4840 |
| ESR, mm/hr | 21 | 66 | 14 | 88 |
| CRP, mg/dL | 0.11 | 0.08 | 0.06 | 0.28 |
| Albumin, g/dL | 4.3 | 4.6 | 4.3 | 4.0 |
| Patient No. | Drug Resistance | Treatment Regimens | Outcome | PET/CT | Highest SUVmax | Mean SUVavg | TMLV (cm3) | TLG (cm3) |
|---|---|---|---|---|---|---|---|---|
| 1 | INH, RFP, EMB, PAS | PZA, KM, MFX, PTO, CS | Success | Baseline | 7.8 | 4.1 | 31.4 | 129.8 |
| 6 mo | 4.8 | 3.1 | 2.0 | 6.1 | ||||
| 12 mo | 2.3 | NA | NA | NA | ||||
| 2 | INH, RFP, FQ, EMB, PAS | KM, PTO, CS, LZD, BDQ, CFZ | Failure | Baseline | 6.6 | 3.3 | 92.3 | 307.3 |
| 6 mo | 3.4 | 2.9 | 0.4 | 1.3 | ||||
| 12 mo | NV | NA | NA | NA | ||||
| 3 | INH, RFP, FQ, EMB | EMB, KM, PTO, CS, LZD, BDQ | Success | Baseline | 5.4 | 3.1 | 11.0 | 34.3 |
| 6 mo | 4.6 | 3.0 | 2.0 | 5.9 | ||||
| 12 mo | 2.3 | NA | NA | NA | ||||
| 4 | INH, RFP, EMB, PAS | PZA, KM, MFX, PTO, CS | Success | Baseline | 4.6 | 3.0 | 4.6 | 14.1 |
| 6 mo | 5.6 | 3.6 | 6.3 | 22.7 | ||||
| 12 mo | 4.7 | 3.3 | 3.7 | 12.2 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Choi, J.Y.; Jhun, B.W.; Hyun, S.H.; Chung, M.J.; Koh, W.-J. 18F-Fluorodeoxyglucose Positron Emission Tomography/Computed Tomography for Assessing Treatment Response of Pulmonary Multidrug-Resistant Tuberculosis. J. Clin. Med. 2018, 7, 559. https://doi.org/10.3390/jcm7120559
Choi JY, Jhun BW, Hyun SH, Chung MJ, Koh W-J. 18F-Fluorodeoxyglucose Positron Emission Tomography/Computed Tomography for Assessing Treatment Response of Pulmonary Multidrug-Resistant Tuberculosis. Journal of Clinical Medicine. 2018; 7(12):559. https://doi.org/10.3390/jcm7120559
Chicago/Turabian StyleChoi, Joon Young, Byung Woo Jhun, Seung Hyup Hyun, Myung Jin Chung, and Won-Jung Koh. 2018. "18F-Fluorodeoxyglucose Positron Emission Tomography/Computed Tomography for Assessing Treatment Response of Pulmonary Multidrug-Resistant Tuberculosis" Journal of Clinical Medicine 7, no. 12: 559. https://doi.org/10.3390/jcm7120559
APA StyleChoi, J. Y., Jhun, B. W., Hyun, S. H., Chung, M. J., & Koh, W.-J. (2018). 18F-Fluorodeoxyglucose Positron Emission Tomography/Computed Tomography for Assessing Treatment Response of Pulmonary Multidrug-Resistant Tuberculosis. Journal of Clinical Medicine, 7(12), 559. https://doi.org/10.3390/jcm7120559





