Aromatherapy for Managing Pain in Primary Dysmenorrhea: A Systematic Review of Randomized Placebo-Controlled Trials
Abstract
1. Introduction
2. Methods
2.1. Data Sources
2.2. Study Selection
2.2.1. Type of Studies
2.2.2. Type of Participants
2.2.3. Types of Interventions
2.2.4. Types of Comparisons
2.3. Outcome Measures
2.4. Data Extraction and Risk-of-Bias Assessment
2.5. Data Synthesis
2.6. Assessing the Overall Quality of the Evidence
3. Results
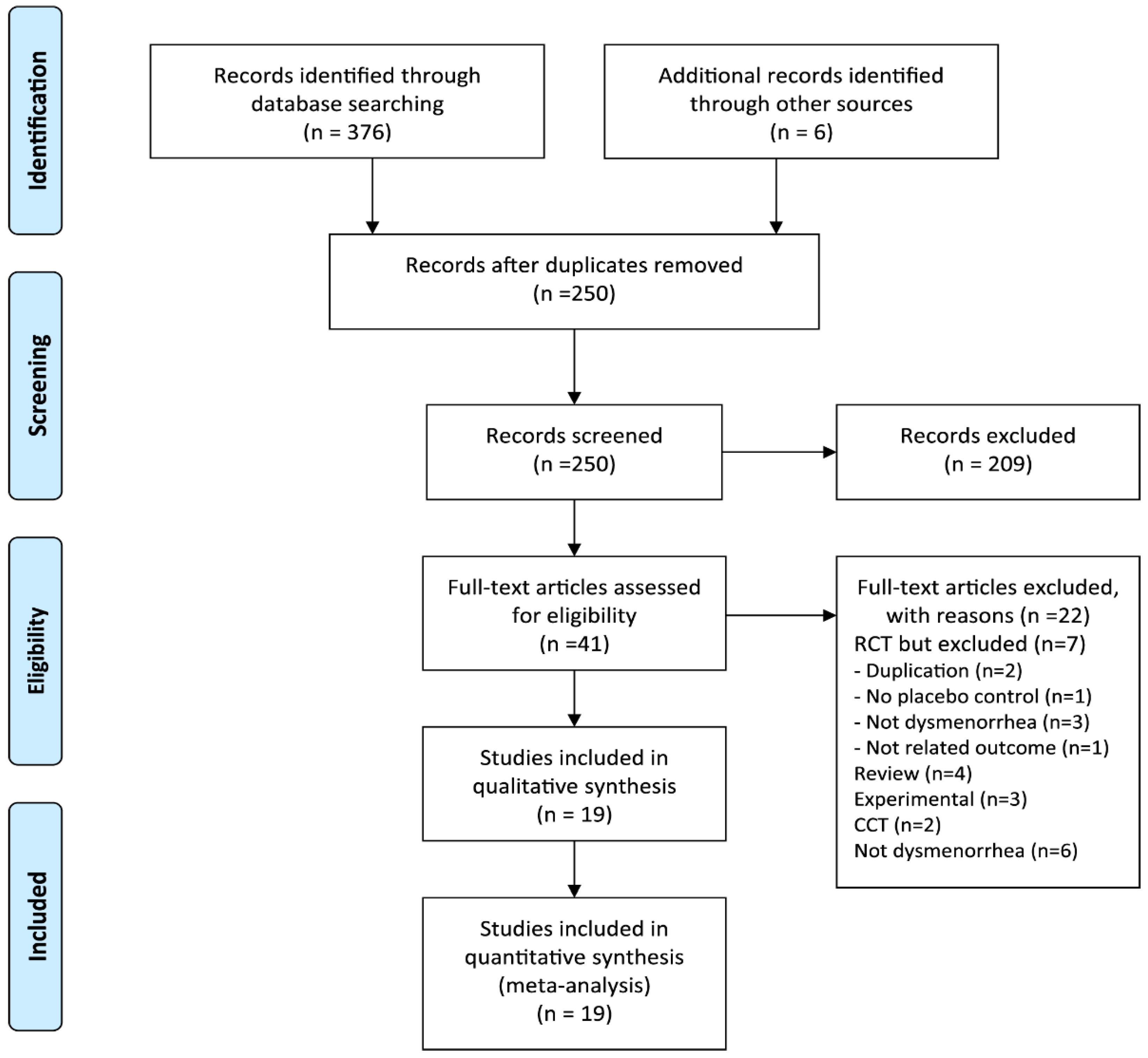
3.1. Description of the Included Trials
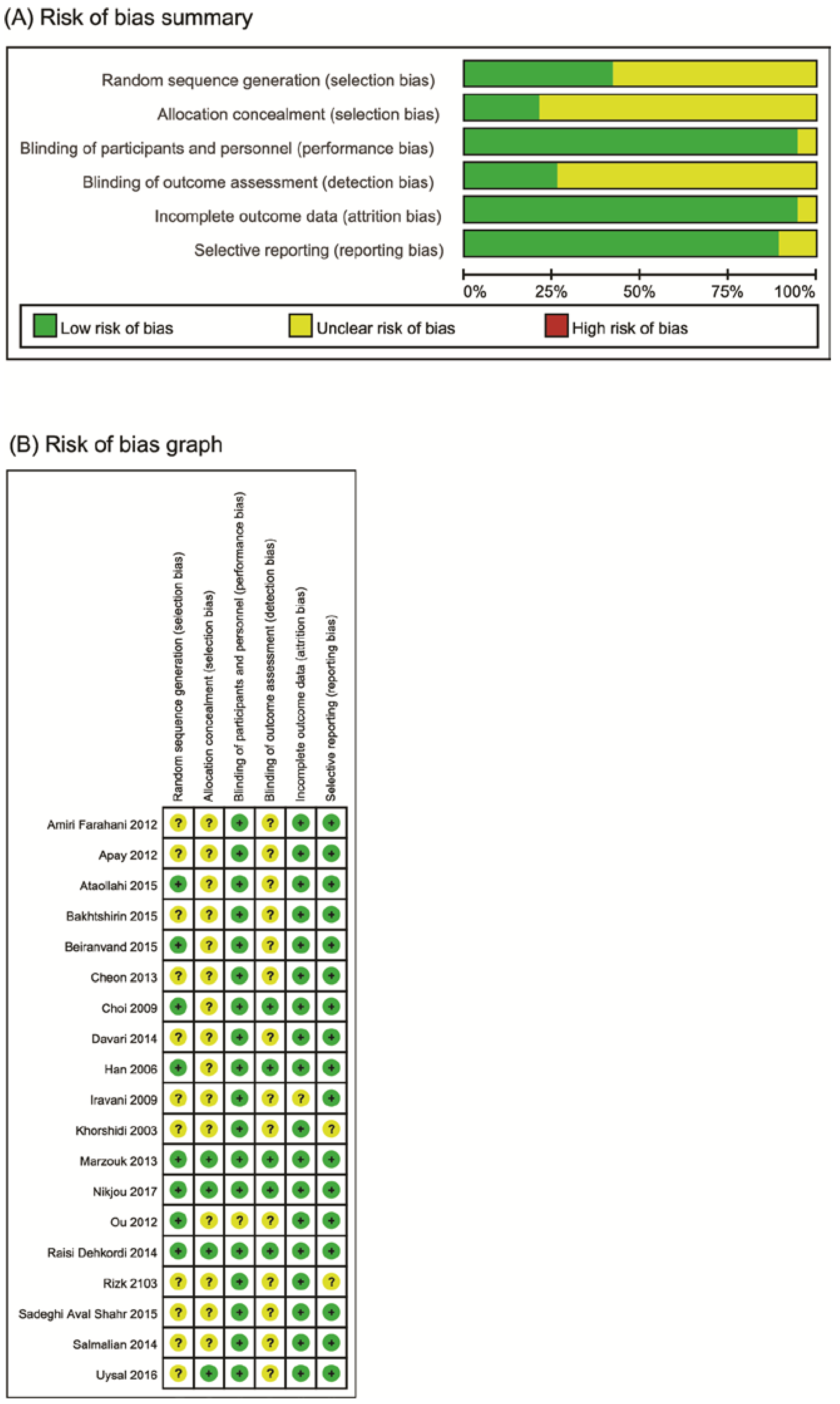
3.2. Risk of Bias
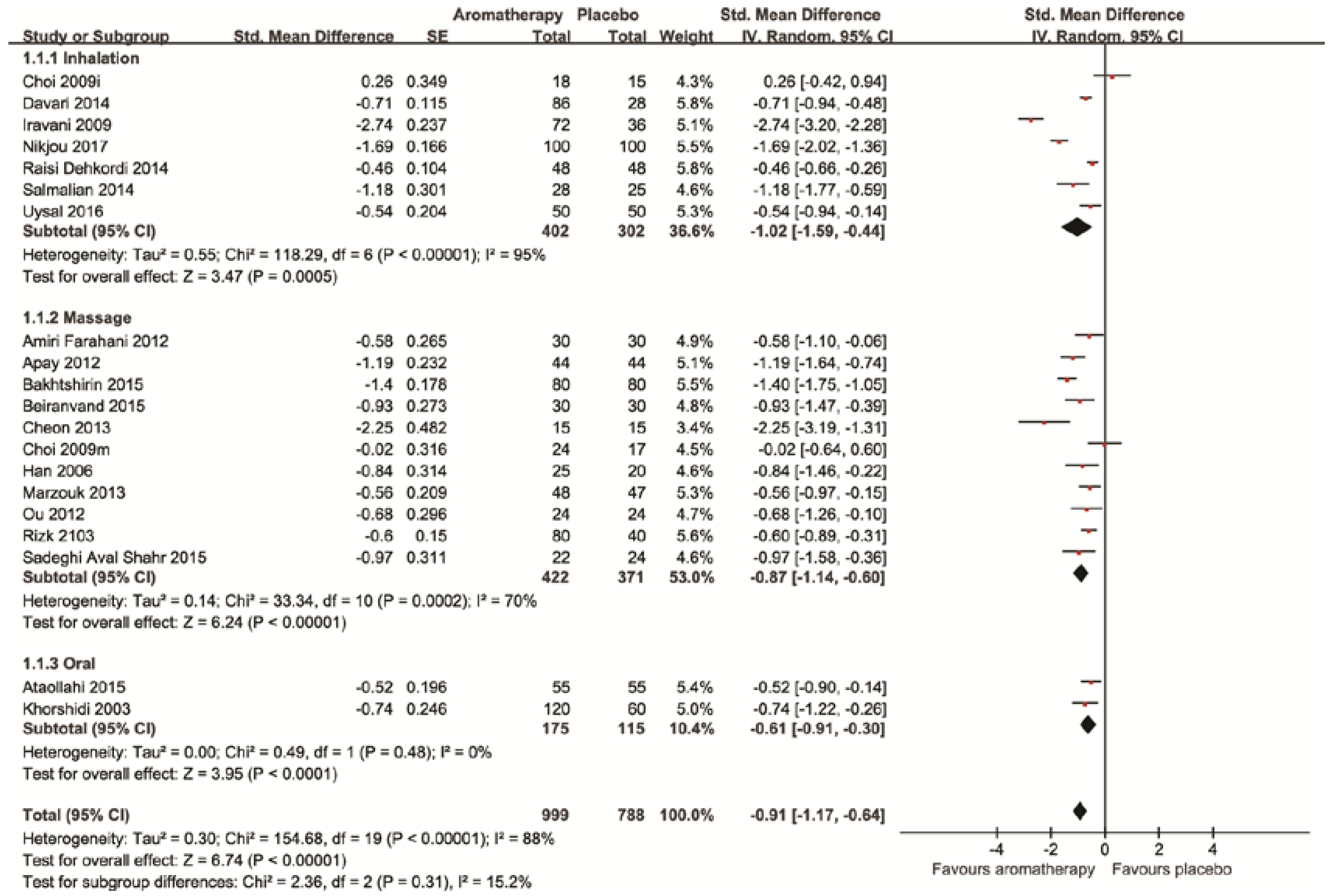
3.3. Outcome Measurements
3.3.1. Pain
3.3.2. Adverse Events
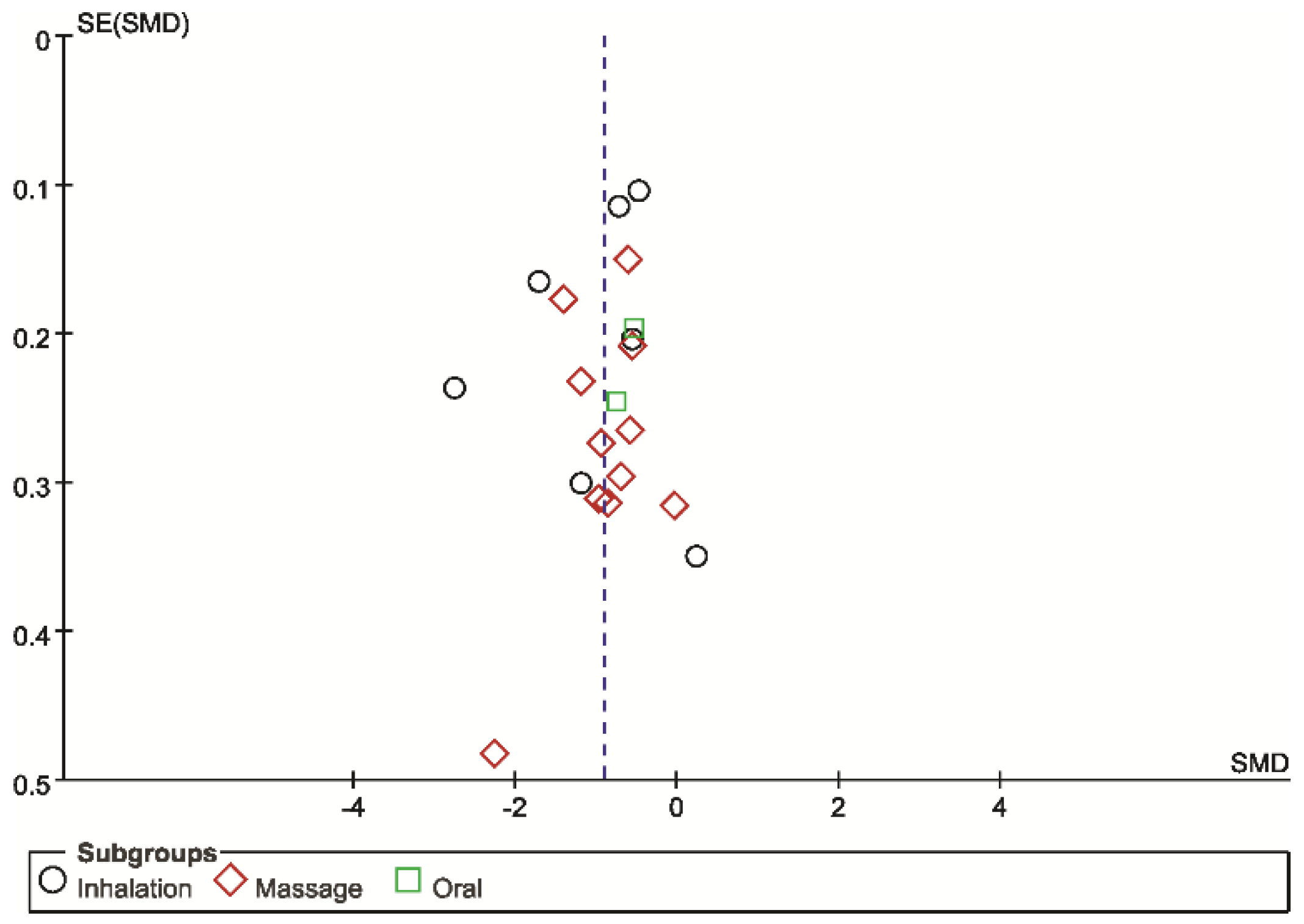
3.3.3. Reporting Bias
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Proctor, M.; Farquhar, C. Diagnosis and management of dysmenorrhoea. BMJ 2006, 332, 1134–1138. [Google Scholar] [CrossRef] [PubMed]
- Latthe, P.M.; Champaneria, R. Dysmenorrhea. Clin. Evid. 2013, 10, 813. [Google Scholar]
- Marjoribanks, J.; Ayeleke, R.O.; Farquhar, C.; Proctor, M. Nonsteroidal anti-inflammatory drugs for dysmenorrhoea. Cochrane Database Syst. Rev. 2015, CD001751Cd001751. [Google Scholar] [CrossRef] [PubMed]
- Wong, C.L.; Farquhar, C.; Roberts, H.; Proctor, M. Oral contraceptive pill for primary dysmenorrhoea. Cochrane Database Syst. Rev. 2009, CD002120. [Google Scholar] [CrossRef]
- Posadzki, P.; Lee, M.S.; Moon, T.W.; Choi, T.Y.; Park, T.Y.; Ernst, E. Prevalence of complementary and alternative medicine (CAM) use by menopausal women: A systematic review of surveys. Maturitas 2013, 75, 34–43. [Google Scholar] [CrossRef] [PubMed]
- Moore, T.R.; Franks, R.B.; Fox, C. Review of Efficacy of Complementary and Alternative Medicine Treatments for Menopausal Symptoms. J. Midwifery Women’s Health 2017, 62, 286–297. [Google Scholar] [CrossRef] [PubMed]
- Ernst, E.; Pittler, M.H.; Wider, B.; Boody, K. The Desktop Guide to Complementary and Alternative Medicine; Elsevier: Philadelpia, PA, USA, 2006. [Google Scholar]
- Conlon, P.M.; Haack, K.M.; Rodgers, N.J.; Dion, L.J.; Cambern, K.L.; Rohlik, G.M.; Nelson, D.E.; Barry, T.A.; Ayres, S.J.; Cutshall, S.M. Introducing Essential Oils into Pediatric and Other Practices at an Academic Medical Center. J. Holist. Nurs. 2017, 35, 389–396. [Google Scholar] [CrossRef] [PubMed]
- Esparham, A.; Herbert, A.; Pierzchalski, E.; Tran, C.; Dilts, J.; Boorigie, M.; Wingert, T.; Connelly, M.; Bickel, J. Pediatric Headache Clinic Model: Implementation of Integrative Therapies in Practice. Children 2018, 5, 74. [Google Scholar] [CrossRef] [PubMed]
- Buckle, J. Clinical Aromatherapy, 3rd ed.; Elsevier: St. Louis, MO, USA, 2015. [Google Scholar]
- Price, S.; Price, L. Aromatherapy for Health Professionals; Elsevier: Edinburgh, UK, 2012. [Google Scholar]
- Lee, M.S.; Choi, J.; Posadzki, P.; Ernst, E. Aromatherapy for health care: An overview of systematic reviews. Maturitas 2012, 71, 257–260. [Google Scholar] [CrossRef] [PubMed]
- Sut, N.; Kahyaoglu-Sut, H. Effect of aromatherapy massage on pain in primary dysmenorrhea: A meta-analysis. Complement. Ther. Clin. Pract. 2017, 27, 5–10. [Google Scholar] [CrossRef] [PubMed]
- Song, J.-A.; Lee, M.-K.; Min, E.; Kim, M.-E.; Fike, G.; Hur, M.-H. Effects of aromatherapy on dysmenorrhea: A systematic review and meta-analysis. Int. J. Nurs. Stud. 2018, 84, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.; Altman, D.; Sterne, J. Chapter 8: Assessing risk of bias in included studies. In Cochrane Handbook for Systematic Reviews of Interventions; Version 510; Higgins, J., Green, S., Eds.; The Cochrane Collaboration: London, UK, 2011. [Google Scholar]
- Uysal, M.; Doğru, H.Y.; Sapmaz, E.; Tas, U.; Çakmak, B.; Ozsoy, A.Z.; Sahin, F.; Ayan, S.; Esen, M. Investigating the effect of rose essential oil in patients with primary dysmenorrhea. Complement. Ther. Clin. Pract. 2016, 24, 45–49. [Google Scholar] [CrossRef] [PubMed]
- Raisi Dehkordi, Z.; Hosseini Baharanchi, F.S.; Bekhradi, R. Effect of lavender inhalation on the symptoms of primary dysmenorrhea and the amount of menstrual bleeding: A randomized clinical trial. Complement. Ther. Med. 2014, 22, 212–219. [Google Scholar] [CrossRef] [PubMed]
- Nikjou, R.; Kazemzadeh, R.; Rostamnegad, M.; Moshfegi, S.; Karimollahi, M.; Salehi, H. The Effect of Lavender Aromatherapy on the Pain Severity of Primary Dysmenorrhea: A Triple-blind Randomized Clinical Trial. Ann. Med. Health Sci. Res. 2016, 6, 211–215. [Google Scholar] [CrossRef] [PubMed]
- Choi, E.H. Comparison of effects of lavender abdominal massage and inhalation on dysmenorrhea, pain, anxiety and depression. Korean J. Fund. Nurs. 2009, 16, 300–306. [Google Scholar]
- Davari, M.; Reihani, M.; Khoshrang, N. The aromatherapy effect of rosemary and lavander on primary dysmenorrhea: A clinical controlled trial. J. Isfahan. Med. Sch. 2014, 32, 929–937. [Google Scholar]
- Iravani, M. Clinical effects of Zataria multiflora essential oil on primary dysmenorrhea. J. Med. Plants 2009, 8, 54–168. [Google Scholar] [CrossRef]
- Salmalian, H.; Saghebi, R.; Moghadamnia, A.A.; Bijani, A.; Faramarzi, M.; Nasiri Amiri, F.; Bakouei, F.; Behmanesh, F.; Bekhradi, R. Comparative effect of thymus vulgaris and ibuprofen on primary dysmenorrhea: A triple-blind clinical study. Caspian J. Intern. Med. 2014, 5, 82–88. [Google Scholar] [PubMed]
- Bakhtshirin, F.; Abedi, S.; YusefiZoj, P.; Razmjooee, D. The effect of aromatherapy massage with lavender oil on severity of primary dysmenorrhea in Arsanjan students. Iran. J. Nurs. Midwifery Res. 2015, 20, 156–160. [Google Scholar] [PubMed]
- Apay, S.E.; Arslan, S.; Akpinar, R.B.; Celebioglu, A. Effect of aromatherapy massage on dysmenorrhea in Turkish students. Pain Manag. Nurs. 2012, 13, 236–240. [Google Scholar] [CrossRef] [PubMed]
- Beiranvand, S.; Hosseinabadi, R.; Anbari, K.; Pirdadeh Beiranvand, S.; Asti, P. The effect of lavender aromatherapy massage on severity and Symptoms of primary dysmenorrhea. Complement. Med. J. 2015, 5, 1028–1041. [Google Scholar]
- Sadeghi Aval Shahr, H.; Saadat, M.; Kheirkhah, M.; Saadat, E. The effect of self-aromatherapy massage of the abdomen on the primary dysmenorrhoea. J. Obstet. Gynaecol. 2015, 35, 382–385. [Google Scholar] [CrossRef] [PubMed]
- Rizk, S.A. Effect of aromatherapy abdominal massage using peppermint versus ginger oils on primary dysmenorrhea among adolescent girls. J. Am. Sci. 2013, 9, 597–605. [Google Scholar]
- Marzouk, T.M.F.; El-Nemer, A.M.R.; Baraka, H.N. The effect of aromatherapy abdominal massage on alleviating menstrual pain in nursing students: A prospective randomized cross-over study. Evid. Based Complement. Alternat. Med. 2013, 2013, 742421. [Google Scholar] [CrossRef] [PubMed]
- Amiri Farahani, L.; Heidari, T.; Roozbahani, N.; Attarha, M.; Akbari Torkestani, N.; Bekhradi, R.; Siyanaki, V. Effect of aromatherapy on pain severity in primary dysmenorrhea. Arak Med. Univ. J. 2012, 15, 1–9. [Google Scholar]
- Han, S.H.; Hur, M.H.; Buckle, J.; Choi, J.; Lee, M.S. Effect of aromatherapy on symptoms of dysmenorrhea in college students: A randomized placebo-controlled clinical trial. J. Altern. Complement. Med. 2006, 12, 535–541. [Google Scholar] [CrossRef] [PubMed]
- Cheon, J.; Lim, M.H. Effect of aromatherapy using nutmeg, fennel and marjoram on menstruation cramps and dysmenorrhea. J. Korean Soc. Cosm. 2013, 19, 1138–1147. [Google Scholar]
- Ou, M.-C.; Hsu, T.-F.; Lai, A.C.; Lin, Y.-T.; Lin, C.-C. Pain relief assessment by aromatic essential oil massage on outpatients with primary dysmenorrhea: A. randomized, double-blind clinical trial. J. Obstet. Gynaecol. Res. 2012, 38, 817–822. [Google Scholar] [CrossRef] [PubMed]
- Khorshidi, N.; Ostad, S.N.; Mahmoud, M.; Soodi, M. Clinical effects of fennel essential oil on primary dysmenorrhea. Iran J. Pharm. Res. 2003, 2, 89–93. [Google Scholar]
- Ataollahi, M.; Akabari, S.A.; Mojab, F.; Roshanaie, G. Effects of aromatherapy by Rosaceous on the severity and systemic symptoms of primary dysmenorrhea. Adv. Nurs. Midwifery 2015, 25, 59–67. [Google Scholar]
- Savović, J.; Jones, H.; Altman, D.; Harris, R.; Jűni, P.; Pildal, J.; Als-Nielsen, B.; Balk, E.; Gluud, C.; Gluud, L.; et al. Influence of reported study design characteristics on intervention effect estimates from randomized controlled trials: Combined analysis of meta-epidemiological studies. Health Technol. Assess. 2012, 16, 81. [Google Scholar] [CrossRef] [PubMed]
- Posadzki, P.; Alotaibi, A.; Ernst, E. Adverse effects of aromatherapy: A systematic review of case reports and case series. Int. J. Risk Saf. Med. 2012, 24, 147–161. [Google Scholar] [CrossRef] [PubMed]
- Dornic, N.; Ficheux, A.S.; Roudot, A.C. Qualitative and quantitative composition of essential oils: A literature-based database on contact allergens used for safety assessment. Regul. Toxicol Pharmacol. 2016, 80, 226–232. [Google Scholar] [CrossRef] [PubMed]
- Diaz, A.; Luque, L.; Badar, Z.; Kornic, S.; Danon, M. Prepubertal gynecomastia and chronic lavender exposure: Report of three cases. J. Pediatr. Endocrinol. Metab. 2016, 29, 103–107. [Google Scholar] [CrossRef] [PubMed]
- Henley, D.V.; Lipson, N.; Korach, K.S.; Bloch, C.A. Prepubertal gynecomastia linked to lavender and tea tree oils. N. Engl. J. Med. 2007, 356, 479–485. [Google Scholar] [CrossRef] [PubMed]
| First Author (Year) [Ref] Country | Sample Size; Age (years); Severity | Intervention | Control | Treatment Duration | Result | Comments Design/AEs |
|---|---|---|---|---|---|---|
| Uysal (2016) [16] Turkey | 105 19–30 Pain on VAS ≥5 | (A) Rose oil (Inhalation, <1 m above patients, n = 52), plus diclofenac sodium (75 mg) | (B) Placebo (saline, <1 m above patients, n = 53), plus diclofenac sodium (75 mg) | 1 time (10–30 min) | p < 0.01 in favor of aromatherapy | Diclofenac 75 mg Treatment time is not clear Parallel/n.r. |
| Raisi Dehkordi (2014) [17] Iran | 96 18–28 Mild or moderate (AMVMS) | (A) Lavender (Inhalation, 7–10 from the nose, rub their palm with aroma oil, n = 48) | (B) Placebo (sesame oil, same methods with A, n = 48) | 5 min (1 h after experiencing dysmenorrhea) every 6 h for the 1st 3 days of menstruation, one or two treatments in 2 consecutive menstrual cycle | p < 0.01 in favor of aromatherapy | Not validated questionnaire Retrospective registration Parallel/n.r. |
| Nikjou (2017) [18] Iran | 200 19–30 n.r. | (A) Lavender (Inhalation, 3 drops, n = 100) | (B) Placebo (diluted milk, same methods with A, n = 100) | 30 min (0.5 h after experiencing dysmenorrhea) for the 1st 3 days of menstruation, one treatment in 2 consecutive menstrual cycle | p < 0.01 in favor of aromatherapy | Registration while recruiting Parallel/n.r. |
| Choi (2009) [19] Korea | 74 not clear Pain on VAS ≥7 | (A) Lavender (Inhalation, n = 18) (B) Lavender (Massage abdomen, n = 24) | (C) Placebo (vitamin C, same methods with A, n = 15) (D) Placebo (Massage, almond oil, n = 17) | Wearing necklace for the daytime during the menstruation Massage was done 10 min once a day for 1 menstrual cycles | NS | Parallel/n.r. |
| Davari (2014) [20] Iran | 150 18–29 Mild or moderate MDQ | (A) Lavender (Inhalation, n = 28) (B) Rosemary (Inhalation, n = 29) (C) Lavender and rosemary (Inhalation, n = 29) | (D) Placebo (n = 30) (E) mefenamic acid (n = 28) † | Twice daily for 3 days the start of menstruation | p < 0.001, in favor of aromatherapy | Parallel/n.r. |
| Iravani (2009) [21] Iran | 108 18–24 Moderate to severe | (A) Zataria Multiflora (Inhalation, 1%, 25 drops, n = 36) (B) Zataria Multiflora (Inhalation, 2%, 25 drops, n = 36) | (C) Placebo (Inhalation, 25 drops, n = 36) | 25 drops every 4 h from the beginning of pain for 3 cycles | p < 0.001 in favor of aromatherapy | Parallel/n.r. |
| Salmalian (2014) [22] Iran | 84 18–24 Mild or moderate | (A) Thymus vulgaris (Inhalation, 25 drops, n = 28), plus placebo capsule | (B) Placebo (Inhalation, n.r., n = 28), plus placebo capsule (C) Ibuprofen (200 mg capsule, n = 28), plus placebo oil | 25 drops every 6 h on 1st day of menstrual cycle and the beginning of pain for 2 consecutive cycles | p < 0.001 in favor of aromatherapy | Triple-blind but no reporting of details Registration while recruiting Parallel/n.r. |
| Bakhtshirin (2015) [23] Iran | 80 18–24 Pain on VAS ≥6 | (A) Lavender oil (Massage abdomen, n = 80) | (B) Placebo (Massage abdomen, n = 80) | 15 min for 1 cycle (2nd and 3rd cycle) by nursing and midwifery students | p < 0.001 in favor of aromatherapy | 1 cycle washout period Cross-over/n.r. |
| Apay (2012) [24] Turkey | 44 20.3 ± 1.1 Pain on VAS ≥6 | (A) Lavender oil (Massage abdomen, n = 44) | (B) Placebo (Massage odorless liquid petrolatum, n = 44) | 15 min for 1 cycle (2nd and 3rd cycle) | p < 0.001 in favor of aromatherapy | 1 cycle washout period Cross-over/n.r. |
| Beiranvand (2015) [25] Iran | 60 18–26 Pain on VAS >5 | (A) Lavender oil (Massage, 2 drops in 2.5 cc of almond oil, 15 min, n = 30) | (B) Placebo (Massage with almond oil only, n = 30) | 48 h before and after menstruation. Massage was done 15 min twice a day for two menstrual cycles above the pubic area | p < 0.001 in favor of aromatherapy | Retrospective registration Parallel/n.r. |
| Sadeghi (2015) [26] Iran | 75 18–35 Pain on VAS ≥5 | (A) Rose oil (Massage, n = 25) | (B) Placebo (Massage, almond oil, unscented, n = 25) (C) Massage only (n = 25) † | 1 time, 5 drops, 15 min on the 1st day of menstruation Self-massage | p < 0.005 in favor of aromatherapy | Registration while recruiting Parallel/None |
| Rizk (2013) [27] Egypt | 120 17–21 Moderate to severe | (A) Peppermint oil (Massage abdomen, n = 40) (B) Ginger oil (massage, n = 40) | (C) Placebo (Massage abdomen, almond, n = 95) | 15 min daily for 5 days for 2 cycles | p < 0.001 in favor of aromatherapy | Parallel/n.r. |
| Marzouk (2013) [28] Egypt | 95 17–20 Pain on VAS ≥6 | (A) Essential oils (cinnamon, clove, rose and lavender, massage, n = 95) | (B) Placebo (Massage abdomen, sweet almond, n = 95) | Once daily, 10 min for 7 days before menstruation by a researcher in a student clinic | p = 0.006 in favor of aromatherapy | 1 cycle washout period Cross-over/n.r. |
| Amiri Farahani (2012) [29] Iran | 90 21 verbal multi-dimensional scoring system ≥2 | (A) Essential oils (Massage, lavender oil 2% (2 drops), peppermint oil 2% (2 drops) in 4 mL of almond oil, n = 30) | (B) Placebo (Massage, almond oil, n = 30) (C) Massage only (n = 30) † | A week before the start of menstrual cycle until the presence of pain, massage at the top of pubic area for 15 min per day. To be repeated for two cycles | p = 0.03 in favor of aromatherapy | Retrospective registration Parallel/n.r. |
| Han (2006) [30] Korea | 75 19–26 Pain on VAS ≥6 | (A) Essential oils (Massage abdomen, lavender, clary sage, rose, n = 25) | (B) Placebo (Massage abdomen, almond oil, n = 25) (C) No treatment (n = 25) † | 1 time, 15 min | p = 0.008 in favor of aromatherapy | Parallel/None |
| Cheon (2013) [31] Korea | 30 Pain on VAS ≥6 | (A) Essential oils (Massage abdomen, nutmeg, fennel, marjoram, n = 15) | (B) Placebo (Massage abdomen, almond oil, n = 15) | 3 mL, 10 min, once a day from 14 days before menstruation to the starting date of next menstruation | p < 0.001 in favor of aromatherapy | Parallel/n.r. |
| Ou (2012) [32] Taiwan | 48 Over 18 Pain on VAS ≥5 | (A) Essential oils cream (Massage abdomen, cream, lavender, clary sage, marjoram, massage, n = 24) | (B) Placebo (Massage abdomen, cream, synthetic fragrance, massage, n = 24) | From the end of the last menstruation continuing to the beginning of the next menstruation Self-massage | p = 0.02 in favor of aromatherapy | Compare the data of 1st cycle (pre) with the data of 2nd cycle (post) Pain measured with NRS did not show significance Parallel/n.r. Pain (VRS) |
| Khorshidi (2003) [33] Iran | 60 17–25 Mild or moderate (AMVMS) | (A) Fennel oil (Oral, 1%, n = 60) (B) Fennel oil (Oral 2%, n = 60) | (C) Placebo (n.r., n = 60) | 1 time, administrated as soon as pain felt | (1) p < 0.05 in favor of aromatherapy (A and B) (2) p < 0.05 in favor of aromatherapy (A and B) | n.r. in details of treatment n.r. in washout period Cross-over/n.r. Pain (LS) |
| Ataollahi (2015) [34] Iran | 110 21.4 Moderate to severe pain (VAS ≥5) | (A) Rosaceous extract (Oral, n = 55) | (B) Placebo (n.r., n = 55) | 10 drops twice daily or the first three days of the cycle for two consecutive cycles | p = 0.001 in favor of aromatherapy | Retrospective registration Parallel/n.r. Pain (VRS) |
| Aromatherapy Compared to Placebo for Pain in Primary Dysmenorrhea | ||||
|---|---|---|---|---|
| Patient or population: patients with pain in primary dysmenorrhea Intervention: Aromatherapy Comparison: placebo Outcomes: Pain | ||||
| Intervention vs. comparator | Anticipated absolute effects * (95% CI) | No. of Participants (studies) | Quality of the evidence (GRADE) | |
| Risk with placebo | Risk with Aromatherapy | |||
| Aromatherapies vs. Placebo | The SMD of in the intervention groups was 0.91 standard deviations lower (1.17 to 0.64 lower) | 1787 (19 studies) | ⊕⊕⊕⊝ moderate a,b | |
| Aromatherapy inhalation vs. Placebo | The SMD in the intervention groups was 1.02 standard deviations lower (1.59 to 0.44 lower) | 704 (7 studies) | ⊕⊕⊕⊝ moderate a,b | |
| Aromatherapy massage vs. Placebo | The SMD of pain in the intervention groups was 0.87 standard deviations lower (1.14 to 0.6 lower) | 793 (11 studies) | ⊕⊕⊕⊝ moderate a,b | |
| Aromatherapy oral use vs. Placebo | The SMD of pain in the intervention groups was 0.61 standard deviations lower (0.91 to 0.3 lower) | 290 (2 studies) | ⊕⊕⊕⊝ moderate a | |
| GRADE Working Group grades of evidence: High quality: We are very confident that the true effect lies close to that of the estimate of the effect. Moderate quality: We are moderately confident in the effect estimate: The true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. Low quality: Our confidence in the effect estimate is limited: The true effect may be substantially different from the estimate of the effect. Very low quality: We have very little confidence in the effect estimate: The true effect is likely to be substantially different from the estimate of effect. | ||||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, M.S.; Lee, H.W.; Khalil, M.; Lim, H.S.; Lim, H.-J. Aromatherapy for Managing Pain in Primary Dysmenorrhea: A Systematic Review of Randomized Placebo-Controlled Trials. J. Clin. Med. 2018, 7, 434. https://doi.org/10.3390/jcm7110434
Lee MS, Lee HW, Khalil M, Lim HS, Lim H-J. Aromatherapy for Managing Pain in Primary Dysmenorrhea: A Systematic Review of Randomized Placebo-Controlled Trials. Journal of Clinical Medicine. 2018; 7(11):434. https://doi.org/10.3390/jcm7110434
Chicago/Turabian StyleLee, Myeong Soo, Hye Won Lee, Mohamed Khalil, Hyun Suk Lim, and Hyun-Ja Lim. 2018. "Aromatherapy for Managing Pain in Primary Dysmenorrhea: A Systematic Review of Randomized Placebo-Controlled Trials" Journal of Clinical Medicine 7, no. 11: 434. https://doi.org/10.3390/jcm7110434
APA StyleLee, M. S., Lee, H. W., Khalil, M., Lim, H. S., & Lim, H.-J. (2018). Aromatherapy for Managing Pain in Primary Dysmenorrhea: A Systematic Review of Randomized Placebo-Controlled Trials. Journal of Clinical Medicine, 7(11), 434. https://doi.org/10.3390/jcm7110434






