Mitochondrial Neurogastrointestinal Encephalomyopathy (MNGIE-MTDPS1)
Abstract
1. Introduction
2. Genetic and Biochemical Findings
3. Clinical and Pathological Aspects
3.1. Classical Phenotype
3.2. Atypical Phenotypes and Clinical Heterogeneity
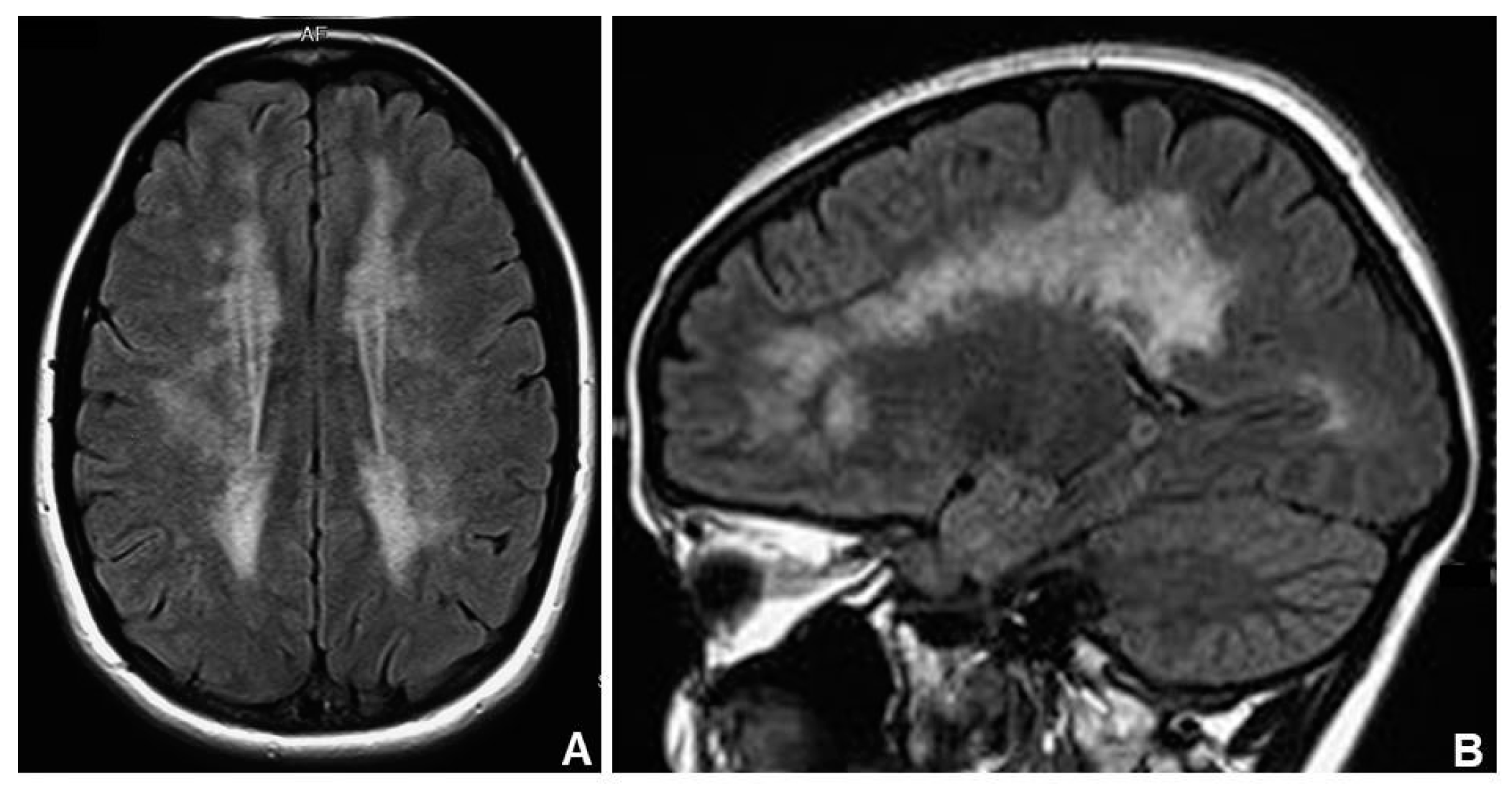
3.3. Other MNGIE-Type Pictures
4. Diagnosis
5. Therapy
6. Conclusions
Conflicts of Interest
References
- Nishino, I.; Spinazzola, A.; Hirano, M. Thymidine phosphorylase gene mutations in MNGIE, a human mitochondrial disorder. Science 1999, 283, 689–692. [Google Scholar] [CrossRef] [PubMed]
- Viscomi, C.; Zeviani, M. mMtDNA-maintenance defects: Syndromes and genes. J. Inherit. Metab. Dis. 2017, 40, 587–599. [Google Scholar] [CrossRef] [PubMed]
- El-Hattab, A.W.; Scaglia, F. SUCLA2-Related Mitochondrial DNA Depletion Syndrome, Encephalomyopathic form with Methylmalonic Aciduria. In GeneReviews®; Adam, M.P., Ardinger, H.H., Pagon, R.A., Wallace, S.E., Bean, L.J.H., Stephens, K., Amemiya, A., Eds.; University of Washington: Seattle, WA, USA, 1993–2018. [Google Scholar]
- Nishino, I.; Spinazzola, A.; Papadimitriou, A.; Hammans, S.; Steiner, I.; Hahn, C.D.; Connolly, A.M.; Verloes, A.; Guimarães, J.; Maillard, I.; et al. Mitochondrial neurogastrointestinal encephalomyopathy: An autosomal recessive disorder due to thymidine phosphorylase mutations. Ann. Neurol. 2000, 47, 792–800. [Google Scholar] [CrossRef]
- Spinazzola, A.; Marti, R.; Nishino, I.; Andreu, A.L.; Naini, A.; Tadesse, S.; Pela, I.; Zammarchi, E.; Donati, M.A.; Oliver, J.A.; et al. Altered thymidine metabolism due to defects of thymidine phosphorylase. J. Biol. Chem. 2002, 277, 4128–4133. [Google Scholar] [CrossRef] [PubMed]
- Kocaefe, Y.C.; Erdem, S.; Ozgüç, M.; Tan, E. Four novel thymidine phosphorylase gene mutations in mitochondrial neurogastrointestinal encephalomyopathy syndrome (MNGIE) patients. Eur. J. Hum. Genet. 2003, 11, 102–104. [Google Scholar] [CrossRef] [PubMed]
- Yadak, R.; Sillevis Smitt, P.; van Gisbergen, M.W.; van Til, N.P.; de Coo, I.I. Mitochondrial Neurogastrointestinal Encephalomyopathy caused by Thymidine Phosphorylase enzyme deficiency: From pathogenesis to emerging therapeutic options. Front. Cell. Neurosci. 2017, 11, 31. [Google Scholar] [CrossRef] [PubMed]
- Lara, M.C.; Valentino, M.L.; Torres-Torronteras, J.; Hirano, M.; Martí, R. Mitochondrial neurogastrointestinal encephalomyopathy (MNGIE): Biochemical features and therapeutic approaches. Biosci. Rep. 2007, 27, 151–163. [Google Scholar] [CrossRef] [PubMed]
- Fairbanks, L.D.; Marinaki, A.M.; Carrey, E.A.; Hammans, S.R.; Duley, J.J. Deoxyuridine accumulation in urine in thymidine phosphorylase deficiency (MNGIE). J. Inherit. Metab. Dis. 2002, 25, 603–604. [Google Scholar] [CrossRef] [PubMed]
- Marti, R.; Spinazzola, A.; Tadesse, S.; Nishino, I.; Nishigaki, Y.; Hirano, M. Definitive diagnosis of mitochondrial neurogastrointestinal encephalomyopathy by biochemical assays. Clin. Chem. 2004, 50, 120–124. [Google Scholar] [CrossRef] [PubMed]
- Hirano, M. Mitochondrial Neurogastrointestinal Encephalopathy Disease. In GeneReviews®; Adam, M.P., Ardinger, H.H., Pagon, R.A., Wallace, S.E., Bean, L.J.H., Stephens, K., Amemiya, A., Eds.; University of Washington: Seattle, WA, USA, 1993–2018. [Google Scholar]
- Filosto, M.; Scarpelli, M.; Tonin, P.; Testi, S.; Cotelli, M.S.; Rossi, M.; Salvi, A.; Grottolo, A.; Vielmi, V.; Todeschini, A.; et al. Pitfalls in diagnosing mitochondrial neurogastrointestinal encephalomyopathy. J. Inherit. Metab. Dis. 2011, 34, 1199–1203. [Google Scholar] [CrossRef] [PubMed]
- Sekino, Y.; Inamori, M.; Yamada, E.; Ohkubo, H.; Sakai, E.; Higurashi, T.; Iida, H.; Hosono, K.; Endo, H.; Nonaka, T.; et al. Characteristics of intestinal pseudo-obstruction in patients with mitochondrial diseases. World J. Gastroenterol. 2012, 18, 4557–4562. [Google Scholar] [CrossRef] [PubMed]
- Granero Castro, P.; Fernández Arias, S.; Moreno Gijón, M.; Alvarez Martínez, P.; Granero Trancón, J.; Álvarez Pérez, J.A.; Lamamie Clairac, E.; González González, J.J. Emergency surgery in chronic intestinal pseudo-obstruction due to mitochondrial neurogastrointestinal encephalomyopathy: Case reports. Int. Arch. Med. 2010, 3, 35. [Google Scholar] [CrossRef] [PubMed]
- Martínez-García, F.A.; Jiménez-Gómez, M.R.; Morsi-Hassan, O.; Fortuna-Alcaraz, M.L.; Sicilia-Guillen, J.; Fernández-Barreiro, A. Acute peritonitis and small bowel diverticula in a patient with mitochondrial neurogastrointestinal encephalomyopathy. Rev. Neurol. 2001, 33, 328–333. [Google Scholar] [PubMed]
- Blondon, H.; Polivka, M.; Joly, F.; Flourie, B.; Mikol, J.; Messing, B. Digestive smooth muscle mitochondrial myopathy in patients with mitochondrial-neuro-gastro-intestinal encephalomyopathy (MNGIE). Gastroenterol. Clin. Biol. 2005, 29, 773–778. [Google Scholar] [CrossRef]
- Giordano, C.; Sebastiani, M.; de Giorgio, R.; Travaglini, C.; Tancredi, A.; Valentino, M.M.; Bellan, M.; Cossarizza, A.; Hirano, M.; d’Amati, G.; et al. Gastrointestinal dysmotility in mitochondrial neurogastrointestinal encephalomyopathy is caused by mitochondrial DNA depletion. Am. J. Pathol. 2008, 173, 1120–1128. [Google Scholar] [CrossRef] [PubMed]
- Szigeti, K.; Wong, L.; Perng, C.; Saifi, G.; Eldin, K.; Adesina, A.; Cass, D.; Hirano, M.; Lupski, J.; Scaglia, F. MNGIE with lack of skeletal muscle involvement and a novel TP splice site mutation. J. Med. Genet. 2004, 41, 125–129. [Google Scholar] [CrossRef] [PubMed]
- Perez-Atayde, A.R. Diagnosis of mitochondrial neurogastrointestinal encephalopathy disease in gastrointestinal biopsies. Hum. Pathol. 2013, 44, 1440–1446. [Google Scholar] [CrossRef] [PubMed]
- Zimmer, V.; Feiden, W.; Becker, G.; Zimmer, A.; Reith, W.; Raedle, J.; Lammert, F.; Zeuzem, S.; Hirano, M.; Menges, M. Absence of the interstitial cell of Cajal network in mitochondrial neurogastrointestinal encephalomyopathy. Neurogastroenterol. Motil. 2009, 21, 627–631. [Google Scholar] [CrossRef] [PubMed]
- Hirano, M.; Martí, R.; Spinazzola, A.; Nishino, I.; Nishigaki, Y. Thymidine phosphorylase deficiency causes MNGIE: An autosomal recessive mitochondrial disorder. Nucleosides Nucleotides Nucleic Acids 2004, 23, 1217–1225. [Google Scholar] [CrossRef] [PubMed]
- Hirano, M.; Nishigaki, Y.; Martí, R. Mitochondrial neurogastrointestinal encephalomyopathy (MNGIE): A disease of two genomes. Neurologist 2004, 10, 8–17. [Google Scholar] [CrossRef] [PubMed]
- Pontarin, G.; Ferraro, P.; Valentino, M.M.; Hirano, M.; Reichard, P.; Bianchi, V. Mitochondrial DNA depletion and thymidine phosphate pool dynamics in a cellular model of mitochondrial neurogastrointestinal encephalomyopathy. J. Biol. Chem. 2006, 281, 22720–22728. [Google Scholar] [CrossRef] [PubMed]
- Man, C.Y.W.; Chinnery, P.P.; Griffiths, P.G. Extraocular muscles have fundamentally distinct properties that make them selectively vulnerable to certain disorders. Neuromuscul. Disord. 2005, 15, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Filosto, M.; Tomelleri, G.; Tonin, P.; Scarpelli, M.; Vattemi, G.; Rizzuto, N.; Padovani, A.; Simonati, A. Neuropathology of mitochondrial diseases. Biosci. Rep. 2007, 27, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Mancuso, M.; Orsucci, D.; Angelini, C.; Bertini, E.; Carelli, V.; Comi, G.G.; Federico, A.; Minetti, C.; Moggio, M.; Mongini, T.; et al. “Mitochondrial neuropathies”: A survey from the large cohort of the Italian Network. Neuromuscul. Disord. 2016, 26, 272–276. [Google Scholar] [CrossRef] [PubMed]
- Said, G.; Lacroix, C.; Planté-Bordeneuve, V.; Messing, B.; Slama, A.; Crenn, P.; Nivelon-Chevallier, A.; Bedenne, L.; Soichot, P.; Manceau, E.; et al. Clinicopathological aspects of the neuropathy of neurogastrointestinal encephalomyopathy (MNGIE) in four patients including two with a Charcot-Marie-Tooth presentation. J. Neurol. 2005, 252, 655–662. [Google Scholar] [CrossRef] [PubMed]
- Bedlack, R.R.; Vu, T.; Hammans, S.; Sparr, S.S.; Myers, B.; Morgenlander, J.; Hirano, M. MNGIE neuropathy: Five cases mimicking chronic inflammatory demyelinating polyneuropathy. Muscle Nerve 2004, 29, 364–368. [Google Scholar] [CrossRef] [PubMed]
- Scarpelli, M.; Ricciardi, G.G.; Beltramello, A.; Zocca, I.; Calabria, F.; Russignan, A.; Zappini, F.; Cotelli, M.M.; Padovani, A.; Tomelleri, G.; et al. The role of brain MRI in mitochondrial neurogastrointestinal encephalomyopathy. Neuroradiol. J. 2013, 26, 520–530. [Google Scholar] [CrossRef] [PubMed]
- Shaibani, A.; Shchelochkov, O.O.; Zhang, S.; Katsonis, P.; Lichtarge, O.; Wong, L.L.; Shinawi, M. Mitochondrial neurogastrointestinal encephalopathy due to mutations in RRM2B. Arch. Neurol. 2009, 66, 1028–1032. [Google Scholar] [CrossRef] [PubMed]
- Tang, S.; Dimberg, E.E.; Milone, M.; Wong, L.J. Mitochondrial neurogastrointestinal encephalomyopathy (MNGIE)-like phenotype: An expanded clinical spectrum of POLG1 mutations. J. Neurol. 2012, 259, 862–868. [Google Scholar] [CrossRef] [PubMed]
- Horváth, R.; Bender, A.; Abicht, A.; Holinski-Feder, E.; Czermin, B.; Trips, T.; Schneiderat, P.; Lochmüller, H.; Klopstock, T. Heteroplasmic mutation in the anticodon-stem of mitochondrial tRNA(Val) causing MNGIE-like gastrointestinal dysmotility and cachexia. J. Neurol. 2009, 256, 810–815. [Google Scholar] [CrossRef] [PubMed]
- Schüpbach, W.W.; Vadday, K.K.; Schaller, A.; Brekenfeld, C.; Kappeler, L.; Benoist, J.J.; Xuan-Huong, C.C.; Burgunder, J.J.; Seibold, F.; Gallati, S.; et al. Mitochondrial neurogastrointestinal encephalomyopathy in three siblings: Clinical, genetic and neuroradiological features. J. Neurol. 2007, 254, 146–153. [Google Scholar] [CrossRef] [PubMed]
- Bianchi, M.M.; Tosetti, M.; Battini, R.; Manca, M.M.; Mancuso, M.; Cioni, G.; Canapicchi, R.; Siciliano, G. Proton MR Spectroscopy of Mitochondrial Diseases: Analysis of Brain Metabolic Abnormalities and Their Possible Diagnostic Relevance. Am. J. Neuroradiol. 2003, 24, 1958–1966. [Google Scholar] [PubMed]
- Rousset, P.; Elmaleh-Bergès, M.; Ogier de Baulny, H.; Viala, J.; Slama, A.; Sebag, G. Mitochondrial neurogastrointestinal encephalomyopathy. J. Neuroradiol. 2008, 35, 121–124. [Google Scholar] [CrossRef] [PubMed]
- Simon, L.; Horoupian, D.; Dorfman, L.; Marks, M.; Herrick, M.M.; Wasserstein, P.; Smith, M.E. Polyneuropathy, ophthalmoplegia, leukoencephalopathy and intestinal pseudo-obstruction: POLIP syndrome. Ann. Neurol. 1990, 28, 349–360. [Google Scholar] [CrossRef] [PubMed]
- Bardosi, A.; Creutzfeldt, W.; DiMauro, S.; Felgenhauer, K.; Friede, R.R.; Goebel, H.H.; Kohlschütter, A.; Mayer, G.; Rahlf, G.; Servidei, S.; et al. Mvo-neuro-gastrointestinal encephalopathy (MNGIE syndrome) due to partial deficiency of cytochrome c oxidase: A new mitochondrial multisystem disorder. Acta Neuropathol. 1987, 74, 248–258. [Google Scholar] [CrossRef] [PubMed]
- Szigeti, K.; Sule, N.; Adesina, A.A.; Armstrong, D.D.; Saifi, G.G.; Bonilla, E.; Hirano, M.; Lupski, J.R. Increased Blood–Brain Barrier Permeability with Thymidine Phosphorylase Deficiency. Ann. Neurol. 2004, 56, 881–886. [Google Scholar] [CrossRef] [PubMed]
- López, L.L.; Akman, H.H.; García-Cazorla, A.; Dorado, B.; Martí, R.; Nishino, I.; Tadesse, S.; Pizzorno, G.; Shungu, D.; Bonilla, E.; et al. Unbalanced deoxynucleotide pools cause mitochondrial DNA instability in thymidine phosphorylase-deficient mice. Hum. Mol. Genet. 2009, 18, 714722. [Google Scholar] [CrossRef] [PubMed]
- Kučerová, L.; Dolina, J.; Dastych, M.; Bartušek, D.; Honzík, T.; Mazanec, J.; Kunovský, L. Mitochondrial neurogastrointestinal encephalomyopathy imitating Crohn’s disease: A rare cause of malnutrition. J. Gastrointestin. Liver Dis. 2018, 27, 321–325. [Google Scholar] [CrossRef] [PubMed]
- Imperatore, N.; Tortora, R.; Gerbino, N.; Caporaso, N.; Rispo, A. Mitochondrial neurogastrointestinal encephalomyopathy (MNGIE) mimicking refractory celiac disease. Dig. Liver Dis. 2017, 49, 1061–1062. [Google Scholar] [CrossRef] [PubMed]
- Needham, M.; Duley, J.; Hammond, S.; Herkes, G.G.; Hirano, M.; Sue, C.M. Mitochondrial disease mimicking Charcot-Marie Tooth disease. BMJ Case Rep. 2009, 2009. [Google Scholar] [CrossRef] [PubMed]
- Bariş, Z.; Eminoğlu, T.; Dalgiç, B.; Tümer, L.; Hasanoğlu, A. Mitochondrial neurogastrointestinal encephalomyopathy (MNGIE): Case report with a new mutation. Eur. J. Pediatr. 2010, 169, 1375–1378. [Google Scholar] [CrossRef] [PubMed]
- Carod-Artal, F.F.; Herrero, M.M.; Lara, M.M.; López-Gallardo, E.; Ruiz-Pesini, E.; Martí, R.; Montoya, J. Cognitive dysfunction and hypogonadotrophic hypogonadism in a Brazilian patient with mitochondrial neurogastrointestinal encephalomyopathy and a novel ECGF1 mutation. Eur. J. Neurol. 2007, 14, 581–585. [Google Scholar] [CrossRef] [PubMed]
- Cardaioli, E.; Da Pozzo, P.; Malfatti, E.; Battisti, C.; Gallus, G.G.; Gaudiano, C.; Macucci, M.; Malandrini, A.; Margollicci, M.; Rubegni, A.; et al. A second MNGIE patient without typical mitochondrial skeletal muscle involvement. Neurol. Sci. 2010, 31, 491–494. [Google Scholar] [CrossRef] [PubMed]
- Massa, R.; Tessa, A.; Margollicci, M.; Micheli, V.; Romigi, A.; Tozzi, G.; Terracciano, C.; Piemonte, F.; Bernardi, G.; Santorelli, F.M. Late-onset MNGIE without peripheral neuropathy due to incomplete loss of thymidine phosphorylase activity. Neuromuscul. Disord. 2009, 19, 837–840. [Google Scholar] [CrossRef] [PubMed]
- Kalkan, I.I.; Tayfur, O.; Oztaş, E.; Beyazit, Y.; Yildiz, H.; Tunç, B. A novel finding in MNGIE (mitochondrial neurogastrointestinal encephalomyopathy): Hypergonadotropic hypogonadism. Hormones 2012, 11, 377–379. [Google Scholar] [CrossRef] [PubMed]
- Hiraki, N.; Udaka, T.; Yamamoto, H.; Kadokawa, Y.; Ohkubo, J.; Suzuki, H. Mitochondrial neurogastrointestinal encephalomyopathy associated with progressive hearing loss. J. Laryngol. Otol. 2010, 124, 1007–1009. [Google Scholar] [CrossRef] [PubMed]
- Garone, C.; Tadesse, S.; Hirano, M. Clinical and genetic spectrum of mitochondrial neurogastrointestinal encephalomyopathy. Brain 2011, 134, 3326–3332. [Google Scholar] [CrossRef] [PubMed]
- Valentino, M.M.; Martí, R.; Tadesse, S.; López, L.L.; Manes, J.J.; Lyzak, J.; Hahn, A.; Carelli, V.; Hirano, M. Thymidine and deoxyuridine accumulate in tissues of patients with mitochondrial neurogastrointestinal encephalomyopathy (MNGIE). FEBS Lett. 2007, 581, 3410–3414. [Google Scholar] [CrossRef] [PubMed]
- Scarpelli, M.; Todeschini, A.; Rinaldi, F.; Rota, S.; Padovani, A.; Filosto, M. Strategies for treating mitochondrial disorders: An update. Mol. Genet. Metab. 2014, 113, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Scarpelli, M.; Cotelli, M.M.; Mancuso, M.; Tomelleri, G.; Tonin, P.; Baronchelli, C.; Vielmi, V.; Gregorelli, V.; Todeschini, A.; Padovani, A.; et al. Current options in the treatment of mitochondrial diseases. Recent Pat. CNS Drug Discov. 2010, 5, 203–209. [Google Scholar] [CrossRef] [PubMed]
- Scarpelli, M.; Todeschini, A.; Volonghi, I.; Padovani, A.; Filosto, M. Mitochondrial diseases: Advances and issues. Appl. Clin. Genet. 2017, 10, 21–26. [Google Scholar] [CrossRef] [PubMed]
- Lara, M.M.; Weiss, B.; Illa, I.; Madoz, P.; Massuet, L.; Andreu, A.A.; Valentino, M.M.; Anikster, Y.; Hirano, M.; Martí, R. Infusion of platelets transiently reduces nucleoside overload in MNGIE. Neurology 2006, 67, 1461–1463. [Google Scholar] [CrossRef] [PubMed]
- Yavuz, H.; Ozel, A.; Christensen, M.; Christensen, E.; Schwartz, M.; Elmaci, M.; Vissing, J. Treatment of mitochondrial neurogastrointestinal encephalomyopathy with dialysis. Arch. Neurol. 2007, 64, 435–438. [Google Scholar] [CrossRef] [PubMed]
- Röeben, B.; Marquetand, J.; Bender, B.; Billing, H.; Haack, T.T.; Sanchez-Albisua, I.; Schöls, L.; Blom, H.H.; Synofzik, M. Hemodialysis in MNGIE transiently reduces serum and urine levels of thymidine and deoxyuridine, but not CSF levels and neurological function. Orphanet. J. Rare Dis. 2017, 12, 135. [Google Scholar] [CrossRef] [PubMed]
- Bax, B.B.; Bain, M.M.; Scarpelli, M.; Filosto, M.; Tonin, P.; Moran, N. Clinical and biochemical improvements in a patient with MNGIE following enzyme replacement. Neurology 2013, 81, 1269–1271. [Google Scholar] [CrossRef] [PubMed]
- Halter, J.J.; Michael, W.; Schüpbach, M.; Mandel, H.; Casali, C.; Orchard, K.; Collin, M.; Valcarcel, D.; Rovelli, A.; Filosto, M.; et al. Allogeneic haematopoietic stem cell transplantation for mitochondrial neurogastrointestinal encephalomyopathy. Brain 2015, 138, 2847–2858. [Google Scholar] [CrossRef] [PubMed]
- Filosto, M.; Scarpelli, M.; Tonin, P.; Lucchini, G.; Pavan, F.; Santus, F.; Parini, R.; Donati, M.M.; Cotelli, M.M.; Vielmi, V.; et al. Course and management of allogeneic stem cell transplantation in patients with mitochondrial neurogastrointestinal encephalomyopathy. J. Neurol. 2012, 259, 2699–2706. [Google Scholar] [CrossRef] [PubMed]
- Halter, J.; Schüpbach, W.; Casali, C.; Elhasid, R.; Fay, K.; Hammans, S.; Illa, I.; Kappeler, L.; Krähenbühl, S.; Lehmann, T.; et al. Allogeneic hematopoietic SCT as treatment option for patients with mitochondrial neurogastrointestinal encephalomyopathy (MNGIE): A consensus conference proposal for a standardized approach. Bone Marrow Transplant. 2011, 46, 330–337. [Google Scholar] [CrossRef] [PubMed]
- Hirano, M.; Martí, R.; Casali, C.; Tadesse, S.; Uldrick, T.; Fine, B.; Escolar, D.D.; Valentino, M.M.; Nishino, I.; Hesdorffer, C.; et al. Allogeneic stem cell transplantation corrects biochemical derangements in MNGIE. Neurology 2006, 67, 1458–1460. [Google Scholar] [CrossRef] [PubMed]
- Scarpelli, M.; Russignan, A.; Zombor, M.; Bereczki, C.; Zappini, F.; Buono, R.; Bax, B.B.; Padovani, A.; Tonin, P.; Filosto, M. Poor Outcome in a Mitochondrial Neurogastrointestinal Encephalomyopathy Patient with a Novel TYMP Mutation: The Need for Early Diagnosis. Case Rep. Neurol. 2012, 4, 248–253. [Google Scholar] [CrossRef] [PubMed]
- Yadak, R.; Cabrera-Pérez, R.; Torres-Torronteras, J.; Bugiani, M.; Haeck, J.J.; Huston, M.M.; Bogaerts, E.; Goffart, S.; Jacobs, E.E.; Stok, M.; et al. Preclinical Efficacy and Safety Evaluation of Hematopoietic Stem Cell Gene Therapy in a Mouse Model of MNGIE. Mol. Ther. Methods Clin. Dev. 2018, 8, 152–165. [Google Scholar] [CrossRef] [PubMed]
- Torres-Torronteras, J.; Cabrera-Pérez, R.; Barba, I.; Costa, C.; de Luna, N.; Andreu, A.A.; Barquinero, J.; Hirano, M.; Cámara, Y.; Martí, R. Long-Term Restoration of Thymidine Phosphorylase Function and Nucleoside Homeostasis Using Hematopoietic Gene Therapy in a Murine Model of Mitochondrial Neurogastrointestinal Encephalomyopathy. Hum. Gene Ther. 2016, 27, 656–667. [Google Scholar] [CrossRef] [PubMed]
- Torres-Torronteras, J.; Cabrera-Pérez, R.; Vila-Julià, F.; Viscomi, C.; Cámara, Y.; Hirano, M.; Zeviani, M.; Martí, R. Long-Term Sustained Effect of Liver-Targeted Adeno-Associated Virus Gene Therapy for Mitochondrial Neurogastrointestinal Encephalomyopathy. Hum. Gene Ther. 2018, 29, 708–718. [Google Scholar] [CrossRef] [PubMed]
- De Giorgio, R.; Pironi, L.; Rinaldi, R.; Boschetti, E.; Caporali, L.; Capristo, M.; Casali, C.; Cenacchi, G.; Contin, M.; D’Angelo, R.; et al. Liver transplantation for mitochondrial neurogastrointestinal encephalomyopathy. Ann. Neurol. 2016, 80, 448–455. [Google Scholar] [CrossRef] [PubMed]
| Phenotype | Gene | Number | Other Clinical Findings |
|---|---|---|---|
| Hepatocerebral | DGUOK | 3 | |
| POLG | 4A | Alpers type | |
| MPV17 | 6 | ||
| TWNK (C10orf2) | 7 | ||
| TFAM | 15 | ||
| Encephalo-myopathic | SUCLA2 | 5 | associated with methylmalonic aciduria |
| FBXL4 | 13 | ||
| SUCLG1 | 9 | associated with methylmalonic aciduria | |
| RRM2B | 8A | associated with renal tubulopathy | |
| OPA1 | 14 | encephalocardiomyopathic type | |
| Neurogastro-intestinal | TYMP | 1 | MNGIE type |
| POLG | 4B | MNGIE type | |
| RRM2B | 8B | MNGIE type | |
| Myopathic | TK2 | 2 | |
| AGK | 10 | cardiomyopathic type | |
| MGME1 | 11 | ||
| SLC25A4 | 12A | autosomal dominant cardiomyopathic type | |
| SLC25A4 | 12B | autosomal recessive cardiomyopathic type |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Filosto, M.; Cotti Piccinelli, S.; Caria, F.; Gallo Cassarino, S.; Baldelli, E.; Galvagni, A.; Volonghi, I.; Scarpelli, M.; Padovani, A. Mitochondrial Neurogastrointestinal Encephalomyopathy (MNGIE-MTDPS1). J. Clin. Med. 2018, 7, 389. https://doi.org/10.3390/jcm7110389
Filosto M, Cotti Piccinelli S, Caria F, Gallo Cassarino S, Baldelli E, Galvagni A, Volonghi I, Scarpelli M, Padovani A. Mitochondrial Neurogastrointestinal Encephalomyopathy (MNGIE-MTDPS1). Journal of Clinical Medicine. 2018; 7(11):389. https://doi.org/10.3390/jcm7110389
Chicago/Turabian StyleFilosto, Massimiliano, Stefano Cotti Piccinelli, Filomena Caria, Serena Gallo Cassarino, Enrico Baldelli, Anna Galvagni, Irene Volonghi, Mauro Scarpelli, and Alessandro Padovani. 2018. "Mitochondrial Neurogastrointestinal Encephalomyopathy (MNGIE-MTDPS1)" Journal of Clinical Medicine 7, no. 11: 389. https://doi.org/10.3390/jcm7110389
APA StyleFilosto, M., Cotti Piccinelli, S., Caria, F., Gallo Cassarino, S., Baldelli, E., Galvagni, A., Volonghi, I., Scarpelli, M., & Padovani, A. (2018). Mitochondrial Neurogastrointestinal Encephalomyopathy (MNGIE-MTDPS1). Journal of Clinical Medicine, 7(11), 389. https://doi.org/10.3390/jcm7110389






