Emotional Prosody Processing in Schizophrenic Patients: A Selective Review and Meta-Analysis
Abstract
1. Introduction
2. Method
2.1. Inclusion and Exclusion Criteria
2.1.1. Types of Studies
- (a)
- cross-sectional studies comparing schizophrenic patients versus healthy controls or/and versus a clinical comparison group (i.e., patients with a different psychiatric disorder or neurological condition);
- (b)
- longitudinal studies comparing participants’ performance before versus after schizophrenia.
2.1.2. Types of Participants
2.1.3. Types of Measures
2.2. Searching Strategies for Identification of Studies
2.3. Data Collection and Analysis
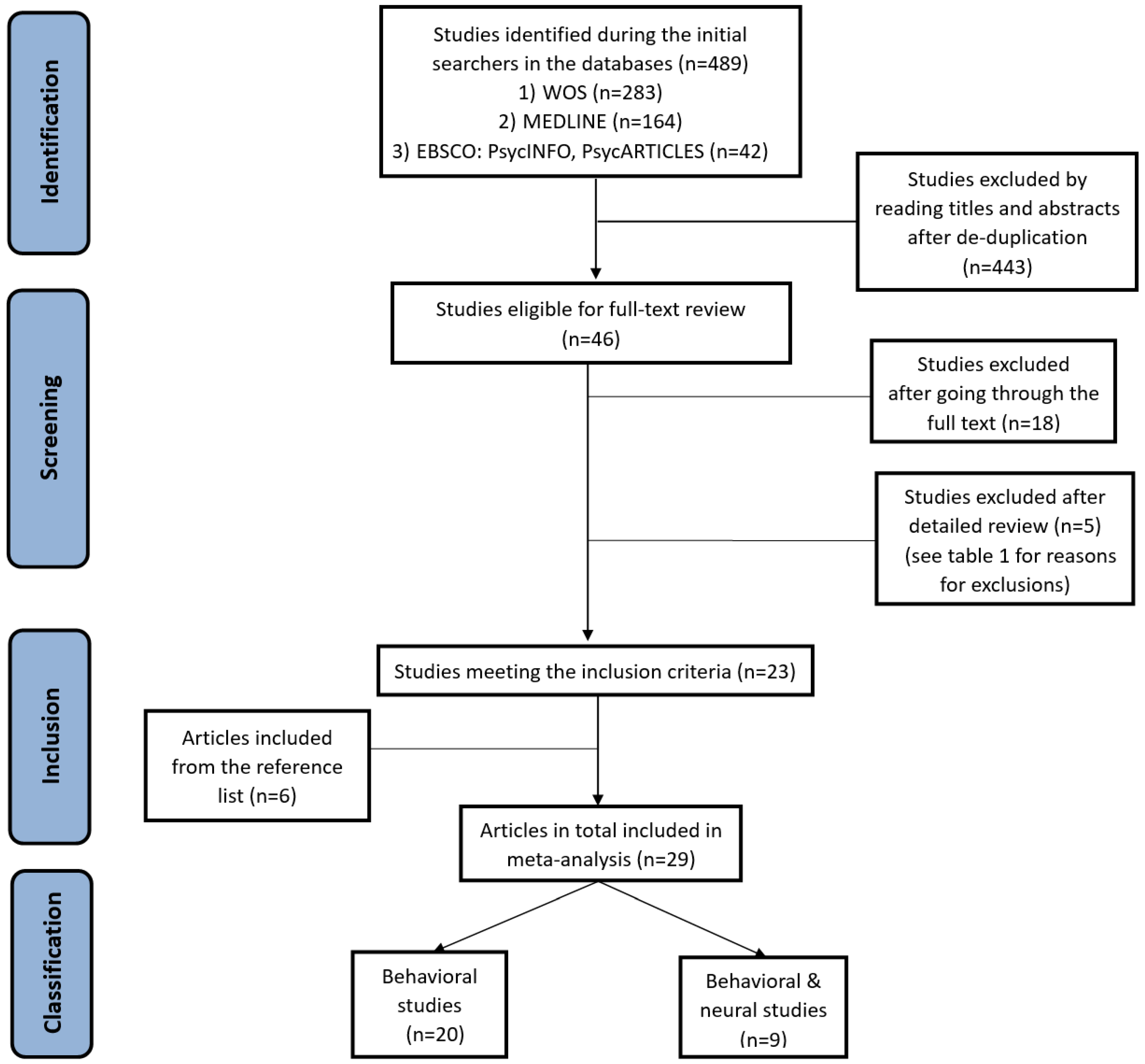
2.3.1. Selection of Studies
2.3.2. Data Extraction
2.3.3. Data Synthesis
3. Results
3.1. General Characteristics of the Included Studies
3.1.1. Study Topics
3.1.2. Task Paradigms, Stimulus Presentation and Response Options
3.1.3. Study Participants
3.1.4. Reporting of Key Demographic and Clinical Information of Schizophrenic Patients
3.1.5. Statistical Analysis Methods and Reporting of the Results
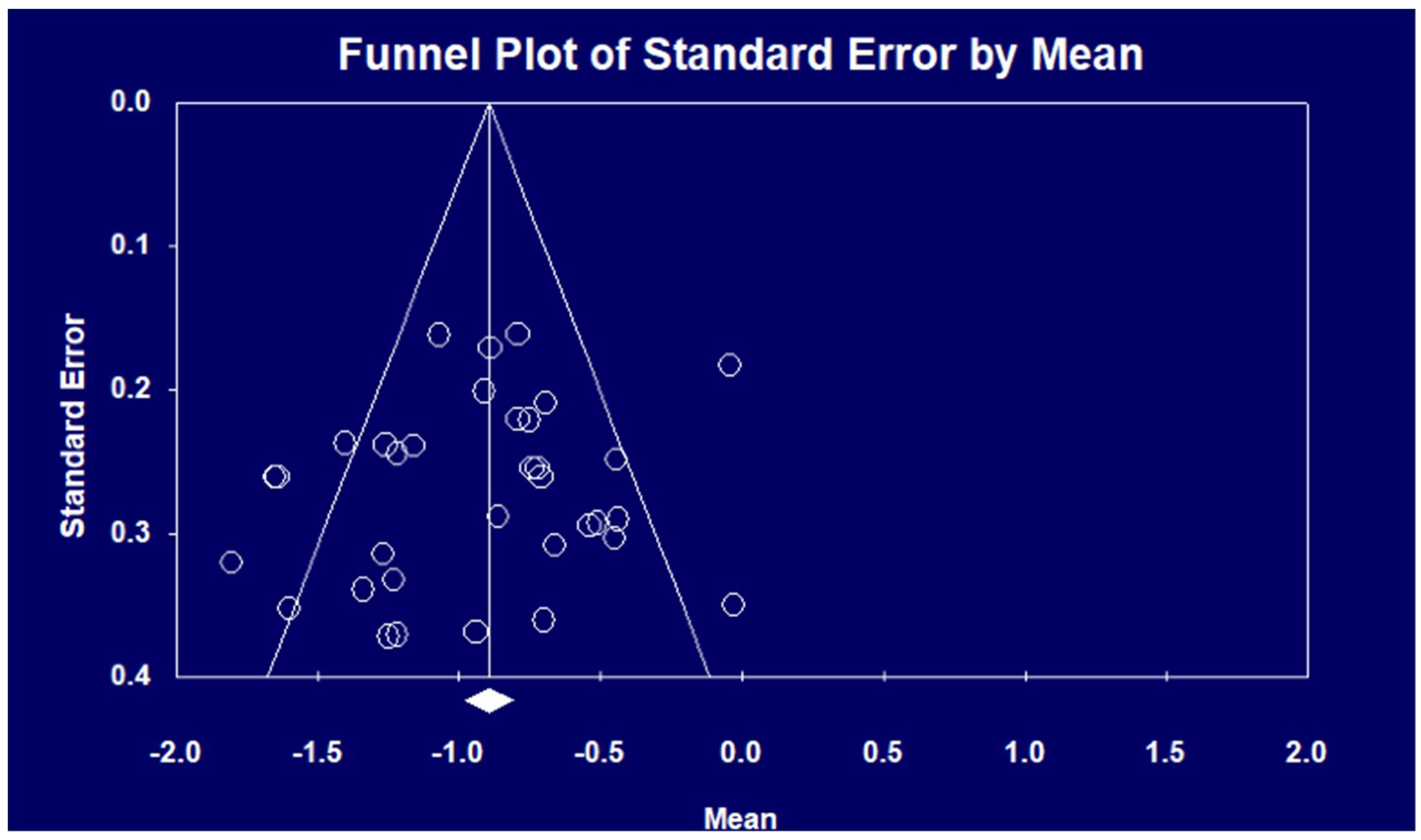
3.2. Performance of Emotional Prosody Processing
3.3. Influential Factors of Emotional Prosody Processing
3.3.1. Factors Related to Participants
Demographic Factors
Cognitive Factors
Clinical Factors
3.3.2. Factors Related to Experiments
Task-Related Factors
Stimulus-Related Factors
3.4. Neural Mechanisms of Emotional Prosody Processing
3.4.1. Hemispheric Processing of Emotional Prosody
3.4.2. Spatial Localization of Brain Networks for Processing Emotional Prosody
3.4.3. Temporal Processing of Emotional Prosody
4. Discussion
4.1. Implications of the Selected Studies
4.1.1. Implications for Practice
4.1.2. Implications for Research
4.2. Limitations of This Selective Review
5. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Schizophrenia Fact Sheet. 2018. Available online: www.who.int/news-room/fact-sheets/detail/schizophrenia (accessed on 15 October 2018).
- Shenton, M.E.; Dickey, C.C.; Frumin, M.; McCarley, R.W. A review of MRI findings in schizophrenia. Schizophr. Res. 2001, 49, 1–52. [Google Scholar] [CrossRef]
- Wible, C.G.; Preus, A.P.; Hashimoto, R. A cognitive neuroscience view of schizophrenic symptoms: Abnormal activation of a system for social perception and communication. Brain Imaging Behav. 2009, 3, 85–110. [Google Scholar] [CrossRef] [PubMed]
- Edwards, J.; Jackson, H.J.; Pattison, P.E. Emotion recognition via facial expression and affective prosody in schizophrenia: A methodological review. Clin. Psychol. Rev. 2002, 22, 789–832. [Google Scholar] [CrossRef]
- Mueser, K.T.; Doonan, R.; Penn, D.L.; Blanchard, J.J.; Bellack, A.S.; Nishith, P.; DeLeon, J. Emotion recognition and social competence in chronic schizophrenia. J. Abnorm. Psychol. 1996, 105, 271–275. [Google Scholar] [CrossRef] [PubMed]
- Penn, D.L.; Sanna, L.J.; Roberts, D.L. Social cognition in schizophrenia: An overview. Schizophr. Bull. 2008, 34, 408–411. [Google Scholar] [CrossRef] [PubMed]
- Pinkham, A.E.; Penn, D.L.; Perkins, D.O.; Lieberman, J. Implications for the neural basis of social cognition for the study of schizophrenia. Am. J. Psychiatry 2003, 160, 815–824. [Google Scholar] [CrossRef] [PubMed]
- Fett, A.K.J.; Viechtbauer, W.; Penn, D.L.; van Os, J.; Krabbendam, L. The relationship between neurocognition and social cognition with functional outcomes in schizophrenia: A meta-analysis. Neurosci. Biobehav. Rev. 2011, 35, 573–588. [Google Scholar] [CrossRef] [PubMed]
- Sterea, R. The relationship between social cognition and functional outcomes in schizophrenia. Procedia Soc. Behav. Sci. 2015, 187, 256–260. [Google Scholar] [CrossRef]
- Penn, D.L.; Spaulding, W.; Reed, D.; Sullivan, M. The relationship of social cognition to ward behavior in chronic schizophrenia. Schizophr. Res. 1996, 20, 327–335. [Google Scholar] [CrossRef]
- Hoertnagl, C.M.; Yalcin-Siedentopf, N.; Baumgartner, S.; Biedermann, F.; Deisenhammer, E.A.; Hausmann, A.; Kaufmann, A.; Kemmler, G.; Mühlbacher, M.; Rauch, A.; et al. Affective prosody perception in symptomatically remitted patients with schizophrenia and bipolar disorder. Schizophr. Res. 2014, 158, 100–104. [Google Scholar] [CrossRef] [PubMed]
- Varga, E.; Herold, R.; Tényi, T.; Bugya, T. Social Cognition Analyzer Application (SCAN)—A new approach to analyse social cognition in schizophrenia. Eur. Neuropsychopharm. 2018, 28, S55–S56. [Google Scholar] [CrossRef]
- Varga, E.; Endre, S.; Bugya, T.; Tényi, T.; Herold, R. Community-based psychosocial treatment has an impact on social processing and functional outcome in schizophrenia. Front. Psychiatry 2018, 9, 247. [Google Scholar] [CrossRef] [PubMed]
- Revell, E.R.; Neill, J.C.; Harte, M.; Khan, Z.; Drake, R.J. A systematic review and meta-analysis of cognitive remediation in early schizophrenia. Schizophr. Res. 2015, 168, 213–222. [Google Scholar] [CrossRef] [PubMed]
- Alba-Ferrara, L.; De Erausquin, G.A.; Hirnstein, M.; Weis, S.; Hausmann, M. Emotional prosody modulates attention in schizophrenia patients with hallucinations. Front. Hum. Neurosci. 2013, 7, 59. [Google Scholar] [CrossRef] [PubMed]
- Minzenberg, M.J.; Poole, J.H.; Vinogradov, S. Social-emotion recognition in borderline personality disorder. Compr. Psychiatry 2006, 47, 468–474. [Google Scholar] [CrossRef] [PubMed]
- Rosenblau, G.; Kliemann, D.; Dziobek, I.; Heekeren, H.R. Emotional prosody processing in autism spectrum disorder. Soc. Cogn. Affect. Neurosci. 2017, 12, 224–239. [Google Scholar] [CrossRef] [PubMed]
- Regenbogen, C.; Kellermann, T.; Seubert, J.; Schneider, D.A.; Gur, R.E.; Derntl, B.; Schneider, F.; Habel, U. Neural responses to dynamic multimodal stimuli and pathology-specific impairments of social cognition in schizophrenia and depression. Br. J. Psychiatry 2015, 206, 198–205. [Google Scholar] [CrossRef] [PubMed]
- Kantrowitz, J.T.; Hoptman, M.J.; Leitman, D.I.; Silipo, G.; Javitt, D.C. The 5% difference: Early sensory processing predicts sarcasm perception in schizophrenia and schizoaffective disorder. Psychol. Med. 2014, 44, 25–36. [Google Scholar] [CrossRef] [PubMed]
- Haskins, B.; Shutty, M.S.; Kellogg, E. Affect processing in chronically psychotic patients: Development of a reliable assessment tool. Schizophr. Res. 1995, 15, 291–297. [Google Scholar] [CrossRef]
- Edwards, J.; Pattison, P.E.; Jackson, H.J.; Wales, R.J. Facial affect and affective prosody recognition in first-episode schizophrenia. Schizophr. Res. 2001, 48, 235–253. [Google Scholar] [CrossRef]
- Kucharska-Pietura, K.; David, A.S.; Masiak, M.; Phillips, M.L. Perception of facial and vocal affect by people with schizophrenia in early and late stages of illness. Br. J. Psychiatry 2005, 187, 523–528. [Google Scholar] [CrossRef] [PubMed]
- Pinkham, A.E.; Penn, D.L. Neurocognitive and social cognitive predictors of interpersonal skill in schizophrenia. Psychiatry Res. 2006, 143, 167–178. [Google Scholar] [CrossRef] [PubMed]
- Hoekert, M.; Kahn, R.S.; Pijnenborg, M.; Aleman, A. Impaired recognition and expression of emotional prosody in schizophrenia: Review and meta-analysis. Schizophr. Res. 2007, 96, 135–145. [Google Scholar] [CrossRef] [PubMed]
- Ekman, P. An Argument for Basic Emotions. Cogn. Emot. 1992, 6, 169–200. [Google Scholar] [CrossRef]
- World Health Organization. The ICD-10 Classification of Mental and Behavioural Disorders: Clinical Description and Diagnostic Guidelines; World Health Organization: Geneva, Switzerland, 1992; pp. 84–97. [Google Scholar]
- Kay, S.R.; Fiszbein, A.; Opler, L.A. The positive and negative syndrome scale (PANSS) for schizophrenia. Schizophr. Bull. 1987, 13, 261–276. [Google Scholar] [CrossRef] [PubMed]
- Ross, E.D.; Orbelo, D.M.; Cartwright, J.; Hansel, S.; Burgard, M.; Testa, J.A.; Buck, R. Affective-prosodic deficits in schizophrenia: Profiles of patients with brain damage and comparison with relation to schizophrenic symptoms. J. Neurol. Neurosurg. Psychiatry 2001, 70, 597–604. [Google Scholar] [CrossRef] [PubMed]
- Hooker, C.; Park, S. Emotion processing and its relationship to social functioning in schizophrenia patients. Psychiatry Res. 2002, 112, 41–50. [Google Scholar] [CrossRef]
- Leitman, D.I.; Foxe, J.J.; Butler, P.D.; Saperstein, A.; Revheim, N.; Javitt, D.C. Sensory contributions to impaired prosodic processing in schizophrenia. Biol. Psychiatry 2005, 58, 56–61. [Google Scholar] [CrossRef] [PubMed]
- Rossell, S.L.; Boundy, C.L. Are auditory-verbal hallucinations associated with auditory affective processing deficits? Schizophr. Res. 2005, 78, 95–106. [Google Scholar] [CrossRef] [PubMed]
- Leitman, D.I.; Hoptman, M.J.; Foxe, J.J.; Saccente, E.; Wylie, G.R.; Nierenberg, J.; Jalbrzikowski, M.; Lim, K.O.; Javitt, D.C. The neural substrates of impaired prosodic detection in schizophrenia and its sensorial antecedents. Am. J. Psychiatry 2007, 164, 474–482. [Google Scholar] [CrossRef] [PubMed]
- Shea, T.L.; Sergejew, A.A.; Burnham, D.; Jones, C.; Rossell, S.L.; Copolov, D.L.; Egan, G.F. Emotional prosodic processing in auditory hallucinations. Schizophr. Res. 2007, 90, 214–220. [Google Scholar] [CrossRef] [PubMed]
- Chan, C.; Wong, R.; Wang, K.; Lee, T. Emotion recognition in Chinese people with schizophrenia. Psychiatry Res. 2008, 157, 67–76. [Google Scholar] [CrossRef] [PubMed]
- Scholten, M.R.; Aleman, A.; Kahn, R.S. The processing of emotional prosody and semantics in schizophrenia: Relationship to gender and IQ. Psychol. Med. 2008, 38, 887–898. [Google Scholar] [CrossRef] [PubMed]
- Bach, D.R.; Buxtorf, K.; Grandjean, D.; Strik, W.K. The influence of emotion clarity on emotional prosody identification in paranoid schizophrenia. Psychol. Med. 2009, 39, 927–938. [Google Scholar] [CrossRef] [PubMed]
- Bach, D.R.; Herdener, M.; Grandjean, D.; Sander, D.; Seifritz, E.; Strik, W.K. Altered lateralisation of emotional prosody processing in schizophrenia. Schizophr. Res. 2009, 110, 180–187. [Google Scholar] [CrossRef] [PubMed]
- Roux, P.; Christophe, A.; Passerieux, C. The emotional paradox: Dissociation between explicit and implicit processing of emotional prosody in schizophrenia. Neuropsychologia 2010, 48, 3642–3649. [Google Scholar] [CrossRef] [PubMed]
- Gold, R.; Butler, P.; Revheim, N.; Leitman, D.I.; Hansen, J.A.; Gur, R.C.; Kantrowitz, J.T.; Laukka, P.; Juslin, P.N.; Silipo, G.S.; et al. Auditory emotion recognition impairments in schizophrenia: Relationship to acoustic features and cognition. Am. J. Psychiatry 2012, 169, 424–432. [Google Scholar] [CrossRef] [PubMed]
- Ito, F.; Matsumoto, K.; Miyakoshi, T.; Ohmuro, N.; Uchida, T.; Matsuoka, H. Emotional processing during speech communication and positive symptoms in schizophrenia. Psychiatry Clin. Neurosci. 2013, 67, 526–531. [Google Scholar] [CrossRef] [PubMed]
- Iwashiro, N.; Yahata, N.; Kawamuro, Y.; Kasai, K.; Yamasue, H. Aberrant interference of auditory negative words on attention in patients with schizophrenia. PLoS ONE 2013, 8, e83201. [Google Scholar] [CrossRef] [PubMed]
- Jahshan, C.; Wynn, J.K.; Green, M.F. Relationship between auditory processing and affective prosody in schizophrenia. Schizophr. Res. 2013, 143, 348–353. [Google Scholar] [CrossRef] [PubMed]
- Kantrowitz, J.T.; Leitman, D.I.; Lehrfeld, J.M.; Laukka, P.; Juslin, P.N.; Butler, P.D.; Silipo, G.; Javitt, D.C. Reduction in tonal discriminations predicts receptive emotion processing deficits in schizophrenia and schizoaffective disorder. Schizophr. Bull. 2013, 39, 86–93. [Google Scholar] [CrossRef] [PubMed]
- McLachlan, N.M.; Phillips, D.S.; Rossell, S.L.; Wilson, S.J. Auditory processing and hallucinations in schizophrenia. Schizophr. Res. 2013, 150, 380–385. [Google Scholar] [CrossRef] [PubMed]
- Pinheiro, A.P.; Del, R.E.; Mezin, J.; Nestor, P.G.; Rauber, A.; Mccarley, R.W.; Gonçalves, O.F.; Niznikiewicz, M.A. Sensory-based and higher-order operations contribute to abnormal emotional prosody processing in schizophrenia: An electrophysiological investigation. Psychol. Med. 2013, 43, 603–618. [Google Scholar] [CrossRef] [PubMed]
- Tseng, H.H.; Chen, S.H.; Liu, C.M.; Howes, O.; Huang, Y.L.; Hsieh, M.H.; Liu, C.C.; Shan, J.C.; Lin, Y.T.; Hwu, H.G. Facial and prosodic emotion recognition deficits associate with specific clusters of psychotic symptoms in schizophrenia. PLoS ONE 2013, 8, e66571. [Google Scholar] [CrossRef] [PubMed]
- Brazo, P.; Beaucousin, V.; Lecardeur, L.; Razafimandimby, A.; Dollfus, S. Social cognition in schizophrenic patients: The effect of semantic content and emotional prosody in the comprehension of emotional discourse. Front. Psychiatry 2014, 5, 120. [Google Scholar] [CrossRef] [PubMed]
- Dondaine, T.; Robert, G.; Péron, J.; Grandjean, D.; Vérin, M.; Drapier, D.; Millet, B. Biases in facial and vocal emotion recognition in chronic schizophrenia. Front. Psychol. 2014, 5, 900. [Google Scholar] [CrossRef] [PubMed]
- Kantrowitz, J.T.; Scaramello, N.; Jakubovitz, A.; Lehrfeld, J.M.; Laukka, P.; Elfenbein, H.A.; Silipo, G.; Javitt, D.C. Amusia and protolanguage impairments in schizophrenia. Psychol. Med. 2014, 44, 2739–2748. [Google Scholar] [CrossRef] [PubMed]
- Müller, V.I.; Kellermann, T.S.; Seligman, S.C.; Turetsky, B.I.; Eickhoff, S.B. Modulation of affective face processing deficits in schizophrenia by congruent emotional sounds. Soc. Cogn. Affect. Neurosci. 2014, 9, 436–444. [Google Scholar] [CrossRef] [PubMed]
- Pinheiro, A.P.; Rezaii, N.; Rauber, A.; Liu, T.; Nestor, P.G.; Mccarley, R.W.; Gonçalves, O.F.; Niznikiewicz, M.A. Abnormalities in the processing of emotional prosody from single words in schizophrenia. Schizophr. Res. 2014, 152, 235–241. [Google Scholar] [CrossRef] [PubMed]
- Corcoran, C.M.; Keilp, J.G.; Kayser, J.; Klim, C.; Butler, P.D.; Bruder, G.E.; Gur, R.C.; Javitt, D.C. Emotion recognition deficits as predictors of transition in individuals at clinical high risk for schizophrenia: A neurodevelopmental perspective. Psychol. Med. 2015, 45, 2959–2973. [Google Scholar] [CrossRef] [PubMed]
- Kantrowitz, J.T.; Hoptman, M.J.; Leitman, D.I.; Morenoortega, M.; Lehrfeld, J.M.; Dias, E.; Sehatpour, P.; Laukka, P.; Silipo, G.; Javitt, D.C. Neural substrates of auditory emotion recognition deficits in schizophrenia. J. Neurosci. 2015, 35, 14909–14921. [Google Scholar] [CrossRef] [PubMed]
- Weisgerber, A.; Vermeulen, N.; Peretz, I.; Samson, S.; Philippot, P.; Maurage, P.; De Graeuwe D’Aoust, C.; De Jaegere, A.; Delatte, B.; Gillain, B.; et al. Facial, vocal and musical emotion recognition is altered in paranoid schizophrenic patients. Psychiatry Res. 2015, 229, 188–193. [Google Scholar] [CrossRef] [PubMed]
- Razafimandimby, A.; Hervé, P.Y.; Marzloff, V.; Brazo, P.; Tzourio-Mazoyer, N.; Dollfus, S. Functional deficit of the medial prefrontal cortex during emotional sentence attribution in schizophrenia. Schizophr. Res. 2016, 178, 86–93. [Google Scholar] [CrossRef] [PubMed]
- Herniman, S.E.; Allott, K.A.; Killackey, E.; Hester, R.; Cotton, S.M. The effect of comorbid depression on facial and prosody emotion recognition in first-episode schizophrenia spectrum. J. Affect. Disord. 2017, 208, 223–229. [Google Scholar] [CrossRef] [PubMed]
- Pawełczyk, A.; Kotlickaantczak, M.; Łojek, E.; Ruszpel, A.; Pawełczyk, T. Schizophrenia patients have higher-order language and extralinguistic impairments. Schizophr. Res. 2018, 192, 274–280. [Google Scholar] [CrossRef] [PubMed]
- Wilson, D. B. Practical meta-analysis effect size calculator. Available online: http://www.campbellcollaboration.org/escalc/html/EffectSizeCalculator-SMD-main.php (accessed on 15 October 2018).
- Borenstein, M.; Hedges, L.V.; Higgins, J.P.T.; Rothstein, H.R. Comprehensive Meta-Analysis 2.0; Biostat: Englewood, NJ, USA, 2005. [Google Scholar]
- Borenstein, M.; Hedges, L.V.; Higgins, J.P.T.; Rothstein, H.R. A basic introduction to fixed-effect and random-effects models for meta-analysis. Res. Synth. Methods 2010, 1, 97–111. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences, 2nd ed.; Lawrence Earlbaum Associates: Hillsdale, NJ, USA, 1988; pp. 40–41. ISBN 0-8058-0283-5. [Google Scholar]
- Juslin, P.N.; Laukka, P. Impact of intended emotion intensity on cue utilization and decoding accuracy in vocal expression of emotion. Emotion 2001, 1, 381–412. [Google Scholar] [CrossRef] [PubMed]
- Ross, E.D.; Thompson, R.D.; Yenkosky, J. Lateralization of affective prosody in brain and the callosal integration of hemispheric language functions. Brain Lang. 1997, 56, 27–54. [Google Scholar] [CrossRef] [PubMed]
- Wildgruber, D.; Riecker, A.; Hertrich, I.; Erb, M.; Grodd, W.; Ethofer, T.; Ackermann, H. Identification of emotional intonation evaluated by fMRI. Neuroimage 2005, 24, 1233–1241. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. ICD-11 for Mortality and Morbidity Statistics. Available online: https://icd.who.int/browse11/l-m/en (accessed on 15 October 2018).
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; American Psychiatric Publishing: Arlington, WA, USA, 2013; pp. 1–51. [Google Scholar]
- Kohler, C.G.; Walker, J.B.; Martin, E.A.; Healey, K.M.; Moberg, P.J. Facial emotion perception in schizophrenia: A meta-analytic review. Schizophr. Bull. 2009, 36, 1009–1019. [Google Scholar] [CrossRef] [PubMed]
- Massaro, D.W.; Egan, P.B. Perceiving affect from the voice and the face. Psychon. Bull. Rev. 1996, 3, 215–221. [Google Scholar] [CrossRef] [PubMed]
- De Silva, L.C.; Miyasato, T.; Nakatsu, R. Facial Emotion Recognition Using Multimodal Information. In Proceedings of the IEEE International Conference on Information, Communications and Signal Processing (ICICS’97), Singapore, 9–12 September 1997; pp. 397–401. [Google Scholar]
- Diamond, E.; Zhang, Y. Cortical processing of phonetic and emotional information in speech: A cross-modal priming study. Neuropsychologia 2016, 82, 110–122. [Google Scholar] [CrossRef] [PubMed]
- Kingdon, D.G.; Ashcroft, K.; Bhandari, B.; Gleeson, S.; Warikoo, N.; Symons, M.; Taylor, L.; Lucas, E.; Mahendra, R.; Ghosh, S.; et al. Schizophrenia and borderline personality disorder: Similarities and differences in the experience of auditory hallucinations, paranoia, and childhood trauma. J. Nerv. Ment. Dis. 2010, 198, 399–403. [Google Scholar] [CrossRef] [PubMed]
| Year | First Author | Study Type (Neurological Technique): Study Topic | Country Language | Task Paradigm | Stimulus Modality: Stimulus Form | Response Option | Patient Information | Statistical Analysis Methods | Notes (Reasons for Exclusion from Meta-Analysis) | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Number (Male%) | Age (S.D.) | Education (years) | IQ | Illness Duration (years) | |||||||||
| 2001 | Ross [28] | Behavioral: Hemispheric dysfunction in schizophrenia patients and the neurology of EPP | the US English | Identification (SSC and SPP) Discrimination (SSC pair) | A: sentences, monosyllables, words and asyllabic sounds | C | 45 (87%) | 42.5 (7.2) | 13.8 (2.3) | N/A | N/A | ANOVA, Chi-square test and PCA | Excluded from the meta-analysis of identification and discrimination task but included in the overall effect size calculation (only reporting the results for comprehension) |
| 2002 | Hooker [29] | Behavioral: EPP and social functioning | the US English | Identification (SSC) | A: sentences, V: facial pictures | C | 20 (75%) | 39.3 (8.5) | 12.7 (2.9) | N/A | 18.8 (10.2) | ANOVA | |
| 2005 | Leitman [30] | Behavioral: Impaired EPP and basic auditory processing deficits | the US English | Identification (SSC) Discrimination (SSC pair) | A: sentences, V: facial pictures | C | 43 (77%) | 39.0 (12.0) | 10.6 (3.2) | N/A | 17.4 (9.6) | ANOVA, Spearman and PCA | |
| 2005 | Rossell [31] | Behavioral: Impaired EPP and auditory-verbal hallucinations | Australia English | Identification (SSC) Discrimination (SSC pair) | A: sentences, words, non-words, syllables V: pictures | R and C | 40 (60%) AVH:20 | 41.5 (9.5) | 13.9 (2.8) | 104.0 (14.7) | 23.9 (20.4) | ANOVA and SNK | |
| NAVH: 20 | 36.9 (8.9) | 14.6 (3.7) | 104.3 (12.6) | 15.7 (20.6) | |||||||||
| 2007 | Leitman [32] | Behavioral and neural (MRI): Neural substrates of impaired EPP | the US English | Identification (SSC) Discrimination (SSC pair) | A: sentences | C | 24 (88%) | 32.5 (10.6) | 11.0 (2.0) | 94.1 (7.5) | N/A | ANOVA, Spearman, voxel wise correlation approach and PCA | |
| 2007 | Shea [33] | Behavioral: EPP and auditory hallucinations | Australia English | Identification (SSC) Discrimination (SSC pair) | A: sentences | C | 67 (69%) AH:38 | 40.0 (10.0) | 14.0 (3.0) | 108.0 (11.0) | 14.0 (9.0) | ANOVA and Tukey‘s HSD | |
| NAH:29 | 44.0 (11.0) | 14 (3) | 109.0 (11.0) | 20.0 (15.0) | |||||||||
| 2008 | Chan [34] | Behavioral: EPP and neuropsychological function | China (HK) Chinese | Identification (SSC) Discrimination (SSC pair) | A: sentences (meaningless) V: facial photos | C | 43 (49%) PS:19 | 34.5 (9.3) | 11.7 (2.1) | N/A | 8.9 (7.9) | MANOVA and Multiple stepwise linear regression | |
| NPS:24 | 40.0 (9.1) | 8.8 (3.4) | |||||||||||
| 2008 | Scholten [35] | Behavioral: Gender differences and IQ in EPP | The Netherlands Dutch | Discrimination (meaning-prosody stroop test) | A: sentences | C | 48 (52%) | M: 29.5 (7.0) | N/A | M: 107.2 (9.6) | 7.0 (4.9) | ANOVA, ANCOVA and Pearson | |
| F: 32.2 (6.6) | F: 110.2 (8.3) | 8.5 (6.6) | |||||||||||
| 2009a | Bach [36] | Behavioral: High clarity and EPP | Switzerland German | Identification (SPP) | A: sentences (meaningless) V: facial photos | C | 25 (52%) | 35.9 (11.8) | N/A | N/A | 6 (N/A) | ANOVA | |
| 2009b | Bach [37] | Behavioral and neural (fMRI): Lateralization of EPP | Switzerland German | Identification (SPP) | A: non-words | C | 15 (53%) | 31.6 (7.9) | N/A | N/A | N/A | ANOVA, ANCOVA, SPM and laterality measures | |
| 2010 | Roux [38] | Behavioral: Implicit and explicit of EPP | France French | Identification (SSC) Discrimination (meaning-prosody stroop test) | A: words | C | 21 (67%) | 38.2 (12.6) | 11.5 (2.9) | 103.7 (7.0) | N/A | ANCOVA, ANOVA and Pearson | |
| 2012 | Gold [39] | Behavioral: The relationship between auditory emotion recognition impairments and acoustic features and cognition | the US English | Identification (SPP) | A: vocal sounds (full version and brief version which contains intensity modulation and pitch modulation) V: facial expressions | C | 92 (86%) | 37.8 (10.4) | N/A | N/A | N/A | ANOVA, multivariate regression and path analysis | Identification of brief version of vocal sounds excluded from meta-analysis (task quite different from other studies’) |
| 2013 | Ito [40] | Behavioral: EPP and positive psychotic symptoms | Japan Japanese | Discrimination (meaning-prosody stroop test) | A: sentences | C | 28 (61%) | 30.9 (8.1) | 13.6 (2.0) | N/A | 6.9 (7.3) | ANOVA, T test and Spearman | |
| 2013 | Iwashiro [41] | Behavioral: Semantic processing of emotional content and auditory attention | Japan Japanese | Discrimination (dichotic listening of SSC pair) | A: words | C | 22 (50%) | 31.6 (5.2) | 13.5 (1.8) | 96.7 (10.0) | 8.7 (6.7) | ANOVA, Paired-samples T test and Spearman | |
| 2013 | Jahshan [42] | Behavioral and neural study (ERP): Auditory processing and EPP | the US English | Identification (SSC) | A: sentences V: photos of facial expressions | C | 36 (69%) | 47.7 (10.0) | 12.6 (1.8) | N/A | 24.3 (11.5) | Independent-samples T test, Chi-square test, One-sample T test, MANOVA, Pearson and Multiple regression analysis | |
| 2013 | Kantrow-itz [43] | Behavioral: Emotion recognition based on tone of voice and basic auditory processing | the US English | Identification (SSC and SPP) | A: FM tones and sentences | C and R | 41 (53%) | 36.5 (10.9) | 12.3 (2.3) | N/A | 16.0 (10.0) | ANOVA, Independent-samples T test, M-W test, multivariate linear regression and Pearson | |
| 2013 | McLach-lan [44] | Behavioral: EPP and auditory hallucinations | Australia English | Identification (SSC) | A: sentences | R | 34 (76%) AVH:19 | 41.2 (9.9) | 13.2 (2.5) | 104.2 (8.8) | N/A | ANOVA, Chi-square test and Independent-samples T test | |
| NAVH:15 | 43.5 (8.8) | 12.9 (2.6) | 101.2 (9.7) | ||||||||||
| 2013 | Pinheiro [45] | Behavioral and neural (ERP): ERP correlates of EPP | the US English | Identification (SSC and SPP) | A: sentences and sentences (meaningless) | C | 15 (100%) | 49.7 (9.1) | 14.33 (1.8) | 92.2 (29.4) | 22.3 (10.5) | MANOVA and Spearman | |
| 2013 | Tseng [46] | Behavioral: EPP across modalities and psychotic symptoms | China (Taiwan) Chinese | Identification (SPP) | A: sounds V: facial photos | C | 111 (46%) | 38.2 (10.1) | 15.9 (3.4) | 92.5 (16.3) | 13.8 (9.7) | Independent-samples T test, Chi-square test, ANCOVA, Pearson, Multiple bidirectional stepwise linear regression, and Multiple linear regression | |
| 2014 | Brazo [47] | Behavioral: EP comprehension and semantic content | France French | Semantic identification (EPP influences) | A: sentences | C | 16 (56%) | 39.7 (8.6) | N/A | 89.8 (11.5) | 13.3 (5.8) | ANOVA, ANCOVA and Paired-samples T test | |
| 2014 | Dondai-ne [48] | Behavioral: facial and vocal emotion recognition biases | France French | Emotion intensity rating | A: non-verbal bursts without semantic content (vowel “ah”) V: facial expressions | R | 23 (65%) | 33.9 (7.3) | 12.7 (2.0) | N/A | 12.4 (6.5) | ANOVA, independent sample T test, Spearman’s correlation analysis | The whole study excluded from meta-analysis (task quite different from other studies’) |
| 2014 | Hoertna-gl [11] | Behavioral: A comparison of EPP between symptomatically remitted patients with schizophrenia and bipolar disorder | Austria N/A | Identification (SSC) | A: sentences | C | 41 (54%) | 40.5 (8.5) | 12.9 (2.9) | N/A | 12.4 (6.9) | Chi-square test, ANOVA, multiple linear regression | |
| 2014a | Kantrow-itz [19] | Behavioral and neural (MRI): early sensory processing and sarcasm perception | the US English | Identification (SSC) Discrimination (sarcasm perception) | A: sentences | C | 76 (63%) | 37.4 (10.1) | 12.1 (2.3) (74 patients) | N/A | 15.3 (9.0) (74 patients) | ANOVA, independent sample T test, Pearson correlations and multivariate linear regression | Discrimination task excluded from meta-analysis (sarcasm is a complex social emotion) |
| 2014b | Kantrow-itz [49] | Behavioral: amusia and protolanguage impairments in schizophrenia | the US English | Identification (emotional categories and intensity of SSC) | A: phrases | R | 31 (87%) | 39.4 (9.9) | 11.4 (2.2) (29 patients) | N/A | 14.8 (8.2) (24 patients) | independent-samples T test, MANOVA, ANOVA, multivariate linear regression and Pearson | Identification of emotional intensity excluded from meta-analysis (tasks quite different from other studies’) |
| 2014 | Müller [50] | Behavioral and neural study (ERP): Neural substrates of auditory emotion recognition deficits | the US English | Discrimination (face-prosody stroop test) | A and V: sounds and pictures of faces | R | 15 (73%) | 35.1 (9.3) | 14.1 (2.2) | N/A | 14.3 (9.1) | ANOVA, MANOVA, Bonferroni correction and Pearson | |
| 2014 | Pinheiro [51] | Behavioral and neural study (ERP): ERP correlates of EPP | the US English | Identification (SSC and SPP) | A: words and non-words | C | 16 (69%) | 48.9 (7.4) | 14.0 (2.4) | >85 | 19.5 (11.0) | ANOVA | |
| 2015 | Corcora-n [52] | Behavioral: emotion recognition deficits as predictors of transition in individual at clinical high risk for schizophrenia | the US English | Identification (SSC) | A: sentences V: facial expressions | C | 7 (57%) | 20.0 (5.2) | N/A | N/A | N/A | ANOVA | The whole study excluded from meta-analysis (involving prodromal patients) |
| 2015 | Kantrow-itz [53] | Behavioral and neural study (ERP and rsfMRI): Neural substrates of auditory emotion recognition deficits | the US English | Identification (SSC and SPP) | A: FM tones and sentences | C | 84 (81%) | 39.4 (10.6) | N/A | N/A | 15.9 (9.4) | ANOVA, MANOVA, Independent-samples T test, multivariate linear regression, Pearson and SPM | |
| 2015 | Regenbo-gen [18] | Behavioral and neural (fMRI): Neural responses to multimodal stimuli and pathology-specific social cognition deficits | Germany German | Empathy rating | A and V: video clips expressing emotion through three channels: facial expression, prosody and content | R | 20 (N/A) | 37.3 (8.4) | N/A | N/A | N/A | ANOVA, Pearson correlation analysis and SPM | The whole study excluded from meta-analysis (involving participants’ empathy, which is a complex social emotion) |
| 2015 | Sterea [9] | Behavioral: the relationship between social cognition and functional outcomes in schizophrenia | Romania Romanian | Definition of emotion and explanation of emotional situations and events | N/A | interview | 15 (60%) | 41.9 (8.4) | N/A | N/A | N/A | M-U test and Kendall correlation | The whole study excluded from meta-analysis (task quite different from other studies’ and involving complex emotions such as surprise and suspiciousness) |
| 2015 | Weisger-ber [54] | Behavioral: Facial, vocal and musical emotion recognition | Belgium French | Identification (SPP) | A: non-verbal vocal affect bursts V: computer-generated faces | R | 30 (37%) | 35.5 (12.7) | 12 (2.2) | N/A | 10.9 (9.4) | ANOVA, MANOVA and Spearman | |
| 2016 | Razafim-andimby [55] | Behavioral and neural study (fMRI): Neural bases of emotional sentence attribution | France French | Identification (SSC) | A: sentences | C | 21 (76%) | 33.9 (7.4) | N/A | N/A | 11.9 (7.9) | MANOVA, Chi-square test, SPM and Wilcoxon Rank Test | |
| 2017 | Hernim-an [56] | Secondary analysis: the effect of comorbid depression on facial and prosody emotion recognition | Australia English | N/A | N/A | N/A | 82 (65.9%) | 21.1 (2.6) | N/A | 93.3 (13.2) | N/A | ANCOVA and partial correlation analysis | The whole study excluded from meta-analysis (involving participants with comorbid disorders and secondary analysis of the data) |
| 2018 | Pawełcz-yk [57] | Behavioral: Extralinguistic and paralinguistic processing meditated by right hemisphere | Poland Polish | Identification (SPP) | A: sentences (meaningless) | C | 40 (58%) | 26.3 (9.3) | 12.0 (2.6) | N/A | 3.9 (4.7) | T test, Chi-square Test and M-U test | |
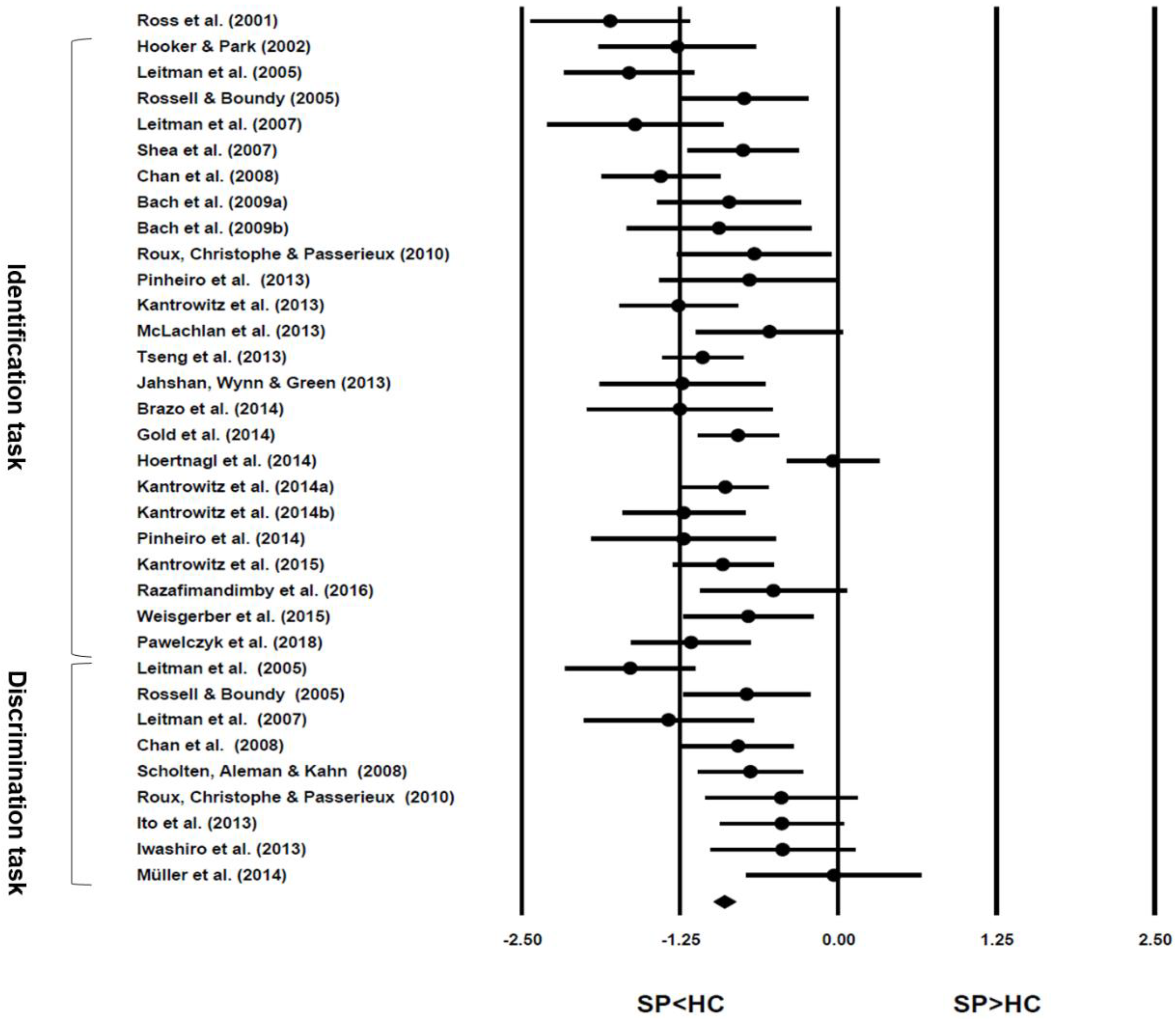
| Year | First Author | Effect Size of Performance in Schizophrenic Patients as Compared to Healthy Control (95% CI) | Task Paradigm and Material | Type of Participants (Number) | Single Emotion Recognition | Overall Emotion Recognition | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Happy | Sad | Angry | Fearful | Surprised | Disgusted | Neutral | ||||||
| 2002 | Hooker | d = −1.27 (−1.91 to −0.64) | Identification (SSC) | SP (20)-HC (27) | ▲ | |||||||
| 2005 | Leitman | d = −1.65 (−2.17 to −1.13) | Identification (SSC) | SP (43)-HC (34) | ▲ | |||||||
| 2005 | Rossell | d = −0.74 (−1.25 to −0.23) | Identification 1 (SSC) | SAVH (20)-HC (26) | ● | ▲ | ● | ▲ | ▲ | |||
| SNAVH (20)-HC (26) | ● | ● | ● | ▲ | ▲ | |||||||
| SAVH (20)-SNAVH (20) | ● | ▲ | ● | ● | ▲ | |||||||
| Identification 2 (SSC) | SAVH (20)-HC (26) | ● | ▲ | ▲ | ▲ | ● | ▲ | |||||
| SNAVH (20)-HC (26) | ● | ▲ | ▲ | ▲ | ● | ▲ | ||||||
| SAVH (20)-SNAVH (20) | ● | ● | ▲ | ● | ● | ● | ||||||
| 2007 | Leitman | d = −1.60 (−2.32 to −0.89) | Identification (SSC) | SP (24)-HC (17) | ▲ | |||||||
| 2007 | Shea | d = −0.75 (−1.18 to −0.31) | Identification (SSC) | SAH (38)-HC (31) | ● | ● | ● | ▲ | ||||
| SAH (38)-SNAH (29) | ● | ● | ● | ▲ | ||||||||
| SNAH (29)-HC (31) | ● | ● | ● | ● | ||||||||
| 2008 | Chan | d = −1.40 (−1.87 to −0.93) | Identification (SSC) | SP (43)-HC (43) | ▲ | |||||||
| 2009a | Bach | d = −0.86 (−1.44 to -0.28) | Identification (SPP) | SP (25)-HC (25) | ● | ● | ● | ● | ● | ● | ▲ | |
| SP (25)-Depression (25) | ● | ● | ● | ▲ | ● | ● | ▲ | |||||
| 2009b | Bach | d = −0.94 (−1.69 to −0.18) | Identification (SPP) | SP (15)-HC (15) | ▲ | |||||||
| 2010 | Roux | d = −0.66 (−1.28 to −0.04) | Identification (SSC) | SP (21)-HC (21) | ▲ | |||||||
| 2012 | Gold | d = −0.79 (−1.11 to −0.47) | Identification (SPP-full version) | SP (92)-HC (73) | ● | ▲ | ● | ▲ | ▲ | ▲ | ▲ | |
| 2013 | Jahshan | d = −1.23 (−1.90 to −0.56) | Identification (SSC) | SP (34)-HC (14) | ● | ● | ● | ● | ● | ● | ▲ | |
| 2013 | Kantrowitz | d = −1.26 (−1.73 to −0.79) | Identification (SSC and SPP) | SP (41)-HC (41) | ▲ | |||||||
| 2013 | McLachlan | d = −0.54 (−1.13 to 0.06) | Identification (SSC) | SP (34)-HC (17) | ▲ | |||||||
| 2013 | Pinheiro | d = −0.70 (−1.44 to 0.04) | Identification (SSC) | SP (15)-HC (15) | ● | ▲ | ● | ▲ | ||||
| Identification (SPP) | SP (15)-HC (15) | ● | ● | ▲ | ▲ | |||||||
| 2013 | Tseng | d = −1.07 (−1.39 to −0.75) | Identification (SPP) | SP (111)-HC (70) | ▲ | |||||||
| 2014 | Brazo | d = −1.25 (−2.01 to −0.49) | Semantic identification (EPP influences) | SP (16)-HC (16) | ▲ | |||||||
| 2014 | Hoertnagl | Identification (SSC) | SP (41)-BD (58) | ● | ● | ● | ● | ● | ||||
| d = −0.04 (−0.41 to 0.33) | Identification (SSC) | SP (41)-HC (85) | ● | ● | ▲ | ● | ● | |||||
| 2014a | Kantrowitz | d = −0.89 (−1.23 to −0.55) | Identification (SSC) | SP (76)-HC (72) | ▲ | |||||||
| 2014b | Kantrowitz | d = −1.22 (−1.72 to −0.72) | Identification (emotional categories of SSC) | SP (31)-HC (44) | ▲ | |||||||
| 2014 | Pinheiro | d = −1.22 (−1.98 to −0.47) | Identification (SSC) | SP (16)-HC (16) | ● | ▲ | ● | ● | ||||
| Identification (SPP) | SP (16)-HC (16) | ▲ | ● | ● | ● | |||||||
| 2015 | Kantrowitz | d = −0.91 (−1.31 to −0.51) | Identification (SSC and SPP) | SP (58)-HC (49) | ▲ | |||||||
| 2015 | Weisgerber | d = −0.71 (−1.23 to −0.19) | Identification (SPP) | SP (30)-HC (30) | ● | ● | ● | ● | ● | ● | ● | ▲ |
| 2016 | Razafimandimby | d = −0.51 (−1.10 to 0.08) | Identification (SSC) | SP (21)-HC (25) | ||||||||
| 2018 | Pawełczyk | d = −1.16 (−1.63 to −0.68) | Identification (SPP) | SP (40)-HC (39) | ▲ | |||||||
| Year | First Author | Effect Size of Performance in Schizophrenic Patients as Compared to Healthy Control (95% CI) | Task Paradigm and Material | Type of Participants (Number) | Single Emotion Recognition | Overall Emotion Recognition | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Happy | Sad | Angry | Fearful | Surprised | Disgusted | Neutral | ||||||
| 2005 | Leitman | d = −1.64 (−2.16 to −1.12) | Discrimination (SSC pair) | SP (43)-HC (34) | ▲ | |||||||
| 2005 | Rossell | d = −0.72 (−1.23 to −0.21) | Discrimination (dichotic listening of SPP pair) | SAVH (20)-HC (26) | ▲ | ● | ● | ▲ | ● | ▲ | ||
| SNAVH (20)-HC (26) | ● | ● | ● | ● | ● | ● | ||||||
| SAVH (20)-SNAVH (20) | ▲ | ● | ● | ▲ | ● | ▲ | ||||||
| 2007 | Leitman | d = − 1.34 (−2.03 to −0.65) | Discrimination (SSC pair) | SP (24)-HC (17) | ▲ | |||||||
| 2008 | Chan | d = −0.79 (−1.23 to −0.35) | Discrimination (SSC pair) | SP (43)-HC (43) | ▲ | |||||||
| 2008 | Scholten | d = −0.69 (−1.11 to −0.28) | Discrimination (meaning-prosody stroop test) | SP (48)-HC (46) | ||||||||
| 2010 | Roux | d = −0.45 (−1.06 to 0.16) | Discrimination (meaning-prosody stroop test) | SP (21)-HC (21) | ▲ | |||||||
| 2013 | Ito | d = −0.44 (−0.94 to 0.05) | Discrimination (meaning-prosody stroop test) | SP (28)-HC (37) | ▲ | |||||||
| 2013 | Iwashiro | d = −0.44 (−1.02 to 0.15) | Discrimination(dichotic listening of SSC pair) | SP (22)-HC (24) | ● | |||||||
| 2014 | Müller | d = −0.03 (−0.75 to 0.69) | Discrimination (face-prosody stroop test) | SP (15)-HC (15) | ● | |||||||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lin, Y.; Ding, H.; Zhang, Y. Emotional Prosody Processing in Schizophrenic Patients: A Selective Review and Meta-Analysis. J. Clin. Med. 2018, 7, 363. https://doi.org/10.3390/jcm7100363
Lin Y, Ding H, Zhang Y. Emotional Prosody Processing in Schizophrenic Patients: A Selective Review and Meta-Analysis. Journal of Clinical Medicine. 2018; 7(10):363. https://doi.org/10.3390/jcm7100363
Chicago/Turabian StyleLin, Yi, Hongwei Ding, and Yang Zhang. 2018. "Emotional Prosody Processing in Schizophrenic Patients: A Selective Review and Meta-Analysis" Journal of Clinical Medicine 7, no. 10: 363. https://doi.org/10.3390/jcm7100363
APA StyleLin, Y., Ding, H., & Zhang, Y. (2018). Emotional Prosody Processing in Schizophrenic Patients: A Selective Review and Meta-Analysis. Journal of Clinical Medicine, 7(10), 363. https://doi.org/10.3390/jcm7100363







