Acute Kidney Injury in Pregnancy: The Need for Higher Awareness. A Pragmatic Review Focused on What Could Be Improved in the Prevention and Care of Pregnancy-Related AKI, in the Year Dedicated to Women and Kidney Diseases
Abstract
1. Introduction
2. Pregnancy-Related AKI and Its Relationship with the Hypertensive Disorders of Pregnancy
3. The Risk of AKI in the Hypertensive Disorders of Pregnancy
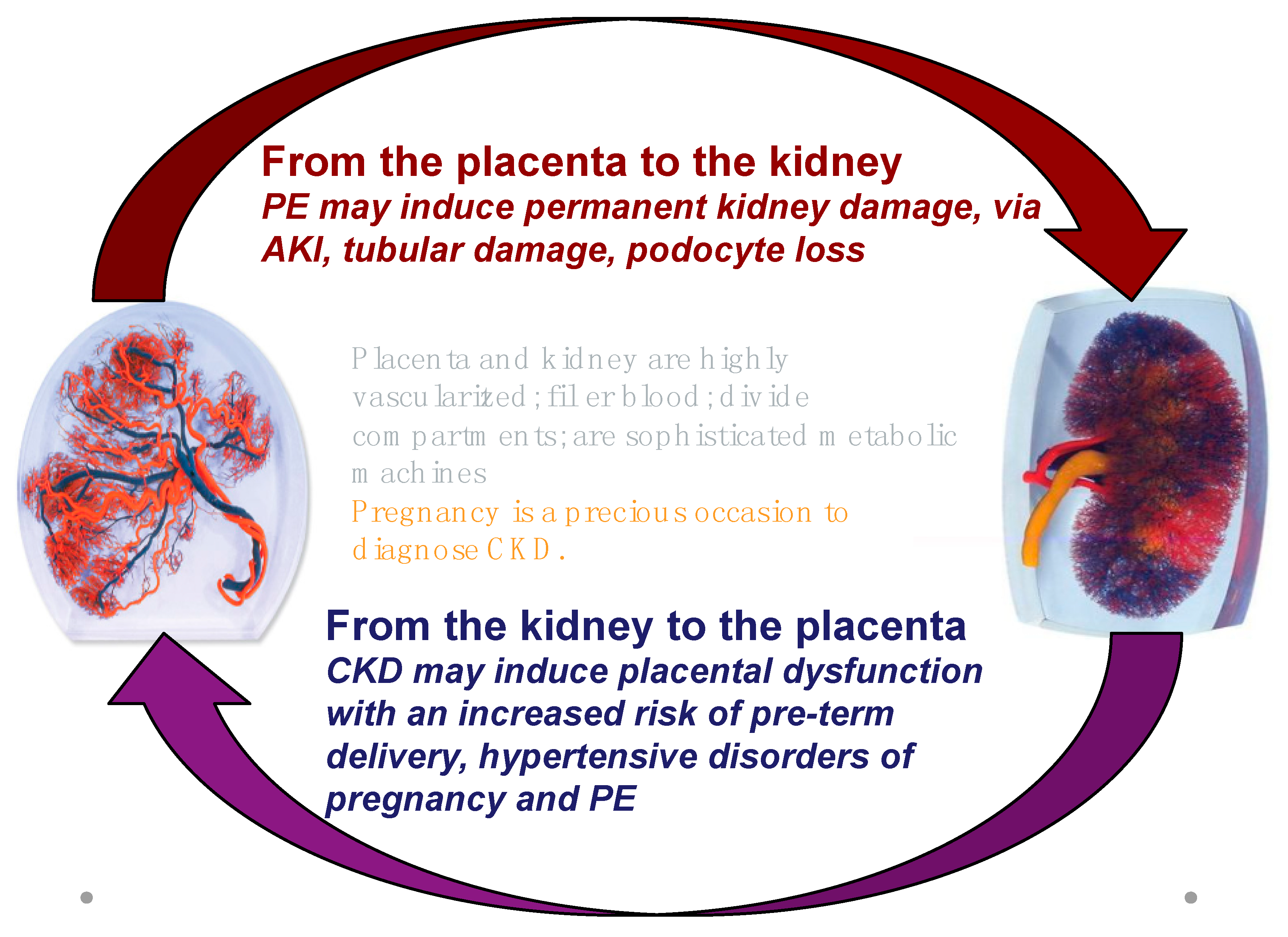
4. The Effect of AKI and the Hypertensive Disorders of Pregnancy on the Development of CKD in Mother and Child
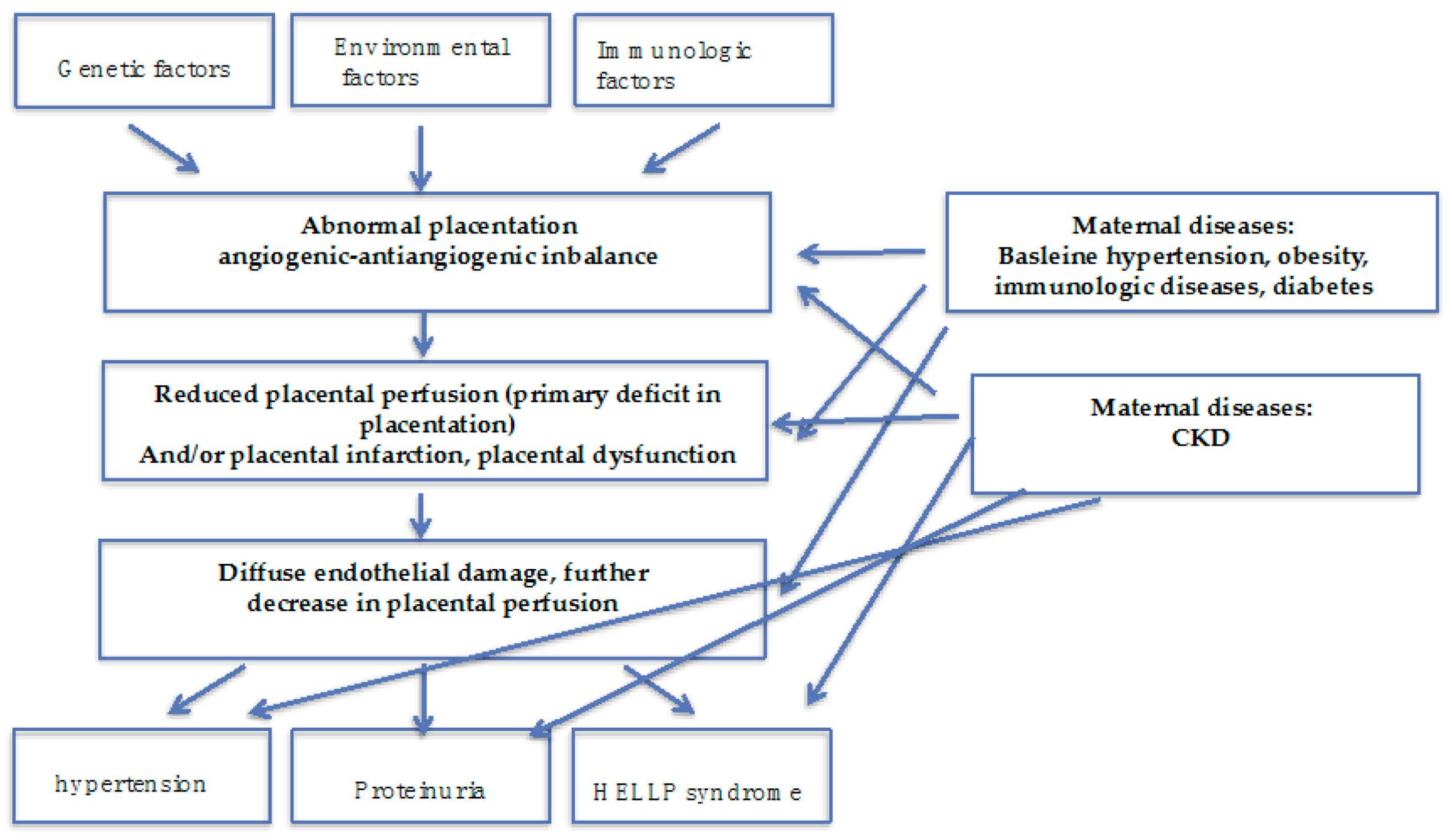
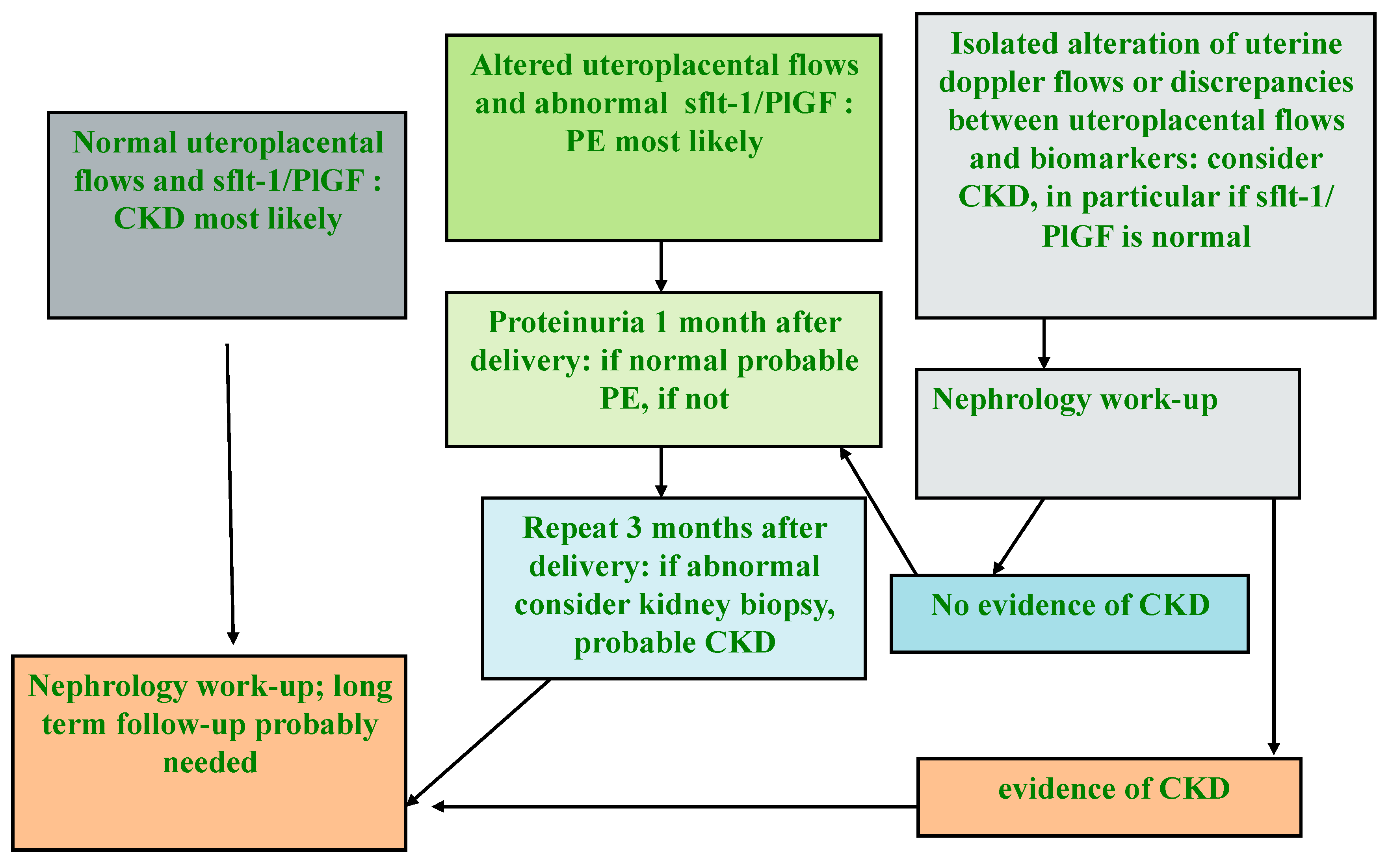
5. The Role of CKD in the Pathogenesis of AKI and Hypertensive Disorders of Pregnancy
6. The Role of Immunologic Diseases in the Pathogenesis of the Hypertensive Disorders of Pregnancy and p-AKI
7. Conclusions: The Need for Research and Clinical Intervention
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Mehta, R.L.; Cerdá, J.; Burdmann, E.A.; Tonelli, M.; García-García, G.; Jha, V. International Society of Nephrology's 0by25 initiative for acute kidney injury (zero preventable deaths by 2025): A human rights case for nephrology. Lancet 2015, 385, 2616–2643. [Google Scholar] [CrossRef]
- Macedo, E.; Mehta, R.L. Preventing Acute Kidney Injury. Crit. Care Clin. 2015, 31, 773–784. [Google Scholar] [CrossRef] [PubMed]
- Perico, N.; Remuzzi, G. Prevention programs for chronic kidney disease in low-income countries. Intern. Emerg. Med. 2016, 11, 385–389. [Google Scholar] [CrossRef] [PubMed]
- Jim, B.; Garovic, V.D. Acute Kidney Injury in Pregnancy. Semin. Nephrol. 2017, 37, 378–385. [Google Scholar] [CrossRef] [PubMed]
- Fakhouri, F.; Deltombe, C. Pregnancy-related acute kidney injury in high income countries: Still a critical issue. J. Nephrol. 2017, 30, 767–771. [Google Scholar] [CrossRef] [PubMed]
- Ibarra-Hernández, M.; Orozco-Guillén, O.A.; de la Alcantar-Vallín, M.L.; Garrido-Roldan, R.; Jiménez-Alvarado, M.P.; Castro, K.B.; Villa-Villagrana, F.; Borbolla, M.; Gallardo-Gaona, J.M.; García-García, G.; et al. Acute kidney injury in pregnancy and the role of underlying CKD: A point of view from México. J. Nephrol. 2017, 30, 773–780. [Google Scholar] [CrossRef] [PubMed]
- Prakash, J.; Pant, P.; Prakash, S.; Sivasankar, M.; Vohra, R.; Doley, P.K.; Pandey, L.K.; Singh, U. Changing picture of acute kidney injury in pregnancy: Study of 259 cases over a period of 33 years. Indian J. Nephrol. 2016, 26, 262–267. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, A.; Ahmed, M.M.; Kedir, S.; Bekele, D. Clinical profile and outcome of patients with acute kidney injury requiring dialysis–An experience from a haemodialysis unit in a developing country. BMC Nephrol. 2016, 17, 91. [Google Scholar] [CrossRef] [PubMed]
- Bentata, Y.; Housni, B.; Mimouni, A.; Azzouzi, A.; Abouqal, R. Acute kidney injury related to pregnancy in developing countries: Etiology and risk factors in an intensive care unit. J. Nephrol. 2012, 25, 764–775. [Google Scholar] [CrossRef] [PubMed]
- Jha, V.; Chugh, K.S. Community-acquired acute kidney injury in Asia. Semin. Nephrol. 2008, 28, 330–347. [Google Scholar] [CrossRef] [PubMed]
- Naicker, S.; Aboud, O.; Gharbi, M.B. Epidemiology of acute kidney injury in Africa. Semin. Nephrol. 2008, 28, 348–353. [Google Scholar] [CrossRef] [PubMed]
- Lombardi, R.; Yu, L.; Younes-Ibrahim, M.; Schor, N.; Burdmann, E.A. Epidemiology of acute kidney injury in Latin America. Semin. Nephrol. 2008, 28, 320–329. [Google Scholar] [CrossRef] [PubMed]
- Cerdá, J.; Bagga, A.; Kher, V.; Chakravarthi, R.M. The contrasting characteristics of acute kidney injury in developed and developing countries. Nat. Clin. Pract. Nephrol. 2008, 4, 138–153. [Google Scholar] [CrossRef] [PubMed]
- Trends in Maternal Mortality: 1990 to 2015. Available online: http://www.who.int/reproductivehealth/publications/monitoring/maternal-mortality-2015/en/ (accessed on 15 July 2018).
- Parr, S.K.; Matheny, M.E.; Abdel-Kader, K.; Greevy, R.A.; Bian, A., Jr.; Fly, J.; Chen, G.; Speroff, T.; Hung, A.M.; Ikizler, T.A.; et al. Acute kidney injury is a risk factor for subsequent proteinuria. Kidney Int. 2018, 93, 460–469. [Google Scholar] [CrossRef] [PubMed]
- Hsu, R.K.; Hsu, C.Y. The Role of Acute Kidney Injury in Chronic Kidney Disease. Semin. Nephrol. 2016, 36, 283–292. [Google Scholar] [CrossRef] [PubMed]
- Mammen, C.; Al Abbas, A.; Skippen, P.; Nadel, H.; Levine, D.; Collet, J.P.; Matsell, D.G. Long-term risk of CKD in children surviving episodes of acute kidney injury in the intensive care unit: A prospective cohort study. Am. J. Kidney Dis. 2012, 59, 523–530. [Google Scholar] [CrossRef] [PubMed]
- Silver, S.A.; Siew, E.D. Follow-up Care in Acute Kidney Injury: Lost in Transition. Adv. Chronic Kidney Dis. 2017, 24, 246–252. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Ma, X.; Zheng, J.; Liu, X.; Yan, T. Pregnancy outcomes in patients with acute kidney injury during pregnancy: A systematic review and meta-analysis. BMC Pregnancy Childbirth 2017, 17, 235. [Google Scholar] [CrossRef] [PubMed]
- Oladapo, O.T.; Adetoro, O.O.; Ekele, B.A.; Chama, C.; Etuk, S.J.; Aboyeji, A.P.; Onah, H.E.; Abasiattai, A.M.; Adamu, A.N.; et al. Nigeria Near-miss and Maternal Death Surveillance Network. When getting there is not enough: A nationwide cross-sectional study of 998 maternal deaths and 1451 near-misses in public tertiary hospitals in a low-income country. BJOG 2016, 123, 928–938. [Google Scholar] [CrossRef] [PubMed]
- Theilen, L.H.; Fraser, A.; Hollingshaus, M.S.; Schliep, K.C.; Varner, M.W.; Smith, K.R.; Esplin, M.S. All-Cause and Cause-Specific Mortality After Hypertensive Disease of Pregnancy. Obstet. Gynecol. 2016, 128, 238–244. [Google Scholar] [CrossRef] [PubMed]
- Acharya, A. Management of Acute Kidney Injury in Pregnancy for the Obstetrician. Obstet. Gynecol. Clin. N. Am. 2016, 43, 747–765. [Google Scholar] [CrossRef] [PubMed]
- Lombardi, R.; Rosa-Diez, G.; Ferreiro, A.; Greloni, G.; Yu, L.; Younes-Ibrahim, M.; Burdmann, E.A. Acute kidney injury in Latin America: A view on renal replacement therapy resources. Nephrol. Dial. Transplant. 2014, 29, 1369–1376. [Google Scholar] [CrossRef] [PubMed]
- Nwoko, R.; Plecas, D.; Garovic, V.D. Acute kidney injury in the pregnant patient. Clin. Nephrol. 2012, 78, 478–486. [Google Scholar] [CrossRef] [PubMed]
- Srinil, S.; Panaput, T. Acute kidney injury complicating septic unsafe abortion: Clinical course and treatment outcomes of 44 cases. J. Obstet. Gynaecol. Res. 2011, 37, 1525–1531. [Google Scholar] [CrossRef] [PubMed]
- Sabban, H.; Zakhari, A.; Patenaude, V.; Tulandi, T.; Abenhaim, H.A. Obstetrical and perinatal morbidity and mortality among in-vitro fertilization pregnancies: A population-based study. Arch. Gynecol. Obstet. 2017, 296, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Storgaard, M.; Loft, A.; Bergh, C.; Wennerholm, U.B.; Söderström-Anttila, V.; Romundstad, L.B.; Aittomaki, K.; Oldereid, N.; Forman, J.; Pinborg, A. Obstetric and neonatal complications in pregnancies conceived after oocyte donation: A systematic review and meta-analysis. BJOG 2017, 124, 561–572. [Google Scholar] [CrossRef] [PubMed]
- Blázquez, A.; García, D.; Rodríguez, A.; Vassena, R.; Figueras, F.; Vernaeve, V. Is oocyte donation a risk factor for preeclampsia? A systematic review and meta-analysis. J. Assist. Reprod. Genet. 2016, 33, 855–863. [Google Scholar] [CrossRef] [PubMed]
- Orozco-Guillen, A.; Merodio-Anguiano, R.; Iyune-Cojab, E.; Piccoli, G.B.; Rodriguez-Bosh, M. A baby and a kidney neoplasia: A question of priorities. Kidney Int. 2016, 90, 1133. [Google Scholar] [CrossRef] [PubMed]
- Mol, B.W.; Roberts, C.T.; Thangaratinam, S.; Magee, L.A.; de Groot, C.J.; Hofmeyr, G.J. Pre-eclampsia. Lancet 2016, 387, 999–1011. [Google Scholar] [CrossRef]
- Brown, M.A.; Magee, L.A.; Kenny, L.C.; Karumanchi, S.A.; McCarthy, F.P.; Saito, S.; Hall, D.R.; Warren, C.E.; Adoyi, G.; Ishaku, S. The hypertensive disorders of pregnancy: ISSHP classification, diagnosis & management recommendations for international practice. Pregnancy Hypertens. 2018, 23, 24–43. [Google Scholar]
- Tranquilli, A.L.; Dekker, G.; Magee, L.; Roberts, J.; Sibai, B.M.; Steyn, W.; Zeeman, G.G.; Brown, M.A. The classification, diagnosis and management of the hypertensive disorders of pregnancy: A revised statement from the ISSHP. Pregnancy Hypertens. 2014, 4, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Brown, M.A.; Lindheimer, M.D.; de Swiet, M.; van Assche, A.; Moutquin, J.M. The classification and diagnosis of the hypertensive disorders of pregnancy: Statement from the International Society for the Study of Hypertension in Pregnancy (ISSHP). Hypertens. Pregnancy 2001, 20. [Google Scholar] [CrossRef] [PubMed]
- Kallela, J.; Jääskeläinen, T.; Kortelainen, E.; Heinonen, S.; Kajantie, E.; Kere, J.; Kivinen, K.; Pouta, A.; Laivuori, H. The diagnosis of pre-eclampsia using two revised classifications in the Finnish Pre-eclampsia Consortium (FINNPEC) cohort. BMC Pregnancy Childbirth 2016, 16, 221. [Google Scholar] [CrossRef] [PubMed]
- Tranquilli, A.L.; Brown, M.A.; Zeeman, G.G.; Dekker, G.; Sibai, B.M. The definition of severe and early-onset preeclampsia. Statements from the International Society for the Study of Hypertension in Pregnancy (ISSHP). Pregnancy Hypertens. 2013, 3, 44–47. [Google Scholar] [CrossRef] [PubMed]
- Helewa, M.E.; Burrows, R.F.; Smith, J.; Williams, K.; Brain, P.; Rabkin, S.W. Report of the Canadian Hypertension Society Consensus Conference: 1. Definitions, evaluation and classification of hypertensive disorders in pregnancy. CMAJ 1997, 157, 715–725. [Google Scholar] [PubMed]
- Ferrazzi, E.; Stampalija, T.; Monasta, L.; di Martino, D.; Vonck, S.; Gyselaers, W. Maternal hemodynamics: A method to classify hypertensive disorders of pregnancy. Am. J. Obstet. Gynecol. 2018, 218, 124. [Google Scholar] [CrossRef] [PubMed]
- Sjaus, A.; McKeen, D.M.; George, R.B. Hypertensive disorders of pregnancy. Can. J. Anaesth. 2016, 63, 1075–1097. [Google Scholar] [CrossRef] [PubMed]
- Magee, L.A.; Pels, A.; Helewa, M.; Rey, E.; von Dadelszen, P. The hypertensive disorders of pregnancy. Best Pract. Res. Clin. Obstet. Gynaecol. 2015, 29, 643–657. [Google Scholar] [CrossRef] [PubMed]
- Wagner, S.J.; Barac, S.; Garovic, V.D. Hypertensive pregnancy disorders: Current concepts. J. Clin. Hypertens. (Greenwich) 2007, 9, 560–566. [Google Scholar] [CrossRef]
- Muñoz-Hernández, R.; Medrano-Campillo, P.; Miranda, M.L.; Macher, H.C.; Praena-Fernández, J.M.; Vallejo-Vaz, A.J.; Dominguez-Simeon, M.J.; Moreno-Luna, R.; Stiefel, P. Total and Fetal Circulating Cell-Free DNA, Angiogenic, and Antiangiogenic Factors in Preeclampsia and HELLP Syndrome. Am. J. Hypertens. 2017, 30, 673–682. [Google Scholar] [CrossRef] [PubMed]
- Schaarschmidt, W.; Rana, S.; Stepan, H. The course of angiogenic factors in early-vs. late-onset preeclampsia and HELLP syndrome. J. Perinat. Med. 2013, 41, 511–516. [Google Scholar] [CrossRef] [PubMed]
- Lind Malte, A.; Uldbjerg, N.; Wright, D.; Tørring, N. Prediction of severe pre-eclampsia/HELLP syndrome by combination of sFlt-1, CT-pro-ET-1 and blood pressure: Exploratory study. Ultrasound Obstet. Gynecol. 2018, 51, 768–774. [Google Scholar] [CrossRef] [PubMed]
- Jim, B.; Karumanchi, S.A. Preeclampsia: Pathogenesis, Prevention, and Long-term Complications. Semin. Nephrol. 2017, 37, 386–397. [Google Scholar] [CrossRef] [PubMed]
- Verlohren, S.; Perschel, F.H.; Thilaganathan, B.; Dröge, L.A.; Henrich, W.; Busjahn, A.; Khalil, A. Angiogenic Markers and Cardiovascular Indices in the Prediction of Hypertensive Disorders of Pregnancy. Hypertension 2017, 69, 1192–1197. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Fierro, M.L.; Hernández-Delgadillo, G.P.; Flores-Morales, V.; Cardenas-Vargas, E.; Mercado-Reyes, M.; Rodriguez-Sanchez, I.P.; Delgado-Enciso, I.; Galván-Tejada, C.E.; Galván-Tejada, J.I.; Celaya-Padilla, J.M. Current model systems for the study of preeclampsia. Exp. Biol. Med. (Maywood) 2018, 243, 576–585. [Google Scholar] [CrossRef] [PubMed]
- Villie, P.; Dommergues, M.; Brocheriou, I.; Piccoli, G.B.; Tourret, J.; Hertig, A. Why kidneys fail post-partum: A tubulocentric viewpoint. J. Nephrol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Wagner, S.J.; Craici, I.M.; Grande, J.P.; Garovic, V.D. From placenta to podocyte: Vascular and podocyte pathophysiology in preeclampsia. Clin. Nephrol. 2012, 78, 241–249. [Google Scholar] [CrossRef] [PubMed]
- Henao, D.E.; Saleem, M.A.; Cadavid, A.P. Glomerular disturbances in preeclampsia: Disruption between glomerular endothelium and podocyte symbiosis. Hypertens Pregnancy 2010, 29, 10–20. [Google Scholar] [CrossRef] [PubMed]
- Koizumi, M.; Yahata, K.; Ohno, S.; Koga, K.; Kikuchi, Y.; Hasegawa, C.; Seta, K.; Kuwahara, T.; Sugawara, A. Glomerular endotheliosis in a pregnant woman with severe gestational proteinuria. Intern. Med. 2013, 52, 1943–1947. [Google Scholar] [CrossRef] [PubMed]
- Von Dadelszen, P.; Magee, L.A. Preventing deaths due to the hypertensive disorders of pregnancy. Best. Pract. Res. Clin. Obstet. Gynaecol. 2016, 36, 83–102. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, I.R.; Silva, W.B.; Cerqueira, G.S.; Novo, N.F.; Almeida, F.A.; Novo, J.L. Maternal and fetal outcome in women with hypertensive disorders of pregnancy: The impact of prenatal care. Ther. Adv. Cardiovasc. Dis. 2015, 9, 140–146. [Google Scholar] [CrossRef] [PubMed]
- Von Dadelszen, P.; Ansermino, J.M.; Dumont, G.; Hofmeyr, G.J.; Magee, L.A.; Mathai, M.; Sawchuck, D.; Teela, K.; Donnay, F.; Roberts, J.M. Improving maternal and perinatal outcomes in the hypertensive disorders of pregnancy: A vision of a community-focused approach. Int. J. Gynaecol. Obstet. 2012, 119, 30–34. [Google Scholar] [CrossRef] [PubMed]
- Cerdá, J.; Mohan, S.; Garcia-Garcia, G.; Jha, V.; Samavedam, S.; Gowrishankar, S.; Bagga, A.; Chakravarthi, R.; Mehta, R. Acute Kidney Injury Recognition in Low- and Middle-Income Countries. Kidney Int. Rep. 2017, 2, 530–543. [Google Scholar] [CrossRef] [PubMed]
- Kashani, K.; Macedo, E.; Burdmann, E.A.; Hooi, L.S.; Khullar, D.; Bagga, A.; Chakravarthi, R.; Mehta, R. Acute Kidney Injury Risk Assessment: Differences and Similarities Between Resource-Limited and Resource-Rich Countries. Kidney Int. Rep. 2017, 2, 519–529. [Google Scholar] [CrossRef] [PubMed]
- Tangren, J.S.; Powe, C.E.; Ankers, E.; Ecker, J.; Bramham, K.; Hladunewich, M.A.; Karumanchi, S.A.; Thadhani, R. Pregnancy Outcomes after Clinical Recovery from AKI. J. Am. Soc. Nephrol. 2017, 28, 1566–1574. [Google Scholar] [CrossRef] [PubMed]
- Chawla, L.S.; Kimmel, P.L. Acute kidney injury and chronic kidney disease: An integrated clinical syndrome. Kidney Int. 2012, 82, 516–524. [Google Scholar] [CrossRef] [PubMed]
- Kaballo, M.A.; Elsayed, M.E.; Stack, A.G. Linking acute kidney injury to chronic kidney disease: The missing links. J. Nephrol. 2017, 30, 461–475. [Google Scholar] [CrossRef] [PubMed]
- Pannu, N. Bidirectional relationships between acute kidney injury and chronic kidney disease. Curr. Opin. Nephrol. Hypertens. 2013, 22, 351–356. [Google Scholar] [CrossRef] [PubMed]
- He, L.; Wei, Q.; Liu, J.; Yi, M.; Liu, Y.; Liu, H.; Sun, L.; Peng, Y.; Liu, F.; Venkatachalam, M.A.; et al. AKI on CKD: Heightened injury, suppressed repair, and the underlying mechanisms. Kidney Int. 2017, 92, 1071–1083. [Google Scholar] [CrossRef] [PubMed]
- Brown, M.C.; Best, K.E.; Pearce, M.S.; Waugh, J.; Robson, S.C.; Bell, R. Cardiovascular disease risk in women with pre-eclampsia: Systematic review and meta-analysis. Eur. J. Epidemiol. 2013, 28, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Bellamy, L.; Casas, J.P.; Hingorani, A.D.; Williams, D.J. Pre-eclampsia and risk of cardiovascular disease and cancer in later life: Systematic review and meta-analysis. BMJ 2007, 335, 974. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, R.; Dunford, J.; Mehran, R.; Robson, S.; Kunadian, V. Pre-eclampsia and future cardiovascular risk among women: A review. J. Am. Coll. Cardiol. 2014, 63, 1815–1822. [Google Scholar] [CrossRef] [PubMed]
- Wu, P.; Haththotuwa, R.; Kwok, C.S.; Babu, A.; Kotronias, R.A.; Rushton, C.; Zaman, A.; Fryer, A.A.; Kadam, U.; Chew-Graham, C.A.; et al. Preeclampsia and Future Cardiovascular Health: A Systematic Review and Meta-Analysis. Circ. Cardiovasc. Qual. Outcomes 2017, 10, e003497. [Google Scholar] [CrossRef] [PubMed]
- McDonald, S.D.; Han, Z.; Walsh, M.W.; Gerstein, H.C.; Devereaux, P.J. Kidney disease after preeclampsia: A systematic review and meta-analysis. Am. J. Kidney. Dis. 2010, 55, 1026–1039. [Google Scholar] [CrossRef] [PubMed]
- Paauw, N.D.; van der Graaf, A.M.; Bozoglan, R.; van der Ham, D.P.; Navis, G.; Gansevoort, R.T.; Groen, H.; Lely, A.T. Kidney Function After a Hypertensive Disorder of Pregnancy: A Longitudinal Study. Am. J. Kidney Dis. 2017, 71, 619–626. [Google Scholar] [CrossRef] [PubMed]
- Van Balen Lopes, V.A.; Spaan, J.J.; Cornelis, T.; Heidema, W.M.; Scholten, R.R.; Spaanderman, M.E.A. Endothelial and kidney function in women with a history of preeclampsia and healthy parous controls: A case control study. Microvasc. Res. 2017, 116, 71–76. [Google Scholar] [CrossRef] [PubMed]
- Tooher, J.; Thornton, C.; Makris, A.; Ogle, R.; Korda, A.; Hennessy, A. All Hypertensive Disorders of Pregnancy Increase the Risk of Future Cardiovascular Disease. Hypertension 2017, 70, 798–803. [Google Scholar] [CrossRef] [PubMed]
- Bokslag, A.; Teunissen, P.W.; Franssen, C.; van Kesteren, F.; Kamp, O.; Ganzevoort, W.; Paulus, W.J.; de Groot, C.J.M. Effect of early-onset preeclampsia on cardiovascular risk in the fifth decade of life. Am. J. Obstet. Gynecol. 2017, 216, 523. [Google Scholar] [CrossRef] [PubMed]
- Paauw, N.D.; Joles, J.A.; Drost, J.T.; Verhaar, M.C.; Franx, A.; Navis, G.; Maas, A.H.; Lely, A.T. High-Normal Estimated Glomerular Filtration Rate in Early-Onset Preeclamptic Women 10 Years Postpartum. Hypertension 2016, 68, 1407–1414. [Google Scholar] [CrossRef] [PubMed]
- Ayansina, D.; Black, C.; Hall, S.J.; Marks, A.; Millar, C.; Prescott, G.J.; Wilde, K.; Bhattacharya, S. Long term effects of gestational hypertension and pre-eclampsia on kidney function: Record linkage study. Pregnancy Hypertens. 2016, 6, 344–349. [Google Scholar] [CrossRef] [PubMed]
- Kessous, R.; Shoham-Vardi, I.; Pariente, G.; Sergienko, R.; Sheiner, E. Long-term maternal atherosclerotic morbidity in women with pre-eclampsia. Heart 2015, 101, 442–446. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.C.; Chen, S.H.; Ho, C.H.; Liang, F.W.; Chu, C.C.; Wang, H.Y.; Lu, Y.H. End-stage renal disease after hypertensive disorders in pregnancy. Am. J. Obstet. Gynecol. 2014, 210, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Sandvik, M.K.; Hallan, S.; Svarstad, E.; Vikse, B.E. Preeclampsia and prevalence of microalbuminuria 10 years later. Clin. J. Am. Soc. Nephrol. 2013, 8, 1126–1134. [Google Scholar] [CrossRef] [PubMed]
- Männistö, T.; Mendola, P.; Vääräsmäki, M.; Järvelin, M.R.; Hartikainen, A.L.; Pouta, A.; Suvanto, E. Elevated blood pressure in pregnancy and subsequent chronic disease risk. Circulation 2013, 127, 681–690. [Google Scholar] [CrossRef] [PubMed]
- Wang, I.K.; Muo, C.H.; Chang, Y.C.; Liang, C.C.; Chang, C.T.; Lin, S.Y.; Yen, T.H.; Chuang, F.R.; Chen, P.C.; Huang, C.C.; et al. Association between hypertensive disorders during pregnancy and end-stage renal disease: A population-based study. CMAJ 2013, 185, 207–213. [Google Scholar] [CrossRef] [PubMed]
- Vikse, B.E.; Irgens, L.M.; Karumanchi, S.A.; Thadhani, R.; Reisæter, A.V.; Skjærven, R. Familial factors in the association between preeclampsia and later ESRD. Clin. J. Am. Soc. Nephrol. 2012, 7, 1819–1826. [Google Scholar] [CrossRef] [PubMed]
- McDonald, S.D.; Yusuf, S.; Walsh, M.W.; Lonn, E.; Teo, K.; Anand, S.S.; Pogue, J.; Islam, S.; Devereaux, P.J.; Gerstein, H.C. Increased cardiovascular risk after pre-eclampsia in women with dysglycaemia. Diabet. Med. 2012, 30, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharya, S.; Prescott, G.J.; Iversen, L.; Campbell, D.M.; Smith, W.C.; Hannaford, P.C. Hypertensive disorders of pregnancy and future health and mortality: A record linkage study. Pregnancy Hypertens. 2011, 2, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Shahbazian, N.; Shahbazian, H.; Ehsanpour, A.; Aref, A.; Gharibzadeh, S. Hypertension and microalbuminuria 5 years after pregnancies complicated by pre-eclampsia. Iran. J. Kidney Dis. 2011, 5, 324–327. [Google Scholar] [PubMed]
- Vikse, B.E.; Hallan, S.; Bostad, L.; Leivestad, T.; Iversen, B.M. Previous preeclampsia and risk for progression of biopsy-verified kidney disease to end-stage renal disease. Nephrol. Dial. Transplant. 2010, 25, 3289–3296. [Google Scholar] [CrossRef] [PubMed]
- Sandvik, M.K.; Iversen, B.M.; Irgens, L.M.; Skjaerven, R.; Leivestad, T.; Søfteland, E.; Vikse, B.E. Are adverse pregnancy outcomes risk factors for development of end-stage renal disease in women with diabetes? Nephrol. Dial. Transplant. 2010, 25, 3600–3607. [Google Scholar] [CrossRef] [PubMed]
- Vikse, B.E.; Irgens, L.M.; Leivestad, T.; Skjaerven, R.; Iversen, B.M. Preeclampsia and the risk of end-stage renal disease. N. Engl. J. Med. 2008, 359, 800–809. [Google Scholar] [CrossRef] [PubMed]
- Lampinen, K.H.; Rönnback, M.; Groop, P.H.; Kaaja, R.J. Renal and vascular function in women with previous preeclampsia: A comparison of low- and high-degree proteinuria. Kidney Int. 2006, 70, 1818–1822. [Google Scholar] [CrossRef] [PubMed]
- Vikse, B.E.; Irgens, L.M.; Bostad, L.; Iversen, B.M. Adverse perinatal outcome and later kidney biopsy in the mother. J. Am. Soc. Nephrol. 2006, 17, 837–845. [Google Scholar] [CrossRef] [PubMed]
- Shammas, A.G.; Maayah, J.F. Hypertension and its relation to renal function 10 years after pregnancy complicated by pre-eclampsia and pregnancy induced hypertension. Saudi Med. J. 2000, 21, 190–192. [Google Scholar] [PubMed]
- Kattah, A.G.; Scantlebury, D.C.; Agarwal, S.; Mielke, M.M.; Rocca, W.A.; Weaver, A.L.; Vaughan, L.E.; Miller, V.M.; Weissgerber, T.L.; White, W.; et al. Preeclampsia and ESRD: The Role of Shared Risk Factors. Am. J. Kidney Dis. 2017, 69, 498–505. [Google Scholar] [CrossRef] [PubMed]
- Wide-Swensson, D.; Strevens, H.; Willner, J. Antepartum percutaneous renal biopsy. Int. J. Gynaecol. Obstet. 2007, 98, 88–92. [Google Scholar] [CrossRef] [PubMed]
- Shiiki, H.; Dohi, K.; Hanatani, M.; Fujii, Y.; Sanai, H.; Ichijo, M.; Shimamoto, I.; Ishikawa, H.; Watanabe, T. Focal and segmental glomerulosclerosis in preeclamptic patients with nephrotic syndrome. Am. J. Nephrol. 1990, 10, 205–212. [Google Scholar] [CrossRef] [PubMed]
- Piccoli, G.B.; Cabiddu, G.; Castellino, S.; Gernone, G.; Santoro, D.; Moroni, G.; Spotti, D.; Giacchino, F.; Attini, R.; Limardo, M.; et al. A best practice position statement on the role of the nephrologist in the prevention and follow-up of preeclampsia: The Italian study group on kidney and pregnancy. J. Nephrol. 2017, 30, 307–317. [Google Scholar] [CrossRef] [PubMed]
- O’Gorman, N.; Wright, D.; Poon, L.C.; Rolnik, D.L.; Syngelaki, A.; de Alvarado, M.; Carbone, I.F.; Dutemeyer, V.; Fiolna, M.; Frick, A. Multicenter screening for pre-eclampsia by maternal factors and biomarkers at 11–13 weeks’ gestation: Comparison with NICE guidelines and ACOG recommendations. Ultrasound Obstet. Gynecol. 2017, 49, 756–760. [Google Scholar] [CrossRef] [PubMed]
- Von Dadelszen, P.; Payne, B.; Li, J.; Ansermino, J.M.; Broughton Pipkin, F.; Côté, A.M.; Douglas, M.J.; Gruslin, A.; Hutcheon, J.A.; Joseph, K.S.; et al. Prediction of adverse maternal outcomes in pre-eclampsia: Development and validation of the fullPIERS model. Lancet 2011, 377, 219–227. [Google Scholar] [CrossRef]
- Linsell, L.; Malouf, R.; Morris, J.; Kurinczuk, J.J.; Marlow, N. Risk Factor Models for Neurodevelopmental Outcomes in Children Born Very Preterm or With Very Low Birth Weight: A Systematic Review of Methodology and Reporting. Am. J. Epidemiol. 2017, 185, 601–612. [Google Scholar] [CrossRef] [PubMed]
- Guellec, I.; Lapillonne, A.; Marret, S.; Picaud, J.C.; Mitanchez, D.; Charkaluk, M.L.; Fresson, J.; Arnaud, C.; Flamand, C.; Cambonie, G. Effect of Intra- and Extrauterine Growth on Long-Term Neurologic Outcomes of Very Preterm Infants. J. Pediatr. 2016, 175, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Moore, T.; Hennessy, E.M.; Myles, J.; Johnson, S.J.; Draper, E.S.; Costeloe, K.L.; Marlow, N. Neurological and developmental outcome in extremely preterm children born in England in 1995 and 2006: The EPICure studies. BMJ 2012. [Google Scholar] [CrossRef] [PubMed]
- Guillén, U.; DeMauro, S.; Ma, L.; Zupancic, J.; Roberts, R.; Schmidt, B.; Kirpalani, H. Relationship between attrition and neurodevelopmental impairment rates in extremely preterm infants at 18 to 24 months: A systematic review. Arch. Pediatr. Adolesc. Med. 2012, 166, 178–184. [Google Scholar] [CrossRef] [PubMed]
- Ranke, M.B.; Schweizer, R.; Rodemann, S.M.; Bevot, A.; Martin, D.D.; Goelz, R.; Wollmann, H.A.; Krägeloh-Mann, I.; Binder, G. Schoolchildren born VLBW or VLGA show height-related changes in body composition and muscle function but no evidence of metabolic syndrome risk factors. Results from the NEOLONG study. J. Pediatr. Endocrinol. Metab. 2016, 29, 163–172. [Google Scholar] [CrossRef] [PubMed]
- Castanys-Muñoz, E.; Kennedy, K.; Castañeda-Gutiérrez, E.; Forsyth, S.; Godfrey, K.M.; Koletzko, B.; Ozanne, S.E.; Rueda, R.; Schoemaker, M.; van der Beek, E.M. Systematic review indicates postnatal growth in term infants born small-for-gestational-age being associated with later neurocognitive and metabolic outcomes. Acta Paediatr. 2017, 106, 1230–1238. [Google Scholar] [CrossRef] [PubMed]
- Ong, K.K.; Kennedy, K.; Castañeda-Gutiérrez, E.; Forsyth, S.; Godfrey, K.M.; Koletzko, B.; Latulippe, M.E.; Ozanne, S.E.; Rueda, R.; Schoemaker, M.H. Postnatal growth in preterm infants and later health outcomes: A systematic review. Acta Paediatr. 2015, 104, 974–986. [Google Scholar] [CrossRef] [PubMed]
- Low Birth Weight and Nephron Number Working Group. The Impact of Kidney Development on the Life Course: A Consensus Document for Action. Nephron Clin. Pract. 2017, 136, 3–49. [Google Scholar]
- Luyckx, V.A.; Bertram, J.F.; Brenner, B.M.; Fall, C.; Hoy, W.E.; Ozanne, S.E.; Vikse, B.E. Effect of fetal and child health on kidney development and long-term risk of hypertension and kidney disease. Lancet 2013, 382, 273–283. [Google Scholar] [CrossRef]
- Luyckx, V.A.; Brenner, B.M. Birth weight, malnutrition and kidney-associated outcomes—A global concern. Nat. Rev. Nephrol. 2015, 11, 135–149. [Google Scholar] [CrossRef] [PubMed]
- Monier, I.; Ancel, P.Y.; Ego, A.; Jarreau, P.H.; Lebeaux, C.; Kaminski, M.; Goffinet, F.; Zeitlin, J. Fetal and neonatal outcomes of preterm infants born before 32 weeks of gestation according to antenatal vs postnatal assessments of restricted growth. Am. J. Obstet. Gynecol. 2017, 216, 516. [Google Scholar] [CrossRef] [PubMed]
- Lee, A.C.; Kozuki, N.; Cousens, S.; Stevens, G.A.; Blencowe, H.; Silveira, M.F.; CHERG Small-for-Gestational-Age-Preterm Birth Working Group. Estimates of burden and consequences of infants born small for gestational age in low and middle income countries with INTERGROWTH-21 standard: Analysis of CHERG datasets. BMJ 2017, 358, 3677. [Google Scholar] [CrossRef] [PubMed]
- Temming, L.A.; Dicke, J.M.; Stout, M.J.; Rampersad, R.M.; Macones, G.A.; Tuuli, M.G.; Cahill, A.G. Early Second-Trimester Fetal Growth Restriction and Adverse Perinatal Outcomes. Obstet. Gynecol. 2017, 130, 865–869. [Google Scholar] [CrossRef] [PubMed]
- Wang, A.Y.; Chughtai, A.A.; Lui, K.; Sullivan, E.A. Morbidity and mortality among very preterm singletons following fertility treatment in Australia and New Zealand, a population cohort study. BMC Pregnancy Childbirth 2017, 17, 50. [Google Scholar] [CrossRef] [PubMed]
- Darmstadt, G.L.; Walker, N.; Lawn, J.E.; Bhutta, Z.A.; Haws, R.A.; Cousens, S. Saving newborn lives in Asia and Africa: Cost and impact of phased scale-up of interventions within the continuum of care. Health Policy Plan 2008, 23, 101–117. [Google Scholar] [CrossRef] [PubMed]
- Bhutta, Z.A.; Das, J.K.; Bahl, R.; Lawn, J.E.; Salam, R.A.; Paul, V.K.; Sankar, M.J.; Blencowe, H.; Rizvi, A.; Chou, V.B.; et al. Can available interventions end preventable deaths in mothers, newborn babies, and stillbirths, and at what cost? Lancet 2014, 384, 347–370. [Google Scholar] [CrossRef]
- Luyckx, V.A. Preterm Birth and its Impact on Renal Health. Semin. Nephrol. 2017, 37, 311–319. [Google Scholar] [CrossRef] [PubMed]
- Abitbol, C.L.; Rodriguez, M.M. The long-term renal and cardiovascular consequences of prematurity. Nat. Rev. Nephrol. 2012, 8, 265–274. [Google Scholar] [CrossRef] [PubMed]
- Sutherland, M.; Ryan, D.; Black, M.J.; Kent, A.L. Long-term renal consequences of preterm birth. Clin. Perinatol. 2014, 41, 561–573. [Google Scholar] [CrossRef] [PubMed]
- Carmody, J.B.; Charlton, J.R. Short-term gestation, long-term risk: Prematurity and chronic kidney disease. Pediatrics 2013, 131, 1168–1179. [Google Scholar] [CrossRef] [PubMed]
- Almasi, O.; Pariente, G.; Kessous, R.; Sergienko, R.; Sheiner, E. Association between delivery of small-for-gestational-age neonate and long-term maternal chronic kidney disease. J. Matern. Fetal Neonatal Med. 2016, 29, 2861–2864. [Google Scholar] [CrossRef] [PubMed]
- Pariente, G.; Sheiner, E.; Kessous, R.; Michael, S.; Shoham-Vardi, I. Association between delivery of a small-for-gestational-age neonate and long-term maternal cardiovascular morbidity. Int. J. Gynaecol. Obstet. 2013, 123, 68–71. [Google Scholar] [CrossRef] [PubMed]
- Neiger, R. Long-Term Effects of Pregnancy Complications on Maternal Health: A Review. J. Clin. Med. 2017, 6, 76. [Google Scholar] [CrossRef] [PubMed]
- Cabiddu, G.; Castellino, S.; Gernone, G.; Santoro, D.; Moroni, G.; Giannattasio, M.; Gregorini, G.; Giacchino, F.; Attini, R.; Loi, V.; et al. A best practice position statement on pregnancy in chronic kidney disease: The Italian Study Group on Kidney and Pregnancy. J. Nephrol. 2016, 29, 277–303. [Google Scholar] [CrossRef] [PubMed]
- Blom, K.; Odutayo, A.; Bramham, K.; Hladunewich, M.A. Pregnancy and Glomerular Disease: A Systematic Review of the Literature with Management Guidelines. Clin. J. Am. Soc. Nephrol. 2017, 18. [Google Scholar] [CrossRef] [PubMed]
- Piccoli, G.B.; Cabiddu, G.; Attini, R.; Vigotti, F.N.; Maxia, S.; Lepori, N.; Tuveri, M.; Massidda, M.; Marchi, C.; Mura, S. Risk of Adverse Pregnancy Outcomes in Women with CKD. J. Am. Soc. Nephrol. 2015, 26, 2011–2022. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.J.; Ma, X.X.; Hao, L.; Liu, L.J.; Lv, J.C.; Zhang, H. A Systematic Review and Meta-Analysis of Outcomes of Pregnancy in CKD and CKD Outcomes in Pregnancy. Clin. J. Am. Soc. Nephrol. 2015, 10, 1964–1978. [Google Scholar] [CrossRef] [PubMed]
- Alsuwaida, A.; Mousa, D.; Al-Harbi, A.; Alghonaim, M.; Ghareeb, S.; Alrukhaimi, M.N. Impact of early chronic kidney disease on maternal and fetal outcomes of pregnancy. J. Matern. Fetal. Neonatal. Med. 2011, 24, 1432–1436. [Google Scholar] [CrossRef] [PubMed]
- De Jesus, G.R.; de Jesus, N.R.; Levy, R.A.; Klumb, E.M. The use of angiogenic and antiangiogenic factors in the differential diagnosis of pre-eclampsia, antiphospholipid syndrome nephropathy and lupus nephritis. Lupus 2014, 23, 1299–1301. [Google Scholar] [CrossRef] [PubMed]
- Morton, A. Imitators of preeclampsia: A review. Pregnancy Hypertens. 2016, 6, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Rolfo, A.; Attini, R.; Tavassoli, E.; Neve, F.V.; Nigra, M.; Cicilano, M.; Nuzzo, A.M.; Giuffrida, D.; Biolcati, M.; Nichelatti, M.; et al. Is It Possible to Differentiate Chronic Kidney Disease and Preeclampsia by means of New and Old Biomarkers? A Prospective Study. Dis. Mark. 2015. [Google Scholar] [CrossRef] [PubMed]
- Piccoli, G.B.; Gaglioti, P.; Attini, R.; Parisi, S.; Bossotti, C.; Olearo, E.; Oberto, M.; Ferraresi, M.; Rolfo, A.; Versino, E.; et al. Pre-eclampsia or chronic kidney disease? The flow hypothesis. Nephrol. Dial. Transplant. 2013, 28, 1199–1206. [Google Scholar] [CrossRef] [PubMed]
- Zeisler, H.; Llurba, E.; Chantraine, F.; Vatish, M.; Staff, A.C.; Sennström, M.; Olovsson, M.; Brennecke, S.P.; Stepan, H.; Allegranza, D.; et al. Predictive Value of the sFlt-1: PlGF Ratio in Women with Suspected Preeclampsia. N. Engl. J. Med. 2016, 374, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Masuyama, H.; Nobumoto, E.; Okimoto, N.; Inoue, S.; Segawa, T.; Hiramatsu, Y. Superimposed preeclampsia in women with chronic kidney disease. Gynecol. Obstet. Invest. 2012, 74, 274–281. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; Zhu, Z.; Liu, C.; Zhang, Z. Dual-cutoff of sFlt-1/PlGF ratio in the stratification of preeclampsia: A systematic review and meta-analysis. Arch. Gynecol. Obstet. 2017, 295, 1079–1087. [Google Scholar] [CrossRef] [PubMed]
- Bartsch, E.; Medcalf, K.E.; Park, A.L.; Ray, J.G. Clinical risk factors for pre-eclampsia determined in early pregnancy: Systematic review and meta-analysis of large cohort studies. BMJ 2016, 353, 1753. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Ma, J.; Zhang, W.H.; Di, W. Management and outcomes of pregnancy with or without lupus nephritis: A systematic review and meta-analysis. Ther. Clin. Risk. Manag. 2018, 14, 885–901. [Google Scholar] [CrossRef] [PubMed]
- Wei, S.; Lai, K.; Yang, Z.; Zeng, K. Systemic lupus erythematosus and risk of preterm birth: A systematic review and meta-analysis of observational studies. Lupus 2017, 26, 563–571. [Google Scholar] [CrossRef] [PubMed]
- Smyth, A.; Oliveira, G.H.; Lahr, B.D.; Bailey, K.R.; Norby, S.M.; Garovic, V.D. A systematic review and meta-analysis of pregnancy outcomes in patients with systemic lupus erythematosus and lupus nephritis. Clin. J. Am. Soc. Nephrol. 2010, 5, 2060–2068. [Google Scholar] [CrossRef] [PubMed]
- Carvalheiras, G.; Vita, P.; Marta, S.; Trovão, R.; Farinha, F.; Braga, J.; Rocha, G.; Almeida, I.; Marinho, A.; Mendonça, T.; et al. Pregnancy and systemic lupus erythematosus: Review of clinical features and outcome of 51 pregnancies at a single institution. Clin. Rev. Allergy Immunol. 2010, 38, 302–306. [Google Scholar] [CrossRef] [PubMed]
- Vellanki, V.S.; Parvathina, S.; Gondi, S.; Yadla, M.; Chenu, K.K.; Vishnubhotla, S. Post-partum bilateral renal cortical necrosis in antiphospholipid syndrome and systemic lupus erythematosus. Saudi J. Kidney Dis. Transpl. 2013, 24, 549–552. [Google Scholar] [PubMed]
- Mizuno, R.; Fujimoto, S.; Fujimoto, T.; Nishino, T.; Shiiki, H.; Hashimoto, T.; Nakamura, S.; Dohi, K. Catastrophic antiphospholipid antibody syndrome in systemic lupus erythematosus: An autopsy case report of a young woman. Intern. Med. 2000, 39, 856–859. [Google Scholar] [CrossRef] [PubMed]
- Moroni, G.; Doria, A.; Giglio, E.; Imbasciati, E.; Tani, C.; Zen, M.; Strigini, F.; Zaina, B.; Tincani, A.; Gatto, M. Maternal outcome in pregnant women with lupus nephritis. A prospective multicenter study. J. Autoimmun. 2016, 74, 194–200. [Google Scholar] [CrossRef] [PubMed]
- Buyon, J.P.; Kim, M.Y.; Guerra, M.M.; Laskin, C.A.; Petri, M.; Lockshin, M.D.; Sammaritano, L.; Branch, D.W.; Porter, T.F.; Sawitzke, A. Predictors of Pregnancy Outcomes in Patients with Lupus: A Cohort Study. Ann. Intern. Med. 2015, 163, 153–163. [Google Scholar] [CrossRef] [PubMed]
- Alchi, B.; Griffiths, M.; Jayne, D. What nephrologists need to know about antiphospholipid syndrome. Nephrol. Dial. Transplant. 2010, 25, 3147–3154. [Google Scholar] [CrossRef] [PubMed]
- Hanouna, G.; Morel, N.; Le Thi Huong, D.; Josselin, L.; Vauthier-Brouzes, D.; Saadoun, D.; Kettaneh, A.; Levesque, K.; Le Guern, V.; Goffinet, F.; et al. Catastrophic antiphospholipid syndrome and pregnancy: An experience of 13 cases. Rheumatology (Oxford) 2013, 52, 1635–1641. [Google Scholar] [CrossRef] [PubMed]
- Dashe, J.S.; Ramin, S.M.; Cunningham, F.G. The long-term consequences of thrombotic microangiopathy (thrombotic thrombocytopenic purpura and hemolytic uremic syndrome) in pregnancy. Obstet. Gynecol. 1998, 91, 662–668. [Google Scholar] [PubMed]
- Silver, R.M. Catastrophic antiphospholipid syndrome and pregnancy. Semin. Perinatol. 2018, 42, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Rao, S.; Jim, B. Acute Kidney Injury in Pregnancy: The Changing Landscape for the 21st Century. Kidney Int. Rep. 2018, 3, 247–257. [Google Scholar] [CrossRef] [PubMed]
- Cervera, R.; Serrano, R.; Pons-Estel, G.J.; Ceberio-Hualde, L.; Shoenfeld, Y.; de Ramón, E. Morbidity and mortality in the antiphospholipid syndrome during a 10-year period: A multicentre prospective study of 1000 patients. Ann. Rheum. Dis. 2015, 74, 1011–1018. [Google Scholar] [CrossRef] [PubMed]
- Pengo, V.; Banzato, A.; Bison, E.; Bracco, A.; Denas, G.; Ruffatti, A. What have we learned about antiphospholipid syndrome from patients and antiphospholipid carrier cohorts? Semin. Thromb. Hemost. 2012, 38, 322–327. [Google Scholar] [CrossRef] [PubMed]
- Ducloy-Bouthors, A.S.; Caron, C.; Subtil, D. Thrombotic thrombocytopenic purpura: Medical and biological monitoring of six pregnancies. Eur. J. Obstet. Gynecol. Reprod. Biol. 2003, 111, 146–152. [Google Scholar] [CrossRef]
- Sanchez-Luceros, A.; Farias, C.E.; Amaral, M.M. Von Willebrand factor cleaving protease (ADAMTS13) activity in normal non-pregnant women, pregnant and post-delivery women. Thromb. Haemost. 2004, 92, 1320–1326. [Google Scholar] [CrossRef] [PubMed]
- Fakhouri, F.; Roumenina, L.; Provot, F.; Sallée, M.; Caillard, S.; Couzi, L. Pregnancy-associated hemolytic uremic syndrome revisited in the era of complement gene mutations. J. Am. Soc. Nephrol. 2010, 21, 859–867. [Google Scholar] [CrossRef] [PubMed]
- Fakhouri, F.; Jablonski, M.; Lepercq, J. Factor H, membrane cofactor protein, and factor I mutations in patients with hemolysis, elevated liver enzymes, and low platelet count syndrome. Blood 2008, 112, 4542–4545. [Google Scholar] [CrossRef] [PubMed]
- Baghli, S.; Abendroth, C.; Farooq, U.; Schaub, J.A. Atypical Presentation of Pregnancy-Related Hemolytic Uremic Syndrome. Am. J. Kidney Dis. 2018, 72, 451–456. [Google Scholar] [CrossRef] [PubMed]
- Barbullushi, M.; Idrizi, A.; Spasovski, G. Familial hemolytic uremic syndrome with occurrence in the postpartum period. Saudi J. Kidney Dis. Transpl. 2017, 28, 1427–1431. [Google Scholar] [CrossRef] [PubMed]
- Gately, R.; San, A.; Kurtkoti, J.; Parnham, A. Life-threatening pregnancy-associated atypical haemolytic uraemic syndrome and its response to eculizumab. Nephrology (Carlton) 2017, 22, 32–35. [Google Scholar] [CrossRef] [PubMed]
- Servais, A.; Devillard, N.; Frémeaux-Bacchi, V.; Hummel, A.; Salomon, L.; Contin-Bordes, C.; Gomer, H.; Legendre, C.; Delmas, Y. Atypical haemolytic uraemic syndrome and pregnancy: Outcome with ongoing eculizumab. Nephrol. Dial. Transplant. 2016, 31, 2122–2130. [Google Scholar] [CrossRef] [PubMed]
- Noris, M.; Bresin, E.; Mele, C.; Remuzzi, G. Genetic Atypical Hemolytic-Uremic Syndrome. In GeneReviews; Adam, M.P., Ardinger, H.H., Pagon, R.A., Wallace, S.E., Bean, L.J.H., Stephens, K., Amemiya, A., Eds.; University of Washington: Seattle, WA, USA, 2007. [Google Scholar]
- Shanmugalingam, R.; Hsu, D.; Makris, A. Pregnancy-induced atypical haemolytic uremic syndrome: A new era with eculizumab. Obstet. Med. 2018, 11, 28–31. [Google Scholar] [CrossRef] [PubMed]
| Main Clinical Feature | Phase | Condition | Main Clinical Features |
|---|---|---|---|
| Prerenal | |||
| Hypovolemic | Early | Hyperemesis gravidarum | May be severe, associated with severe nutritional deficits, so more common in patients with a nutritional disorder or malnutrition; may reflect psychological problems. More commonly diagnosed in developed countries, it is probably underestimated in developing ones. |
| Any time | Other causes of hypovolemia | Infectious diseases, nutritional disorders; acute fatty liver of pregnancy; metabolic acidosis, uremia. | |
| Haemorrhagic | Early | Abortion | Early foetal loss can cause severe haemorrhaging, but unsafe, illegal abortions are the most common cause of massive bleeding, usually associated with sepsis. More common in low- and middle-income countries, and where abortion is illegal. |
| Late | Placental abruption | Can cause massive bleeding, as well as foetal loss, usually occurs in late pregnancy. | |
| Hypotensive | Any time | Hypovolemia, cardiopathy, sepsis | Hypotension is usually a concomitant cause and a marker of severity of the above. The rare, but sometimes severe, cardiomyopathy of pregnancy can lead to severe hypotension and AKI. Sepsis (any cause, any phase) is often associated with hypotension up to hypotensive shock, and associated with tubular necrosis (see below). |
| Combined pathogenesis | Any time | Septic abortion, placental abruption, puerperal sepsis | Severe bleeding is associated with hypovolemia and hypotension. While the “usual” classification of AKI may be of help, focusing on one element only may avert attention from treating all associated factors. |
| Parenchymal (for PE, and related disorders, see further tables) | |||
| Glomerular | Any time | CKD (known or undiagnosed) | The presence of CKD is associated with adverse pregnancy outcomes starting from the early stages. Immunologic diseases may relapse or appear in pregnancy. CKD worsening is described in 20–80% of patients. |
| Usually late | Microangiopaties | Haemolytic uremic syndrome and related diseases are an emerging concern in particular in developed countries probably because they fail to be diagnosed in low-income settings. | |
| Interstitial damage | Any time | Iatrogenic, associated with other causes of AKI | The causes are the same encountered outside of pregnancy but the consequences may be more severe. Whether the “pregnant kidney” is associated with increased risk is a matter of debate. History of obstructive renal disease and vesicoureteral reflux may contribute to loss of parenchymal volume which for still unknown reasons may be facilitate PE and possibly AKI |
| Any time | Pyelonephritis and upper urinary tract infections | These infections seldom cause AKI, even though they can be severe and life threatening. In this context, AKI is usually linked to sepsis or is iatrogenic. | |
| Combined pathogenesis | Any time | Tubular necrosis- cortical necrosis | Tubular necrosis may result from any severe AKI, and may be multifactorial. |
| Postrenal-Obstructive | |||
| Obstruction of the urinary tract | Any time | Stone disease | Hypercalciuria can occur in pregnancy. Pain due to the passage of a stone may be misinterpreted, especially at term; infection and undiagnosed obstruction may be life- and function-threatening. |
| Post partum | Iatrogenic | Ligature of the ureters is a rare but serious iatrogenic complication of caesarean section or reinterventions. | |
| Any time | Neoplasia | Kidney and urinary tract neoplasia are rare in young women. Diagnosis should be considered in macroscopic haematuria, in particular if there are clots. | |
| Usually late | ADPKD and other cystic diseases of the kidney | Cystic diseases of the kidney may go unrecognized in younger women. Large non-symptomatic cysts may become symptomatic, cause pain or obstruction, or become infected in pregnancy. | |
| Functional | Usually late | Functional obstruction and hydronephrosis | Mild urinary tract dilatation (usually on the right side) is common, and usually without consequences; pyelo-ureteral junction anomaly, may decompensate, leading to giant hydronephrosis. Severe urinary tract dilatation is described in patients with reflux nephropathy. |
| Combined pathogenesis | Any time | Infection, bleeding in obstructed kidney | Different causes may be associated; when sepsis (mainly Gram negative), is superimposed, AKI may be of combined parenchymal (septic), prerenal (hypovolemia, shock) and obstructive pathogenesis. |
| Term | Definition | Main Problems and Limits |
|---|---|---|
| Preeclampsia (PE) | Hypertension and proteinuria or end-organ damage in a previously healthy woman; involvement is reversible 1–3 months after delivery | Usually defined as new onset of proteinuria above 300 mg/day and hypertension after the 20th gestational week (GW) in a patient who was previously normotensive and without proteinuria or kidney disease. The syndrome resolves within three months from delivery. New definitions include hypertension in the absence of proteinuria but in the presence of end-organ damage, including creatinine increase. Does not apply to patients with chronic kidney disease or on dialysis, due to baseline hypertension, proteinuria, or to no urine output. |
| Eclampsia | Same as above, with neurological damage and convulsions | This definition is no longer universally accepted. Some authors consider it synonymous with untreated (or inadequately treated) PE. |
| Mild PE | PA ≥140/90 <160/110 proteinuria ≥0.3 <5.0 g/day in the absence of the criteria set forth above | The definition of mild and severe PE is somewhat static; mild PE can abruptly evolve into severe PE. Therefore, this definition should be used to: identify ALL cases of severe PE, that should be followed accordingly; identify cases in which the disease is not severe, allowing pregnancy to continue under careful surveillance, keeping in mind that mild PE can abruptly progress to severe PE. |
| Severe PE | Central nervous system involvement Liver damage Poorly controlled hypertension Proteinuria ≥5 g/24 h Platelets <100.000 Oliguria <500 mL/24 h Pulmonary oedema Intrauterine growth restriction (IUGR) | |
| Early PE | Before 34 completed GW | This pragmatic definition has the advantage of simplicity, and makes the early identification of cases possible; however a late diagnosis of early PE may in fact be identified as “late” PE. |
| Late PE | After 34 completed GW | |
| Maternal PE | With maternal predisposing disease | This pragmatic definition is clinically relevant (maternal PE is usually, but not uniformly milder); the definition of maternal disease is elusive, and the equation early = placental = severe; late = maternal = mild is imperfect and may be misleading. |
| Placental PE | Severe placental involvement in the absence of the above | |
| Angiogenic PE | Altered angiogenic/non-angiogenic balance | This is a promising approach, with the advantage of simplicity and of employing numeric values, and is possibly more objective; however, there is no agreement on how best to test for the condition: cut-points are not univocal, the levels and the ratio between factors may change over time and the availability of the tests is limited. |
| Non angiogenic PE | Absence of the above | |
| Superimposed PE | PE with underlying CKD | Some but not all CKD are proteinuric or hypertensive before pregnancy; this definition tries to correct for the baseline values. However, there is no agreed level of “worsening” of proteinuria or hypertension, in part because of the adaptation of anti-hypertensive treatments in pregnancy. Albeit of potential interest, this definition is ambiguous and less frequently encountered. |
| Postpartum PE | A clinical syndrome with the clinical features of PE, occurring postpartum | A rare occurrence, probably accounting for less than 5% of PE. The clinical picture is often severe and onset is abrupt; diagnosis may be difficult and delayed, since it may occur after hospital discharge. |
| Other related definitions (other hypertensive disorders of pregnancy) | ||
| Pregnancy induced hypertension (PIH) | “Isolated” hypertension in pregnancy | Hypertension occurring after the 20th week of pregnancy in a previously normotensive woman; the definition is the same as for PE; the absence of proteinuria is however no longer sufficient to exclude PE (see above). |
| Pregnancy induced proteinuria | “Isolated” proteinuria in pregnancy. | Proteinuria above 300 mg/day occurring after the 20th week of pregnancy in a previously non-proteinuric woman. Often a sign of underlying CKD. |
| HELLP syndrome | A virulent syndrome of endothelial damage | The acronym stands for hypertension, elevated liver enzymes, low platelets (HELLP); it is a severe syndrome, often abrupt, potentially occurring in the immediate postpartum period. Some authors consider it as the end of the spectrum of PE (severest disease); others consider it a separate disease, due to the frequent lack of prodromal PE. |
| IUGR | Intrauterine growth restriction | Some authors relate IUGR to the hypertensive disorders of pregnancy, due to its pathogenesis, which is related to insufficient placental vascularization. See Table 3 for details. |
| Structure | Main Clinical Feature | Pathologic Findings | Long-Term Effects |
|---|---|---|---|
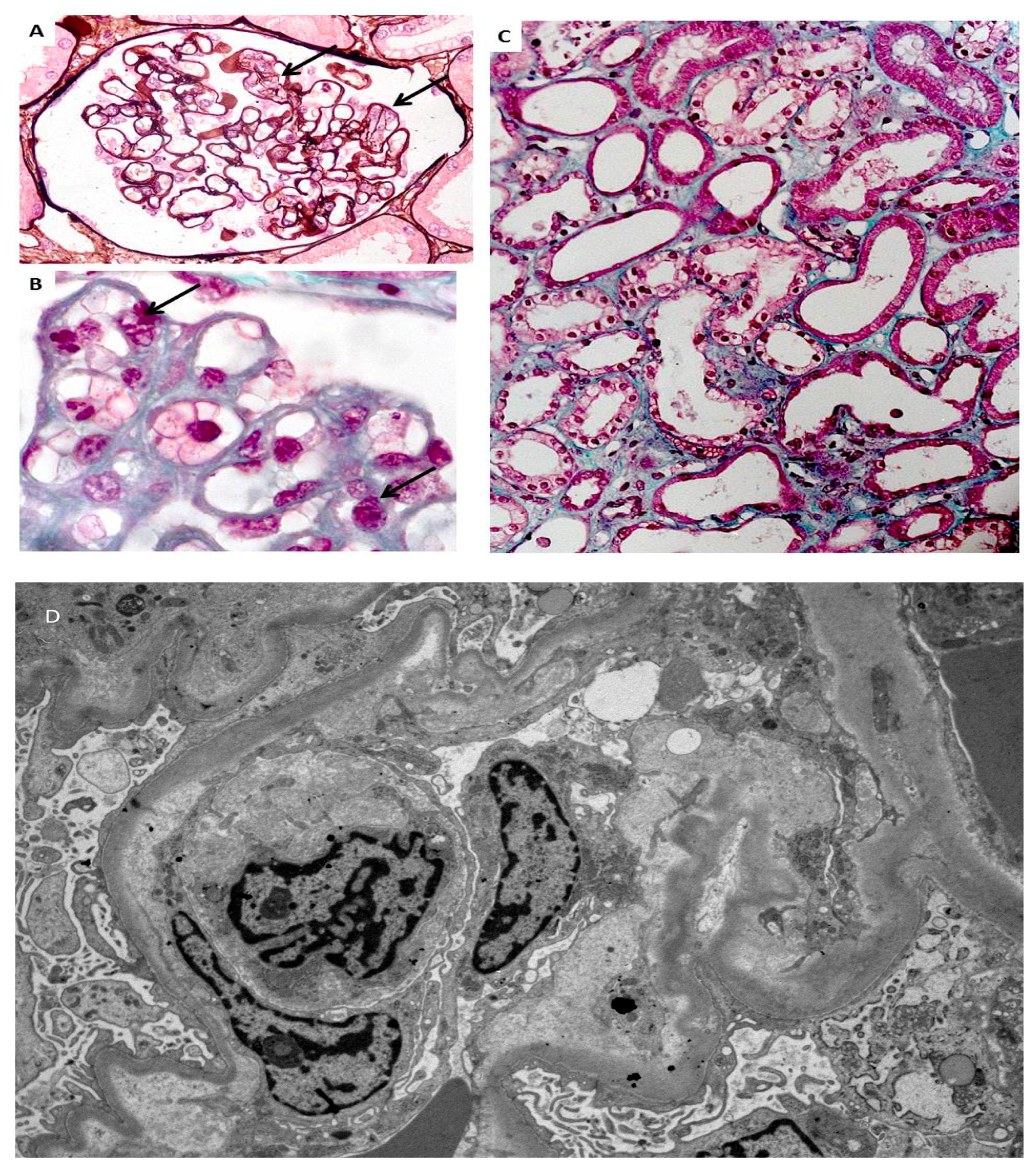
| Glomerular | Isolated proteinuria, PE, AKI | Glomerular endotheliosis (PE), focal-segmental glomerulosclerosis (FSGS); podocyte loss. Differential diagnosis with CKD | Endotheliosis is the hallmark of PE. It is considered reversible, and may also be found in normal pregnancies. FSGS is not reversible. Its frequency has been differently assessed, and the symptoms of severe endotheliosis may merge with it. Podocyte loss, with permanent damage to the basal membrane seems to be the unifying cellular lesion. |
| Tubular | AKI, severe PE, HELLP | Acute tubular necrosis, tubulitis, no alteration | AKI can be an outcome of ischemic, hypovolemic. septic or toxic (iatrogenic) damage. The long-term effects of AKI are not fully understood, but severe dialysis-requiring AKI even when reversible in the short term, is associated with CKD and ESRD later in life. |
| Vascular | Hypertensive disorders, PE, AKI | Vascular hypertrophy, up to “onion skin” lesions; ischemic or necrotic glomerular changes | Hypertensive crises in pregnancy may exhibit the features of “malignant” or accelerated hypertension. While onion-skin changes indicate a progressive onset, necrotic changes are the hallmark of the abrupt development of severe hypertension. |
| Global | AKI followed by CKD | Cortical necrosis | The irreversible loss of function associated with necrotic changes is often multifactorial (hypovolemic or haemorrhagic shock, sepsis, HELLP); can be diagnosed using magnetic resonance imaging. |
| Term | Definition | Main Problems and Limits |
|---|---|---|
| Small baby | A baby weighing less than 2500 g at birth | While there is a trend towards an inverse association between birth weight and clinical problems, the outcome of “small babies” depends on the pathogenesis of low birth weight; hence, measures adjusted for gestational age and growth patterns show a better relationship with short- and long-term outcomes. |
| Very small baby | A baby weighing less than 1500 g at birth | |
| Pre-term delivery | Delivery before 37 completed gestational weeks; late pre-term: 34–37 gestational weeks | These three widely-used terms were defined in relation to “normal development” (at term); higher risk for mild developmental or intellectual deficits (late pre-term is mainly associated with school problems); extreme preterm is associated with higher frequency of mild deficits, and higher incidence of severe developmental neurologic problems. The risk for metabolic syndrome, hypertension and CKD, albeit less well explored, may follow a similar pattern. Once more, these definitions should be associated with data on intrauterine growth; outcome may be modified by follow-up. |
| Early pre-term delivery | Delivery before 34 completed gestational weeks | |
| Very early (or extreme) pre-term delivery | Delivery before 28 completed gestational weeks | |
| Small for gestational age baby (SGA) | A baby below the 5th or the 10th centile for gestational age, adjusted to local growth curves | This finer definition takes into account the relationship between being small or preterm and the “quality” of intrauterine growth. This definition does not distinguish between harmonic growth of genetically small children (not pathologic) and stunted growth of children not attaining their genetic growth target. Given the wide ethnic differences in growth curves, the absence of local data may impair precise interpretation. Other relevant terms are AGA (adequate for gestational age) and LGA (large for gestational age: above the 90th or the 95th centile) |
| Intrauterine growth restriction (IUGR) | A baby that does not reach its growth potential, i.e. one below the 5th centile for gestational age, or whose growth curve flattens in pregnancy | Growth restriction is a dynamic concept that indicates flattening of the growth curve. Data are emerging on the pivotal importance of the “quality” of intrauterine growth, which may be more important than actual birth weight, or prematurity; while babies who are SGA below the 5th centile are so often IUGR that they are included in the definitions, a baby may still be adequate for gestational age, be born at term, but be IUGR, if its growth was severely reduced in the last gestational weeks. |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Piccoli, G.B.; Zakharova, E.; Attini, R.; Ibarra Hernandez, M.; Covella, B.; Alrukhaimi, M.; Liu, Z.-H.; Ashuntantang, G.; Orozco Guillen, A.; Cabiddu, G.; et al. Acute Kidney Injury in Pregnancy: The Need for Higher Awareness. A Pragmatic Review Focused on What Could Be Improved in the Prevention and Care of Pregnancy-Related AKI, in the Year Dedicated to Women and Kidney Diseases. J. Clin. Med. 2018, 7, 318. https://doi.org/10.3390/jcm7100318
Piccoli GB, Zakharova E, Attini R, Ibarra Hernandez M, Covella B, Alrukhaimi M, Liu Z-H, Ashuntantang G, Orozco Guillen A, Cabiddu G, et al. Acute Kidney Injury in Pregnancy: The Need for Higher Awareness. A Pragmatic Review Focused on What Could Be Improved in the Prevention and Care of Pregnancy-Related AKI, in the Year Dedicated to Women and Kidney Diseases. Journal of Clinical Medicine. 2018; 7(10):318. https://doi.org/10.3390/jcm7100318
Chicago/Turabian StylePiccoli, Giorgina Barbara, Elena Zakharova, Rossella Attini, Margarita Ibarra Hernandez, Bianca Covella, Mona Alrukhaimi, Zhi-Hong Liu, Gloria Ashuntantang, Alejandra Orozco Guillen, Gianfranca Cabiddu, and et al. 2018. "Acute Kidney Injury in Pregnancy: The Need for Higher Awareness. A Pragmatic Review Focused on What Could Be Improved in the Prevention and Care of Pregnancy-Related AKI, in the Year Dedicated to Women and Kidney Diseases" Journal of Clinical Medicine 7, no. 10: 318. https://doi.org/10.3390/jcm7100318
APA StylePiccoli, G. B., Zakharova, E., Attini, R., Ibarra Hernandez, M., Covella, B., Alrukhaimi, M., Liu, Z.-H., Ashuntantang, G., Orozco Guillen, A., Cabiddu, G., Li, P. K. T., Garcia-Garcia, G., & Levin, A. (2018). Acute Kidney Injury in Pregnancy: The Need for Higher Awareness. A Pragmatic Review Focused on What Could Be Improved in the Prevention and Care of Pregnancy-Related AKI, in the Year Dedicated to Women and Kidney Diseases. Journal of Clinical Medicine, 7(10), 318. https://doi.org/10.3390/jcm7100318










