Mechanisms Involved in the Improvement of Lipotoxicity and Impaired Lipid Metabolism by Dietary α-Linolenic Acid Rich Salvia hispanica L (Salba) Seed in the Heart of Dyslipemic Insulin-Resistant Rats
Abstract
:1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Experimental Design
| Diet Ingredients | Control Diet (CD) | Sucrose-Rich Diet (SRD) | SRD+chia Seed (SRD+chia) | |||
|---|---|---|---|---|---|---|
| % w/w | % Energy | % w/w | % Energy | % w/w | % Energy | |
| Carbohydrates | ||||||
| Corn starch | 58.0 | 60.0 | 2.5 | 2.6 | -- | -- |
| Sucrose | -- | -- | 55.5 | 57.4 | 55.5 | 57.4 |
| Chia seed 2 | -- | -- | -- | -- | 2.5 | 2.6 |
| Fat | ||||||
| Corn oil | 10.5 | 23.0 | 10.5 | 23.0 | 0.1 | 0.2 |
| Chia seed | -- | -- | -- | -- | 10.4 | 22.8 |
| Protein | ||||||
| Casein (vitamin free) | 16.3 | 17.0 | 16.3 | 17.0 | 8.6 | 9.0 |
| Chia seed | -- | -- | -- | -- | 7.7 | 8.0 |
| Fatty Acids 1 | CD and SRD | SRD+chia Seed |
|---|---|---|
| 16:0 | 10.92 | 6.96 |
| 18:0 | 2.73 | 2.42 |
| 18:1 n-9 | 33.71 | 7.39 |
| 18:2 n-6 | 54.10 | 19.85 |
| 18:3 n-3 | 0.80 | 67.26 |
| 20:1 n-9 | 0.47 | 0.36 |
| Total saturated | 13.65 | 9.38 |
| Monounsaturated | 34.18 | 7.75 |
| Polyunsaturated | ||
| n-6 | 54.10 | 19.85 |
| n-3 | 0.80 | 67.26 |
| n-6/n-3 | 67.62 | 0.295 |
2.3. Analytical Methods
2.4. Determination of Blood Pressure
2.5. Enzymatic Activity Assays of CPT and PDHc
2.6. Determination of FAT/CD36 Protein Mass Level (Euglycemic-Hyperinsulinemic Clamp Studies)
2.7. Determination of M-CPT1 and PPARα Protein Mass Levels
2.8. Determination of UCP2 Protein Mass Level
2.9. Statistical Analysis
3. Results
3.1. Body Weight, Energy Intake and Visceral Adiposity Index
| Diet | Body Weight (g) | Energy Intake (kJ/Day) | Diet | Body Weight (g) | Energy Intake (kJ/Day) | Visceral Adiposity Index (%) | |
|---|---|---|---|---|---|---|---|
| Initial | 3 Months | Initial to 3 Months | 6 Months | 3 to 6 Months | 6 Months | ||
| CD (8) | 184.3 ± 2.6 | 414.5 ± 5.5 | 294.5 ± 12.5 | CD (8) | 476.3 ± 7.6 b | 292.0 ± 7.2 b | 4.1 ± 0.3 b |
| SRD (16) | 186.0 ± 1.6 | 428.0 ± 6.0 | 292.0 ± 7.2 | SRD (8) | 545.0 ± 10.0 a | 374.0 ± 9.5 a | 6.2 ± 0.4 a |
| SRD+chia (8) | 524.0 ± 7.3 a | 356.5 ± 13.0 a | 4.5 ± 0.2 b | ||||
3.2. Total and Relative Heart Weight and Systolic Blood Pressure
| CD | SRD | SRD+chia | |
|---|---|---|---|
| Heart tissue | |||
| Total wet weight, g | 1.24 ± 0.01 b (8) | 1.31 ± 0.03 a (8) | 1.28 ± 0.02 a (8) |
| g wet weight/100 g body weight | 0.260 ± 0.003 (8) | 0.250 ± 0.004 (8) | 0.250 ± 0.005 (8) |
| mg wet weight/mm tibia length | 27.2 ± 1.0 (8) | 30.1 ± 1.3 (8) | 28.3 ± 1.0 (8) |
| Systolic blood pressure, mmHg | |||
| Initial | 115.0 ± 4.6 (8) | 116.0 ± 2.4 (16) | |
| 3 months | 123.3 ± 1.0 b (8) | 130.9 ± 1.2 a (16) | |
| 6 months | 120.2 ± 4.8 b (8) | 138.4 ± 1.6 a (8) | 118.8 ± 1.6 b (8) |
3.3. Plasma Metabolites, Insulin Levels and GIR
| Diet | Time on Diet (Months) | Triglyceride (mM) | Free Fatty Acids (μM) | Cholesterol (mM) | Glucose (mM) | Insulin (μU/mL) |
|---|---|---|---|---|---|---|
| CD | 3 | 0.69 ± 0.04 b | 300.1 ± 16.0 b | 1.85 ± 0.10 b | 6.5 ± 0.2 b | 64.1 ± 3.2 |
| SRD | 3 | 1.98 ± 0.08 a | 716.0 ± 8.1 a | 3.21 ± 0.14 a | 7.9 ± 0.1 a | 60.1 ± 4.2 |
| CD | 6 | 0.72 ± 0.03 b | 335.0 ± 13.0 b | 1.92 ± 0.11 b | 6.6 ± 0.1 b | 62.0 ± 2.9 |
| SRD | 6 | 2.06 ± 0.17 a | 760.4 ± 16.3 a | 3.60 ± 0.04 a | 8.3 ± 0.1 a | 65.0 ± 3.2 |
| SRD+chia | 3 to 6 | 0.72 ± 0.05 b | 363.0 ± 35.4 b | 1.75 ± 0.21 b | 6.9 ± 0.1 b | 67.4 ± 6.5 |
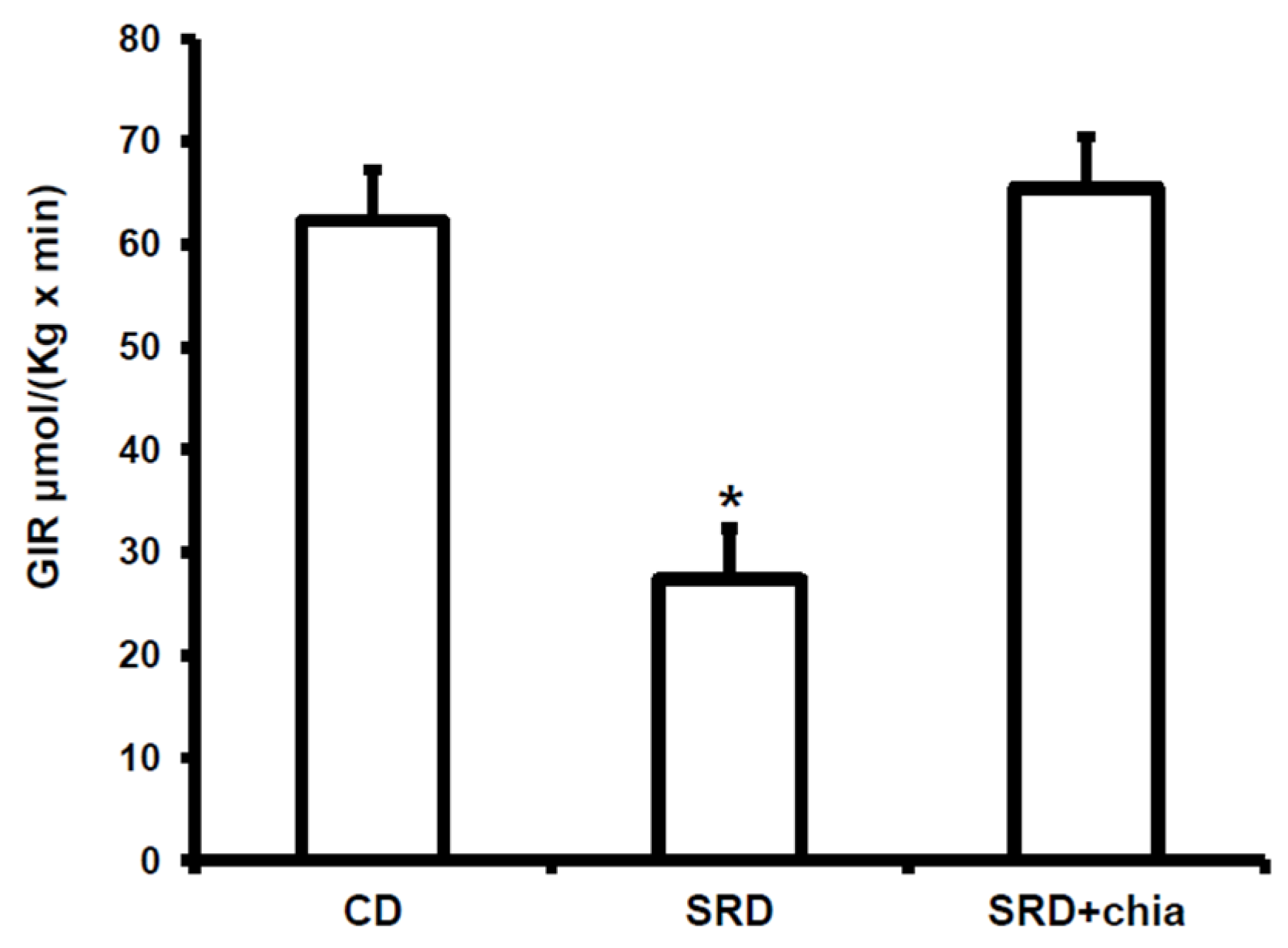
3.4. Heart Muscle Metabolites Concentrations and PDHc Activity
| CD | SRD | SRD+chia | |
|---|---|---|---|
| Triglyceride (μmol/g wet tissue) | 3.60 ± 0.22 b | 6.03 ± 0.34 a | 4.44 ± 0.60 b |
| LC ACoA (nmol/g wet tissue) | 31.4 ± 5.5 b | 68.0 ± 4.0 a | 45.8 ± 5.5 b |
| DAG (nmol/g wet tissue) | 250.8 ± 19.5 b | 355.3 ± 15.4 a | 276.6 ± 24.0 b |
| PDHa (% of total PDHc) | 59.7 ± 5.8 a | 26.6 ± 4.6 b | 45.1 ± 4.9 a |
3.5. FAT/CD36 Protein Mass Level in Heart Muscle at the Beginning and at the End of the Euglycemic-Hyperinsulinemic Clamp Studies

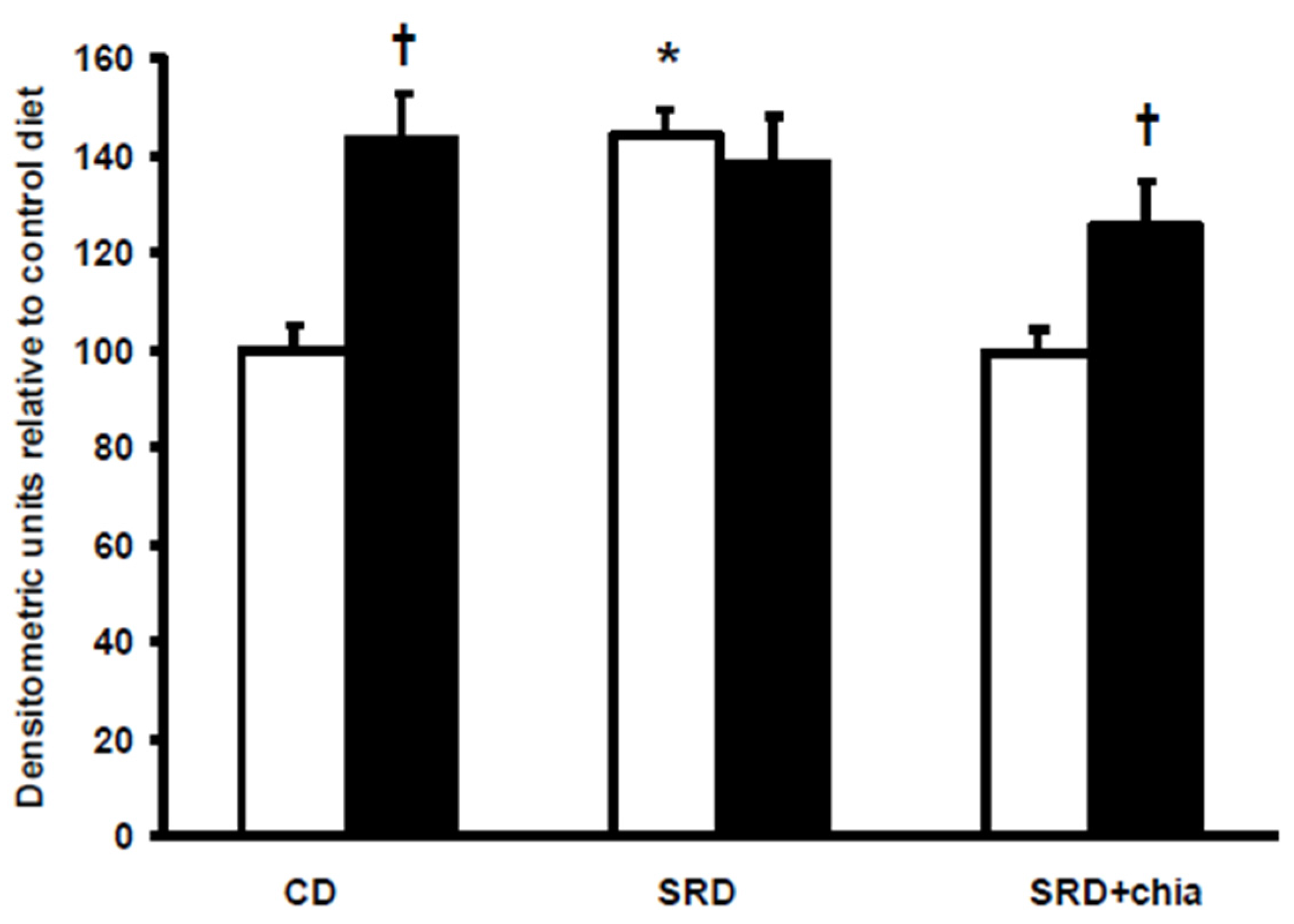
3.6. M-CPT1 Activity and Protein Mass Level

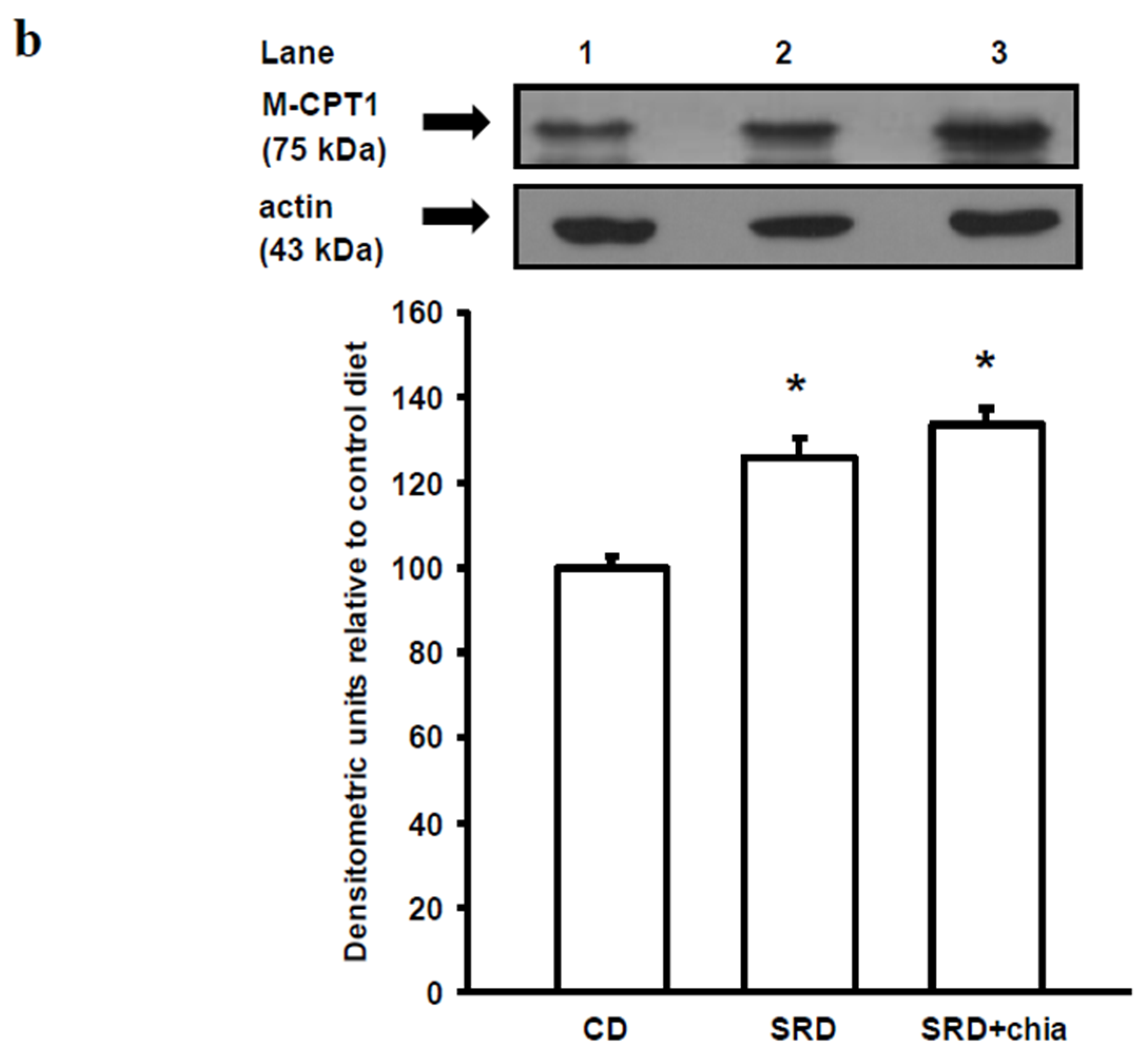
3.7. PPARα Protein Mass Level
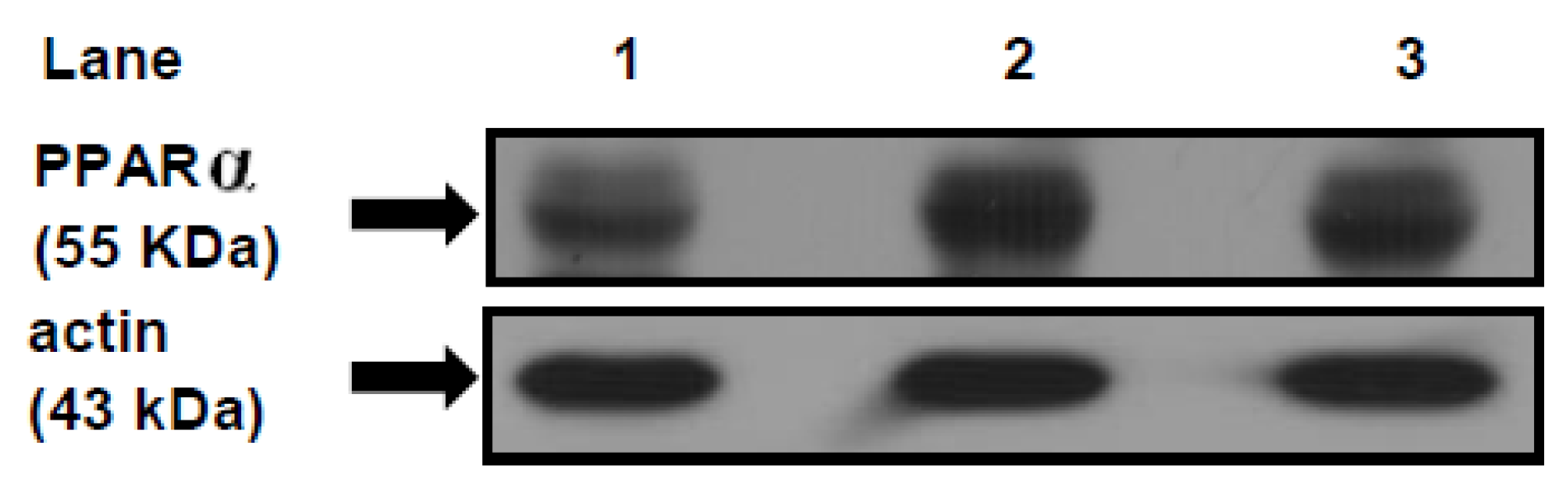
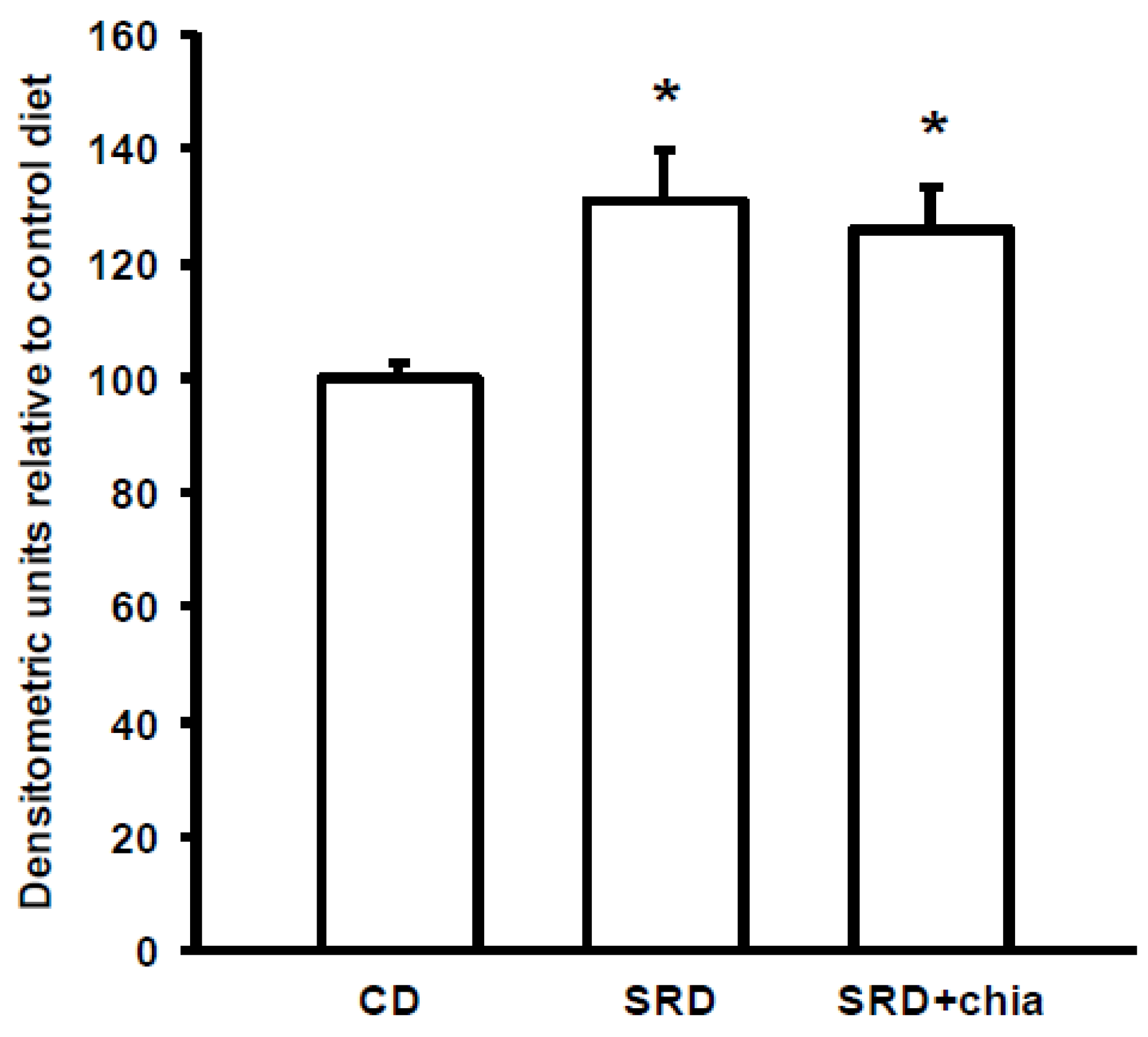
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Pollex, R.L.; Hegele, R.A. Genetic determinants of the metabolic syndrome. Nat. Clin. Pract. Cardiovasc. Med. 2006, 3, 482–489. [Google Scholar] [CrossRef] [PubMed]
- Gaziano, T.A.; Bitton, A.; Anand, S.; Abrahams-Gessel, S.; Murphy, A. Growing epidemic of coronary heart disease in low- and middle- income countries. Curr. Probl. Cardiol. 2010, 35, 72–115. [Google Scholar] [CrossRef] [PubMed]
- Mazumder, P.K.; O’Neill, B.T.; Roberts, M.W.; Buchanan, J.; Yun, U.J.; Cooksey, R.C.; Boudina, S.; Abel, E.D. Impaired cardiac efficiency and increased fatty acid oxidation in insulin-resistant Ob/Ob mouse hearts. Diabetes 2004, 53, 2366–2374. [Google Scholar] [CrossRef] [PubMed]
- Glatz, J.F.; Angin, Y.; Steinbusch, L.K.; Schwenk, R.W.; Luiken, J.J. CD36 as a target to prevent cardiac lipotoxicity and insulin resistance. Prostaglandins Leukot. Essent. Fatty Acids 2013, 88, 71–77. [Google Scholar] [CrossRef] [PubMed]
- Ouwens, D.M.; Diamant, M.; Fodor, M.; Habets, D.D.; Pelsers, M.M.; El, H.M.; Dang, Z.C.; van den Brom, C.E.; Vlasblom, R.; Rietdijk, A.; et al. Cardiac contractile dysfunction in insulin-resistant rats fed a high-fat diet is associated with elevated CD36-mediated fatty acid uptake and esterification. Diabetologia 2007, 50, 1938–1948. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.T.; Grayburn, P.; Karim, A.; Shimabukuro, M.; Higa, M.; Baetens, D.; Orci, L.; Unger, R.H. Lipotoxic heart disease in obese rats: Implications for human obesity. Proc. Natl. Acad. Sci. USA 2000, 97, 1784–1789. [Google Scholar] [CrossRef] [PubMed]
- Paulson, D.J.; Crass, M.F., III. Endogenous triacylglycerol metabolism in diabetic heart. Am. J. Physiol. 1982, 242, H1084–H1094. [Google Scholar] [PubMed]
- Van Oort, M.M.; van Doorn, J.M.; Bonen, A.; Glatz, J.F.; van der Horst, D.J.; Rodenburg, K.W.; Luiken, J.J. Insulin-induced translocation of CD36 to the plasma membrane is reversible and shows similarity to that of GLUT4. Biochim. Biophys. Acta 2008, 1781, 61–71. [Google Scholar] [CrossRef] [PubMed]
- Chiu, H.C.; Kovacs, A.; Ford, D.A.; Hsu, F.F.; Garcia, R.; Herrero, P.; Saffitz, J.E.; Schaffer, J.E. A novel mouse model of lipotoxic cardiomyopathy. J. Clin. Investig. 2001, 107, 813–822. [Google Scholar] [CrossRef] [PubMed]
- Coort, S.L.; Hasselbaink, D.M.; Koonen, D.P.; Willems, J.; Coumans, W.A.; Chabowski, A.; van der Vusse, G.J.; Bonen, A.; Glatz, J.F.; Luiken, J.J. Enhanced sarcolemmal FAT/CD36 content and triacylglycerol storage in cardiac myocytes from obese Zucker rats. Diabetes 2004, 53, 1655–1663. [Google Scholar] [CrossRef] [PubMed]
- Finck, B.N.; Han, X.; Courtois, M.; Aimond, F.; Nerbonne, J.M.; Kovacs, A.; Gross, R.W.; Kelly, D.P. A critical role for PPARalpha-mediated lipotoxicity in the pathogenesis of diabetic cardiomyopathy: Modulation by dietary fat content. Proc. Natl. Acad. Sci. USA 2003, 100, 1226–1231. [Google Scholar] [CrossRef] [PubMed]
- Barger, P.M.; Kelly, D.P. PPAR signaling in the control of cardiac energy metabolism. Trends Cardiovasc. Med. 2000, 10, 238–245. [Google Scholar] [CrossRef]
- Brandt, J.M.; Djouadi, F.; Kelly, D.P. Fatty acids activate transcription of the muscle carnitine palmitoyltransferase I gene in cardiac myocytes via the peroxisome proliferator-activated receptor alpha. J. Biol. Chem. 1998, 273, 23786–23792. [Google Scholar] [CrossRef] [PubMed]
- Lombardo, Y.B.; Chicco, A.G. Effects of dietary polyunsaturated n-3 fatty acids on dyslipidemia and insulin resistance in rodents and humans. A review. J. Nutr. Biochem. 2006, 17, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Montes, M.; Chicco, A.; Lombardo, Y.B. The effect of insulin on the uptake and metabolic fate of glucose in isolated perfused hearts of dyslipemic rats. J. Nutr. Biochem. 2000, 11, 30–37. [Google Scholar] [CrossRef]
- D’Alessandro, M.E.; Chicco, A.; Lombardo, Y.B. Dietary fish oil reverses lipotoxicity, altered glucose metabolism, and nPKCepsilon translocation in the heart of dyslipemic insulin-resistant rats. Metabolism 2008, 57, 911–919. [Google Scholar] [CrossRef] [PubMed]
- Jump, D.B.; Depner, C.M.; Tripathy, S. Omega-3 fatty acid supplementation and cardiovascular disease. J. Lipid Res. 2012, 53, 2525–2545. [Google Scholar] [CrossRef] [PubMed]
- Djousse, L.; Arnett, D.K.; Carr, J.J.; Eckfeldt, J.H.; Hopkins, P.N.; Province, M.A.; Ellison, R.C. Dietary linolenic acid is inversely associated with calcified atherosclerotic plaque in the coronary arteries: The National Heart, Lung, and Blood Institute Family Heart Study. Circulation 2005, 111, 2921–2926. [Google Scholar] [CrossRef] [PubMed]
- Mozaffarian, D.; Ascherio, A.; Hu, F.B.; Stampfer, M.J.; Willett, W.C.; Siscovick, D.S.; Rimm, E.B. Interplay between different polyunsaturated fatty acids and risk of coronary heart disease in men. Circulation 2005, 111, 157–164. [Google Scholar] [CrossRef] [PubMed]
- Folino, A.; Sprio, A.E.; di Scipio, F.; Berta, G.N.; Rastaldo, R. Alpha-linolenic acid protects against cardiac injury and remodeling induced by beta-adrenergic overstimulation. Food Funct. 2015, 6, 2231–2239. [Google Scholar] [CrossRef] [PubMed]
- Poudyal, H.; Panchal, S.K.; Ward, L.C.; Waanders, J.; Brown, L. Chronic high-carbohydrate, high-fat feeding in rats induces reversible metabolic, cardiovascular, and liver changes. Am. J. Physiol. Endocrinol. Metab. 2012, 302, E1472–E1482. [Google Scholar] [CrossRef] [PubMed]
- Poudyal, H.; Panchal, S.K.; Ward, L.C.; Brown, L. Effects of ALA, EPA and DHA in high-carbohydrate, high-fat diet-induced metabolic syndrome in rats. J. Nutr. Biochem. 2013, 24, 1041–1052. [Google Scholar] [CrossRef] [PubMed]
- Chicco, A.G.; D’Alessandro, M.E.; Hein, G.J.; Oliva, M.E.; Lombardo, Y.B. Dietary chia seed (Salvia hispanica L.) rich in alpha-linolenic acid improves adiposity and normalises hypertriacylglycerolaemia and insulin resistance in dyslipaemic rats. Br. J. Nutr. 2009, 101, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Rossi, A.S.; Oliva, M.E.; Ferreira, M.R.; Chicco, A.; Lombardo, Y.B. Dietary chia seed induced changes in hepatic transcription factors and their target lipogenic and oxidative enzyme activities in dyslipidaemic insulin-resistant rats. Br. J. Nutr. 2013, 109, 1617–1627. [Google Scholar] [CrossRef] [PubMed]
- Oliva, M.E.; Ferreira, M.R.; Chicco, A.; Lombardo, Y.B. Dietary salba (Salvia hispanica L) seed rich in alpha-linolenic acid improves adipose tissue dysfunction and the altered skeletal muscle glucose and lipid metabolism in dyslipidemic insulin-resistant rats. Prostaglandins Leukot. Essent. Fatty Acids 2013, 89, 279–289. [Google Scholar] [CrossRef] [PubMed]
- Laurell, S. A method for routine determination of plasma triglycerides. Scan. J. Clin. Lab. Investig. 1966, 18, 668–672. [Google Scholar] [CrossRef]
- Lowenstein, J.M. Citric Acid cycle. In Methods in Enzymology; Academic Press: New York, NY, USA, 1969; pp. 450–468. [Google Scholar]
- Schmitz-Peiffer, C.; Browne, C.L.; Oakes, N.D.; Watkinson, A.; Chisholm, D.J.; Kraegen, E.W.; Biden, T.J. Alterations in the expression and cellular localization of protein kinase C isozymes ε and θ are associated with insulin resistance in skeletal muscle of the high-fat-fed rat. Diabetes 1997, 46, 169–178. [Google Scholar] [CrossRef] [PubMed]
- Ling, B.; Aziz, C.; Alcorn, J. Systematic evaluation of key l-carnitine homeostasis mechanisms during postnatal development in rat. Nutr. Metab (Lond.) 2012, 9, 66. [Google Scholar] [CrossRef] [PubMed]
- Chicco, A.; D’Alessandro, M.E.; Karabatas, L.; Pastorale, C.; Basabe, J.C.; Lombardo, Y.B. Muscle lipid metabolism and insulin secretion are altered in insulin-resistant rats fed a high sucrose diet. J. Nutr. 2003, 133, 127–133. [Google Scholar] [PubMed]
- Rodnick, K.J.; Slot, J.W.; Studelska, D.R.; Hanpeter, D.E.; Robinson, L.J.; Geuze, H.J.; James, D.E. Immunocytochemical and biochemical studies of GLUT4 in rat skeletal muscle. J. Biol. Chem. 1992, 267, 6278–6285. [Google Scholar] [PubMed]
- Bogazzi, F.; Raggi, F.; Ultimieri, F.; Russo, D.; D’Alessio, A.; Manariti, A.; Brogioni, S.; Manetti, L.; Martino, E. Regulation of cardiac fatty acids metabolism in transgenic mice overexpressing bovine GH. J. Endocrinol. 2009, 201, 419–427. [Google Scholar] [CrossRef] [PubMed]
- Pecqueur, C.; Alves-Guerra, M.C.; Gelly, C.; Levi-Meyrueis, C.; Couplan, E.; Collins, S.; Ricquier, D.; Bouillaud, F.; Miroux, B. Uncoupling protein 2, in vivo distribution, induction upon oxidative stress, and evidence for translational regulation. J. Biol. Chem. 2001, 276, 8705–8712. [Google Scholar] [CrossRef] [PubMed]
- Glantz, S.A. Primer of Biostatistics; McGraw Hill: New York, NY, USA, 2005. [Google Scholar]
- Snedecor, G.W.; Cochran, W.G. Factorial Experiments, in Statistical Methods Applied to Experimental in Agriculture and Biology; Iowa State University Press: Ames, IA, USA, 1967; pp. 339–350. [Google Scholar]
- Chess, D.J.; Stanley, W.C. Role of diet and fuel overabundance in the development and progression of heart failure. Cardiovasc. Res. 2008, 79, 269–278. [Google Scholar] [CrossRef] [PubMed]
- Duncan, J.G. Peroxisome proliferator activated receptor-alpha (PPARalpha) and PPAR gamma coactivator-1alpha (PGC-1alpha) regulation of cardiac metabolism in diabetes. Pediatr. Cardiol. 2011, 32, 323–328. [Google Scholar] [CrossRef] [PubMed]
- Georgiadi, A.; Boekschoten, M.V.; Muller, M.; Kersten, S. Detailed transcriptomics analysis of the effect of dietary fatty acids on gene expression in the heart. Physiol. Genom. 2012, 44, 352–361. [Google Scholar] [CrossRef] [PubMed]
- Buchanan, J.; Mazumder, P.K.; Hu, P.; Chakrabarti, G.; Roberts, M.W.; Yun, U.J.; Cooksey, R.C.; Litwin, S.E.; Abel, E.D. Reduced cardiac efficiency and altered substrate metabolism precedes the onset of hyperglycemia and contractile dysfunction in two mouse models of insulin resistance and obesity. Endocrinology 2005, 146, 5341–5349. [Google Scholar] [CrossRef] [PubMed]
- Stanley, W.C.; Dabkowski, E.R.; Ribeiro, R.F., Jr.; O’Connell, K.A. Dietary fat and heart failure: Moving from lipotoxicity to lipoprotection. Circ. Res. 2012, 110, 764–776. [Google Scholar] [CrossRef] [PubMed]
- Franekova, V.; Angin, Y.; Hoebers, N.T.; Coumans, W.A.; Simons, P.J.; Glatz, J.F.; Luiken, J.J.; Larsen, T.S. Marine omega-3 fatty acids prevent myocardial insulin resistance and metabolic remodeling as induced experimentally by high insulin exposure. Am. J. Physiol. Cell Physiol. 2015, 308, C297–C307. [Google Scholar] [CrossRef] [PubMed]
- Ayerza, R.; Coates, W. Ground chia seed and chia oil effects on plasma lipids and fatty acids in the rat. J. Endocrinol. 2005, 25, 995–1003. [Google Scholar] [CrossRef]
- Li, N.; Wang, J.; Gao, F.; Tian, Y.; Song, R.; Zhu, S.J. The role of uncoupling protein 2 in the apoptosis induced by free fatty acid in rat cardiomyocytes. J. Cardiovasc. Pharmacol. 2010, 55, 161–167. [Google Scholar] [CrossRef] [PubMed]
- Rousseau, D.; Helies-Toussaint, C.; Moreau, D.; Raederstorff, D.; Grynberg, A. Dietary n-3 PUFAs affect the blood pressure rise and cardiac impairments in a hyperinsulinemia rat model in vivo. Am. J. Physiol. Heart Circ. Physiol. 2003, 285, H1294–H1302. [Google Scholar] [CrossRef] [PubMed]
- Vuksan, V.; Whitham, D.; Sievenpiper, J.L.; Jenkins, A.L.; Rogovik, A.L.; Bazinet, R.P.; Vidgen, E.; Hanna, A. Supplementation of conventional therapy with the novel grain Salba (Salvia hispanica L.) improves major and emerging cardiovascular risk factors in Type 2 diabetes: Results of a randomized controlled trial. Diabetes Care 2007, 30, 2804–2810. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Creus, A.; Ferreira, M.R.; Oliva, M.E.; Lombardo, Y.B. Mechanisms Involved in the Improvement of Lipotoxicity and Impaired Lipid Metabolism by Dietary α-Linolenic Acid Rich Salvia hispanica L (Salba) Seed in the Heart of Dyslipemic Insulin-Resistant Rats. J. Clin. Med. 2016, 5, 18. https://doi.org/10.3390/jcm5020018
Creus A, Ferreira MR, Oliva ME, Lombardo YB. Mechanisms Involved in the Improvement of Lipotoxicity and Impaired Lipid Metabolism by Dietary α-Linolenic Acid Rich Salvia hispanica L (Salba) Seed in the Heart of Dyslipemic Insulin-Resistant Rats. Journal of Clinical Medicine. 2016; 5(2):18. https://doi.org/10.3390/jcm5020018
Chicago/Turabian StyleCreus, Agustina, María R. Ferreira, María E. Oliva, and Yolanda B. Lombardo. 2016. "Mechanisms Involved in the Improvement of Lipotoxicity and Impaired Lipid Metabolism by Dietary α-Linolenic Acid Rich Salvia hispanica L (Salba) Seed in the Heart of Dyslipemic Insulin-Resistant Rats" Journal of Clinical Medicine 5, no. 2: 18. https://doi.org/10.3390/jcm5020018
APA StyleCreus, A., Ferreira, M. R., Oliva, M. E., & Lombardo, Y. B. (2016). Mechanisms Involved in the Improvement of Lipotoxicity and Impaired Lipid Metabolism by Dietary α-Linolenic Acid Rich Salvia hispanica L (Salba) Seed in the Heart of Dyslipemic Insulin-Resistant Rats. Journal of Clinical Medicine, 5(2), 18. https://doi.org/10.3390/jcm5020018





