Integrated Exposure-Based Therapy for Co-Occurring Post Traumatic Stress Disorder (PTSD) and Substance Dependence: Predictors of Change in PTSD Symptom Severity
Abstract
:1. Introduction
2. Methods
2.1. Procedure
2.2. Structured Interviews
2.3. Interventions
2.3.1. COPE
2.3.2. TAU
2.4. Statistical Analyses
3. Results
3.1. Participant Baseline Characteristics
3.1.1. Demographics
3.1.2. Substance Use
3.1.3. Trauma History
3.1.4. Mental Health
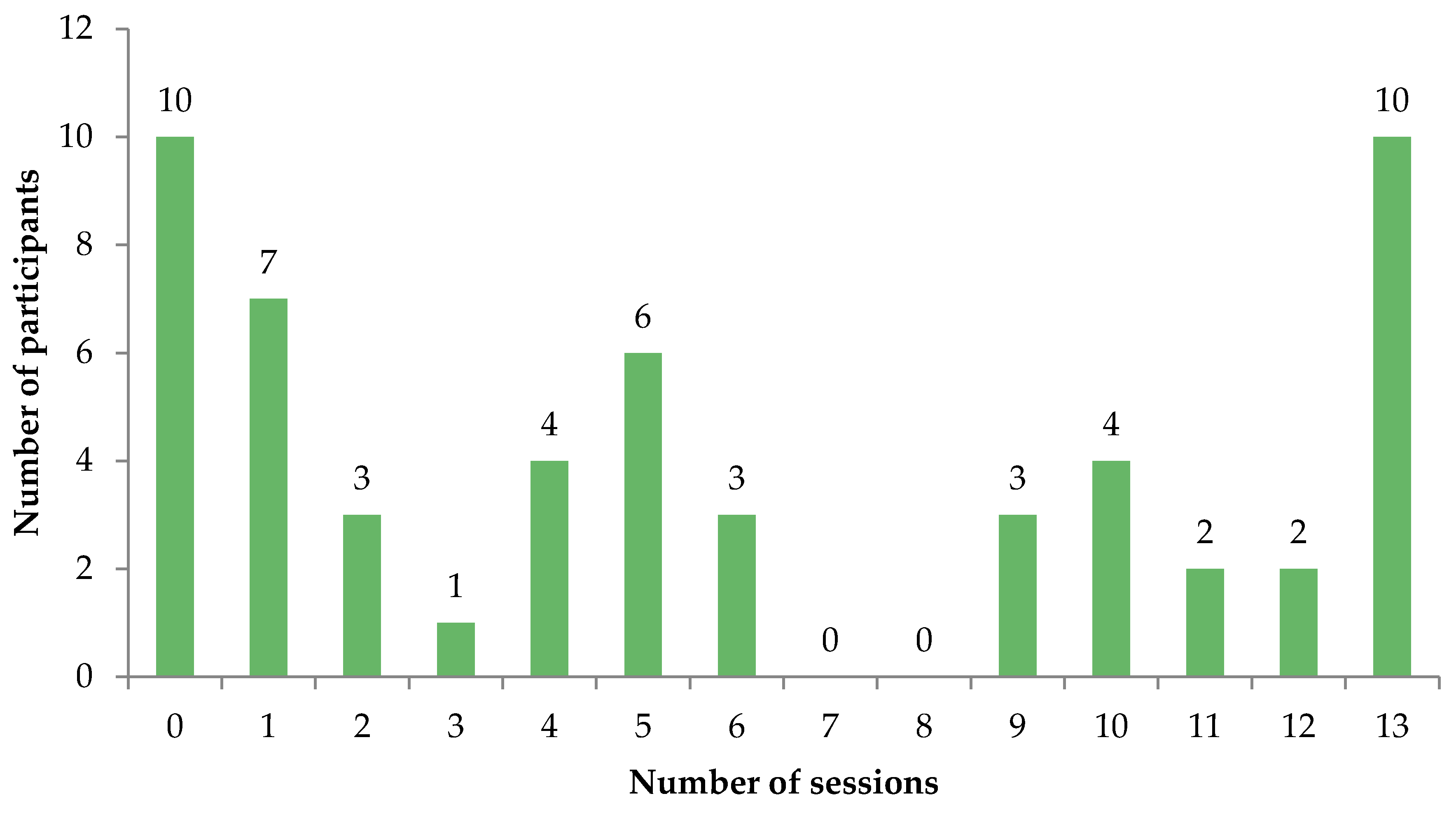
3.2. COPE Treatment Characteristics
3.3. Events over Follow-Up
3.3.1. Substance Use Treatment
3.3.2. New Trauma Exposure
3.4. Change in PTSD Symptom Severity
3.5. Characteristics Associated with Change in PTSD Symptom Severity
3.5.1. Participant Characteristics
3.5.2. COPE Treatment Characteristics
3.5.3. Events over Follow-Up
3.6. Independent Predictors of Change in PTSD Symptom Severity
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Dore, G.; Mills, K.; Murray, R.; Teesson, M.; Farrugia, P. Post-traumatic stress disorder, depression and suicidality in inpatients with substance use disorders. Drug Alcohol Rev. 2012, 31, 294–302. [Google Scholar] [CrossRef] [PubMed]
- Mills, K.L.; Lynskey, M.; Teesson, M.; Ross, J.; Darke, S. Post-traumatic stress disorder among people with heroin dependence in the Australian treatment outcome study (ATOS): Prevalence and correlates. Drug Alcohol Depend. 2005, 77, 243–249. [Google Scholar] [CrossRef] [PubMed]
- Kingston, R.; Marel, C.; Mills, K. A systematic review of the prevalence of comorbid mental health disorders in people presenting for substance use treatment in Australia. Drug Alcohol Rev. 2016. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, M.; Hinchliffe, K.; Asamoah, V.; Kouimtsidis, C. Trauma and post-traumatic stress disorder in a drug treatment community service. Psychiatrist 2011, 35, 256–260. [Google Scholar] [CrossRef]
- Driessen, M.; Schulte, S.; Luedecke, C.; Schaefer, I.; Sutmann, F.; Ohlmeier, M.; Kemper, U.; Koesters, G.; Chodzinski, C.; Schneider, U.; et al. Trauma and PTSD in patients with alcohol, drug, or dual dependence: A multi-center study. Alcohol Clin. Exp. Res. 2008, 32, 481–488. [Google Scholar] [CrossRef] [PubMed]
- Bonin, M.F.; Norton, G.R.; Asmundson, G.J.; Dicurzio, S.; Pidlubney, S. Drinking away the hurt: The nature and prevalence of PTSD in substance abuse patients attending a community-based treatment program. J. Behav. Ther. Exp. Psychiatry 2000, 31, 55–66. [Google Scholar] [CrossRef]
- Brown, P.J.; Stout, R.L.; Mueller, T. Substance use disorder and posttraumatic stress disorder comorbidity: Addiction and psychiatric treatment rates. Psychol. Addict. Behav. 1999, 13, 115–122. [Google Scholar] [CrossRef]
- Clark, H.W.; Masson, C.L.; Delucchi, K.L.; Hall, S.M.; Sees, K.L. Violent traumatic events and drug abuse severity. J. Subst. Abuse Treat. 2001, 20, 121–127. [Google Scholar] [CrossRef]
- Mills, K.L.; Teesson, M.; Ross, J.; Peters, L. Trauma, PTSD, and substance use disorders: Findings from the Australian National Survey of Mental Health and Well-Being. Am. J. Psychiatry 2006, 163, 652–658. [Google Scholar] [CrossRef] [PubMed]
- Najavits, L.M.; Gastfriend, D.R.; Barber, J.P.; Reif, S.; Muenz, L.R.; Blaine, J.; Frank, A.; Crits-Christoph, P.; Thase, M.; Weiss, R.D. Cocaine dependence with and without PTSD among subjects in the National Institute on Drug Abuse Collaborative Cocaine Treatment Study. Am. J. Psychiatry 1998, 155, 214–219. [Google Scholar] [PubMed]
- Ouimette, P.C.; Brown, P.J.; Najavits, L.M. Course and treatment of patients with both substance use and posttraumatic stress disorders. Addict. Behav. 1998, 23, 785–795. [Google Scholar] [CrossRef]
- Read, J.P.; Brown, P.J.; Kahler, C.W. Substance use and posttraumatic stress disorders: Symptom interplay and effects on outcome. Addict. Behav. 2004, 29, 1665–1672. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, M.; Mezey, G.; Chapman, M.; Wheeler, M.; Drummond, C.; Baldacchino, A. Co-morbid post-traumatic stress disorder in a substance misusing clinical population. Drug Alcohol. Depend. 2005, 77, 251–258. [Google Scholar] [CrossRef] [PubMed]
- Villagomez, R.E.; Meyer, T.J.; Lin, M.M.; Brown, L.S., Jr. Post-traumatic stress disorder among inner city methadone maintenance patients. J. Subst. Abuse Treat. 1995, 12, 253–257. [Google Scholar] [CrossRef]
- Wasserman, D.A.; Havassy, B.E.; Boles, S.M. Traumatic events and post-traumatic stress disorder in cocaine users entering private treatment. Drug Alcohol. Depend 1997, 46, 1–8. [Google Scholar] [CrossRef]
- Mills, K.L.; Teesson, M.; Ross, J.; Darke, S. The impact of post-traumatic stress disorder on treatment outcomes for heroin dependence. Addiction 2007, 102, 447–454. [Google Scholar] [CrossRef] [PubMed]
- Ouimette, P.C.; Finney, J.W.; Moos, R.H. Two-year posttreatment functioning and coping of substance abuse patients with posttraumatic stress disorder. Psychol. Addict. Behav. 1999, 13, 105–114. [Google Scholar] [CrossRef]
- Back, S.E.; Brady, K.T.; Jaanimagi, U.; Jackson, J.L. Cocaine dependence and PTSD: A pilot study of symptom interplay and treatment preferences. Addict. Behav. 2006, 31, 351–354. [Google Scholar] [CrossRef] [PubMed]
- Hien, D.A.; Jiang, H.; Campbell, A.N.; Hu, M.-C.; Miele, G.M.; Cohen, L.R.; Brigham, G.S.; Capstick, C.; Kulaga, A.; Robinson, J.; et al. Do treatment improvements in PTSD severity affect substance use outcomes? A secondary analysis from a randomized clinical trial in NIDA’s Clinical Trials Network. Am. J. Psychiatry 2010, 167, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Phoenix Australia–Centre for Posttraumatic Mental Health. Australian Guidelines for the Treatment of Acute Stress Disorder and Posttraumatic Stress Disorder; Phoenix Australia: Melbourne, Australia, 2013. [Google Scholar]
- Foa, E.B.; Keane, T.M.; Friedman, M.J.; Cohen, J.A. Effective treatments for PTSD: Practice Guidelines from the International Society for Traumatic Stress Studies; Guilford Press: New York, NY, USA, 2008. [Google Scholar]
- Back, S.E.; Killeen, T.K.; Teer, A.P.; Hartwell, E.E.; Federline, A.; Beylotte, F.; Cox, E. Substance use disorders and PTSD: An exploratory study of treatment preferences among military veterans. Addict. Behav. 2014, 39, 369–373. [Google Scholar] [CrossRef] [PubMed]
- Najavits, L.M.; Sullivan, T.P.; Schmitz, M.; Weiss, R.D.; Lee, C.S.N. Treatment utilization by women with PTSD and substance dependence. Am. J. Addict. 2004, 13, 215–224. [Google Scholar] [CrossRef] [PubMed]
- Roberts, N.P.; Roberts, P.A.; Jones, N.; Bisson, J.I. Psychological interventions for post-traumatic stress disorder and comorbid substance use disorder: A systematic review and meta-analysis. Clin. Psychol. Rev. 2015, 38, 25–38. [Google Scholar] [CrossRef] [PubMed]
- Najavits, L.M.; Hien, D. Helping vulnerable populations: A comprehensive review of the treatment outcome literature on substance use disorder and PTSD. J. Clin. Psychol. 2013, 69, 433–479. [Google Scholar] [CrossRef] [PubMed]
- Van Dam, D.; Vedel, E.; Ehring, T.; Emmelkamp, P.M. Psychological treatments for concurrent posttraumatic stress disorder and substance use disorder: A systematic review. Clin. Psychol. Rev. 2012, 32, 202–214. [Google Scholar] [CrossRef] [PubMed]
- Ballenger, J.C.; Davidson, J.R.; Lecrubier, Y.; Nutt, D.J.; Marshall, R.D.; Nemeroff, C.B.; Shalev, A.Y.; Yehuda, R. Consensus statement on posttraumatic stress disorder from the international consensus group on depression and anxiety. J. Clin. Psychiatry 2000, 61 (Suppl. 5), 60–66. [Google Scholar] [PubMed]
- Van Minnen, A.; Harned, M.S.; Zoellner, L.; Mills, K. Examining potential contraindications for prolonged exposure therapy for PTSD. Eur. J. Psychotraumatol. 2012, 3. [Google Scholar] [CrossRef] [PubMed]
- Becker, C.B.; Zayfert, C.; Anderson, E. A survey of psychologists’ attitudes towards and utilization of exposure therapy for PTSD. Behav. Res. Ther. 2004, 42, 277–292. [Google Scholar] [CrossRef]
- Killeen, T.K.; Back, S.E.; Brady, K.T. The use of exposure-based treatment among individuals with PTSD and co-occurring substance use disorders: Clinical considerations. J. Dual Diagn. 2011, 7, 194–206. [Google Scholar] [CrossRef] [PubMed]
- Ouimette, P.; Moos, R.H.; Brown, P.J.; Ouimette, P.; Brown, P.J. Substance use disorder-posttraumatic stress disorder comorbidity: A survey of treatments and proposed practice guidelines. In Trauma and Substance Abuse: Causes, Consequences, and Treatment of Comorbid Disorders; American Psychological Association: Washington, WA, USA, 2003; pp. 91–110. [Google Scholar]
- Brady, K.T.; Dansky, B.S.; Back, S.E.; Foa, E.B.; Carroll, K.M. Exposure therapy in the treatment of PTSD among cocaine-dependent individuals: Preliminary findings. J. Subst. Abuse Treat. 2001, 21, 47–54. [Google Scholar] [CrossRef]
- Mills, K.L.; Teesson, M.; Back, S.E.; Brady, K.T.; Baker, A.L.; Hopwood, S.; Sannibale, C.; Barrett, E.L.; Merz, S.; Rosenfeld, J.; et al. Integrated exposure-based therapy for co-occurring posttraumatic stress disorder and substance dependence: A randomized controlled trial. JAMA 2012, 308, 690–699. [Google Scholar] [CrossRef] [PubMed]
- Najavits, L.M.; Schmitz, M.; Gtthardt, S.; Weiss, R.D. Seeking safety plus exposure therapy: An outcome study on dual diagnosis men. J. Psychoact. Drugs 2005, 37, 425–435. [Google Scholar] [CrossRef] [PubMed]
- Triffleman, E. Gender differences in a controlled pilot study of psychosocial treatments in substance dependent patients with post-traumatic stress disorder: Design considerations and outcomes. Alcohol Treat. Q. 2000, 18, 113–126. [Google Scholar] [CrossRef]
- Sannibale, C.; Teesson, M.; Creamer, M.; Sitharthan, T.; Bryant, R.A.; Sutherland, K.; Taylor, K.; Bostock-Matusko, D.; Visser, A.; Peek-O’Leary, M. Randomized controlled trial of cognitive behaviour therapy for comorbid post-traumatic stress disorder and alcohol use disorders. Addiction 2013, 108, 1397–1410. [Google Scholar] [CrossRef]
- Hien, D.A.; Ruglass, L.M.; Lopez-Castro, T.; Papini, S.; Killeen, T.; Back, S.E. Concurrent treatment with prolonged exposure for co-occurring PTSD and substance use disorders: A randomized clinical trial. In Proceedings of the annual meeting of the College on Problems of Drug Dependence, Palm Springs, CA, USA, 15 June 2016.
- Back, S.E.; Foa, E.B.; Killeen, T.K.; Mills, K.L.; Teesson, M.; Cotton, B.D.; Carroll, K.M.; Brady, K.T. Concurrent Treatment of PTSD and Substance Use Disorders Using Prolonged Exposure (COPE): Therapist Guide; Oxford University Press: New York, NY, USA, 2014. [Google Scholar]
- Back, S.E.; Foa, E.B.; Killeen, T.K.; Mills, K.L.; Teesson, M.; Cotton, B.D.; Carroll, K.M.; Brady, K.T. Concurrent Treatment of PTSD and Substance Use Disorders Using Prolonged Exposure (COPE): Patient Workbook; Oxford University Press: New York, NY, USA, 2014. [Google Scholar]
- Blake, D.; Weathers, F.; Nagy, L.; Kaloupek, D.; Klauminzer, G.; Charney, D. A clinician rating scale for assessing current and lifetime PTSD: The CAPS-1. J. Trauma Stress. 1995, 8, 75–90. [Google Scholar] [CrossRef] [PubMed]
- Darke, S.; Hall, W.; Wodak, A.; Heather, N.; Ward, J. Development and validation of a multi-dimensional instrument for assessing outcome of treatment among opiate users: The Opiate Treatment Index. Br. J. Addict. 1992, 87, 733–742. [Google Scholar] [CrossRef] [PubMed]
- Kessler, R.C.; Ustun, T.B. The World Mental Health (WMH) Survey Initiative Version of the World Health Organization (WHO) Composite International Diagnostic Interview (CIDI). Int. J. Methods Psychiatr. Res. 2004, 13, 93–121. [Google Scholar] [CrossRef] [PubMed]
- World Health Organisation. Composite International Diagnostic Interview (CIDI) Core Version 2.1, 12 Month Version; World Health Organisation: Geneva, Switzerland, 1997. [Google Scholar]
- Beck, A.; Steer, R.; Brown, G. Manual for the Beck Depression Inventory-II; Psychological Corp: San Antonio, TX, USA, 1996. [Google Scholar]
- Spielberger, C. Manual for the State-Trait Anxiety Inventory; Consulting Psychologists Press: Palo Alto, CA, USA, 1983. [Google Scholar]
- Loranger, A.; Janca, A.; Sartorius, N. Assessment and Diagnosis of Personality Disorders; Cambridge University Press: Cambridge, UK, 1997. [Google Scholar]
- Back, S.E.; Dansky, B.S.; Carroll, K.M.; Foa, E.B.; Brady, K.T. Exposure therapy in the treatment of PTSD among cocaine-dependent individuals: Description of procedures. J. Subst. Abuse Treat. 2001, 21, 35–45. [Google Scholar] [CrossRef]
- Carroll, K. A Cognitive-Behavioral Approach: Treating Cocaine Addiction; US Dept of Health and Human Services, National Institute on Drug Abuse: Rockville, MD, USA, 1998.
- Foa, E.; Rothbaum, B. Treating the Trauma of Rape: Cognitive-Behavioral Therapy for PTSD; Guilford Press: New York, NY, USA, 1998. [Google Scholar]
- Baker, A.; Kay-Lambkin, F.; Lee, N.; Claire, M.; Jenner, L. A Brief Cognitive Behavioural Intervention for Regular Amphetamine Users; Australian Government Department of Health and Ageing: Canberra, Australia, 2003.
- Jacobson, N.S.; Truax, P. Clinical significance: A statistical approach to defining meaningful change in psychotherapy research. J. Consult. Clin. Psychol. 1991, 59, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Weathers, F.W.; Keane, T.M.; Davidson, J.R. Clinician-Administered PTSD Scale: A review of the first ten years of research. Depress Anxiety 2001, 13, 132–156. [Google Scholar] [CrossRef] [PubMed]
- Bradley, R.; Greene, J.; Russ, E.; Dutra, L.; Westen, D. A multidimensional meta-analysis of psychotherapy for PTSD. Am. J. Psychiatry 2005, 162, 214–227. [Google Scholar] [CrossRef] [PubMed]
- Schnurr, P.P.; Lunney, C.A. Symptom benchmarks of improved quality of life in PTSD. Depress Anxiety 2016, 33, 247–255. [Google Scholar] [CrossRef] [PubMed]
- Schnurr, P.P.; Lunney, C.A. Work-related outcomes among female veterans and service members after treatment of posttraumatic stress disorder. Psychiatr. Serv. 2012, 63, 1072–1079. [Google Scholar] [CrossRef] [PubMed]
- Mills, K.L.; Ewer, P.; Dore, G.; Teesson, M.; Baker, A.; Kay-Lambkin, F.; Sannibale, C. The feasibility and acceptability of a brief intervention for clients of substance use services experiencing symptoms of post traumatic stress disorder. Addict. Behav. 2014, 39, 1094–1099. [Google Scholar] [CrossRef] [PubMed]
- Foa, E.B.; Rothbaum, B.O.; Furr, J.M. Augmenting exposure therapy with other CBT procedures. Psychiatr. Ann. 2003, 33, 47–53. [Google Scholar] [CrossRef]
- McLean, C.P.; Foa, E.B. Prolonged exposure therapy for post-traumatic stress disorder: A review of evidence and dissemination. Exp. Rev. Neurotherap. 2011, 11, 1151–1163. [Google Scholar] [CrossRef] [PubMed]
- Van Minnen, A.; Hendriks, L.; Olff, M. When do trauma experts choose exposure therapy for PTSD patients? A controlled study of therapist and patient factors. Behav. Res. Ther. 2010, 48, 312–320. [Google Scholar] [CrossRef] [PubMed]
- Larsen, S.E.; Stirman, S.W.; Smith, B.N.; Resick, P.A. Symptom exacerbations in trauma-focused treatments: Associations with treatment outcome and non-completion. Behav. Res. Ther. 2016, 77, 68–77. [Google Scholar] [CrossRef] [PubMed]
- Hembree, E.A.; Foa, E.B.; Dorfan, N.M.; Street, G.P.; Kowalski, J.; Tu, X. Do patients drop out prematurely from exposure therapy for PTSD? J. Trauma Stress 2003, 16, 555–562. [Google Scholar] [CrossRef] [PubMed]
- Schottenbauer, M.A.; Glass, C.R.; Arnkoff, D.B.; Tendick, V.; Gray, S.H. Nonresponse and dropout rates in outcome studies on PTSD: Review and methodological considerations. Psychiatry 2008, 71, 134–168. [Google Scholar] [CrossRef] [PubMed]
- Gutner, C.A.; Gallagher, M.W.; Baker, A.S.; Sloan, D.M.; Resick, P.A. Time course of treatment dropout in cognitive–behavioral therapies for posttraumatic stress disorder. Psychol. Trauma Theory Res. Pract. Policy 2016, 8, 115. [Google Scholar] [CrossRef] [PubMed]
- Elliott, D.E.; Bjelajac, P.; Fallot, R.D.; Markoff, L.S.; Reed, B.G. Trauma-informed or trauma-denied: Principles and implementation of trauma-informed services for women. J. Commun. Psychol. 2005, 33, 461–477. [Google Scholar] [CrossRef]
- Vanderplasschen, W.; Wolf, J.; Rapp, R.C.; Broekaert, E. Effectiveness of different models of case management for substance-abusing populations. J. Psychoact. Drugs 2007, 39, 81–95. [Google Scholar] [CrossRef] [PubMed]
- Marel, C.; Mills, K.L.; Kingston, R.; Gournay, K.; Deady, M.; Kay-Lambkin, F.; Baker, A.; Teesson, M. Guidelines on the Management of Co-Occurring Alcohol and Other Drug and Mental Health Conditions in Alcohol and Other Drug Treatment Settings, 2nd ed.; National Drug and Alcohol Research Centre: Sydney, Australia, 2016. [Google Scholar]
- Cloitre, M.; Petkova, E.; Su, Z.; Weiss, B. Patient characteristics as a moderator of post-traumatic stress disorder treatment outcome: Combining symptom burden and strengths. Br. J. Psychiatry Open 2016, 2, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Ehlers, A.; Grey, N.; Wild, J.; Stott, R.; Liness, S.; Deale, A.; Handley, R.; Albert, I.; Cullen, D.; Hackmann, A. Implementation of Cognitive Therapy for PTSD in routine clinical care: Effectiveness and moderators of outcome in a consecutive sample. Behav. Res. Ther. 2013, 51, 742–752. [Google Scholar] [CrossRef] [PubMed]
- Teesson, M.; Slade, T.; Swift, W.; Mills, K.; Memedovic, S.; Mewton, L.; Grove, R.; Newton, N.; Hall, W. Prevalence, correlates and comorbidity of DSM-IV cannabis use and cannabis use disorders in Australia. ANZJP 2012, 46, 1182–1192. [Google Scholar] [CrossRef] [PubMed]
- AIHW. Alcohol and Other Drug Treatment Services in Australia 2014–2015; Drug Treatment Series No. 27. Cat. No. HSE 173; AIHW: Canberra, Australia, 2016.
- Bonn-Miller, M.O.; Babson, K.A.; Vandrey, R. Using cannabis to help you sleep: Heightened frequency of medical cannabis use among those with PTSD. Drug Alcohol Depend 2014, 136, 162–115. [Google Scholar] [CrossRef] [PubMed]
- Greer, G.R.; Grob, C.S.; Halberstadt, A.L. PTSD symptom reports of patients evaluated for the New Mexico Medical Cannabis Program. J. Psychoact. Drugs 2014, 46, 73–77. [Google Scholar] [CrossRef] [PubMed]
- Darke, S. Self-report among injecting drug users: A review. Drug Alcohol Depend. 1998, 51, 253–263. [Google Scholar] [CrossRef]
| Change in PTSD Symptom Severity | Clinically Significant Change in PTSD Symptom Severity | |||||
|---|---|---|---|---|---|---|
| β | SE | p | Improvement (n = 27) | No change (n = 28) | OR (95% CI) | |
| Demographic characteristics | ||||||
| Age | 0.21 | 0.52 | 0.691 | 33.64 (SE 1.51) | 33.93 (SE 1.41) | 1.00 (0.92–1.08) |
| Female sex | 9.17 | 8.49 | 0.282 | 63.6 | 56.3 | 1.82 (0.54–0.61) |
| Born in Australia | 6.44 | 12.02 | 0.593 | 90.0 | 80.7 | 3.53 (0.55–22.67) |
| Years of school completed | 6.06 | 2.68 | 0.024 | 10.59 (0.24) | 10.14 (0.31) | 1.36 (0.81–2.26) |
| Unemployed | 2.81 | 9.10 | 0.309 | 83.0 | 70.0 | 1.74 (0.44–6.86) |
| History of imprisonment | −15.82 | 10.32 | 0.137 | 18.5 | 42.9 | 0.37 (0.09–1.51) |
| Substance use | ||||||
| Age of first intoxication | 2.19 | 1.25 | 0.084 | 13.68 (0.77) | 12.36 (0.53) | 1.27 (0.98–1.65) |
| History of injecting drug use | 7.04 | 9.44 | 0.456 | 28.1 | 15.7 | 1.63 (0.34–7.76) |
| Number of drug classes used (last month | −0.18 | 2.88 | 0.950 | 3.67 (0.31) | 3.75 (0.31) | 1.07 (0.69–1.66) |
| Main drug of concern alcohol | 1.33 | 13.30 | 0.920 | 14.1 | 11.4 | 1.25 (0.16–9.53) |
| Trauma and PTSD | ||||||
| Age of first trauma exposure | 0.69 | 0.46 | 0.132 | 13.77 (1.82) | 9.82 (1.52) | 1.08 (0.99–1.17) |
| Age of worst trauma exposure | 0.10 | 0.10 | 0.302 | 21.6 (2.15) | 23.55 (2.37) | 0.98 (0.93–1.03) |
| Childhood trauma | −7.69 | 9.28 | 0.410 | 60.7 | 77.1 | 0.39 (0.11–1.42) |
| Types of trauma | ||||||
| Life threatening accident | −.312 | 9.30 | 0.973 | 65.2 | 69.3 | 0.82 (0.18–3.75) |
| Natural disaster | −10.04 | 9.32 | 0.283 | 22.2 | 32.1 | 0.66 (0.15–2.86) |
| Witness serious injury or death | −13.43 | 10.44 | 0.20 | 80.0 | 87.1 | 0.65 (0.13–3.2) |
| Sexual assault | −3.80 | 10.48 | 0.719 | 71.9 | 80.7 | 0.66 (0.13–3.21) |
| Physical assault | −13.43 | 16.82 | 0.424 | 96.3 | 92.9 | 2.05 (0.15–27.58) |
| Threatened | 6.81 | 13.6 | 0.617 | 92.6 | 89.3 | 1.15 (0.15–8.65) |
| Torture | −7.15 | 9.22 | 0.439 | 28.9 | 25.7 | .93 (0.25–3.49) |
| Other | 12.26 | 8.84 | 0.170 | 41.5 | 31.4 | 1.53 (0.33–7.13) |
| Great shock | −2.11 | 8.31 | 0.799 | 57.8 | 51.4 | 1.54 (0.40–5.91) |
| Number of trauma types experienced | −2.70 | 2.71 | 0.323 | 6.02 (0.32) | 6.23 (0.29) | 0.95 (0.66–1.37) |
| Number of traumas experienced | −0.324 | 0.159 | 0.042 | 16.99 (3.50) | 23.02 (5.43) | 0.99 (0.96–1.02) |
| Trauma exposure preceded age of first intoxication | −7.02 | 8.51 | 0.412 | 44.4 | 64.3 | 0.37 (0.09–1.50) |
| Duration of PTSD symptoms | −0.064 | 0.41 | 0.709 | 11.85 (1.98) | 8.57 (1.83) | 1.04 (1.98–1.11) |
| Severity of PTSD symptoms | 0.64 | 0.25 | 0.010 | 95.57 (3.01) | 86.89 (2.91) | 1.04 (1.00–1.08) |
| Mental health | ||||||
| Severity of depression | −0.19 | 0.48 | 0.687 | 36.69 (2.42) | 35.44 (2.07) | 0.98 (0.90–1.06) |
| State anxiety | 0.134 | 0.305 | 0.660 | 56.13 (2.99) | 53.33 (2.48) | 1.01 (0.96–1.06) |
| Trait anxiety | −0.025 | 0.495 | 0.960 | 61.76 (2.34) | 62.62 (1.67) | 0.97 (0.89–1.06) |
| Screened positive for BPD | −8.23 | 8.75 | 0.348 | 63.7 | 74.3 | 0.55 (0.12–2.43) |
| Treatment history | ||||||
| History of SUD treatment | 0.47 | 13.72 | 0.973 | 91.9 | 90.0 | 1.17 (0.13–10.28) |
| History of PTSD treatment | 2.67 | 10.16 | 0.794 | 27.4 | 34.3 | 0.62 (0.13–2.96) |
| Engaged in current SUD treatment | −4.48 | 10.82 | 0.680 | 84.4 | 75.7 | 1.39 (0.27–7.10) |
| Engaged in current PTSD treatment | 5.97 | 14.34 | 0.677 | 7.4 | 10.7 | 0.27 (0.03–2.37) |
| Current use of antidepressants | 1.51 | 9.21 | 0.870 | 57.0 | 52.1 | 1.29 (0.32–5.21) |
| Change in PTSD Symptom Severity | Clinically Significant Change in PTSD Symptom Severity | |||||
|---|---|---|---|---|---|---|
| β | SE | p | Improvement (n = 27) | No change (n = 28) | OR (95% CI) | |
| COPE treatment characteristics | ||||||
| Therapist | 4.13 | 7.89 | 0.601 | 50.4 | 51.4 | 0.99 (0.29–3.40) |
| Started COPE therapy | 22.76 | 11.04 | 0.044 | 94.8 | 69.3 | – |
| Number of sessions attended | 2.14 | 0.88 | 0.019 | 6.27 (0.91) | 5.53 (1.14) | 1.07 (0.92–1.25) |
| Received imaginal and/or in vivo exposure | 19.75 | 8.81 | 0.031 | 60.7 | 48.6 | 2.26 (0.46–11.03) |
| Events over follow-up | ||||||
| Proportion of time spent in SUD treatment | −4.26 | 11.08 | 0.701 | 0.51 | 0.57 | 0.90 (0.19–4.27) |
| Use of antidepressants | 8.56 | 10.19 | 0.401 | 63.7 | 49.3 | 1.22 (0.28–5.29) |
| Trauma exposure over follow-up | −13.50 | 8.90 | 0.132 | 50.4 | 55.0 | 0.60 (0.16–2.28) |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mills, K.L.; Barrett, E.L.; Merz, S.; Rosenfeld, J.; Ewer, P.L.; Sannibale, C.; Baker, A.L.; Hopwood, S.; Back, S.E.; Brady, K.T.; et al. Integrated Exposure-Based Therapy for Co-Occurring Post Traumatic Stress Disorder (PTSD) and Substance Dependence: Predictors of Change in PTSD Symptom Severity. J. Clin. Med. 2016, 5, 101. https://doi.org/10.3390/jcm5110101
Mills KL, Barrett EL, Merz S, Rosenfeld J, Ewer PL, Sannibale C, Baker AL, Hopwood S, Back SE, Brady KT, et al. Integrated Exposure-Based Therapy for Co-Occurring Post Traumatic Stress Disorder (PTSD) and Substance Dependence: Predictors of Change in PTSD Symptom Severity. Journal of Clinical Medicine. 2016; 5(11):101. https://doi.org/10.3390/jcm5110101
Chicago/Turabian StyleMills, Katherine L., Emma L. Barrett, Sabine Merz, Julia Rosenfeld, Philippa L. Ewer, Claudia Sannibale, Amanda L. Baker, Sally Hopwood, Sudie E. Back, Kathleen T. Brady, and et al. 2016. "Integrated Exposure-Based Therapy for Co-Occurring Post Traumatic Stress Disorder (PTSD) and Substance Dependence: Predictors of Change in PTSD Symptom Severity" Journal of Clinical Medicine 5, no. 11: 101. https://doi.org/10.3390/jcm5110101
APA StyleMills, K. L., Barrett, E. L., Merz, S., Rosenfeld, J., Ewer, P. L., Sannibale, C., Baker, A. L., Hopwood, S., Back, S. E., Brady, K. T., & Teesson, M. (2016). Integrated Exposure-Based Therapy for Co-Occurring Post Traumatic Stress Disorder (PTSD) and Substance Dependence: Predictors of Change in PTSD Symptom Severity. Journal of Clinical Medicine, 5(11), 101. https://doi.org/10.3390/jcm5110101






