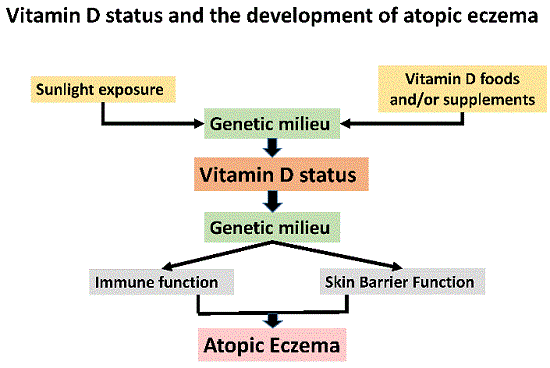
Vitamin D and the Development of Atopic Eczema
Abstract
:1. Introduction
2. Pathogenesis of Eczema
3. Vitamin D Sources and Status
4. Vitamin D Status in Utero and Eczema Development
| Reference and Study Location | Study Design | Study Population and Latitude | 25(OH)D Levels | Main Results | Higher Vitamin D Level |
|---|---|---|---|---|---|
| Gale et al. 2008 [38] Southampton, UK | Prospective birth cohort | 466 mother-child pairs Latitude 51°N | Maternal antenatal blood during third trimester of pregnancy median = 50 nmol/L (IQR 30–75) | Higher maternal 25(OH)D levels of >75 nmol/L (compared with lower levels <30 nmol/L) were associated with an increased risk of eczema at 9 months of age | Eczema c |
| Weisse et al. 2013 [45] Leipzig, Germany | Prospective birth cohort | 378 mother-child pairs Latitude 51°N | Maternal antenatal blood during third trimester of pregnancy median = 55 nmol/L (IQR 36–78) | No associations between maternal antenatal 25(OH)D levels with risk of eczema in the children to 2 years of age. | Eczema b |
| Wills et al. 2013 [46] South West of England | Prospective birth cohort | 5513 mother-child pairs Latitude 51°N | Maternal antenatal blood during first, second or third trimester of pregnancy median = 62 nmol/L (IQR 46–81) | No associations between maternal antenatal 25(OH)D levels with risk of eczema in the children at 7 years of age. | Eczema b |
| Baiz et al. 2014 [36] Poitiers and Nancy, France | Prospective birth cohort | 239 mother-child pairs. Latitude 46–48°N | Cord blood mean = 44 nmol/L (IQR = 38 nmol/L) | Inverse association between cord blood 25(OH)D levels with eczema in the children by age 1, 3, 5 years. | Eczema a |
| Chawes et al. 2014 [47] Copenhagen, Denmark | Prospective birth cohort | 257 mother-child pairs Latitude 55°N | Cord blood median = 48 nmol/L | No association between cord blood 25(OH)D levels with eczema in the children to 6–7 years old. | Eczema b |
| Jones et al. 2012 [37] Perth, Australia | Prospective birth cohort | 231 mother-child pairs Latitude 32°S | Cord blood mean (SD) = 58 nmol/L ± 24.1 nmol/L | Inverse association between cord blood 25(OH)D levels with risk for eczema in the children at 12 months of age. | Eczema a |
| Weisse et al. 2013 [45] Leipzig, Germany | Prospective birth cohort | 378 mother-child pairs Latitude 51°N | Cord blood median = 27 nmol/L (IQR 17–43) | No associations between cord blood 25(OH)D levels with risk of eczema in the children to 2 years of age. | Eczema b |
5. Vitamin D Status in Early Infancy and Eczema Development
6. Vitamin D as a Treatment Approach for Eczema Symptoms
7. Vitamin D Status and Allergen Sensitisation
8. Future Directions
8.1. Genotype Variations
8.2. Vitamin D or Sunlight?
9. Conclusions
Acknowledgements
Conflicts of Interest
References
- Holick, M.F. Vitamin D deficiency. N. Engl. J. Med. 2007, 357, 266–281. [Google Scholar] [CrossRef] [PubMed]
- Franco, J.M.; Gurgel, R.; Sole, D.; Lucia Franca, V.; Brabin, B.; Brazilian, I.G. Socio-environmental conditions and geographical variability of asthma prevalence in northeast Brazil. Allergol. Immunopathol. 2009, 37, 116–121. [Google Scholar] [CrossRef]
- Hoyos-Bachiloglu, R.; Morales, P.S.; Cerda, J.; Talesnik, E.; Gonzalez, G.; Camargo, C.A., Jr.; Borzutzky, A. Higher latitude and lower solar radiation influence on anaphylaxis in Chilean children. Pediatr. Allergy Immunol. 2014, 25, 338–343. [Google Scholar] [CrossRef] [PubMed]
- Krstic, G. Asthma prevalence associated with geographical latitude and regional insolation in the United States of America and Australia. PLoS ONE 2011, 6, e18492. [Google Scholar] [CrossRef] [PubMed]
- Osborne, N.J.; Ukoumunne, O.C.; Wake, M.; Allen, K.J. Prevalence of eczema and food allergy is associated with latitude in Australia. J. Allergy Clin. Immunol. 2012, 129, 865–867. [Google Scholar] [CrossRef] [PubMed]
- Weiland, S.K.; Husing, A.; Strachan, D.P.; Rzehak, P.; Pearce, N.; ISAAC Phase One Study Group. Climate and the prevalence of symptoms of asthma, allergic rhinitis, and atopic eczema in children. Occup. Environ. Med. 2004, 61, 609–615. [Google Scholar] [CrossRef] [PubMed]
- Camargo, C.A., Jr.; Clark, S.; Kaplan, M.S.; Lieberman, P.; Wood, R.A. Regional differences in epipen prescriptions in the United States: The potential role of vitamin D. J. Allergy Clin. Immunol. 2007, 120, 131–136. [Google Scholar] [CrossRef] [PubMed]
- Mullins, R.J.; Clark, S.; Camargo, C.A., Jr. Regional variation in epinephrine autoinjector prescriptions in Australia: More evidence for the vitamin D-anaphylaxis hypothesis. Ann. Allergy Asthma Immunol. 2009, 103, 488–495. [Google Scholar] [CrossRef] [PubMed]
- Silverberg, J.I.; Hanifin, J.; Simpson, E.L. Climatic factors are associated with childhood eczema prevalence in the United States. J. Investig. Dermatol. 2013, 133, 1752–1759. [Google Scholar] [CrossRef] [PubMed]
- Byremo, G.; Rod, G.; Carlsen, K.H. Effect of climatic change in children with atopic eczema. Allergy 2006, 61, 1403–1410. [Google Scholar] [CrossRef] [PubMed]
- Williams, H.; Stewart, A.; von Mutius, E.; Cookson, W.; Anderson, H. Is eczema really on the increase worldwide? J. Allergy Clin. Immunol. 2008, 121, 947–954. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, R.; Menon, C.; Hoffstad, O.; Bilker, W.; Leclerc, P.; Margolis, D.J. The prevalence of atopic triad in children with physician-confirmed atopic dermatitis. J. Am. Acad. Dermatol. 2008, 58, 68–73. [Google Scholar] [CrossRef] [PubMed]
- Spergel, J.M. From atopic dermatitis to asthma: The atopic march. Ann. Allergy Asthma Immunol. 2010, 105, 99–106. [Google Scholar] [CrossRef] [PubMed]
- Heimbeck, I.; Wjst, M.; Apfelbacher, C.J. Low vitamin D serum level is inversely associated with eczema in children and adolescents in Germany. Allergy 2013, 68, 906–910. [Google Scholar] [CrossRef] [PubMed]
- Garg, N.K.; Silverberg, J.I. Eczema is associated with osteoporosis and fractures in adults: A US population-based study. J. Allergy Clin. Immunol. 2015, 135, 1085–1087. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.S.; Hon, K.L.; Kong, A.P.; Pong, H.N.; Wong, G.W.; Leung, T.F. Vitamin D deficiency is associated with diagnosis and severity of childhood atopic dermatitis. Pediatr. Allergy Immunol. 2014, 25, 30–35. [Google Scholar] [CrossRef] [PubMed]
- Baek, J.H.; Shin, Y.H.; Chung, I.H.; Kim, H.J.; Yoo, E.G.; Yoon, J.W.; Jee, H.M.; Chang, Y.E.; Han, M.Y. The link between serum vitamin D level, sensitization to food allergens, and the severity of atopic dermatitis in infancy. J. Pediatr. 2014, 165, 849–854. [Google Scholar] [CrossRef] [PubMed]
- Peroni, D.G.; Piacentini, G.L.; Cametti, E.; Chinellato, I.; Boner, A.L. Correlation between serum 25-hydroxyvitamin D levels and severity of atopic dermatitis in children. Br. J. Dermatol. 2011, 164, 1078–1082. [Google Scholar] [CrossRef] [PubMed]
- Noh, S.; Park, C.O.; Bae, J.M.; Lee, J.; Shin, J.U.; Hong, C.S.; Lee, K.H. Lower vitamin D status is closely correlated with eczema of the head and neck. J. Allergy Clin. Immunol. 2014, 133, 1767–1770. [Google Scholar] [CrossRef] [PubMed]
- Chiu, Y.E.; Havens, P.L.; Siegel, D.H.; Ali, O.; Wang, T.; Holland, K.E.; Galbraith, S.S.; Lyon, V.B.; Drolet, B.A. Serum 25-hydroxyvitamin D concentration does not correlate with atopic dermatitis severity. J. Am. Acad. Dermatol. 2013, 69, 40–46. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.A.; Hong, S.; Kim, H.J.; Lee, S.H.; Yum, H.Y. Correlation between serum vitamin D level and the severity of atopic dermatitis associated with food sensitization. Allergy Asthma Immunol. Res 2013, 5, 207–210. [Google Scholar] [CrossRef] [PubMed]
- Akan, A.; Azkur, D.; Ginis, T.; Toyran, M.; Kaya, A.; Vezir, E.; Ozcan, C.; Ginis, Z.; Kocabas, C.N. Vitamin D level in children is correlated with severity of atopic dermatitis but only in patients with allergic sensitizations. Pediatr. Dermatol. 2013, 30, 359–363. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.S.; Hon, K.L.; Kong, A.P.; Tang, M.F.; Sy, H.Y.; Chan, J.C.; Leung, T.F. Eczema phenotypes are associated with multiple vitamin D pathway genes in Chinese children. Allergy 2014, 69, 118–124. [Google Scholar] [CrossRef] [PubMed]
- Sugarman, J.L. The epidermal barrier in atopic dermatitis. Semin. Cutan. Med. Surg. 2008, 27, 108–114. [Google Scholar] [CrossRef] [PubMed]
- Bikle, D.D. Vitamin D metabolism and function in the skin. Mol. Cell. Endocrinol. 2011, 347, 80–89. [Google Scholar] [CrossRef] [PubMed]
- Schauber, J.; Dorschner, R.A.; Yamasaki, K.; Brouha, B.; Gallo, R.L. Control of the innate epithelial antimicrobial response is cell-type specific and dependent on relevant microenvironmental stimuli. Immunology 2006, 118, 509–519. [Google Scholar] [PubMed]
- Biggs, L.; Yu, C.; Fedoric, B.; Lopez, A.F.; Galli, S.J.; Grimbaldeston, M.A. Evidence that vitamin D(3) promotes mast cell-dependent reduction of chronic UVB-induced skin pathology in mice. J. Exp. Med. 2010, 207, 455–463. [Google Scholar] [CrossRef] [PubMed]
- Mora, J.R.; Iwata, M.; von Andrian, U.H. Vitamin effects on the immune system: Vitamins A and D take centre stage. Nat. Rev. Immunol. 2008, 8, 685–698. [Google Scholar] [CrossRef] [PubMed]
- Mullins, R.J.; Camargo, C.A., Jr. Shining a light on vitamin D and its impact on the developing immune system. Clin. Exp. Allergy 2011, 41, 766–768. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.C.; Chimeh, F.; Lu, Z.; Mathieu, J.; Person, K.S.; Zhang, A.; Kohn, N.; Martinello, S.; Berkowitz, R.; Holick, M. Factors that influence the cutaneous synthesis and dietary sources of vitamin D. Arch. Biochem. Biophys. 2007, 460, 213–217. [Google Scholar] [CrossRef] [PubMed]
- Wagner, C.L.; Greer, F.R. American Academy of Pediatrics Committee on Nutrition: Prevention of rickets and vitamin D deficiency in infants, children, and adolescents. Pediatrics 2008, 122, 1142–1152. [Google Scholar] [CrossRef] [PubMed]
- Lai, J.K.; Lucas, R.M.; Clements, M.S.; Harrison, S.L.; Banks, E. Assessing vitamin D status: Pitfalls for the unwary. Mol. Nutr. Food Res. 2010, 54, 1062–1071. [Google Scholar] [PubMed]
- Paxton, G.A.; Teale, G.R.; Nowson, C.A.; Mason, R.S.; McGrath, J.J.; Thompson, M.J.; Siafarikas, A.; Rodda, C.P.; Munns, C.F.; et al. Vitamin D and health in pregnancy, infants, children and adolescents in Australia and New Zealand: A position statement. Med. J. Aust. 2013, 198, 142–143. [Google Scholar] [CrossRef] [PubMed]
- Vogiatzi, M.G.; Jacobson-Dickman, E.; DeBoer, M.D. Vitamin D supplementation and risk of toxicity in pediatrics: A review of current literature. J. Clin. Endocrinol. Metab. 2014, 99, 1132–1141. [Google Scholar] [CrossRef] [PubMed]
- Schlingmann, K.P.; Kaufmann, M.; Weber, S.; Irwin, A.; Goos, C.; John, U.; Misselwitz, J.; Klaus, G.; Kuwertz-Bröking, E.; Fehrenbach, H.; et al. Mutations in CYP24A1 and idiopathic infantile hypercalcemia. N. Engl. J. Med. 2011, 365, 410–421. [Google Scholar] [CrossRef] [PubMed]
- Baiz, N.; Dargent-Molina, P.; Wark, J.D.; Souberbielle, J.C.; Annesi-Maesano, I.; EDEN Mother-Child Cohort Study Group. Cord serum 25-hydroxyvitamin D and risk of early childhood transient wheezing and atopic dermatitis. J. Allergy Clin. Immunol. 2014, 133, 147–153. [Google Scholar] [CrossRef] [PubMed]
- Jones, A.P.; Palmer, D.; Zhang, G.; Prescott, S.L. Cord blood 25-hydroxyvitamin D3 and allergic disease during infancy. Pediatrics 2012, 130, e1128–e1135. [Google Scholar] [CrossRef] [PubMed]
- Gale, C.R.; Robinson, S.M.; Harvey, N.C.; Javaid, M.K.; Jiang, B.; Martyn, C.N.; Godfrey, K.M.; Cooper, C.; Princess Anne Hospital Study Group; et al. Maternal vitamin D status during pregnancy and child outcomes. Eur. J. Clin. Nutr. 2008, 62, 68–77. [Google Scholar] [CrossRef] [PubMed]
- Salle, B.L.; Delvin, E.E.; Lapillonne, A.; Bishop, N.J.; Glorieux, F.H. Perinatal metabolism of vitamin D. Am. J. Clin. Nutr. 2000, 71, 1317S–1324S. [Google Scholar] [PubMed]
- Miyake, Y.; Sasaki, S.; Tanaka, K.; Hirota, Y. Dairy food, calcium and vitamin D intake in pregnancy, and wheeze and eczema in infants. Eur. Respir. J. 2010, 35, 1228–1234. [Google Scholar] [CrossRef] [PubMed]
- Dunstan, J.A.; Mori, T.A.; Barden, A.; Beilin, L.J.; Taylor, A.L.; Holt, P.G.; Prescott, S.L. Fish oil supplementation in pregnancy modifies neonatal allergen-specific immune responses and clinical outcomes in infants at high risk of atopy: A randomized, controlled trial. J. Allergy Clin. Immunol. 2003, 112, 1178–1184. [Google Scholar] [CrossRef] [PubMed]
- Furuhjelm, C.; Warstedt, K.; Larsson, J.; Fredriksson, M.; Bottcher, M.F.; Falth-Magnusson, K.; Duchen, K. Fish oil supplementation in pregnancy and lactation may decrease the risk of infant allergy. Acta Paediatr. 2009, 98, 1461–1467. [Google Scholar] [CrossRef] [PubMed]
- Olsen, S.F.; Osterdal, M.L.; Salvig, J.D.; Mortensen, L.M.; Rytter, D.; Secher, N.J.; Henriksen, T.B. Fish oil intake compared with olive oil intake in late pregnancy and asthma in the offspring: 16 y of registry-based follow-up from a randomized controlled trial. Am. J. Clin. Nutr. 2008, 88, 167–175. [Google Scholar] [PubMed]
- Palmer, D.J.; Sullivan, T.; Gold, M.S.; Prescott, S.L.; Heddle, R.; Gibson, R.A.; Makrides, M. Effect of n−3 long chain polyunsaturated fatty acid supplementation in pregnancy on infants’ allergies in first year of life: Randomised controlled trial. BMJ 2012, 344. [Google Scholar] [CrossRef] [PubMed]
- Weisse, K.; Winkler, S.; Hirche, F.; Herberth, G.; Hinz, D.; Bauer, M.; Röder, S.; Rolle-Kampczyk, U.; von Bergen, M.; Olek, S.; et al. Maternal and newborn vitamin D status and its impact on food allergy development in the german lina cohort study. Allergy 2013, 68, 220–228. [Google Scholar] [CrossRef] [PubMed]
- Wills, A.K.; Shaheen, S.O.; Granell, R.; Henderson, A.J.; Fraser, W.D.; Lawlor, D.A. Maternal 25-hydroxyvitamin D and its association with childhood atopic outcomes and lung function. Clin. Exp. Allergy 2013, 43, 1180–1188. [Google Scholar] [PubMed]
- Chawes, B.L.; Bonnelykke, K.; Jensen, P.F.; Schoos, A.M.; Heickendorff, L.; Bisgaard, H. Cord blood 25(OH)-vitamin D deficiency and childhood asthma, allergy and eczema: The COPSAC 2000 birth cohort study. PLoS ONE 2014, 9, e99856. [Google Scholar] [CrossRef] [PubMed]
- Goldring, S.T.; Griffiths, C.J.; Martineau, A.R.; Robinson, S.; Yu, C.; Poulton, S.; Kirkby, J.C.; Stocks, J.; Hooper, R.; Shaheen, S.O.; et al. Prenatal vitamin D supplementation and child respiratory health: A randomised controlled trial. PLoS ONE 2013, 8, e66627. [Google Scholar] [CrossRef] [PubMed]
- Heaney, R.P.; Recker, R.R.; Grote, J.; Horst, R.L.; Armas, L.A. Vitamin D(3) is more potent than vitamin D(2) in humans. J. Clin. Endocrinol. Metab. 2011, 96, E447–E452. [Google Scholar] [CrossRef] [PubMed]
- Hollis, B.W.; Roos, B.A.; Draper, H.H.; Lambert, P.W. Vitamin D and its metabolites in human and bovine milk. J. Nutr. 1981, 111, 1240–1248. [Google Scholar] [PubMed]
- Greer, F.R.; Hollis, B.W.; Cripps, D.J.; Tsang, R.C. Effects of maternal ultraviolet B irradiation on vitamin D content of human milk. J. Pediatr. 1984, 105, 431–433. [Google Scholar] [CrossRef] [PubMed]
- Hollis, B.W.; Wagner, C.L. The role of the parent compound vitamin D with respect to metabolism and function: Why clinical dose intervals can affect clinical outcomes. J. Clin. Endocrinol. Metab. 2013, 98, 4619–4628. [Google Scholar] [CrossRef] [PubMed]
- Hollis, B.W.; Wagner, C.L. Vitamin D requirements during lactation: High-dose maternal supplementation as therapy to prevent hypovitaminosis d for both the mother and the nursing infant. Am. J. Clin. Nutr. 2004, 80, 1752S–1758S. [Google Scholar] [PubMed]
- Wagner, C.L.; Hulsey, T.C.; Fanning, D.; Ebeling, M.; Hollis, B.W. High-dose vitamin D3 supplementation in a cohort of breastfeeding mothers and their infants: A 6-month follow-up pilot study. Breastfeed Med. 2006, 1, 59–70. [Google Scholar] [CrossRef] [PubMed]
- Gallo, S.; Comeau, K.; Vanstone, C.; Agellon, S.; Sharma, A.; Jones, G.; L’Abbé, M.; Khamessan, A.; Rodd, C.; Weiler, H.; et al. Effect of different dosages of oral vitamin D supplementation on vitamin D status in healthy, breastfed infants: A randomized trial. JAMA 2013, 309, 1785–1792. [Google Scholar] [CrossRef] [PubMed]
- Ziegler, E.E.; Nelson, S.E.; Jeter, J.M. Vitamin D supplementation of breastfed infants: A randomized dose-response trial. Pediatr. Res. 2014, 76, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Braegger, C.; Campoy, C.; Colomb, V.; Decsi, T.; Domellof, M.; Fewtrell, M.; Hojsak, I.; Mihatsch, W.; Molgaard, C.; Shamir, R.; et al. Vitamin D in the healthy European paediatric population. J. Pediatr. Gastroenterol. Nutr. 2013, 56, 692–701. [Google Scholar] [CrossRef] [PubMed]
- Godel, J.C. Vitamin D supplementation: Recommendations for Canadian mothers and infants. Paediatr. Child Health 2007, 12, 583–598. [Google Scholar] [PubMed]
- Back, O.; Blomquist, H.K.; Hernell, O.; Stenberg, B. Does vitamin D intake during infancy promote the development of atopic allergy? Acta Derm. Venereol. 2009, 89, 28–32. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, N.J.; Franklin, V.; Gray, J.C.; Diffey, B.L.; Farr, P.M. Narrow-band ultraviolet B and broad-band ultraviolet A phototherapy in adult atopic eczema: A randomised controlled trial. Lancet 2001, 357, 2012–2016. [Google Scholar] [CrossRef] [PubMed]
- Norizoe, C.; Akiyama, N.; Segawa, T.; Tachimoto, H.; Mezawa, H.; Ida, H.; Urashima, M. Increased food allergy and vitamin D: Randomized, double-blind, placebo-controlled trial. Pediatr. Int. 2014, 56, 6–12. [Google Scholar] [CrossRef] [PubMed]
- Camargo, C.A., Jr.; Ganmaa, D.; Sidbury, R.; Erdenedelger, K.; Radnaakhand, N.; Khandsuren, B. Randomized trial of vitamin D supplementation for winter-related atopic dermatitis in children. J. Allergy Clin. Immunol. 2014, 134, 831–835. [Google Scholar] [CrossRef] [PubMed]
- Sidbury, R.; Sullivan, A.F.; Thadhani, R.I.; Camargo, C.A., Jr. Randomized controlled trial of vitamin D supplementation for winter-related atopic dermatitis in boston: A pilot study. Br. J. Dermatol. 2008, 159, 245–247. [Google Scholar] [CrossRef] [PubMed]
- Amestejani, M.; Salehi, B.S.; Vasigh, M.; Sobhkhiz, A.; Karami, M.; Alinia, H.; Kamrava, S.K.; Shamspour, N.; Ghalehbaghi, B.; Behzadi, A.H.; et al. Vitamin D supplementation in the treatment of atopic dermatitis: A clinical trial study. J. Drugs Dermatol. 2012, 11, 327–330. [Google Scholar] [PubMed]
- Javanbakht, M.H.; Keshavarz, S.A.; Djalali, M.; Siassi, F.; Eshraghian, M.R.; Firooz, A.; Seirafi, H.; Ehsani, A.H.; Chamari, M.; Mirshafiey, A.; et al. Randomized controlled trial using vitamins E and D supplementation in atopic dermatitis. J. Dermatol. Treat. 2011, 22, 144–150. [Google Scholar] [CrossRef]
- Hata, T.R.; Audish, D.; Kotol, P.; Coda, A.; Kabigting, F.; Miller, J.; Alexandrescu, D.; Boguniewicz, M.; Taylor, P.; Aertker, L.; et al. A randomized controlled double-blind investigation of the effects of vitamin D dietary supplementation in subjects with atopic dermatitis. J. Eur. Acad. Dermatol. Venereol. 2014, 28, 781–789. [Google Scholar] [CrossRef] [PubMed]
- Turner, M.J.; DaSilva-Arnold, S.C.; Yi, Q.; Mehrotra, P.; Kaplan, M.H.; Travers, J.B. Topical application of a vitamin D analogue exacerbates atopic dermatitis and induces the atopic dermatitis-like phenotype in Stat6VT mice. Pediatr. Dermatol. 2013, 30, 574–578. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Hener, P.; Zhang, Z.; Kato, S.; Metzger, D.; Chambon, P. Topical vitamin D3 and low-calcemic analogs induce thymic stromal lymphopoietin in mouse keratinocytes and trigger an atopic dermatitis. Proc. Natl. Acad. Sci. U.S.A. 2006, 103, 11736–11741. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Hener, P.; Zhang, Z.; Ganti, K.P.; Metzger, D.; Chambon, P. Induction of thymic stromal lymphopoietin expression in keratinocytes is necessary for generating an atopic dermatitis upon application of the active vitamin D3 analogue MC903 on mouse skin. J. Investig. Dermatol. 2009, 129, 498–502. [Google Scholar] [CrossRef] [PubMed]
- Nwaru, B.I.; Ahonen, S.; Kaila, M.; Erkkola, M.; Haapala, A.M.; Kronberg-Kippila, C.; Veijola, R.; Ilonen, J.; Simell, O.; Knip, M.; et al. Maternal diet during pregnancy and allergic sensitization in the offspring by 5 yrs of age: A prospective cohort study. Pediatr. Allergy Immunol. 2010, 21, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Rothers, J.; Wright, A.L.; Stern, D.A.; Halonen, M.; Camargo, C.A., Jr. Cord blood 25-hydroxyvitamin D levels are associated with aeroallergen sensitization in children from Tucson, Arizona. J. Allergy Clin. Immunol. 2011, 128, 1093–1099. [Google Scholar] [CrossRef] [PubMed]
- Hypponen, E.; Berry, D.J.; Wjst, M.; Power, C. Serum 25-hydroxyvitamin D and IgE—A significant but nonlinear relationship. Allergy 2009, 64, 613–620. [Google Scholar] [CrossRef] [PubMed]
- Matheu, V.; Back, O.; Mondoc, E.; Issazadeh-Navikas, S. Dual effects of vitamin D-induced alteration of TH1/TH2 cytokine expression: Enhancing IgE production and decreasing airway eosinophilia in murine allergic airway disease. J. Allergy Clin. Immunol. 2003, 112, 585–592. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Wang, G.; Hong, X.; Wang, D.; Tsai, H.J.; Zhang, S.; Arguelles, L.; Kumar, R.; Wang, H.; Liu, R.; et al. Gene-vitamin D interactions on food sensitization: A prospective birth cohort study. Allergy 2011, 66, 1442–1448. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Arguelles, L.; Zhou, Y.; Wang, G.; Chen, Q.; Tsai, H.J.; Hong, X.; Liu, R.; Price, H.E.; Pearson, C.; et al. Longitudinal trajectory of vitamin D status from birth to early childhood in the development of food sensitization. Pediatr. Res. 2013, 74, 321–326. [Google Scholar] [CrossRef] [PubMed]
- Heine, G.; Hoefer, N.; Franke, A.; Nothling, U.; Schumann, R.R.; Hamann, L.; Worm, M. Association of vitamin D receptor gene polymorphisms with severe atopic dermatitis in adults. Br. J. Dermatol. 2013, 168, 855–858. [Google Scholar] [CrossRef] [PubMed]
- Weller, R. Nitric oxide—A newly discovered chemical transmitter in human skin. Br. J. Dermatol. 1997, 137, 665–672. [Google Scholar] [CrossRef] [PubMed]
- Hart, P.H.; Gorman, S. Exposure to UV wavelengths in sunlight suppresses immunity. To what extent is UV-induced vitamin D3 the mediator responsible? Clin. Biochem. Rev. 2013, 34, 3–13. [Google Scholar] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Palmer, D.J. Vitamin D and the Development of Atopic Eczema. J. Clin. Med. 2015, 4, 1036-1050. https://doi.org/10.3390/jcm4051036
Palmer DJ. Vitamin D and the Development of Atopic Eczema. Journal of Clinical Medicine. 2015; 4(5):1036-1050. https://doi.org/10.3390/jcm4051036
Chicago/Turabian StylePalmer, Debra J. 2015. "Vitamin D and the Development of Atopic Eczema" Journal of Clinical Medicine 4, no. 5: 1036-1050. https://doi.org/10.3390/jcm4051036
APA StylePalmer, D. J. (2015). Vitamin D and the Development of Atopic Eczema. Journal of Clinical Medicine, 4(5), 1036-1050. https://doi.org/10.3390/jcm4051036