Alterations in Skin Temperature and Sleep in the Fear of Harm Phenotype of Pediatric Bipolar Disorder
Abstract
:1. Introduction
2. Experimental Section
2.1. Subjects
2.2. Protocol
2.2.1. Diary
2.2.2. Nocturnal Skin Temperature
2.2.3. Actigraphy
2.2.4. Data Analysis
2.2.5. DPG/DPG0
3. Results and Discussion
3.1. Sleep
| Sleep Parameter (Source) | Control | FOH |
|---|---|---|
| Bedtime (d) | 21:04 ± 0:40 | 21:10 ± 1:01 |
| Risetime (d) | 06:32 ± 0:50 | 07:23 ± 1:04 |
| Sleep Onset Latency (d) | 9 ± 5 min | 27 ± 20 min |
| Sleep Onset Latency (a) | 8 ± 4 min | 37 ± 38 min |
| Total Sleep Time (a) | 7 h 36 min ± 41 min | 8 h 07 min ± 1 h 17 min |
| Sleep Efficiency * (a) | 85.7% ± 7.1% | 87.0% ± 6.2% |
| Sleep Period Duration (d) | 9 h 13 min ± 44 min | 10 h 04 min ± 1 h 28 min |
| Parasomnias reported | 0 | 8 |
3.2. Parasomnias
3.3. Skin Temperature and Sleep
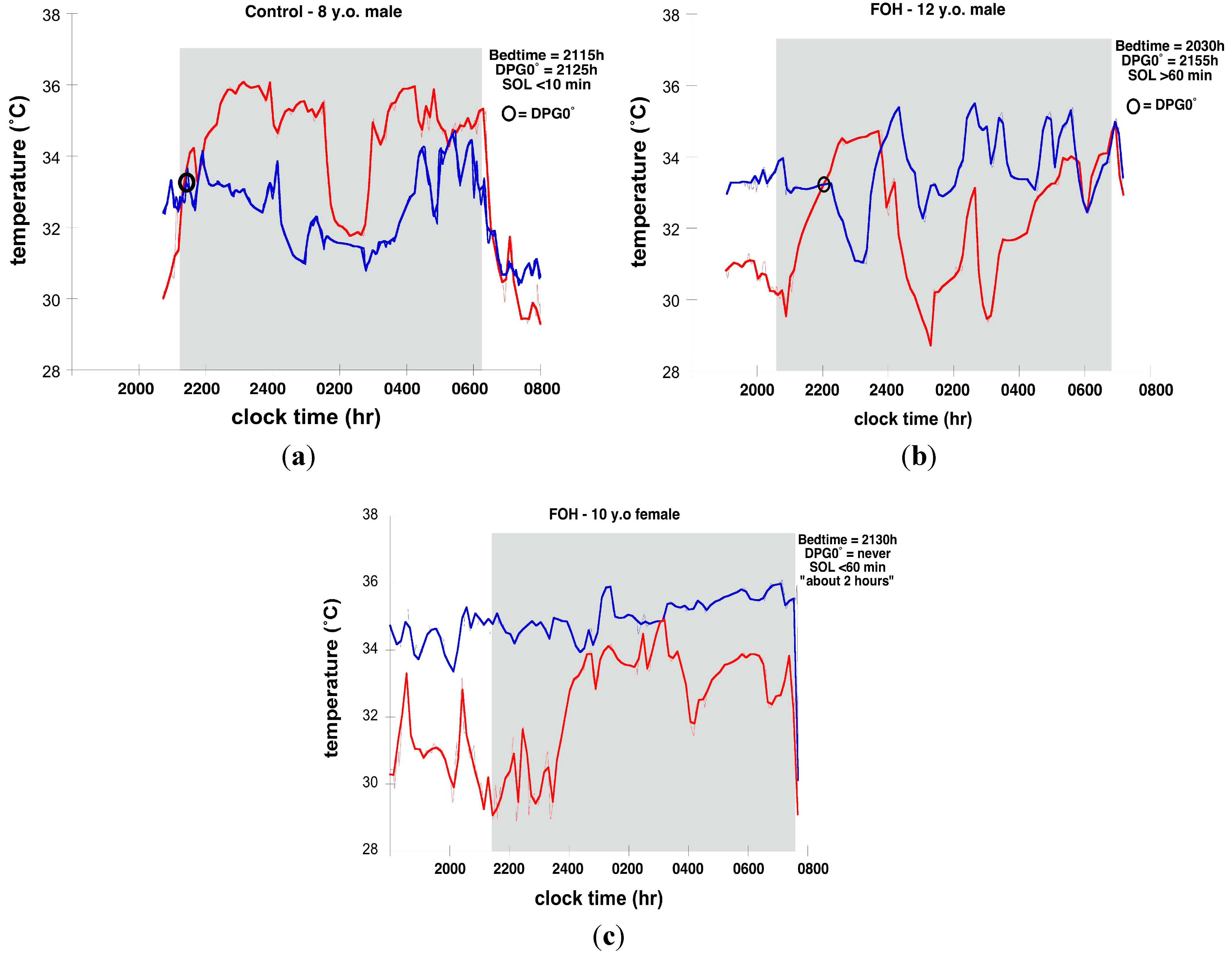
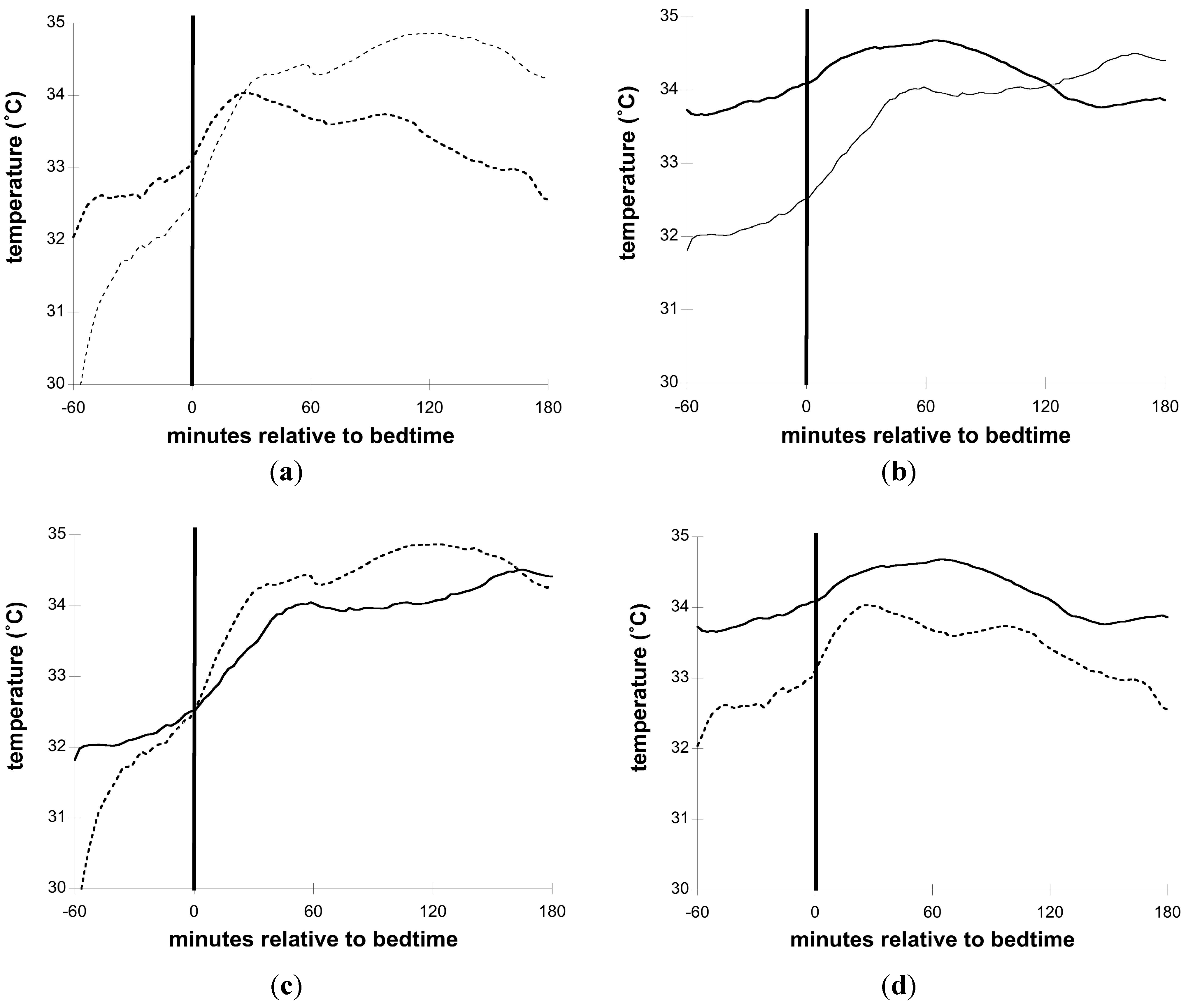
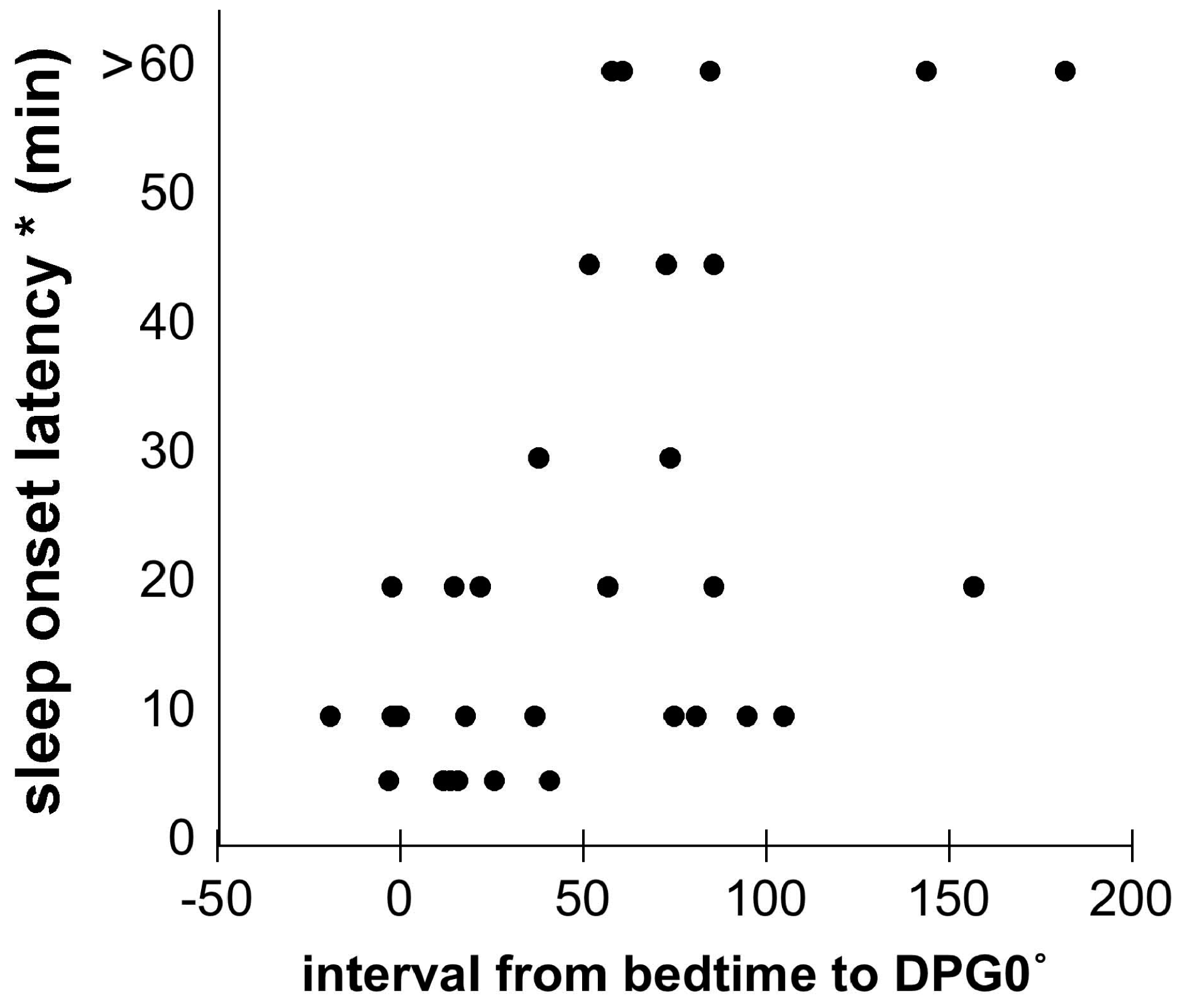
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Baroni, A.; Hernandez, M.; Grant, M.C.; Faedda, G.L. Sleep Disturbances in Pediatric Bipolar Disorder: A Comparison between Bipolar I and Bipolar NOS. Front. Psychiatry 2012, 3. [Google Scholar] [CrossRef]
- Berry, E.; Heaton, P.C.; Kelton, C.M. PMH29 National Estimates of the Inpatient Burden of Pediatric Bipolar Disorder. Value Health 2010, 14, 115–123. [Google Scholar]
- Chang, K.D. Course and Impact of Bipolar Disorder in Young Patients. J. Clin. Psychiatry 2010, 71. [Google Scholar] [CrossRef]
- Dilsaver, S.C.; Akiskal, H.S. “Mixed Hypomania” in Children and Adolescents: Is It a Pediatric Bipolar Phenotype with Extreme Diurnal Variation between Depression and Hypomania? J. Affect. Disord. 2009, 116, 12–17. [Google Scholar] [CrossRef]
- Faedda, G.L.; Baldessarini, R.J.; Glovinsky, I.P.; Austin, N.B. Pediatric Bipolar Disorder: Phenomenology and Course of Illness. Bipolar Disord. 2004, 6, 305–313. [Google Scholar] [CrossRef] [PubMed]
- Harvey, A.G.; Mullin, B.C.; Hinshaw, S.P. Sleep and Circadian Rhythms in Children and Adolescents with Bipolar Disorder. Dev. Psychopathol. 2006, 18, 1147–1168. [Google Scholar] [CrossRef] [PubMed]
- Harvey, A.G. The Adverse Consequences of Sleep Disturbance in Pediatric Bipolar Disorder: Implications for Intervention. Child Adolesc. Psychiatr. Clin. N. Am. 2009, 18, 321–338. [Google Scholar] [CrossRef] [PubMed]
- Ivanenko, A.; Johnson, K. Sleep Disturbances in Children with Psychiatric Disorders. Semin. Pediatr. Neurol. 2008, 15, 70–78. [Google Scholar] [CrossRef] [PubMed]
- Lofthouse, N.; Gilchrist, R.; Splaingard, M. Mood-Related Sleep Problems in Children and Adolescents. Child Adolesc. Psychiatr. Clin. N. Am. 2009, 18, 893–916. [Google Scholar] [CrossRef] [PubMed]
- Luby, J.L.; Navsaria, N. Pediatric Bipolar Disorder: Evidence for Prodromal States and Early Markers. J. Child Psychol. Psychiatry Allied Discip. 2010, 51, 459–471. [Google Scholar] [CrossRef]
- Mehl, R.C.; O’Brien, L.M.; Jones, J.H.; Dreisbach, J.K.; Mervis, C.B.; Gozal, D. Correlates of Sleep and Pediatric Bipolar Disorder. Sleep 2006, 29, 193–197. [Google Scholar] [PubMed]
- Staton, D. The Impairment of Pediatric Bipolar Sleep: Hypotheses Regarding a Core Defect and Phenotype-Specific Sleep Disturbances. J. Affect. Disord. 2008, 108, 199–206. [Google Scholar] [CrossRef] [PubMed]
- Staton, D.; Hill, C.M.; Chen, W. ADHD, Sleep Onset, and Pediatric Bipolar Disorder. J. Sleep Res. 2008, 71. [Google Scholar] [CrossRef]
- Youngstrom, E.A.; Birmaher, B.; Findling, R.L. Pediatric Bipolar Disorder: Validity, Phenomenology, and Recommendations for Diagnosis. Bipolar Disord. 2008, 10, 194–214. [Google Scholar] [CrossRef] [PubMed]
- Dienstbier, R.A.; Laguardia, R.L.; Wilcox, N.S. The Relationship of Temperament to Tolerance of Cold and Heat: Beyond “Cold Hands-Warm Heart”. Motiv. Emot. 1987, 11, 269–294. [Google Scholar] [CrossRef]
- Papolos, D.F.; Bronsteen, A. Bipolar disorder in children: Assessment in general pediatric practice. Curr. Opin. Pediatr. 2013, 25, 419–426. [Google Scholar] [PubMed]
- Geller, B.; Luby, J. Child and adolescent bipolar disorder: A review of the past 10 years. J. Am. Acad. Child Adolesc. Psychiatry 1997, 36, 1168–1176. [Google Scholar] [CrossRef] [PubMed]
- Birmaher, B. Bipolar disorder in children and adolescents. Child Adolesc. Ment. Health 2013, 18, 140–148. [Google Scholar] [CrossRef]
- Moreno, C.; Laje, G.; Blanco, C.; Jiang, H.; Schmidt, A.B.; Olfson, M. National Trends in the Outpatient Diagnosis and Treatment of Bipolar Disorder in Youth. Arch. Gen. Psychiatry 2007, 64, 1032–1039. [Google Scholar] [CrossRef] [PubMed]
- Papolos, D. Bipolar Disorder and Comorbid Disorders: The Case for a Dimensional Nosology. In Bipolar Disorder in Childhood and Early Adolescence; Geller, B., DelBello, M.P., Eds.; Guilford Press: New York, NY, USA, 2003; pp. 76–106. [Google Scholar]
- Papolos, D.; Hennen, J.; Cockerham, M.S.; Lachman, H. A Strategy for Identifying Phenotypic Subtypes: Concordance of Symptom Dimensions Between Sibling Pairs Who Met Screening Criteria for a Genetic Linkage Study of Childhood-Onset Bipolar Disorder Using the Child Bipolar Questionnaire. J. Affect. Disord. 2007, 99, 27–36. [Google Scholar] [CrossRef] [PubMed]
- Papolos, D.; Mattis, S.; Golshan, S.; Molay, F. Fear of Harm, a Possible Phenotype of Pediatric Bipolar Disorder: A Dimensional Approach to Diagnosis for Genotyping Psychiatric Syndromes. J. Affect. Disord. 2009, 118, 28–38. [Google Scholar] [CrossRef] [PubMed]
- Papolos, D.; Hennen, J.; Cockerham, M.S.; Thode, H.C.; Youngstrom, E.A. The Child Bipolar Questionnaire: A Dimensional Approach to Screening for Pediatric Bipolar Disorder. J. Affect. Disord. 2006, 95, 149–158. [Google Scholar] [CrossRef] [PubMed]
- Kaufman, J.; Birmaher, B.; Brent, D.; Rao, U.; Flynn, C.; Moreci, P.; Williamson, D.; Ryan, N. Schedule for Affective Disorders and Schizophrenia for School-Age Children-Present and Lifetime Version (K-SADS-PL): Initial reliability and validity data. J. Am. Acad. Child Adolesc. Psychiatry 1997, 36, 980–988. [Google Scholar] [CrossRef] [PubMed]
- Kräuchi, K.; Cajochen, C.; Werth, E.; Wirz-Justice, A. Functional Link between Distal Vasodilation and Sleep-Onset Latency? Am. J. Physiol. Regul. Integr. Comp. Physiol. 2000, 278, R741–R748. [Google Scholar]
- Kräuchi, K.; Cajochen, C.; Werth, E.; Wirz-Justice, A. Warm Feet Promote the Rapid Onset of Sleep. Nature 1999, 401, 36–37. [Google Scholar]
- Krauchi, K.; Deboer, T. The Interrelationship between Sleep Regulation and Thermoregulation. Front. Biosci. 2010, 15, 604–625. [Google Scholar] [CrossRef]
- Murphy, P.J.; Campbell, S.S. Nighttime Drop in Body Temperature: A Physiological Trigger for Sleep Onset? Sleep 1997, 20, 505–511. [Google Scholar] [PubMed]
- Raymann, R.J.E.M.; Swaab, D.F.; van Someren, E.J.W. Skin Deep: Enhanced Sleep Depth by Cutaneous Temperature Manipulation. Brain 2008, 131, 500–513. [Google Scholar] [CrossRef] [PubMed]
- Raymann, R.J.E.M.; Swaab, D.F.; van Someren, E.J.W. Skin Temperature and Sleep-Onset Latency: Changes with Age and Insomnia. Physiol. Behav. 2007, 90, 257–266. [Google Scholar] [CrossRef] [PubMed]
- Van Someren, E.J.W. More than a Marker: Interaction between the Circadian Regulation of Temperature and Sleep, Age-Related Changes, and Treatment Possibilities. Chronobiol. Int. 2000, 17, 313–354. [Google Scholar]
- Van Someren, E.J.W.; Raymann, R.J.E.M.; Scherder, E.J.A.; Daanen, H.A.M.; Swaab, D.F. Circadian and Age-Related Modulation of Thermoreception and Temperature Regulation: Mechanisms and Functional Implications. Ageing Res. Rev. 2002, 17, 313–354. [Google Scholar]
- Kräuchi, K.; Gasio, P.F.; Vollenweider, S.; von Arb, M.; Dubler, B.; Orgül, S.; Flammer, J.; Stutz, E.Z. Cold extremities and difficulties initiating sleep: Evidence of co-morbidity from a random sample of a Swiss urban population. J. Sleep Res. 2008, 17, 420–426. [Google Scholar]
- Gilbert, S.S.; van den Heuvel, C.J.; Kennaway, D.J.; Dawson, D. Peripheral Heat Loss: A Predictor of the Hypothermic Response to Melatonin Administration in Young and Older Women. Physiol. Behav. 1999, 66, 365–370. [Google Scholar] [CrossRef] [PubMed]
- Avery, D.H.; Shah, S.H.; Eder, D.N.; Wildschiødtz, G. Nocturnal Sweating and Temperature in Depression. Acta Psychiatr. Scand. 1999, 100, 295–301. [Google Scholar] [CrossRef] [PubMed]
- Avery, D.H.; Wildshiodtz, G.; Rafaelsen, O.J. Nocturnal Temperature in Affective Disorder. J. Affect. Disord. 1982, 4, 61–71. [Google Scholar] [CrossRef] [PubMed]
- Avery, D.H.; Dahl, K.; Savage, M.V.; Brengelmann, G.L.; Larsen, L.H.; Kenny, M.A.; Eder, D.N.; Vitiello, M.V.; Prinz, P.N. Circadian Temperature and Cortisol Rhythms during a Constant Routine Are Phase-Delayed in Hypersomnic Winter Depression. Biol. Psychiatry 1997, 41, 1109–1123. [Google Scholar] [CrossRef] [PubMed]
- Lack, L.C.; Gradisar, M.; van Someren, E.J.W.; Wright, H.R.; Lushington, K. The Relationship between Insomnia and Body Temperatures. Sleep Med. Rev. 2008, 12, 307–317. [Google Scholar] [CrossRef] [PubMed]
- Lushington, K.; Dawson, D.; Lack, L. Core body temperature is elevated during constant wakefulness in elderly poor sleepers. Sleep 2000, 23, 504–510. [Google Scholar] [PubMed]
- Van Den Heuvel, C.J.; Ferguson, S.A.; Gilbert, S.S.; Dawson, D. Thermoregulation in Normal Sleep and Insomnia: The Role of Peripheral Heat Loss and New Applications for Digital Thermal Infrared Imaging (DITI). J. Therm. Biol. 2004, 29, 457–461. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Murphy, P.J.; Frei, M.G.; Papolos, D. Alterations in Skin Temperature and Sleep in the Fear of Harm Phenotype of Pediatric Bipolar Disorder. J. Clin. Med. 2014, 3, 959-971. https://doi.org/10.3390/jcm3030959
Murphy PJ, Frei MG, Papolos D. Alterations in Skin Temperature and Sleep in the Fear of Harm Phenotype of Pediatric Bipolar Disorder. Journal of Clinical Medicine. 2014; 3(3):959-971. https://doi.org/10.3390/jcm3030959
Chicago/Turabian StyleMurphy, Patricia J., Mark G. Frei, and Demitri Papolos. 2014. "Alterations in Skin Temperature and Sleep in the Fear of Harm Phenotype of Pediatric Bipolar Disorder" Journal of Clinical Medicine 3, no. 3: 959-971. https://doi.org/10.3390/jcm3030959
APA StyleMurphy, P. J., Frei, M. G., & Papolos, D. (2014). Alterations in Skin Temperature and Sleep in the Fear of Harm Phenotype of Pediatric Bipolar Disorder. Journal of Clinical Medicine, 3(3), 959-971. https://doi.org/10.3390/jcm3030959




