Dynamics of Glycemic Status and Glucose Metabolism Markers 12 Months After Coronary Artery Bypass Grafting and Their Relationship with the Annual Prognosis of Patients
Abstract
1. Introduction
2. Methods
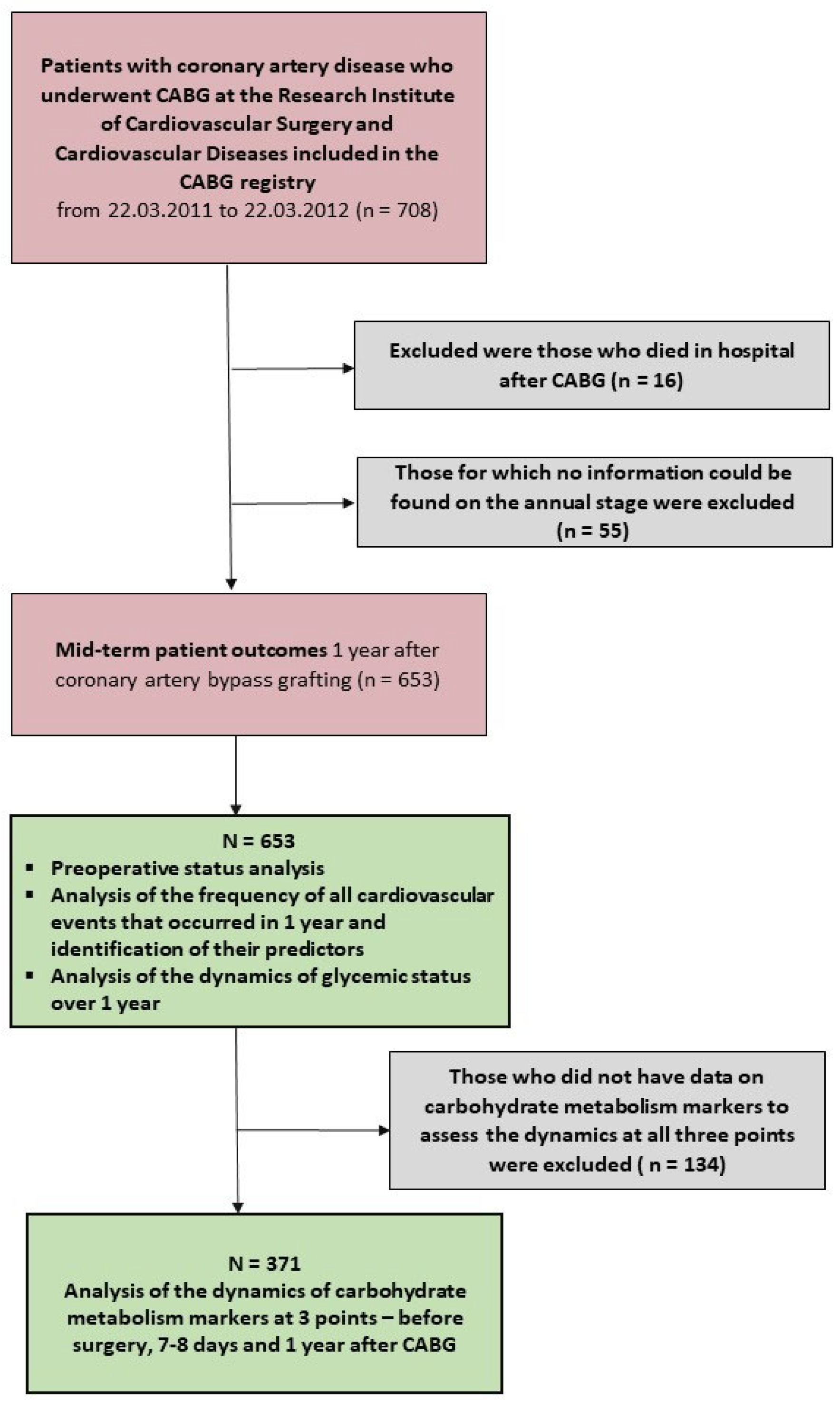
2.1. Study Population
2.2. Data Collection
2.3. Follow-Up Measures
2.4. Statistical Analyses
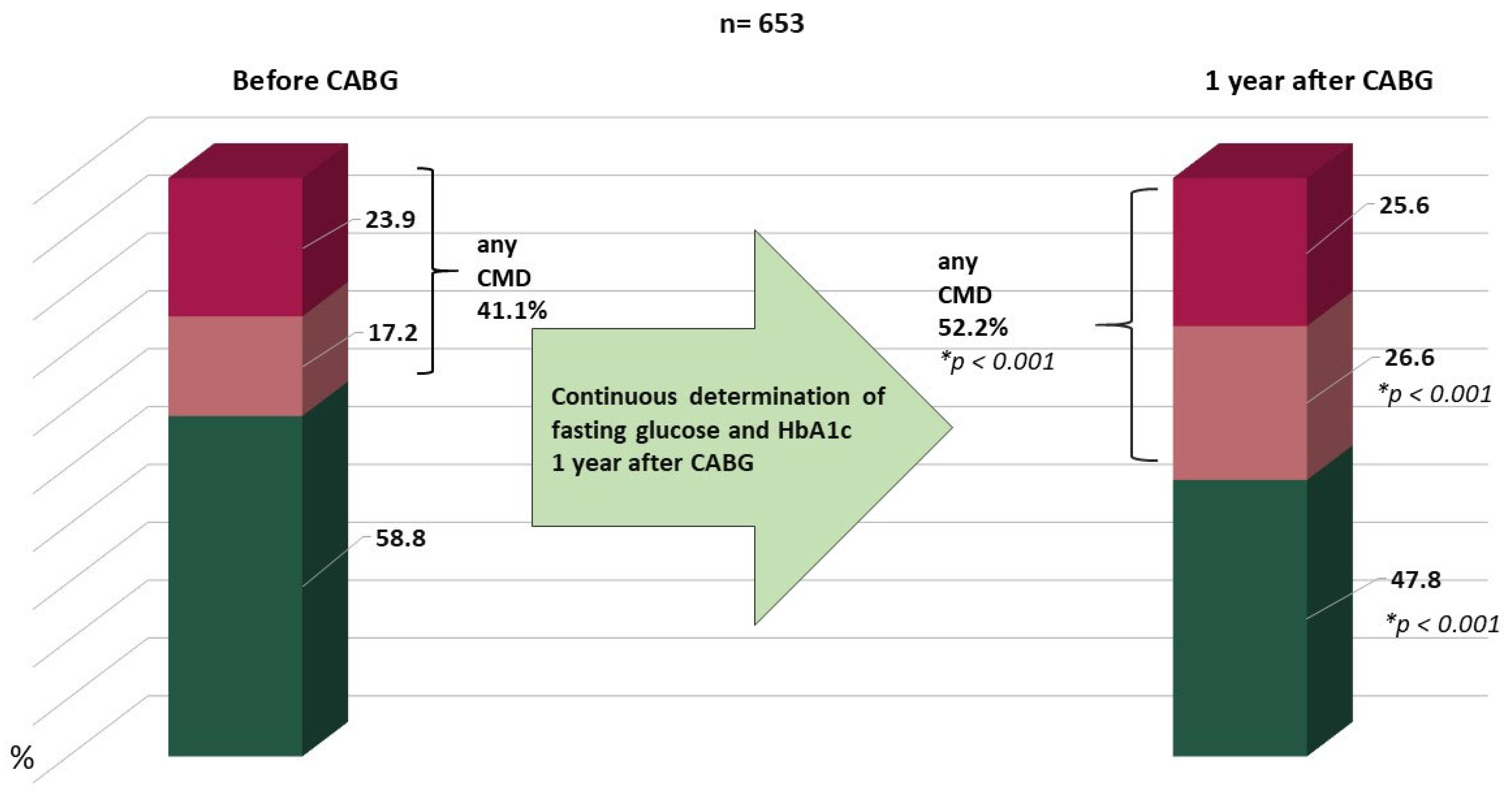
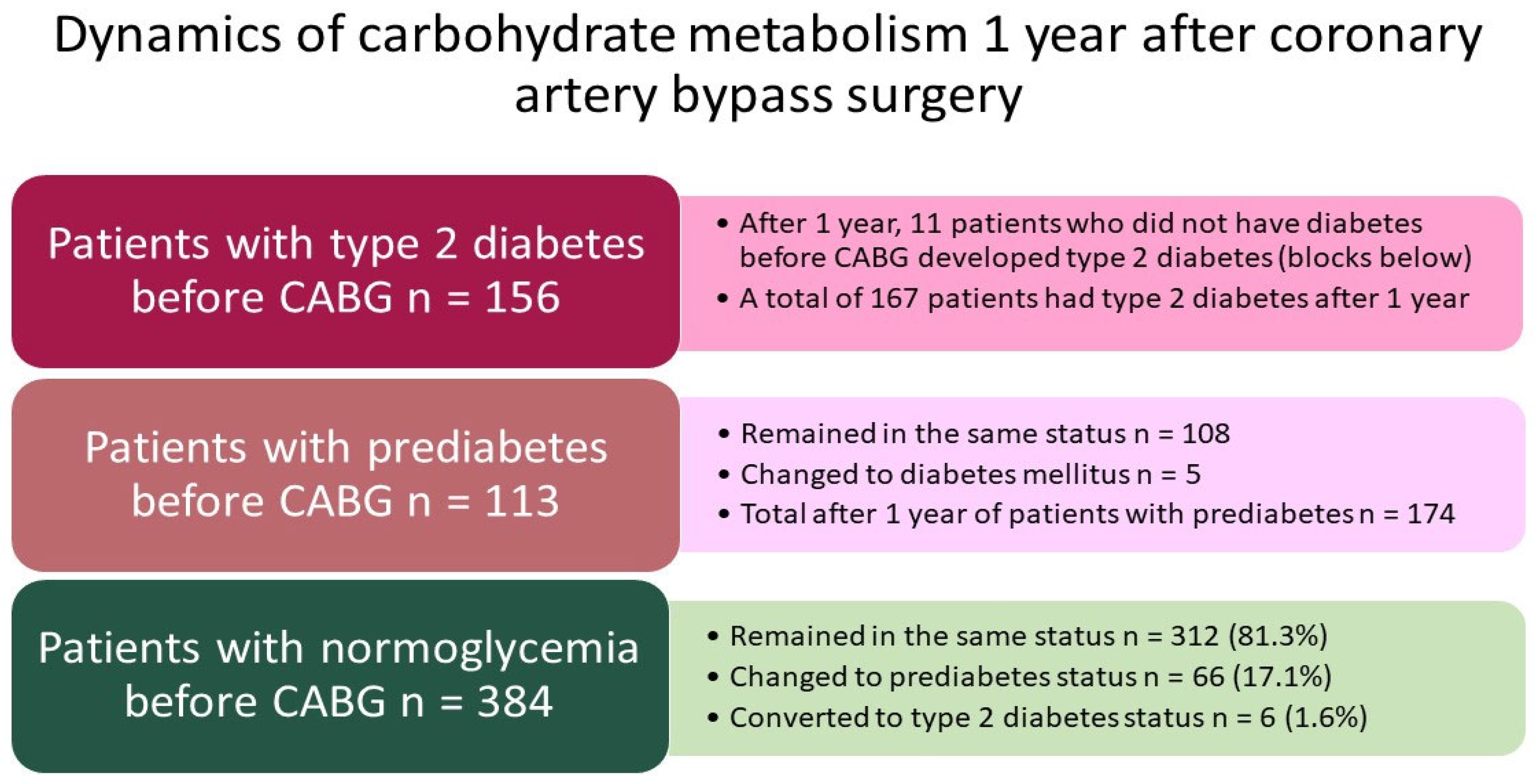
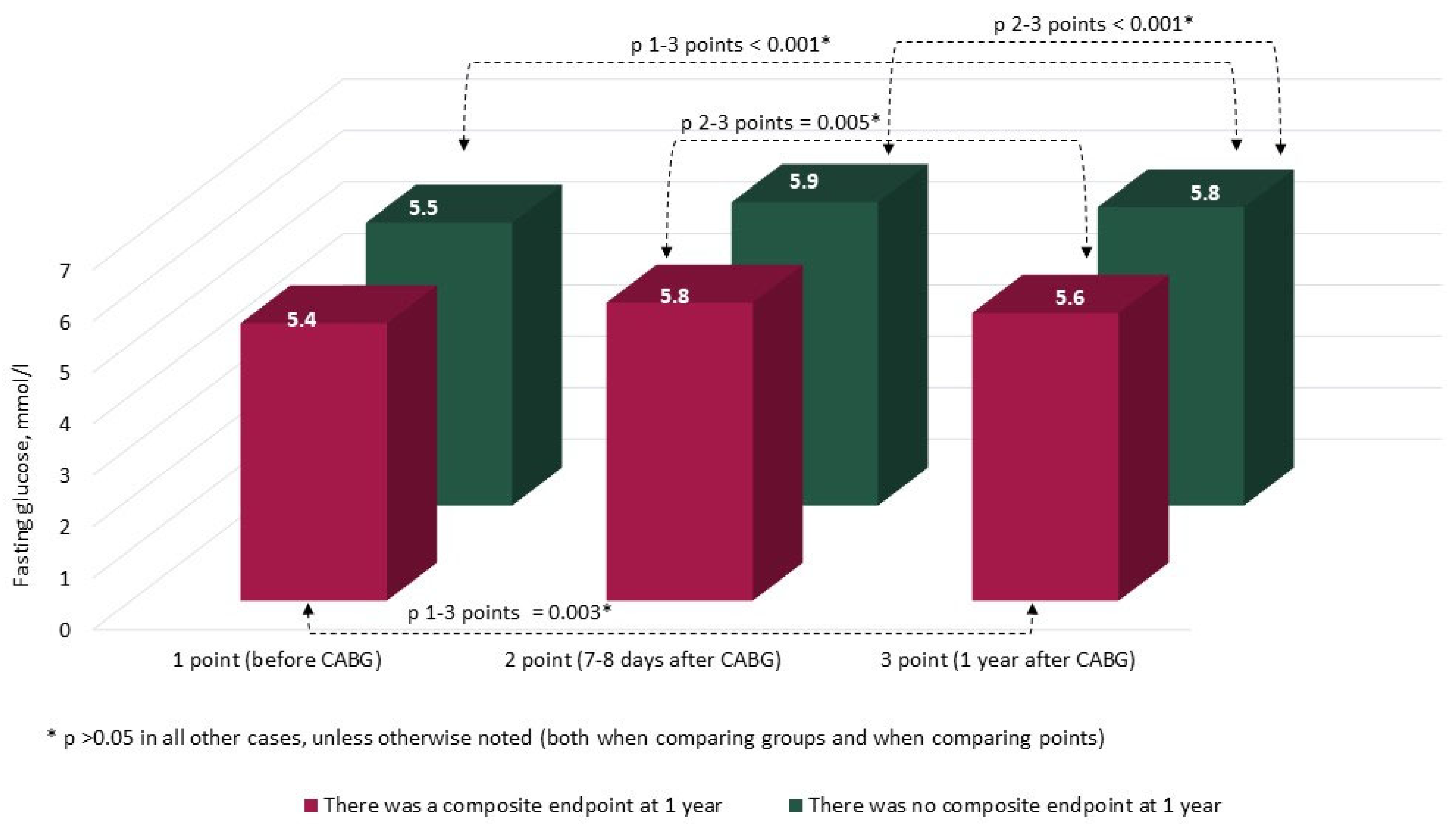
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Neumann, F.J.; Sousa-Uva, M.; Ahlsson, A.; Alfonso, F.; Banning, A.P.; Benedetto, U.; Byrne, R.A.; Collet, J.P.; Falk, V.; Head, S.J.; et al. 2018 ESC/EACTS Guidelines on myocardial revascularization. Eur. Heart J. 2019, 40, 87–165. [Google Scholar] [CrossRef]
- Farkouh, M.E.; Domanski, M.; Sleeper, L.A.; Siami, F.S.; Dangas, G.; Mack, M.; Yang, M.; Cohen, D.J.; Rosenberg, Y.; Solomon, S.D.; et al. Strategies for multivessel revascularization in patients with diabetes. N. Engl. J. Med. 2012, 367, 2375–2384. [Google Scholar] [CrossRef]
- Turgeon, R.D.; Koshman, S.L.; Youngson, E.; Pearson, G.J. Association Between Hemoglobin A1c and Major Adverse Coronary Events in Patients with Diabetes Following Coronary Artery Bypass Surgery. Pharmacotherapy 2020, 40, 116–124. [Google Scholar] [CrossRef]
- Wang, F.; Mei, X. Association of blood glucose change with postoperative delirium after coronary artery bypass grafting in patients with diabetes mellitus: A study of the MIMIC-IV database. Front. Endocrinol. 2024, 15, 1400207. [Google Scholar] [CrossRef]
- Fukuda, Y.; Ushigome, E.; Yamazaki, M.; Fukui, M. Postoperative Insulin Dose for Cardiac Artery Bypass Graft and Other Cardiac Surgeries in Patients with Type 2 Diabetes: A Retrospective Study. Vasc. Health Risk Manag. 2024, 20, 59–68. [Google Scholar] [CrossRef]
- Lam, S.; Kumar, B.; Loke, Y.K.; Orme, S.E.; Dhatariya, K. Glycated haemoglobin and the risk of postoperative complications in people without diabetes: A prospective population-based study in UK Biobank. Anaesthesia 2022, 77, 659–667. [Google Scholar] [CrossRef] [PubMed]
- Sumin, A.N.; Bezdenezhnykh, N.A.; Bezdenezhnykh, A.V.; Kuzmina, A.A.; Dyleva, Y.A.; Barbarash, O.L. Association of Alternative Markers of Carbohydrate Metabolism (Fructosamine and 1,5-Anhydroglucitol) with Perioperative Characteristics and In-Hospital Complications of Coronary Artery Bypass Grafting in Patients with Type 2 Diabetes Mellitus, Prediabetes, and Normoglycemia. Diagnostics 2023, 13, 969. [Google Scholar] [CrossRef]
- You, H.; Hou, X.; Zhang, H.; Li, X.; Feng, X.; Qian, X.; Shi, N.; Guo, R.; Wang, X.; Sun, H.; et al. Effect of glycemic control and glucose fluctuation on in-hospital adverse outcomes after on-pump coronary artery bypass grafting in patients with diabetes: A retrospective study. Diabetol. Metab. Syndr. 2023, 15, 20. [Google Scholar] [CrossRef]
- Robich, M.P.; Iribarne, A.; Leavitt, B.J.; Malenka, D.J.; Quinn, R.D.; Olmstead, E.M.; Ross, C.S.; Sawyer, D.B.; Klemperer, J.D.; Clough, R.A.; et al. Intensity of Glycemic Control Affects Long-Term Survival After Coronary Artery Bypass Graft Surgery. Ann. Thorac. Surg. 2019, 107, 477–484. [Google Scholar] [CrossRef]
- Alshair, F.M.; Baghaffar, A.H.; Fatani, M.A.; Alqahtani, A.K.; Al Assiri, A.K.; Alsulymani, B.M.; Sanedi, A.M.; Bamousa, S.M. Glycosylated Hemoglobin (HbA1C) as a Predictor of Early Postoperative Outcomes After Coronary Artery Bypass Grafting: A Single-Center Observational Study. Cureus 2024, 16, e65567. [Google Scholar] [CrossRef]
- Bezdenezhnykh, N.A.; Sumin, A.N.; Bezdenezhnykh, A.V.; Sinitskaya, A.V.; Kuzmina, A.A.; Koshelev, V.A.; Briukhanov, Y.a.I.; Fokin, F.A.; Barbarash, O.L. Association between insulin resistance indices and perioperative functional status and immediate prognosis in patients with carbohydrate metabolism disorders undergoing coronary artery bypass grafting. Complex Issues Cardiovasc. Dis. 2022, 11, 6–24. [Google Scholar] [CrossRef]
- Sumin, A.N.; Bezdenezhnykh, N.A.; Bezdenezhnykh, A.V.; Osokina, A.V.; Kuzmina, A.A.; Sinitskaya, A.V.; Barbarash, O.L. The Role of Insulin Resistance in the Development of Complications after Coronary Artery Bypass Grafting in Patients with Coronary Artery Disease. Biomedicines 2023, 11, 2977. [Google Scholar] [CrossRef]
- Abu Tailakh, M.; Ishay, S.Y.; Awesat, J.; Poupko, L.; Sahar, G.; Novack, V. Hemoglobin A1c in Patients with Diabetes Predict Long-Term Mortality Following Coronary Artery Surgery. J. Clin. Med. 2021, 10, 2739. [Google Scholar] [CrossRef]
- Skendelas, J.P.; Phan, D.K.; Friedmann, P.; Rodriguez, C.J.; Stein, D.; Arbab-Zadeh, A.; Forest, S.J.; Slipczuk, L. Perioperative Cardiometabolic Targets and Coronary Artery Bypass Surgery Mortality in Patients With Diabetes. J. Am. Hear. Assoc. 2022, 11, e023558. [Google Scholar] [CrossRef]
- Chen, J.; Yin, D.; Dou, K. Intensified glycemic control by HbA1c for patients with coronary heart disease and Type 2 diabetes: A review of findings and conclusions. Cardiovasc. Diabetol. 2023, 22, 146. [Google Scholar] [CrossRef]
- Sumin, A.N.; Bezdenezhnykh, N.A.; Bezdenezhnykh, A.V.; Osokina, A.V.; Kuz’mina, A.A.; Tsepokina, A.V.; Barbarash, O.L. Screening for Glucose Metabolism Disorders, Assessment the Disse Insulin Resistance Index and Hospital Prognosis of Coronary Artery Bypass Surgery. J. Pers. Med. 2021, 11, 802. [Google Scholar] [CrossRef]
- Dedov, I.I.; Shestakova, M.V.; Aleksandrov, A.A.; Galstyan, G.R.; Grigoryan, O.R.; Esayan, R.M.; Kalashnikov, V.Y.; Kuraeva, T.L.; Lipatov, D.V.; Mayorov, A.Y.; et al. Algorithms of specialized medical care for patients with diabetes mellitus patients specialized medical care to patients with diabetes, 5 edition. Diabetes Mellit. 2011, 14, 2–72. [Google Scholar] [CrossRef]
- World Health Organization. Definition and Diagnosis of Diabetes Mellitus and Intermediate Hyperglycemia: Report of a WHO/IDF.; Report of a WHO/IDF Consultation; World Health Organization: Geneva, Switzerland, 2006; pp. 1–50. [Google Scholar]
- Deo, S.; Sundaram, V.; Sheikh, M.A.; Sahadevan, J.; Selvaganesan, P.; Madan Mohan, S.K.; Rubelowsky, J.; Elgudin, Y.; Josephson, R.; Davierwala, P.M.; et al. Pre-operative glycaemic control and long-term survival in diabetic patients after coronary artery bypass grafting. Eur. J. Cardiothorac Surg. 2021, 60, 1169–1177. [Google Scholar] [CrossRef]
- Liu, B.; Li, Z.; Chen, X.; Wang, S.; Cheng, K. Influence of preoperative HbA1c levels on the occurrence of postoperative complications in patients undergoing coronary artery bypass grafting. J. Cardiothorac. Surg. 2024, 19, 593. [Google Scholar] [CrossRef]
- Zhu, Y.; Hu, J.; Zhang, M.; Xue, Q.; Liu, H.; Wang, R.; Wang, X.; Cheng, Z.; Zhao, Q. Influence of Baseline HbA1c and Antiplatelet Therapy on 1-Year Vein Graft Outcome. JACC Asia 2022, 2, 197–206. [Google Scholar] [CrossRef]


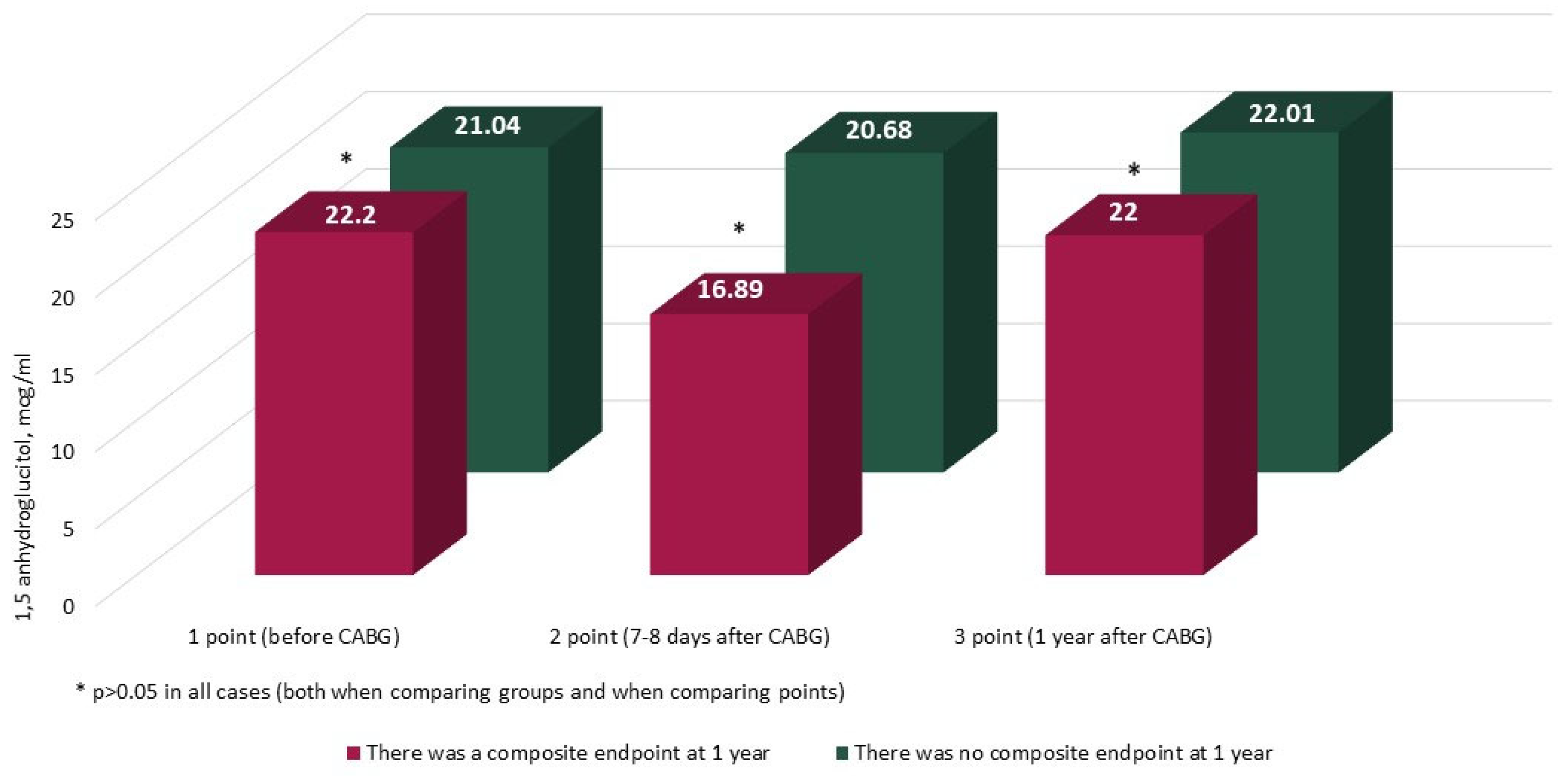
| Indicator | Group With CEP at 1 Year (n = 59) | Group Without CEP at 1 Year (n = 594) | p |
|---|---|---|---|
| Men (n,%) | 51 (86.4) | 467 (78.6) | 0.157 |
| Women (n, %) | 8 (13.6) | 127 (21.4) | 0.157 |
| Age (years, Me [LQ; UQ]) | 59.0 (54.0; 65.5) | 58.0 (54.1; 64.0) | 0.561 |
| Mental work (n, %) | 12 (20.3) | 209 (35.1) | <0.001 |
| Physical labor (n, %) | 47 (79.7) | 385 (64.8) | <0.001 |
| Higher education (n, %) | 6 (10.2) | 134 (22.5) | 0.027 |
| BMI (kg/m2, Me [LQ;UQ]) | 27.2 (24.2; 29.6) | 28.0 (25.2; 31.19) | 0.086 |
| Obesity (n, %) | 14 (23.7) | 208 (35.0) | 0.044 |
| Type 2 diabetes mellitus before CABG (n, %) | 10 (20.3) | 146 (24.6) | 0.524 |
| Prediabetes before CABG (IFG or IGT) (n, %) | 12 (18.6) | 101 (17.0) | 0.646 |
| Any disturbance of carbohydrate metabolism before CABG (n, %) | 19 (32.2) | 226 (38.1) | 0.472 |
| Arterial hypertension (n, %) | 55 (93.2) | 521 (87.7) | 0.210 |
| III—IV FC angina (n, %) | 24 (40.7) | 238 (40.1) | 0.927 |
| III-IV NYHA FC heart failure (n, %) | 16 (27.1) | 173 (29.1) | 0.746 |
| Smoking at the time of CABG (n, %) | 25 (42.4) | 205 (34.5) | 0.228 |
| Intermittent claudication (n, %) | 16 (27.1) | 67 (11.3) | <0.001 |
| Pain-free walking distance (m, Me [LQ; UQ]) | 150 (70.0; 300.0) | 450.0 (200.0; 1000.0) | 0.007 |
| History of myocardial infarction (n, %) | 38 (64.4) | 383 (64.5) | 0.936 |
| History of stroke (n, %) | 8 (13.5) | 40 (6.7) | 0.061 |
| History of PCI (n, %) | 6 (10.2) | 51 (8.6) | 0.711 |
| History of CABG (n, %) | 0 (0) | 5 (0.8) | 0.682 |
| History of surgery on carotid arteries (n, %) | 4 (6.8) | 13 (2.2) | 0.037 |
| History of surgery on arteries of the lower extremities (n, %) | 1 (1.7) | 3 (0.5) | 0.271 |
| History of thyroid disease (n, %) | 2 (3.4) | 34 (5.7) | 0.439 |
| EuroSCORE 2 (%, Me [LQ; UQ]) | 2.4 (1.7; 4.1) | 1.7 (1.1; 2.5) | <0.001 |
| Indicator | n, % |
|---|---|
| Visit to the study center after 1 year | 505 (77.3) |
| Telephone contact or other means of communication after 1 year, except for a visit | 148 (22.7) |
| All cardiovascular events at 1 year (combined endpoint) | 59 (9.0) |
| Major adverse cardiovascular events in 1 year | 29 (4.4) |
| Death from all causes in 1 year | 18 (2.6) |
| Myocardial infarction | 4 (0.06) |
| Stroke | 7 (1.1) |
| Percutaneous coronary intervention | 6 (0.1) |
| Carotid artery intervention | 10 (1.5) |
| Intervention in the arteries of the lower extremities | 12 (1.8) |
| Recurrence of angina | 6 (0.1) |
| Emergency hospitalizations for CVD | 18 (2.7) |
| Indicator | Group with CEP at 1 Year (n = 59) | Group Without CEP at 1 Year (n = 594) | p |
|---|---|---|---|
| On-pump heart surgery (n, %) | 47 (79.6) | 525 (88.4) | 0.053 |
| Cardiopulmonary bypass laminar mode (n, % of the number of patients on pump) | 37 (78.7) | 431 (82.1) | 0.600 |
| Cardiopulmonary bypass pulse mode (n, % of the number of patients on pump) | 10 (21.2) | 94 (17.9) | 0.600 |
| Off-pump heart surgery (n, %) | 12 (20.3) | 69 (11.6) | 0.053 |
| Isolated CABG (n, %) | 49 (83.0) | 546 (91.9) | 0.022 |
| Combined operations (n, %) | 10 (17.0) | 48 (8.1) | 0.022 |
| 2 (3.4) | 9 (1.5) | 0.296 |
| 4 (6.7) | 26 (4.4) | 0.401 |
| 1 (1.7) | 16 (2.7) | 0.633 |
| 1 (1.7) | 2 (0.3) | 0.146 |
| 2 (3.4) | 4 (0.7) | 0.037 |
| Thrombectomy (n, %) | 3 (5.1) | 14 (2.4) | 0.220 |
| Total duration of surgery (min, Me [LQ; UQ]) | 246.0 [192.0; 282.0] | 246.0 [204.0; 300.0] | 0.472 |
| Cardiopulmonary bypass duration (min, Me [LQ; UQ]) | 98.0 [78.0; 125.0] | 95 [78.0; 110.0] | 0.207 |
| Duration of aortic clamping (min, Me [LQ; UQ]) | 65.0 [52.0; 77.0] | 60.0 [50.0; 73.0] | 0.215 |
| Indicator (n, %, Unless Otherwise Stated) | Group with CEP at 1 Year (n = 59) | Group Without CEP at 1 Year (n = 594) | p # | |||
|---|---|---|---|---|---|---|
| Acetylsalicylic acid (n, %) | preoperatively | 32 (54.2) | 374 (63.0) | 0.225 | ||
| pre-hospital | 28 (47.4) | p * < 0.001 | 394 (66.3) | p * < 0.001 | 0.007 | |
| in 1 year | 14 (23.7) | 286 (48.1) | <0.001 | |||
| β blockers (n, %) | preoperatively | 58 (98.3) | 564 (94.9) | 0.309 | ||
| pre-hospital | 32 (54.2) | p * < 0.001 | 374 (63.0) | p * < 0.001 | 0.225 | |
| in 1 year | 15 (25.4) | 284 (47.8) | 0.032 | |||
| Angiotensin-converting enzyme inhibitors (n, %) | preoperatively | 51 (86.4) | 506 (85.2) | 0.928 | ||
| pre-hospital | 31 (52.5) | p * = 0.001 | 273 (45.9) | p * = 0.005 | 0.290 | |
| in 1 year | 12 (20.3) | 214 (36.0) | 0.013 | |||
| Angiotensin 2 receptor antagonists (n, %) | preoperatively | 1 (1.7) | 28 (4.7) | 0.283 | ||
| pre-hospital | 1 (1.7) | p * = 0.917 | 42 (7.0) | p * = 0.883 | 0.154 | |
| in 1 year | 1 (1.7) | 43 (7.2) | 0.488 | |||
| Statins (n, %) | preoperatively | 40 (67.8) | 443 (74.6) | 0.227 | ||
| pre-hospital | 27 (45.8) | p * = 0.021 | 279 (47.0) | p * = 0.861 | 0.835 | |
| in 1 year | 15 (25.4) | 282 (47.5) | 0.032 | |||
| Calcium channel blockers (n, %) | preoperatively | 30 (50.9) | 368 (62.0) | 0.095 | ||
| pre-hospital | 13 (22.0) | p * = 0.674 | 135 (22.7) | p * = 0.834 | 0.871 | |
| in 1 year | 11 (18.6) | 132 (22.2) | 0.981 | |||
| Mineralocorticoid receptor antagonists (n, %) | preoperatively | 15 (25.4) | 95 (16.0) | 0.171 | ||
| pre-hospital | 5 (9.1) | p * = 0.751 | 51 (8.6) | p * = 0.454 | 0.839 | |
| in 1 year | 6 (10.7) | 44 (7.4) | 0.251 | |||
| Thiazide-like diuretics (n, %) | preoperatively | 3 (5.1) | 57 (9.5) | 0.308 | ||
| pre-hospital | 6 (10.1) | p * = 0.298 | 32 (5.3) | p * = 0.101 | 0.434 | |
| in 1 year | 3 (5.1) | 46 (7.7) | 0.491 | |||
| Loop diuretics (n, %) | preoperatively | 4 (6.7) | 39 (6.5) | 0.831 | ||
| pre-hospital | 1 (1.7) | p * = 0.558 | 12 (2.0) | p * = 0.667 | 0.905 | |
| in 1 year | 2 (3.4) | 10 (1.8) | 0.711 | |||
| Insulin (n, %) | preoperatively | 5 (8.5) | 65 (10.9) | 0.558 | ||
| pre-hospital | 3 (5.0) | p * = 0.675 | 24 (4.0) | p * = 0.773 | 0.870 | |
| in 1 year | 3 (5.0) | 26 (4.3) | 0.365 | |||
| Median insulin dose (U/day, Me [LQ; UQ]) | preoperatively | 28.0 (18.0; 36.0) | 28.0 (18.0; 38.0) | 0.733 | ||
| pre-hospital | 34.0 (24.0; 44.0) | p * = 0.047 | 34.0 (20.0; 38.0) | p * = 0.018 | 0.733 | |
| in 1 year | 36.0 (22.0;46.0) | 38.0 (26.0;48.0) | 0.801 | |||
| Oral medications for the treatment of diabetes mellitus (n,%) | pre-hospital | 4 (5.1) | p * = 0.551 | 57 (9.6) | p * = 0.842 | 0.258 |
| in 1 year | 5 (8.5) | 55 (9.2) | 0.586 | |||
| Sulfonylurea drugs (n, %) | pre-hospital | 3 (5.1) | p * = 0.674 | 32 (5.4) | p * = 0.794 | 0.900 |
| in 1 year | 2 (3.4) | 30 (5.0) | 0.636 | |||
| Metformin (n, %) | pre-hospital | 1 (1.7) | p * = 0.611 | 25 (4.4) | p * = 0.754 | 0.260 |
| in 1 year | 3 (5.1) | 32 (5.4) | 0.754 | |||
| DPP-4 inhibitors (n, %) | pre-hospital | 1 (1.7) | p * = 0.560 | 4 (0.7) | p * = 0.758 | 0.657 |
| in 1 year | 2 (3.4) | 6 (1.0) | 0.562 | |||
| GLP-1 agonists (n, %) | pre-hospital | 1 (1.7) | p * = 0.999 | 2 (0.3) | p * = 0.654 | 0.789 |
| in 1 year | 1 (1.7) | 3 (0.5) | 0.638 | |||
| Indicator | Group with CEP at 1 Year (n = 59) | Group Without CEP at 1 Year (n = 594) | p |
|---|---|---|---|
| Coronary angiography results (n, %) | |||
| 1 vessel disease | 15 (25.4) | 134 (22.6) | 0.617 |
| 2 vessel diseases | 13 (22.6) | 162 (27.3) | 0.386 |
| 3 vessel diseases | 27 (45.8) | 255 (42.9) | 0.675 |
| Stenosis of the left main coronary artery >50% | 10 (16.9) | 117 (19.7) | 0.611 |
| 3 vessels and left main coronary artery | 34 (6.7) | 34 (5.7) | 0.554 |
| 3 vessels or left main coronary artery | 33 (55.9) | 320 (53.9) | 0.762 |
| Aggregate results of instrumental examinations of non-coronary arterial basins (n, %, unless otherwise stated) | |||
| Multifocal atherosclerosis, lesion of 3 arterial basins | 21 (35.6) | 87 (14.7) | <0.001 |
| Multifocal atherosclerosis, lesion of 2 or more arterial basins | 32 (54.2) | 157 (44.8) | <0.001 |
| Multifocal atherosclerosis, lesion of 2 arterial basins | 25 (42.3) | 199 (33.5) | 0.171 |
| Absence of multifocal atherosclerosis (damage to the coronary bed only) | 13 (22.0) | 308 (51.9) | <0.001 |
| Carotid artery stenosis ≥50% before CABG | 21 (16.2) | 19 (12.1) | 0.354 |
| Stenosis of the arteries of the lower extremities before CABG | 37 (28.7) | 46 (29.0) | 0.355 |
| Intima-media thickness, mm (Me [LQ; UQ]) | 1.2 (1.0; 1.3) | 1.1 (1.0; 1.2) | 0.043 |
| Echocardiography data, extended protocol (Me [LQ; UQ]) | |||
| LV end-diastolic volume (ml) | 160.0 (133.0; 180.0) | 155.0 (135.0; 188.0) | 0.759 |
| LV end-diastolic dimension (cm) | 5.7 (5.3; 6.0) | 5.6 (5.3 6.1) | 0.692 |
| LV end-systolic volume (ml) | 66.0 (47.0; 104.0) | 62 (50.0; 92) | 0.816 |
| LV end-systolic dimension (cm) | 3.9 (3.5; 4.7) | 3.8 (3.5; 4.6) | 0.667 |
| Left atrium (cm) | 4.15 (3.8; 4.5) | 4.2 (3.9; 4.5) | 0.144 |
| Interventricular septum (mm) | 1.1 (1.0; 1.2) | 1.1 (1.0; 1.2) | 0.854 |
| Posterior wall of the left ventricle (mm) | 1.1 (1.0; 1.2) | 1.1 (1.0; 1.2) | 0.638 |
| Right ventricle, cm | 1.8 (1.75; 1.8) | 1.8 (1.8; 1.9) | 0.336 |
| Right atrium, cm | 4.3 (4.0; 4.6) | 4.2 (3.8; 4.6) | 0.374 |
| Aorta, cm | 3.4 (3.2; 3.7) | 3.5 (3.3; 3.7) | 0.334 |
| LV ejection fraction, % | 58.0 (46.0; 65.0) | 60 (50.0; 64.0) | 0.798 |
| Pulmonary artery pressure, systolic, mmHg | 12.0 (10.0; 30.0) | 18 (12.0; 28.0) | 0.257 |
| E (cm/sec) | 53.0 (46.0; 73.0) | 61.0 (50.0; 75.0) | 0.140 |
| A (cm/sec) | 68.0 (56.0; 88.0) | 65.0 (53.0; 78.0) | 0.438 |
| E/A | 0.7 (0.6; 1.2) | 0.8 (0.7; 1.2) | 0.039 |
| DT, sec | 190.0 (140.0; 204.0) | 200.0 (180.0; 235.0) | 0.004 |
| IRT—isovolumic relaxation time, sec | 111.0 (93.0; 122.0) | 100.0 (90.0; 114.0) | 0.059 |
| Flow velocity (cm/sec)—Vf | 59.0 (46.0; 61.0) | 48.0 (42.0; 60.0) | 0.254 |
| LV myocardial mass according to Deveraux and Reichek, g | 314.0 (246.3; 375.2) | 306, 4 (250.4; 372.7) | 0.395 |
| LV myocardial mass index | 166.1 (129.3; 198.6) | 161.7 (134.2; 190.5) | 0.181 |
| Preoperative laboratory data (except carbohydrate metabolism) Me (LQ; UQ) | |||
| Aspartate aminotransferase (IU/L) | 23.0 (20.0; 31.0) | 24.0 (19.0; 31.0) | 0.626 |
| Alanine aminotransferase (IU/L) | 26.0 (17.0; 37.0) | 25.0 (18.0; 36.9) | 0.665 |
| Total cholesterol (mmol/l) | 5.0 (4.3; 6.0) | 4.9 (4.2; 5.9) | 0.691 |
| Triglycerides (mmol/l) | 1.8 (1.3; 2.3) | 1.7 (1.3; 2.3) | 0.393 |
| HDL cholesterol (mmol/l) | 0.97 (0.88; 1.15) | 0.99 (0.83; 1.19) | 0.937 |
| LDL cholesterol (mmol/l) | 3.02 (2.5; 4.1) | 2.95 (2.3; 3.7) | 0.290 |
| Creatinine (µmol/l) | 77.5 (65.0; 94.0) | 76.0 (64.0; 91.0) | 0.082 |
| GFR CKD—EPI (ml/min/1.73 m2) | 76.4 (62.9; 90.3) | 82.4 (66.7; 102.6) | 0.106 |
| SFMCs (g/l) | 7.5 (4.8; 12.0) | 5.0 (4.0; 8.0) | <0.001 |
| Fibrinogen (g/l) | 5.3 (3.8; 6.2) | 4.2 (3.5; 5.5) | 0.004 |
| 95% CI.for EXP(B) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| B | Lower | Upper | df | Sig. | Exp(B) | Lower | Upper | ||
| Step 1 | EuroSCORE | 0.183 | 0.059 | 9.650 | 1 | 0.002 | 1.201 | 1.070 | 1.348 |
| Constant | −3.441 | 0.275 | 156.543 | 1 | 0.000 | 0.032 | |||
| Step 2 | Fibrinogen | 0.243 | 0.113 | 4.590 | 1 | 0.032 | 1.275 | 1.021 | 1.591 |
| EuroSCORE | 0.184 | 0.060 | 9.562 | 1 | 0.002 | 1.202 | 1.070 | 1.351 | |
| Constant | −4.645 | 0.659 | 49.680 | 1 | 0.000 | 0.010 | |||
| Step 3 | Fibrinogen | 0.263 | 0.115 | 5.246 | 1 | 0.022 | 1.300 | 1.039 | 1.628 |
| EuroSCORE | 0.183 | 0.060 | 9.415 | 1 | 0.002 | 1.201 | 1.069 | 1.351 | |
| CABG on pump | −0.976 | 0.470 | 4.316 | 1 | 0.038 | 0.377 | 0.150 | 0.946 | |
| Constant | −3.943 | 0.723 | 29.764 | 1 | 0.000 | 0.019 | |||
| Step 4 | PAD | 0.949 | 0.437 | 4.711 | 1 | 0.030 | 2.583 | 1.096 | 6.083 |
| Fibrinogen | 0.276 | 0.116 | 5.676 | 1 | 0.017 | 1.317 | 1.050 | 1.653 | |
| EuroSCORE | 0.173 | 0.061 | 8.090 | 1 | 0.004 | 1.189 | 1.055 | 1.340 | |
| CABG on pump | −1.038 | 0.476 | 4.764 | 1 | 0.029 | 0.354 | 0.139 | 0.900 | |
| Constant | −5.102 | 0.930 | 30.088 | 1 | 0.000 | 0.006 | |||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sumin, A.N.; Bezdenezhnykh, N.A.; Belik, E.V.; Dyleva, Y.A.; Bezdenezhnykh, A.V.; Gruzdeva, O.V.; Barbarash, O.L. Dynamics of Glycemic Status and Glucose Metabolism Markers 12 Months After Coronary Artery Bypass Grafting and Their Relationship with the Annual Prognosis of Patients. J. Clin. Med. 2025, 14, 351. https://doi.org/10.3390/jcm14020351
Sumin AN, Bezdenezhnykh NA, Belik EV, Dyleva YA, Bezdenezhnykh AV, Gruzdeva OV, Barbarash OL. Dynamics of Glycemic Status and Glucose Metabolism Markers 12 Months After Coronary Artery Bypass Grafting and Their Relationship with the Annual Prognosis of Patients. Journal of Clinical Medicine. 2025; 14(2):351. https://doi.org/10.3390/jcm14020351
Chicago/Turabian StyleSumin, Alexey N., Natalia A. Bezdenezhnykh, Ekaterina V. Belik, Yulia A. Dyleva, Andrey V. Bezdenezhnykh, Olga V. Gruzdeva, and Olga L. Barbarash. 2025. "Dynamics of Glycemic Status and Glucose Metabolism Markers 12 Months After Coronary Artery Bypass Grafting and Their Relationship with the Annual Prognosis of Patients" Journal of Clinical Medicine 14, no. 2: 351. https://doi.org/10.3390/jcm14020351
APA StyleSumin, A. N., Bezdenezhnykh, N. A., Belik, E. V., Dyleva, Y. A., Bezdenezhnykh, A. V., Gruzdeva, O. V., & Barbarash, O. L. (2025). Dynamics of Glycemic Status and Glucose Metabolism Markers 12 Months After Coronary Artery Bypass Grafting and Their Relationship with the Annual Prognosis of Patients. Journal of Clinical Medicine, 14(2), 351. https://doi.org/10.3390/jcm14020351






