Post-Traumatic Stress and Stressor-Related Disorders in Hematological Malignancies: A Review
Abstract
1. Introduction
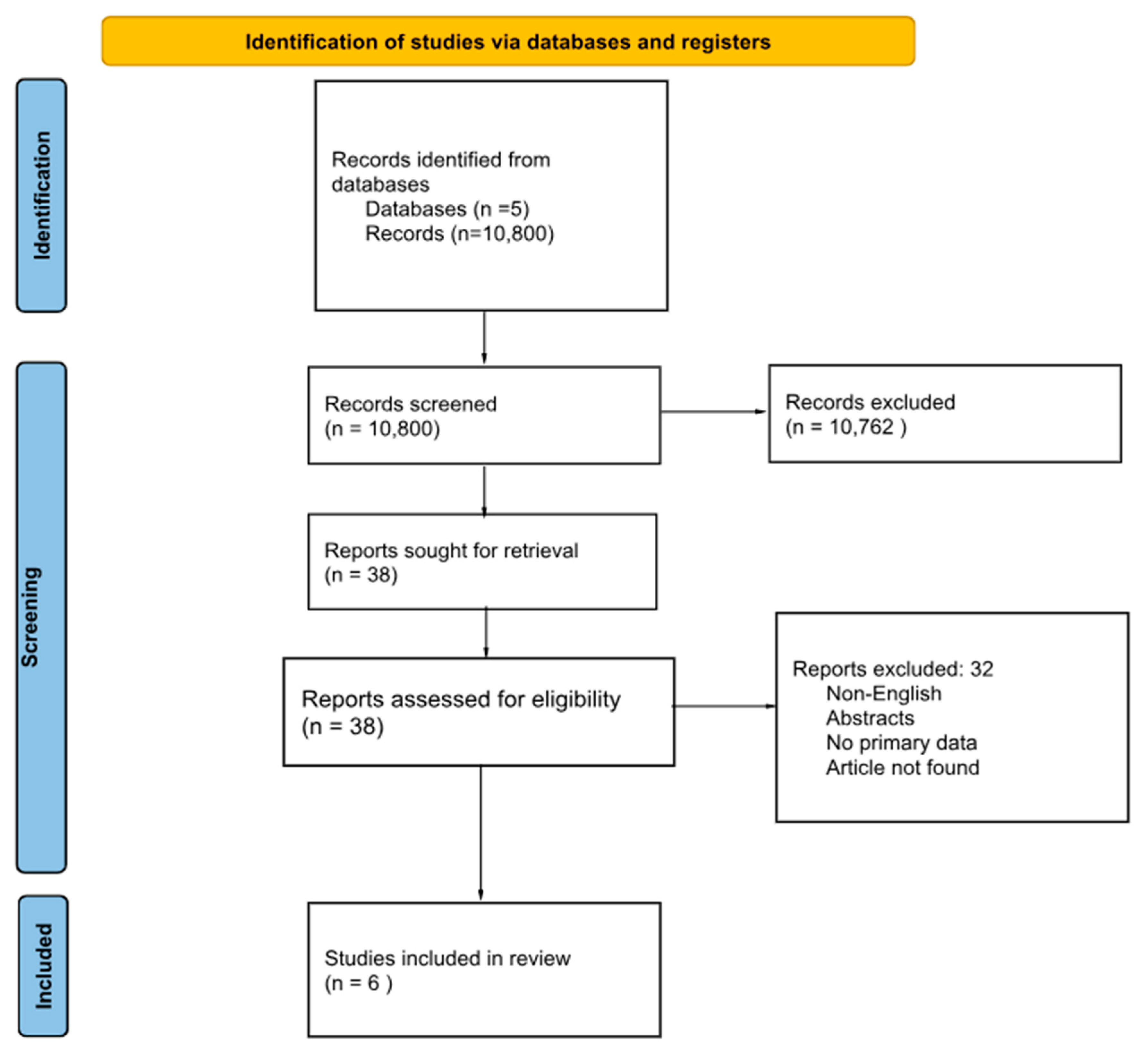
2. Methods
3. Results
4. Discussion
4.1. Assessment and Screening
- Post-Traumatic Stress Disorder Checklist–Civilian Version (PCL-C)—a self-administered questionnaire widely used in cancer-related PTSD screening due to its brevity, alignment with DSM-IV criteria, and sensitivity to changes in symptom severity over time [25]. It can be used for both provisional diagnosis and monitoring of symptom progression in clinical or research settings.
- Impact of Event Scale–Revised (IES-R)—a self-administered measure capturing subjective distress in response to a specific traumatic event, assessing intrusion, avoidance, and hyperarousal [26]. While useful for screening, it does not assess all DSM-5 PTSD criteria and should be supplemented with structured diagnostic interview.
- Stanford Acute Stress Reaction Questionnaire (SASRQ)—a self-administered tool measuring acute stress symptoms in the immediate post-diagnosis phase, including dissociation, re-experiencing, avoidance, and arousal [27]. It is particularly relevant during the early post-diagnosis period, when symptoms may not yet meet the threshold or duration required for a PTSD diagnosis.
- Impact of Event Scale–Revised (IES-R)—a self-administered measure capturing subjective distress in response to a specific traumatic event, assessing intrusion, avoidance, and hyperarousal [26]. While useful for screening, it does not assess all DSM-5 PTSD criteria and should be supplemented with structured diagnostic interviews.
- Adjustment Disorder New Module 20 (ADNM-20)—a self-administered questionnaire designed to assess adjustment disorder symptoms and severity across multiple stress-related domains [28]. It is well-suited to oncology settings for identifying patients experiencing significant distress who do not meet criteria for PTSD or acute stress disorder.
- Posttraumatic Diagnostic Scale (PDS)—a self-administered questionnaire assessing DSM-IV PTSD criteria, including trauma exposure, symptom severity, and functional impairment [25]. Originally developed for non-medical trauma populations, it has demonstrated utility in oncology settings for identifying both full and subthreshold PTSD.
- PTSD Checklist–Specific (PCL-S)—a self-administered questionnaire measuring PTSD symptoms related to a specific traumatic event, used in French-validated form in Study 6 [19].
4.2. Prevalence of PTSD in Hematological Malignancies
4.3. Comparative Perspective with Solid-Tumor Research
4.4. Special Populations—PTSD in HSCT
4.5. Treatment Approaches and Interventions
4.5.1. Psychological Interventions
4.5.2. Integrative Palliative Care
4.5.3. Pharmacological Treatments
4.5.4. The Role of Social Support
5. Limitations
6. Future Directions and Clinical Implications
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Veenstra, C.; Bruce, D.; Timbs, A.; Hamblin, A. Application of genomics to clinical practice in haematological malignancy. Curr. Genet. Med. Rep. 2019, 7, 179–186. [Google Scholar] [CrossRef]
- Waddington, F.; Amerikanou, M.; Brett, J.; Watson, E.; Abbots, V.; Dawson, P.; Henshall, C. A systematic review to explore the effectiveness of physical health and psychosocial interventions on anxiety, depression and quality of life in people living with blood cancer. J. Psychosoc. Oncol. 2023, 42, 113–147. [Google Scholar] [CrossRef]
- Jadoon, N.A.; Munir, W.; Shahzad, M.A.; Choudhry, Z.S. Assessment of depression and anxiety in adult cancer outpatients: A cross-sectional study. BMC Cancer 2010, 10, 594. [Google Scholar] [CrossRef]
- Jairam, V.; Lee, V.; Park, H.S.; Thomas, C.R.; Melnick, E.R.; Gross, C.P.; Presley, C.J.; Adelson, K.B.; Yu, J.B. Treatment-related complications of systemic therapy and radiotherapy. JAMA Oncol. 2019, 5, 1028–1035. [Google Scholar] [CrossRef]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; text rev.; DSM-5-TR; American Psychiatric Publishing: Washington, DC, USA, 2022. [Google Scholar]
- Cordova, M.J.; Riba, M.B.; Spiegel, D. Post-traumatic stress disorder and cancer. Lancet Psychiatry 2017, 4, 330–338. [Google Scholar] [CrossRef] [PubMed]
- Rodin, G.; Lloyd-Williams, M.; Zimmermann, C.; Lo, C. Traumatic stress in acute leukemia. Psycho-Oncology 2013, 22, 299–307. [Google Scholar] [CrossRef]
- Gao, C.; Wang, J.; Li, Y.; Zhao, H.; Li, R.; Hou, L.; Zhang, Y.; Tian, S.; Liang, H.; Wang, C.; et al. Incidence and risk of hematologic toxicities with hypomethylating agents in the treatment of myelodysplastic syndromes and acute myeloid leukopenia: A systematic review and meta-analysis. Medicine 2018, 97, e11860. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kuhn, J.G. Chemotherapy-associated hematopoietic toxicity. Am. J. Health-Syst. Pharm. 2002, 59 (Suppl. S4), S4–S7. [Google Scholar] [CrossRef]
- O’Brien, S.N.; Blijlevens, N.M.; Mahfouz, T.H.; Anaissie, E.J. Infections in patients with hematological cancer: Recent developments. Hematol. Am. Soc. Hematol. Educ. Program 2003, 2003, 438–472. [Google Scholar] [CrossRef] [PubMed]
- Williams, A.M.; Zent, C.S.; Janelsins, M.C. What is known and unknown about chemotherapy-related cognitive impairment in patients with haematological malignancies and areas of needed research. Br. J. Haematol. 2016, 174, 835–846. [Google Scholar] [CrossRef] [PubMed]
- Hofer, F.; Koinig, K.A.; Nagl, L.; Borjan, B.; Stauder, R. Fatigue at baseline is associated with geriatric impairments and represents an adverse prognostic factor in older patients with a hematological malignancy. Ann. Hematol. 2018, 97, 2235–2243. [Google Scholar] [CrossRef]
- Springer, F.; Friedrich, M.; Kuba, K.; Ernst, J.; Glaesmer, H.; Platzbecker, U.; Vucinic, V.; Heyne, S.; Mehnert-Theuerkauf, A.; Esser, P. New progress in an old debate? Applying the DSM-5 criteria to assess traumatic events and stressor-related disorders in cancer survivors. Psycho-Oncology 2023, 32, 1616–1624. [Google Scholar] [CrossRef]
- Liu, L.; Yang, Y.-L.; Wang, Z.-Y.; Wu, H.; Wang, Y.; Wang, L. Prevalence and positive correlates of posttraumatic stress disorder symptoms among Chinese patients with hematological malignancies: A cross-sectional study. PLoS ONE 2015, 10, e0145103. [Google Scholar] [CrossRef]
- Springer, F.; Kuba, K.; Ernst, J.; Friedrich, M.; Glaesmer, H.; Platzbecker, U.; Vucinic, V.; Heyne, S.; Mehnert-Theuerkauf, A.; Esser, P. Symptoms of posttraumatic stress disorder and adjustment disorder in hematological cancer patients with different treatment regimes. Acta Oncol. 2023, 62, 1110–1117. [Google Scholar] [CrossRef]
- Amonoo, H.L.; LeBlanc, T.W.; Kavanaugh, A.R.; Webb, J.A.; Traeger, L.N.; Jagielo, A.D.; Vaughn, D.M.; Elyze, M.; Longley, R.M.; Fathi, A.T.; et al. Posttraumatic stress disorder symptoms in patients with acute myeloid leukemia. Cancer 2021, 127, 2500–2506. [Google Scholar] [CrossRef]
- Ehooman, F.; Biard, L.; Lemiale, V.; Contou, D.; de Prost, N.; Mokart, D.; Pène, F.; Kouatchet, A.; Mayaux, J.; Demoule, A.; et al. Long-term health-related quality of life of critically ill patients with haematological malignancies: A prospective observational multicenter study. Ann. Intensive Care 2019, 9, 2. [Google Scholar] [CrossRef] [PubMed]
- Romito, F.; Dellino, M.; Loseto, G.; Opinto, G.; Silvestris, E.; Cormio, C.; Guarini, A.; Minoia, C. Psychological distress in outpatients with lymphoma during the COVID-19 pandemic. Front. Oncol. 2020, 10, 1270. [Google Scholar] [CrossRef]
- Camille, C.; Dimeglio, C.; Yrondi, A.; Compaci, G.; Delmas, E.; Gauché, M.; Laurent, G.; Birmes, P. Posttraumatic stress disorder symptoms in lymphoma patients: A prospective study. Front. Psychiatry 2020, 11, 201. [Google Scholar] [CrossRef] [PubMed]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. PLOS Med. 2021, 18, e1003583. [Google Scholar] [CrossRef]
- Fenech, A.L.; Van Benschoten, O.; Jagielo, A.D.; Ufere, N.N.; Topping, C.E.; Clay, M.; Jones, B.T.; Traeger, L.; Temel, J.S.; El-Jawahri, A. Post-traumatic stress symptoms in hematopoietic stem cell transplant recipients. Transplant. Cell Ther. 2021, 27, 341.e1–341.e6. [Google Scholar] [CrossRef] [PubMed]
- El-Jawahri, A.R.; Bs, H.B.V.; Traeger, L.N.; Ba, J.N.F.; Keenan, T.; Gallagher, E.R.; Greer, J.A.; Pirl, W.F.; Jackson, V.A.; Spitzer, T.R.; et al. Quality of life and mood predict posttraumatic stress disorder after hematopoietic stem cell transplantation. Cancer 2016, 122, 806–812. [Google Scholar] [CrossRef] [PubMed]
- Jackson, B.N.; Weathers, F.W.; Jeffirs, S.M.; Preston, T.J.; Brydon, C.M. The revised Clinician-Administered PTSD scale for DSM-5 (CAPS-5-R): Initial psychometric evaluation in a trauma-exposed community sample. J. Trauma. Stress 2025, 38, 40–52. [Google Scholar] [CrossRef] [PubMed]
- LeardMann, C.A.; McMaster, H.S.; Warner, S.; Esquivel, A.P.; Porter, B.; Powell, T.M.; Tu, X.M.; Lee, W.W.; Rull, R.P.; Hoge, C.W.; et al. Comparison of Posttraumatic Stress Disorder Checklist Instruments from Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition vs. Fifth Edition in a Large Cohort of US Military Service Members and Veterans. JAMA Netw. Open 2021, 4, e218072. [Google Scholar] [CrossRef] [PubMed]
- Foa, E.B. Posttraumatic Diagnostic Scale (PDS) manual. In National Computer Systems; U.S. Department of Veterans Affairs: Washington, DC, USA, 1995. [Google Scholar]
- Weiss, D.S.; Marmar, C.R. The Impact of Event Scale–Revised. In Assessing Psychological Trauma and PTSD; Wilson, J.P., Keane, T.M., Eds.; The Guilford Press: New York, NY, USA, 1997; pp. 399–411. [Google Scholar]
- Cardeña, E.; Koopman, C.; Classen, C.C.; Waelde, L.C.; Spiegel, D. Psychometric properties of the Stanford Acute Stress Reaction Questionnaire (SASRQ): A valid and reliable measure of acute stress. J. Trauma. Stress 2000, 13, 719–734. [Google Scholar] [CrossRef]
- Lorenz, L.; Bachem, R.; Maercker, A. The Adjustment Disorder–New Module 20 as a screening instrument: Cluster analysis and cut-off values. Int. J. Occup. Environ. Med. 2016, 7, 215. [Google Scholar] [CrossRef]
- First, M.B.; Williams, J.B.W.; Karg, R.S.; Spitzer, R.L. Structured Clinical Interview for DSM-5 Disorders: Research Version (SCID-5-RV); American Psychiatric Association Publishing: Washington, DC, USA, 2015. [Google Scholar]
- Parker, A.M.; Sricharoenchai, T.; Raparla, S.; Schneck, K.W.; Bienvenu, O.J.; Needham, D.M. Posttraumatic stress disorder in critical illness survivors: A meta-analysis. Crit. Care Med. 2015, 43, 1121–1129. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Wang, J.; Cofie, R.; Kaminga, A.C.; Liu, A. Prevalence of Posttraumatic Stress Disorder among Breast Cancer Patients: A Meta-analysis. Iran. J. Public Health 2016, 45, 1533–1544. [Google Scholar] [PubMed] [PubMed Central]
- Weiss, O.; Runnels, J.; Dickstein, D.R.; Hsieh, K.; Jacobs, L.; Shah, A.; Arons, D.; Reed, S.; Sindhu, K.K.; Bakst, R.; et al. PTSD in Patients Who Undergo Head and Neck Cancer Treatment: A Systematic Review. Curr. Oncol. 2025, 32, 134. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Yahi, F.; Lequesne, J.; Rigal, O.; Morel, A.; Leheurteur, M.; Grellard, J.-M.; Leconte, A.; Clarisse, B.; Joly, F.; Lefèvre-Arbogast, S. Post-traumatic stress disorder symptoms and associated factors in breast cancer patients during the first COVID-19 lockdown in France. Front. Psychol. 2022, 13, 768043. [Google Scholar] [CrossRef]
- Esser, P.; Kuba, K.; Ernst, J.; Mehnert-Theuerkauf, A. Trauma- and stressor-related disorders among hematological cancer patients with and without stem cell transplantation: Protocol of an interview-based study according to updated diagnostic criteria. BMC Cancer 2019, 19, 870. [Google Scholar] [CrossRef]
- Wiener, L.; Sannes, T.S.; Randall, J.; Blosser, C.D.; Shaw, R.J.; Syrjala, K.L.; Bevans, M.; Jacobsen, P.B. Psychosocial assessment practices for hematopoietic stem cell transplantation: A national survey study. Bone Marrow Transplant. 2023, 58, 1314–1321. [Google Scholar] [CrossRef]
- DuHamel, K.N.; Mosher, C.E.; Winkel, G.; Labay, L.E.; Rini, C.; Meschian, Y.; Papadopoulos, E.; Brunstein, C.; Bartell, A.; Burkhalter, J.; et al. Randomized clinical trial of telephone-administered cognitive-behavioral therapy to reduce post-traumatic stress disorder and distress symptoms after hematopoietic stem-cell transplantation. J. Clin. Oncol. 2010, 28, 3754–3761. [Google Scholar] [CrossRef]
- Dimitrov, L.; Moschopoulou, E.; Korszun, A. Interventions for the treatment of cancer-related traumatic stress symptoms: A systematic review of the literature. Psycho-Oncology 2019, 28, 970–979. [Google Scholar] [CrossRef]
- Capezzani, L.; Ostacoli, L.; Cavallo, M.; Carletto, S.; Fernandez, I.; Solomon, R.; Pagani, M.; Cantelmi, T. EMDR and CBT for cancer patients: Comparative study of effects on PTSD, anxiety, and depression. J. EMDR Pract. Res. 2013, 7, 134–143. [Google Scholar] [CrossRef]
- Amonoo, H.L.; Johnson, P.C.; Nelson, A.M.; Clay, M.A.; Daskalakis, E.; Newcomb, R.A.; Deary, E.C.; Mattera, E.F.; Yang, D.; Cronin, K.; et al. Coping in caregivers of patients with hematologic malignancies undergoing hematopoietic stem cell transplantation. Blood Adv. 2023, 7, 1108–1116. [Google Scholar] [CrossRef] [PubMed]
- Carlson, L.E.; Toivonen, K.; Flynn, M.; Deleemans, J.; Piedalue, K.-A.; Tolsdorf, E.; Subnis, U. The role of hypnosis in cancer care. Curr. Oncol. Rep. 2018, 20, 93. [Google Scholar] [CrossRef]
- Salhofer, I.; Will, A.; Monsef, I.; Skoetz, N. Meditation for adults with haematological malignancies. Cochrane Database Syst. Rev. 2016, 2016, CD011157. [Google Scholar] [CrossRef] [PubMed]
- Compernolle, M.C.; Sledge, J.A. Effects of a mindfulness intervention on hospitalized patients with hematologic malignancies and their caregivers. Oncol. Nurs. Forum 2020, 47, 70–78. [Google Scholar] [CrossRef]
- Davidson, C.A.; Kennedy, K.; Jackson, K.T. Trauma-Informed Approaches in the Context of Cancer Care in Canada and the United States: A Scoping Review. Trauma Violence Abus. 2023, 24, 2983–2996. [Google Scholar] [CrossRef] [PubMed]
- Glaser, K.M.; McDaniel, D.C.; Hess, S.M.; Flores, T.F.; Rokitka, D.A.; Reid, M.E. Implementing an integrative survivorship program at a comprehensive cancer center: A multimodal approach to life after cancer. J. Altern. Complement. Med. 2019, 25 (Suppl. S1), S106–S111. [Google Scholar] [CrossRef]
- Miller, K.D.; Pandey, M.; Jain, R.; Mehta, R. Cancer survivorship and models of survivorship care. Am. J. Clin. Oncol. 2015, 38, 627–633. [Google Scholar] [CrossRef]
- Xunlin, N.; Lau, Y.; Klainin-Yobas, P. The effectiveness of mindfulness-based interventions among cancer patients and survivors: A systematic review and meta-analysis. Support. Care Cancer 2020, 28, 1563–1578. [Google Scholar] [CrossRef]
- Liu, X.; Wang, L.; Zhang, Q.; Wang, R.; Xu, W. Less mindful, more struggle and growth. J. Nerv. Ment. Dis. 2018, 206, 621–627. [Google Scholar] [CrossRef] [PubMed]
- El-Jawahri, A.; Leblanc, T.W.; Kavanaugh, A.R.; Webb, J.A.; Jackson, V.; Campbell, T.; O’Connor, N.R.; Luger, S.M.; Gafford, E.; Gustin, J.; et al. Multi-site randomized trial of integrated palliative and oncology care for patients with acute myeloid leukemia (AML). Blood 2020, 136 (Suppl. S1), 26–27. [Google Scholar] [CrossRef]
- Paiva, H.S.; Filho, I.J.Z.; Da Silva Cais, C.F. Using prazosin to treat posttraumatic stress disorder and associations: A systematic review. Psychiatry Investig. 2021, 18, 365–372. [Google Scholar] [CrossRef]
- Mendes, T.P.; Pereira, B.G.; Coutinho, E.S.F.; Melani, M.S.; Neylan, T.C.; Berger, W. Factors impacting prazosin efficacy for nightmares and insomnia in PTSD patients: A systematic review and meta-regression analysis. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2025, 136, 111253. [Google Scholar] [CrossRef]
- Lappas, A.S.; Glarou, E.; Polyzopoulou, Z.A.; Goss, G.; Huhn, M.; Samara, M.T.; Christodoulou, N.G. Pharmacotherapy for sleep disturbances in post-traumatic stress disorder (PTSD): A network meta-analysis. Sleep Med. 2024, 119, 467–479. [Google Scholar] [CrossRef] [PubMed]
- Geldenhuys, C.; van den Heuvel, L.L.; Steyn, P.; Seedat, S. Pharmacological Management of Nightmares Associated with Posttraumatic Stress Disorder. CNS Drugs 2022, 36, 721–737. [Google Scholar] [CrossRef] [PubMed]
- Marchi, M.; Grenzi, P.; Boks, M.P. Clonidine for post-traumatic stress disorder: A systematic review of the current evidence. Eur. J. Psychotraumatol. 2024, 15, 2366049. [Google Scholar] [CrossRef]
- Anderer, S. Combination drug therapy may better alleviate PTSD symptoms than SSRIs alone. JAMA 2025, 333, 559. [Google Scholar] [CrossRef]
- Bajor, L.A.; Balsara, C.; Osser, D.N. An evidence-based approach to psychopharmacology for posttraumatic stress disorder (PTSD): 2022 update. Psychiatry Res. 2022, 317, 114840. [Google Scholar] [CrossRef] [PubMed]
- Geue, K.; Götze, H.; Friedrich, M. Perceived social support and associations with health-related quality of life in young versus older adult patients with haematological malignancies. Health Qual. Life Outcomes 2019, 17, 145. [Google Scholar] [CrossRef]
- Kim, S.H.; Kim, J.Y. Effects of social support on quality of life in cancer patients. Asia Oncol. Nurs. 2022, 22, 29–38. [Google Scholar] [CrossRef]
- Zell, E.; Stockus, C.A. Social support and psychological adjustment: A quantitative synthesis of 60 meta-analyses. Am. Psychol. 2025, 80, 33–46. [Google Scholar] [CrossRef]
- Gianfredi, V.; Beran, M.; Koster, A.; Eussen, S.J.; Odone, A.; Signorelli, C.; Schaper, N.C.; Köhler, S.; Bosma, H.; Dagnelie, P.C.; et al. Association between social network characteristics and prevalent and incident depression: The Maastricht Study. J. Affect. Disord. 2021, 293, 338–346. [Google Scholar] [CrossRef] [PubMed]
- Shahsavar, Y.; Choudhury, A. Examining influential factors in newly diagnosed cancer patients and survivors: Emphasizing distress, self-care ability, peer support, health perception, daily life activity, and the role of time since diagnosis. PLoS ONE 2023, 18, e0291064. [Google Scholar] [CrossRef] [PubMed]

| Serial Number | Study | Design | Sample (n) | Assessment—Scales Used | Timing of Assessment | PTSD Prevalence | Other Observations | Country |
|---|---|---|---|---|---|---|---|---|
| 1 | Prevalence and Positive Correlates of Posttraumatic Stress Disorder Symptoms among Chinese Patients with Hematological Malignancies: A Cross-Sectional Study (2015) [14] | Observational cross-sectional study | 225 | PTSD Checklist-Civilian Version (PCL-C) | Not reported | 10.7% | Optimism was measured as a personality trait in the present study and shown to have a positive effect on PTSD symptoms. | China |
| 2 | Symptoms of posttraumatic stress disorder and adjustment disorder in hematological cancer patients with different treatment regimes (2023) [15] | Observational cross-sectional study | 285 | PCL-C Adjustment Disorder–New Module 20 (ADMN-20) | Note reported | 26 (9.3%) met criteria for PTSD 66 (23.7%) met criteria for subthreshold PTSD 40 (14.2%) met criteria for adjustment disorder (AjD) | Factors associated with elevated symptomatology were younger age, physical comorbidity and active disease | Germany |
| 3 | Posttraumatic stress disorder symptoms in patients with acute myeloid leukemia (2021) [16] | Secondary analysis/supportive care trial | 160 | PCL-C | 1 month after diagnosis | 40 patients—25% | The checklist was used to assess PTSD symptoms at 1 month after AML diagnosis. The Brief COPE and the Functional Assessment of Cancer Therapy-Leukemia were to assess coping and quality of life (QOL), respectively. | United States of America (USA) |
| 4 | Long-term health-related quality of life of critically ill patients with haematological malignancies: a prospective observational multicenter study (2019) [17] | Cross-sectional study | 269 | Impact of Event Scale (IES-R) | Not reported | 22 (8%) patients had an IES score greater than 35 points, which is considered the threshold for PTSD. | The use of an ICU diary in many participating centers may have participated in the low incidence of PTSD as an ICU diary has been associated with a significant reduction in PTSD symptoms in critical illness survivors | France, Belgium |
| 5 | Psychological Distress in Outpatients with Lymphoma During the COVID-19 Pandemic (2020) [18] | Prospective observational study | 77 | IES-R | Not reported | 36% (n. 28) | Study conducted during COVID-19 pandemic | Italy |
| 6 | Posttraumatic Stress Disorder Symptoms in Lymphoma Patients: A Prospective Study (2020) [19] | Prospective observational study | 129 | PCL-C | Not reported | 0 patients met full PTSD criteria 29 (23%) subthreshold PTSD | 13.4% related it to receiving the diagnosis of lymphoma, 8% to telling family members, and 1.6% to adverse effects. | France |
| Serial Number | Study | Design | Sample (n) | Assessment—Scales Used | PTSD Prevalence | Timing of Assessment | Country |
|---|---|---|---|---|---|---|---|
| 1 | Post-Traumatic Stress Symptoms in Hematopoietic Stem Cell Transplant Recipients (2021) [21] | Secondary analysis of randomized controlled trial | 206 | PCL-C | 18.9% (39) | 6 months after transplantation | USA |
| 2 | Quality of life and mood predict posttraumatic stress disorder after hematopoietic stem cell transplantation (2016) [22] | Prospective observational study | 90 | PCL-C | At 6 months, 28.4% of participants met the criteria for PTSD | Data at 6 months were available for 67 participants. | USA |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Buciuc, A.G.; Espinel, Z.; Weber, M.; Tran, S.; Rueda-Lara, M. Post-Traumatic Stress and Stressor-Related Disorders in Hematological Malignancies: A Review. J. Clin. Med. 2025, 14, 6132. https://doi.org/10.3390/jcm14176132
Buciuc AG, Espinel Z, Weber M, Tran S, Rueda-Lara M. Post-Traumatic Stress and Stressor-Related Disorders in Hematological Malignancies: A Review. Journal of Clinical Medicine. 2025; 14(17):6132. https://doi.org/10.3390/jcm14176132
Chicago/Turabian StyleBuciuc, Adela Georgiana, Zelde Espinel, Mary Weber, Sabrina Tran, and Maria Rueda-Lara. 2025. "Post-Traumatic Stress and Stressor-Related Disorders in Hematological Malignancies: A Review" Journal of Clinical Medicine 14, no. 17: 6132. https://doi.org/10.3390/jcm14176132
APA StyleBuciuc, A. G., Espinel, Z., Weber, M., Tran, S., & Rueda-Lara, M. (2025). Post-Traumatic Stress and Stressor-Related Disorders in Hematological Malignancies: A Review. Journal of Clinical Medicine, 14(17), 6132. https://doi.org/10.3390/jcm14176132






