Current Status and Future Perspectives of Superior Mesenteric Artery Dissection in Robotic Pancreaticoduodenectomy: A Scoping Review of Technical Variations in the Robotic Era
Abstract
1. Introduction
2. Methods
2.1. Search Strategy
2.2. Eligibility Criteria
- Addressed technical aspects of SMA dissection during robotic pancreaticoduodenectomy.
- Included original data (case series, cohort studies, or technical reports).
- Were published in peer-reviewed English journals.
- Focused exclusively on open or laparoscopic pancreaticoduodenectomy.
- Did not describe the SMA dissection technique in sufficient detail.
- Were editorials, commentaries, conference abstracts, or non-English articles.
2.3. Selection Process
2.4. Data Extraction and Synthesis
- Type and name of the SMA approach (e.g., right, left, posterior, anterior).
- Procedural details and scope position.
- Technical tips regarding retraction when available.
- Robotic platform used.
2.5. PRISMA-ScR Compliance
3. Results
- -
- Exclusive focus on open or laparoscopic pancreaticoduodenectomy (n = 1);
- -
- Insufficient description of the SMA dissection technique (n = 7);
- -
- Overlapping reports from the same institution within the same timeframe (n = 2)
4. Types of SMA Approach During PD
5. Right-Sided SMA Approach During RPD
5.1. Conventional Right-Sided Approach in RPD
5.2. Right Posterior Approach in RPD
5.3. Right Lateral Approach in RPD
5.4. Extended Indications of Right-Sided Approaches in RPD
6. Left-Sided SMA Approach During RPD
6.1. Left-Posterior Approach in RPD
6.2. Normal Left-Sided Approaches in RPD
6.3. Extended Indications of Left-Sided Approaches in RPD
7. Other SMA Approach During RPD
8. Technical Issues Associated with Right- and Left-Sided Approaches
8.1. Strengths and Limitations of the Right-Sided Approach
8.2. Strengths and Limitations of the Left-Sided Approach
9. Future Perspectives
10. Limitations
11. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| PD | pancreaticoduodenectomy |
| RPD | robotic pancreaticoduodenectomy |
| SMA | superior mesenteric artery |
| OPD | open pancreaticoduodenectomy |
| SMV | superior mesenteric vein |
| LPD | laparoscopic pancreaticoduodenectomy |
| ST | scope transition |
| JA | jejunal artery |
| IPDA | inferior pancreaticoduodenal artery |
References
- Napoli, N.; Cacace, C.; Kauffmann, E.F.; Jones, L.; Ginesini, M.; Gianfaldoni, C.; Salamone, A.; Asta, F.; Ripolli, A.; Di Dato, A.; et al. The PD-ROBOSCORE: A difficulty score for robotic pancreatoduodenectomy. Surgery 2023, 173, 1438–1446. [Google Scholar] [CrossRef] [PubMed]
- Zureikat, A.H.; Beane, J.D.; Zenati, M.S.; Al Abbas, A.I.; Boone, B.A.; Moser, A.J.; Bartlett, D.L.; Hogg, M.E.; Zeh, H.J., 3rd. 500 Minimally Invasive Robotic Pancreatoduodenectomies: One Decade of Optimizing Performance. Ann. Surg. 2021, 273, 966–972. [Google Scholar] [CrossRef] [PubMed]
- Tol, J.A.; Gouma, D.J.; Bassi, C.; Dervenis, C.; Montorsi, M.; Adham, M.; Andrén-Sandberg, A.; Asbun, H.J.; Bockhorn, M.; Büchler, M.W.; et al. Definition of a standard lymphadenectomy in surgery for pancreatic ductal adenocarcinoma: A consensus statement by the International Study Group on Pancreatic Surgery (ISGPS). Surgery 2014, 156, 591–600. [Google Scholar] [CrossRef] [PubMed]
- Sanjay, P.; Takaori, K.; Govil, S.; Shrikhande, S.V.; Windsor, J.A. ‘Artery-first’ approaches to pancreatoduodenectomy. Br. J. Surg. 2012, 99, 1027–1035. [Google Scholar] [CrossRef] [PubMed]
- Tricco, A.C.; Lillie, E.; Zarin, W.; O’Brien, K.K.; Colquhoun, H.; Levac, D.; Moher, D.; Peters, M.D.J.; Horsley, T.; Weeks, L.; et al. PRISMA Extension for Scoping Reviews (PRISMA-ScR): Checklist and Explanation. Ann. Intern. Med. 2018, 169, 467–473. [Google Scholar] [CrossRef]
- Ohigashi, H.; Ishikawa, O.; Eguchi, H.; Yamada, T.; Sasaki, Y.; Noura, S.; Takachi, K.; Miyashiro, I.; Murata, K.; Doki, Y.; et al. Early ligation of the inferior pancreaticoduodenal artery to reduce blood loss during pancreaticoduodenectomy. Hepatogastroenterology 2004, 51, 4–5. [Google Scholar]
- Horiguchi, A.; Ishihara, S.; Ito, M.; Nagata, H.; Shimizu, T.; Furusawa, K.; Kato, R.; Katada, K.; Miyakawa, S. Pancreatoduodenectomy in which dissection of the efferent arteries of the head of the pancreas is performed first. J. Hepatobiliary Pancreat. Surg. 2007, 14, 575–578. [Google Scholar] [CrossRef]
- Kurosaki, I.; Minagawa, M.; Takano, K.; Takizawa, K.; Hatakeyama, K. Left posterior approach to the superior mesenteric vascular pedicle in pancreaticoduodenectomy for cancer of the pancreatic head. JOP 2011, 12, 220–229. [Google Scholar]
- Nagakawa, Y.; Hosokawa, Y.; Osakabe, H.; Sahara, Y.; Takishita, C.; Nakajima, T.; Hijikata, Y.; Kasahara, K.; Kazuhiko, K.; Saito, K.; et al. Pancreaticoduodenectomy with right-oblique posterior dissection of superior mesenteric nerve plexus is logical procedure for pancreatic cancer with extrapancreatic nerve plexus invasion. Hepatogastroenterology 2014, 61, 2371–2376. [Google Scholar]
- Nagakawa, Y.; Watanabe, Y.; Kozono, S.; Boggi, U.; Palanivelu, C.; Liu, R.; Wang, S.E.; He, J.; Nishino, H.; Ohtsuka, T.; et al. Surgical approaches to the superior mesenteric artery during minimally invasive pancreaticoduodenectomy: A systematic review. J. Hepato-Biliary-Pancreat. Sci. 2022, 29, 114–123. [Google Scholar] [CrossRef]
- Giulianotti, P.C.; Sbrana, F.; Bianco, F.M.; Elli, E.F.; Shah, G.; Addeo, P.; Caravaglios, G.; Coratti, A. Robot-assisted laparoscopic pancreatic surgery: Single-surgeon experience. Surg. Endosc. 2010, 24, 1646–1657. [Google Scholar] [CrossRef]
- Nguyen, K.T.; Zureikat, A.H.; Chalikonda, S.; Bartlett, D.L.; Moser, A.J.; Zeh, H.J. Technical aspects of robotic-assisted pancreaticoduodenectomy (RAPD). J. Gastrointest. Surg. 2011, 15, 870–875. [Google Scholar] [CrossRef] [PubMed]
- Boggi, U.; Signori, S.; De Lio, N.; Perrone, V.G.; Vistoli, F.; Belluomini, M.; Cappelli, C.; Amorese, G.; Mosca, F. Feasibility of robotic pancreaticoduodenectomy. Br. J. Surg. 2013, 100, 917–925. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Chen, J.Z.; Zhan, Q.; Deng, X.X.; Shen, B.Y.; Peng, C.H.; Li, H.W. Robot-assisted laparoscopic versus open pancreaticoduodenectomy: A prospective, matched, mid-term follow-up study. Surg. Endosc. 2015, 29, 3698–3711. [Google Scholar] [CrossRef] [PubMed]
- Memeo, R.; De Blasi, V.; Perotto, O.; Mutter, D.; Marescaux, J.; Pessaux, P. Robotic Lymphadenectomy During Pancreatoduodenectomy with First Superior Mesenteric Artery Dissection. Ann. Surg. Oncol. 2016, 23, 968. [Google Scholar] [CrossRef]
- Liu, R.; Zhang, T.; Zhao, Z.M.; Tan, X.L.; Zhao, G.D.; Zhang, X.; Xu, Y. The surgical outcomes of robot-assisted laparoscopic pancreaticoduodenectomy versus laparoscopic pancreaticoduodenectomy for periampullary neoplasms: A comparative study of a single center. Surg. Endosc. 2017, 31, 2380–2386. [Google Scholar] [CrossRef]
- Marino, M.; Gulotta, G.; Komorowski, A.L. Robotic Pancreaticoduodenectomy: Technical Considerations. Indian J. Surg. 2018, 80, 118–122. [Google Scholar] [CrossRef]
- Goh, B.K.P.; Low, T.Y.; Lee, S.Y.; Chan, C.Y.; Chung, A.Y.F.; Ooi, L. Initial experience with robotic pancreatic surgery in Singapore: Single institution experience with 30 consecutive cases. ANZ J. Surg. 2019, 89, 206–210. [Google Scholar] [CrossRef]
- Ikoma, N.; Kim, M.P.; Tzeng, C.D.; Tran Cao, H.S.; Lee, J.E.; Katz, M.H.G. External Retraction Technique for Robotic Pancreatoduodenectomy. J. Am. Coll. Surg. 2020, 231, e8–e10. [Google Scholar] [CrossRef]
- Machado, M.A.; Mattos, B.V.; Lobo Filho, M.M.; Makdissi, F.F. Robotic Artery-First Approach During Pancreatoduodenectomy. Ann. Surg. Oncol. 2021, 28, 6257–6261. [Google Scholar] [CrossRef]
- Azagra, J.S.; Rosso, E.; Pascotto, B.; de Blasi, V.; Henrard, A.; González González, L. Real robotic total mesopancreas excision (TMpE) assisted by hanging manoeuver (HM): Standardised technique. Int. J. Med. Robot. Comput. Assist. Surg. MRCAS 2021, 17, e2259. [Google Scholar] [CrossRef]
- Kinny-Köster, B.; Habib, J.R.; Javed, A.A.; Shoucair, S.; van Oosten, A.F.; Fishman, E.K.; Lafaro, K.J.; Wolfgang, C.L.; Hackert, T.; He, J. Technical progress in robotic pancreatoduodenectomy: TRIANGLE and periadventitial dissection for retropancreatic nerve plexus resection. Langenbeck’s Arch. Surg. 2021, 406, 2527–2534. [Google Scholar] [CrossRef] [PubMed]
- Shyr, B.U.; Shyr, B.S.; Chen, S.C.; Shyr, Y.M.; Wang, S.E. Mesopancreas level 3 dissection in robotic pancreaticoduodenectomy. Surgery 2021, 169, 362–368. [Google Scholar] [CrossRef]
- Takagi, K.; Umeda, Y.; Yoshida, R.; Fuji, T.; Yasui, K.; Kimura, J.; Hata, N.; Mishima, K.; Yagi, T.; Fujiwara, T. Surgical Strategies to Dissect around the Superior Mesenteric Artery in Robotic Pancreatoduodenectomy. J. Clin. Med. 2022, 11, 7112. [Google Scholar] [CrossRef]
- Kauffmann, E.F.; Napoli, N.; Ginesini, M.; Gianfaldoni, C.; Asta, F.; Salamone, A.; Amorese, G.; Vistoli, F.; Boggi, U. Feasibility of “cold” triangle robotic pancreatoduodenectomy. Surg. Endosc. 2022, 36, 9424–9434. [Google Scholar] [CrossRef]
- Wei, Z.G.; Liang, C.J.; Du, Y.; Zhang, Y.P.; Liu, Y. Learning curve for a surgeon in robotic pancreaticoduodenectomy through a “G”-shaped approach: A cumulative sum analysis. World J. Clin. Cases 2022, 10, 4357–4367. [Google Scholar] [CrossRef] [PubMed]
- Zwart, M.J.W.; Jones, L.R.; Hogg, M.E.; Tol, J.; Abu Hilal, M.; Daams, F.; Festen, S.; Busch, O.R.; Besselink, M.G. Robotic Pancreatoduodenectomy for Pancreatic Head Cancer: A Case Report of a Standardized Technique. J. Vis. Exp. JoVE 2022, 184, e62863. [Google Scholar] [CrossRef]
- Nakata, K.; Abe, T.; Ideno, N.; Nakamura, S.; Ikenaga, N.; Nagayoshi, K.; Mizuuchi, Y.; Moriyama, T.; Ohuchida, K.; Nakamura, M. A left-sided approach for wide mobilization of the pancreas with complete dissection of the Treitz ligament (with video). Surg. Endosc. 2023, 37, 4982–4989. [Google Scholar] [CrossRef] [PubMed]
- Ninomiya, R.; Komagome, M.; Abe, S.; Chiyoda, T.; Kogure, R.; Kimura, A.; Maki, A.; Beck, Y. Right lateral approach to the superior mesenteric artery in robotic pancreaticoduodenectomy. J. Hepato-Biliary-Pancreat. Sci. 2023, 30, e73–e74. [Google Scholar] [CrossRef]
- Torres, M.B.; Maxwell, J.E.; Snyder, R.A.; Cao, H.S.T.; Kim, M.P.; Tzeng, C.D.; Lee, J.E.; Katz, M.H.G.; Ikoma, N. Total Venous Control and Vein-to-the-Right Superior Mesenteric Artery Approach in Robotic Pancreatoduodenectomy. Ann. Surg. Oncol. 2024, 31, 8721–8722. [Google Scholar] [CrossRef]
- Takagi, K.; Yamada, M.; Umeda, Y. Left posterior superior mesenteric artery approach using a hanging maneuver in robotic pancreaticoduodenectomy. J. Gastrointest. Surg. 2024, 28, 786–788. [Google Scholar] [CrossRef] [PubMed]
- Takagi, K.; Fuji, T.; Yasui, K.; Yamada, M.; Nishiyama, T.; Nagai, Y.; Kanehira, N.; Fujiwara, T. Robot-Assisted Pancreaticoduodenectomy Using the Anterior Superior Mesenteric Artery-First Approach for Pancreatic Cancer. Ann. Surg. Oncol. 2024, 31, 8741–8743. [Google Scholar] [CrossRef] [PubMed]
- Bhandare, M.S.; Varty, G.P.; Chaudhari, V.A.; Shrikhande, S.V. Robotic Pancreaticoduodenectomy with ‘SMA-First Approach (Posterior and Right Medial)’ and ‘Triangle Operation’. Ann. Surg. Oncol. 2024, 31, 4112. [Google Scholar] [CrossRef]
- Mizumoto, T.; Takahara, T.; Nishimura, A.; Mii, S.; Uchida, Y.; Iwama, H.; Kojima, M.; Kato, Y.; Uyama, I.; Suda, K. Challenge in optimizing robotic pancreaticoduodenectomy including nerve plexus hanging maneuver: A Japanese single center experience of 76 cases. Surg. Endosc. 2024, 38, 1077–1087. [Google Scholar] [CrossRef]
- Mourthadhoi, F.; Le Roy, B.; Blanc, P.Y. Robotic minimally invasive duodenopancreatectomy with superior mesenteric artery approach (with video). J. Visc. Surg. 2025, 162, 157–158. [Google Scholar] [CrossRef]
- Kiritani, S.; Inoue, Y.; Sato, T.; Sawa, Y.; Kobayashi, K.; Oba, A.; Ono, Y.; Ito, H.; Takahashi, Y. A left-posterior approach to the superior mesenteric artery during robot-assisted pancreaticoduodenectomy. J. Hepato-Biliary-Pancreat. Sci. 2025, 32, 317–326. [Google Scholar] [CrossRef]
- Inoue, Y.; Saiura, A.; Yoshioka, R.; Ono, Y.; Takahashi, M.; Arita, J.; Takahashi, Y.; Koga, R. Pancreatoduodenectomy With Systematic Mesopancreas Dissection Using a Supracolic Anterior Artery-first Approach. Ann. Surg. 2015, 262, 1092–1101. [Google Scholar] [CrossRef]
- Hackert, T.; Strobel, O.; Michalski, C.W.; Mihaljevic, A.L.; Mehrabi, A.; Muller-Stich, B.; Berchtold, C.; Ulrich, A.; Buchler, M.W. The TRIANGLE operation—Radical surgery after neoadjuvant treatment for advanced pancreatic cancer: A single arm observational study. HPB Off. J. Int. Hepato Pancreato Biliary Assoc. 2017, 19, 1001–1007. [Google Scholar] [CrossRef] [PubMed]
- Kawabata, Y.; Tanaka, T.; Nishi, T.; Monma, H.; Yano, S.; Tajima, Y. Appraisal of a total meso-pancreatoduodenum excision with pancreaticoduodenectomy for pancreatic head carcinoma. Eur. J. Surg. Oncol. 2012, 38, 574–579. [Google Scholar] [CrossRef]
- Inoue, Y.; Sato, T.; Kato, T.; Oba, A.; Ono, Y.; Ito, H.; Makuuchi, R.; Takahashi, Y. How Can We Optimize Surgical View During Robotic-Assisted Pancreaticoduodenectomy? Feasibility of Multiple Scope Transition Method. J. Am. Coll. Surg. 2022, 235, e1–e7. [Google Scholar] [CrossRef]
- Kitano, Y.; Inoue, Y.; Kobayashi, H.; Kobayashi, K.; Oba, A.; Ono, Y.; Sato, T.; Ito, H.; Takahashi, Y. Development of the multiple scope transition method in robotic pancreaticoduodenectomy. Surg. Endosc. 2024, 38, 6169–6176. [Google Scholar] [CrossRef] [PubMed]
- Omiya, K.; Inoue, Y.; Kobayashi, K.; Oba, A.; Ono, Y.; Sato, T.; Ito, H.; Takahashi, Y. Robot-Assisted Pancreaticoduodenectomy with Hemicircumferential Dissection of Nerve Plexus Around the Superior Mesenteric Artery. Ann. Surg. Oncol. 2024, 31, 7064–7065. [Google Scholar] [CrossRef]
- Kobayashi, K.; Inoue, Y.; Takahashi, Y. Efficacy of Robot-assisted Pancreaticoduodenectomy with Left Posterior Approach for Pancreas Head Cancer with Venous Invasion. J. Gastrointest. Surg. 2025, 29, 101996. [Google Scholar] [CrossRef]
- Ikoma, N.; Seo, Y.D.; Newhook, T.E.; Maxwell, J.E.; Kim, M.P.; Tran Cao, H.S.; Tzeng, C.D.; Chun, Y.S.; Lee, J.E.; Vauthey, J.N.; et al. Quality of superior mesenteric and hepatic artery dissection in robotic pancreatoduodenectomy for pancreatic cancer. J. Hepato-Biliary-Pancreat. Sci. 2023, 30, e1–e2. [Google Scholar] [CrossRef]
- Kiguchi, G.; Sugioka, A.; Kato, Y.; Uyama, I. Use of a novel semi-derotation technique for artery-first approach in laparoscopic pancreaticoduodenectomy. Surg. Oncol. 2020, 33, 141–142. [Google Scholar] [CrossRef]
- Irie, S.; Inoue, Y.; Oba, A.; Ono, Y.; Sato, T.; Mise, Y.; Ito, H.; Saiura, A.; Takahashi, Y. Technical Guidelines for Safe Mesojejunum Dissection During Pancreaticoduodenectomy: Unveiling Critical Techniques in a Complex Procedure. Ann. Surg. Oncol. 2024, 32, 1850–1857. [Google Scholar] [CrossRef]
- Klotz, R.; Mihaljevic, A.L.; Kulu, Y.; Sander, A.; Klose, C.; Behnisch, R.; Joos, M.C.; Kalkum, E.; Nickel, F.; Knebel, P.; et al. Robotic versus open partial pancreatoduodenectomy (EUROPA): A randomised controlled stage 2b trial. Lancet Reg. Health Eur. 2024, 39, 100864. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Zhao, Z.; Zhang, X.; Wang, W.; Han, B.; Chen, X.; Tan, X.; Xu, S.; Zhao, G.; Gao, Y.; et al. Perioperative and Oncological Outcomes of Robotic Versus Open Pancreaticoduodenectomy in Low-Risk Surgical Candidates: A Multicenter Propensity Score-Matched Study. Ann. Surg. 2023, 277, e864–e871. [Google Scholar] [CrossRef] [PubMed]
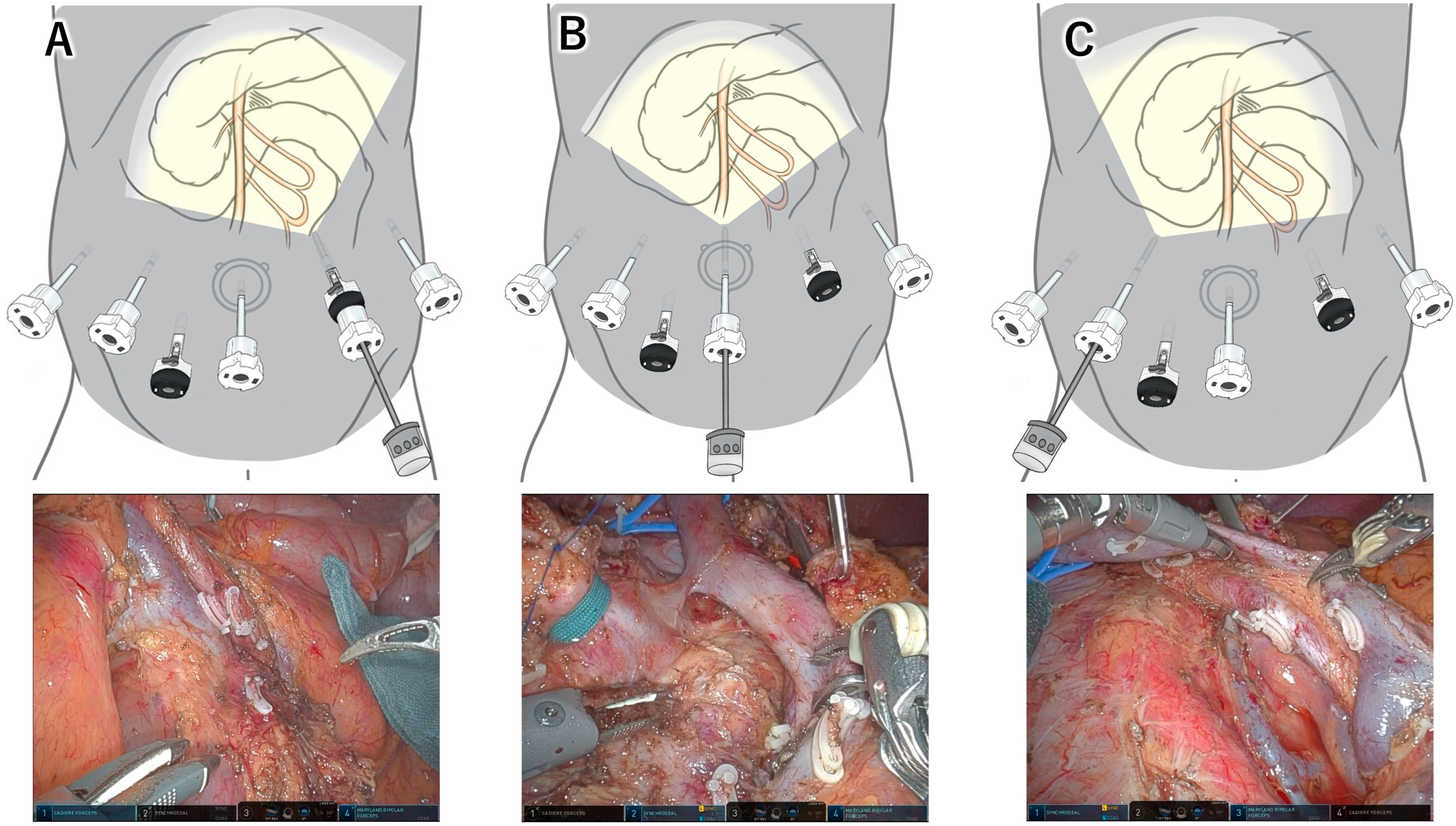
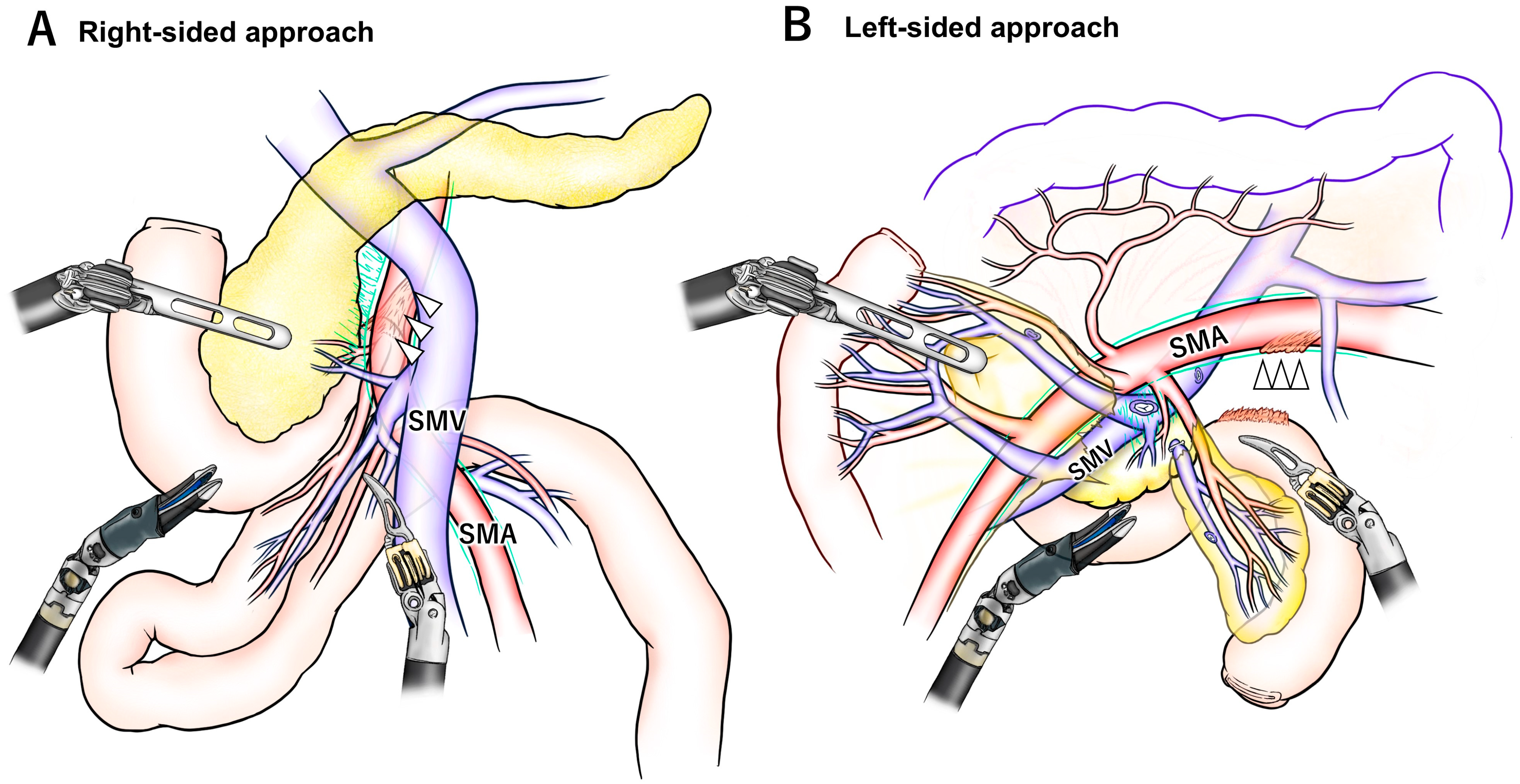
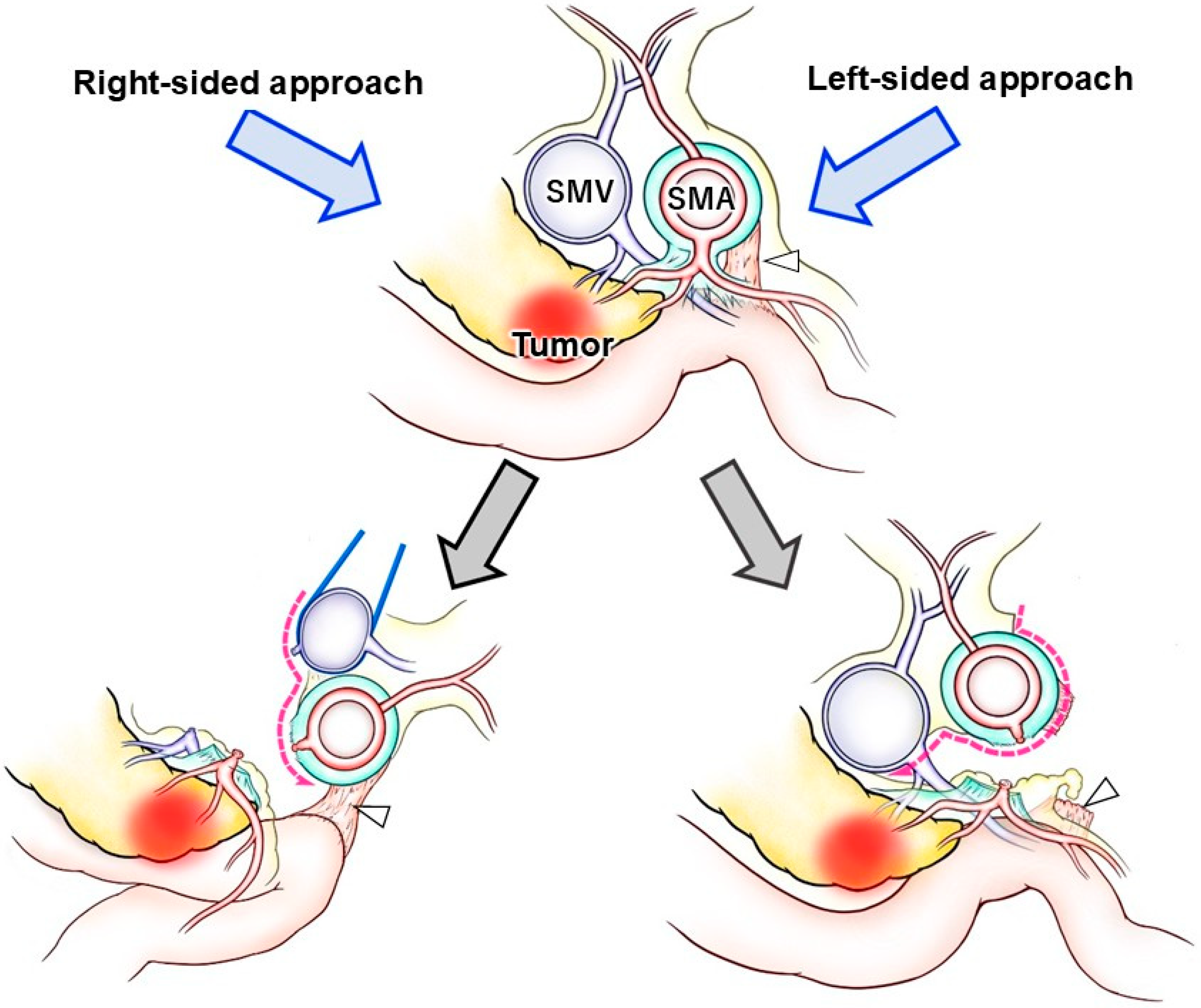
| Author | Year | Article Type | N | da Vinci Type | SMA Approach | Combination | Scope Position |
|---|---|---|---|---|---|---|---|
| Giulianotti [11] | 2010 | Retrospective analysis | 50 | Standard/S | Right | ND | Right |
| Nguyen [12] | 2011 | Case report | 1 | S | Right | ND | Midline |
| Boggi [13] | 2013 | Retrospective analysis | 34 | S/Si | Right | ND | Right |
| Chen [14] | 2015 | Retrospective analysis | 60 | S | Right | ND | Midline |
| Memeo [15] | 2016 | Case report | 1 | Si | Right posterior | ND | Midline/right |
| Liu [16] | 2017 | Retrospective analysis | 27 | S | Right | ND | Midline |
| Marino [17] | 2018 | Retrospective analysis | 8 | Si | Right | ND | Midline |
| Goh [18] | 2019 | Retrospective analysis | 8 | Si | Right | ND | Midline |
| Ikoma [19] | 2020 | Case report | 1 | Xi | Right | ND | Right |
| Machado [20] | 2021 | Retrospective analysis | 73 | Si/Xi | Right posterior | ND | Right |
| Azagra [21] | 2021 | Case report | 1 | Xi | Right posterior | ND | Right |
| Kinny-Köster [22] | 2021 | Case report | 3 | Xi | Right | ND | Right |
| Shyr [23] | 2021 | Retrospective analysis | 289 | Si/Xi | Right | ND | Midline/Right |
| Inoue [22] | 2022 | Retrospective analysis | 14 | Xi | Left posterior | Left & right | Left/Midline/Right |
| Takagi [24] | 2022 | Case report | 1 | Si | Left | Left & right | Right |
| Kaufman [25] | 2022 | Retrospective analysis | 127 | Si/Xi | Right posterior | ND | Right |
| Wei [26] | 2022 | Retrospective analysis | 60 | Si | Right | ND | Midline |
| Zwart [27] | 2022 | Case report | 1 | Xi | Right | ND | Right |
| Nakata [28] | 2023 | Retrospective analysis | 43 | Xi | Left | Left & right | Midline |
| Ninomiya [29] | 2023 | Retrospective analysis | 9 | Xi | Right lateral | ND | Right |
| Torres [30] | 2024 | Case report | 1 | Xi | Right | ND | Right |
| Takagi [31] | 2024 | Case report | 1 | Xi | Left posterior | Left & right | Midline |
| Takagi [32] | 2024 | Case report | 1 | Xi | Anterior | Left & Anterior | Midline |
| Bhandare [33] | 2024 | Case report | 1 | Xi | Right posterior | ND | Right |
| Mizumoto [34] | 2024 | Retrospective analysis | 73 | Si/Xi | Right | ND | Midline |
| Mourthadhoi [35] | 2025 | Case report | 1 | Xi | Right posterior | ND | Right |
| Kiritani [36] | 2025 | Retrospective analysis | 83 | Xi | Left posterior | Left & right | Left/Midline/Right |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Inoue, Y.; Kobayashi, K.; Kato, T.; Kiritani, S.; Oba, A.; Ono, Y.; Ito, H.; Takahashi, Y. Current Status and Future Perspectives of Superior Mesenteric Artery Dissection in Robotic Pancreaticoduodenectomy: A Scoping Review of Technical Variations in the Robotic Era. J. Clin. Med. 2025, 14, 6084. https://doi.org/10.3390/jcm14176084
Inoue Y, Kobayashi K, Kato T, Kiritani S, Oba A, Ono Y, Ito H, Takahashi Y. Current Status and Future Perspectives of Superior Mesenteric Artery Dissection in Robotic Pancreaticoduodenectomy: A Scoping Review of Technical Variations in the Robotic Era. Journal of Clinical Medicine. 2025; 14(17):6084. https://doi.org/10.3390/jcm14176084
Chicago/Turabian StyleInoue, Yosuke, Kosuke Kobayashi, Tomotaka Kato, Sho Kiritani, Atsushi Oba, Yoshihiro Ono, Hiromichi Ito, and Yu Takahashi. 2025. "Current Status and Future Perspectives of Superior Mesenteric Artery Dissection in Robotic Pancreaticoduodenectomy: A Scoping Review of Technical Variations in the Robotic Era" Journal of Clinical Medicine 14, no. 17: 6084. https://doi.org/10.3390/jcm14176084
APA StyleInoue, Y., Kobayashi, K., Kato, T., Kiritani, S., Oba, A., Ono, Y., Ito, H., & Takahashi, Y. (2025). Current Status and Future Perspectives of Superior Mesenteric Artery Dissection in Robotic Pancreaticoduodenectomy: A Scoping Review of Technical Variations in the Robotic Era. Journal of Clinical Medicine, 14(17), 6084. https://doi.org/10.3390/jcm14176084






