May Spasticity-Related Unpleasant Sensations Interfere with Daily Activities in People with Stroke and Traumatic Brain Injury? Secondary Analysis from the CORTOX Study
Abstract
1. Introduction
2. Materials and Methods
Analysis of Data
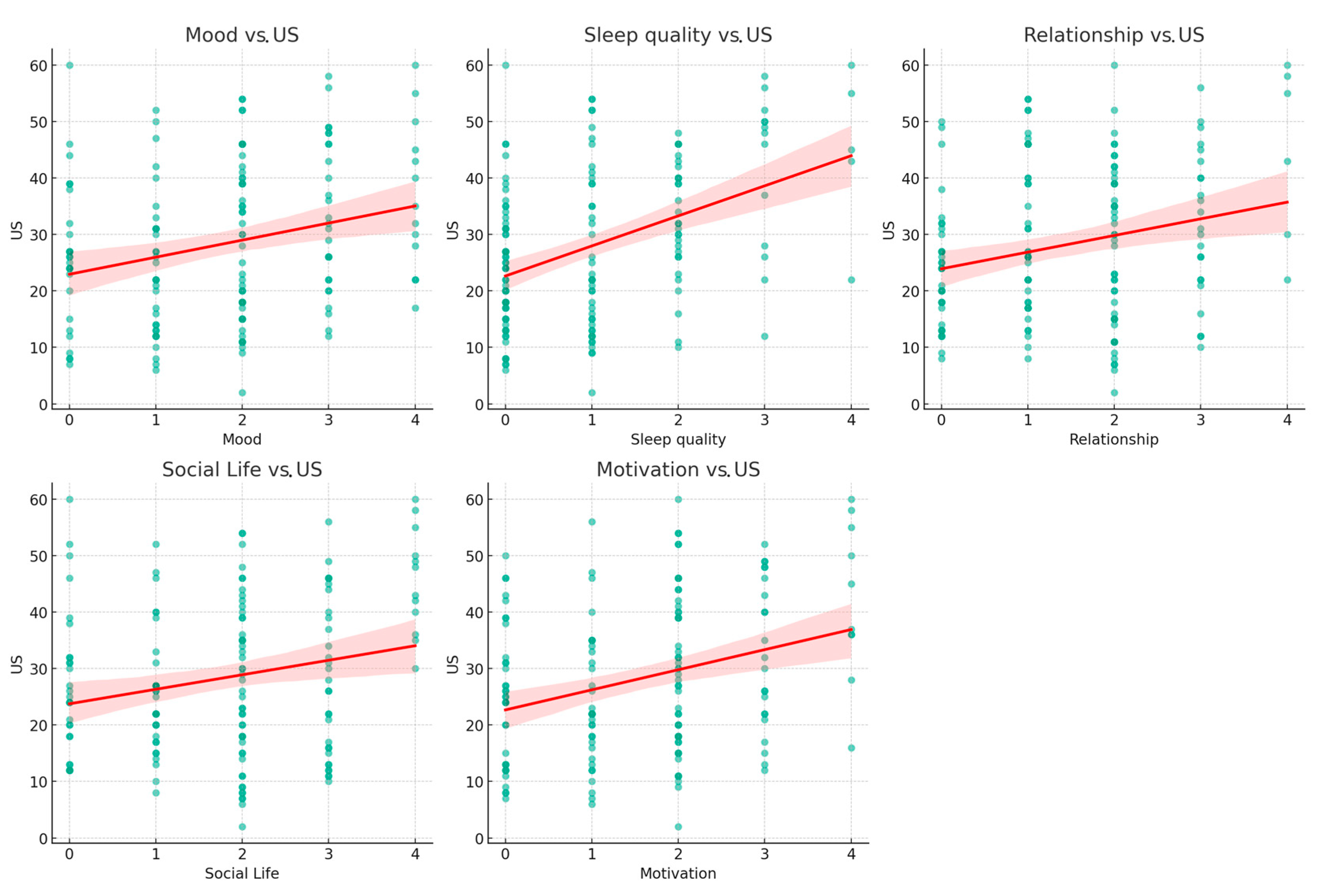
3. Results
4. Discussion
Study Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Trompetto, C.; Marinelli, L.; Mori, L.; Pelosin, E.; Currà, A.; Molfetta, L.; Abbruzzese, G. Pathophysiology of Spasticity: Implications for Neurorehabilitation. Biomed. Res. Int. 2014, 2014, 354906. [Google Scholar] [CrossRef]
- Watkins, C.L.; Leathley, M.J.; Gregson, J.M.; Moore, A.P.; Smith, T.L.; Sharma, A.K. Prevalence of Spasticity Post Stroke. Clin. Rehabil. 2002, 16, 515–522. [Google Scholar] [CrossRef]
- Dragojlovic, N.; Romanoski, N.L.; Verduzco-Gutierrez, M.; Francisco, G.E. Prevalence and Treatment Characteristics of Spastic Hypertonia on First-Time Admission to Acute Inpatient Rehabilitation. Am. J. Phys. Med. Rehabil. 2022, 101, 348–352. [Google Scholar] [CrossRef] [PubMed]
- Goldman, L.; Siddiqui, E.M.; Khan, A.; Jahan, S.; Rehman, M.U.; Mehan, S.; Sharma, R.; Budkin, S.; Kumar, S.N.; Sahu, A.; et al. Understanding Acquired Brain Injury: A Review. Biomedicines 2022, 10, 2167. [Google Scholar] [CrossRef]
- Qin, C.; Yang, S.; Chu, Y.H.; Zhang, H.; Pang, X.W.; Chen, L.; Zhou, L.Q.; Chen, M.; Tian, D.S.; Wang, W. Signaling Pathways Involved in Ischemic Stroke: Molecular Mechanisms and Therapeutic Interventions. Signal Transduct. Target. Ther. 2022, 7, 215. [Google Scholar] [CrossRef]
- Chang, E.; Ghosh, N.; Yanni, D.; Lee, S.; Alexandru, D.; Mozaffar, T. A Review of Spasticity Treatments: Pharmacological and Interventional Approaches. Crit. Rev. Phys. Rehabil. Med. 2013, 25, 11. [Google Scholar] [CrossRef]
- O’Dwyer, N.J.; Ada, L.; Neilson, P.D. Spasticity and Muscle Contracture Following Stroke. Brain 1996, 119, 1737–1749. [Google Scholar] [CrossRef]
- Khan, F.; Amatya, B.; Bensmail, D.; Yelnik, A. Non-Pharmacological Interventions for Spasticity in Adults: An Overview of Systematic Reviews. Ann. Phys. Rehabil. Med. 2019, 62, 265–273. [Google Scholar] [CrossRef]
- Crowe, C.S.; Pino, P.A.; Rhee, P.C.; Boyce, D.; Fridén, J.; Horwitz, M.; Lam, W.L.; Leclercq, C.; Luria, S.; McArthur, P.; et al. Expert Consensus on the Surgical Evaluation and Management of Upper Extremity Spasticity in Adults. J. Hand Surg. Eur. Vol. 2023, 48, 986–997. [Google Scholar] [CrossRef]
- Salem, S.M.; El-Saadany, W.F.; Fouad, W.A.; Abdel Ghany, W.A. Evaluation of Selective Peripheral Neurotomies in the Treatment of Refractory Lower Limb Spasticity in Adults. Alex. J. Med. 2018, 54, 229–233. [Google Scholar] [CrossRef]
- Renzenbrink, G.J.; Buurke, J.H.; Nene, A.V.; Geurts, A.C.H.; Kwakkel, G.; Rietman, J.S. Improving Walking Capacity by Surgical Correction of Equinovarus Foot Deformity in Adult Patients with Stroke or Traumatic Brain Injury: A Systematic Review. J. Rehabil. Med. 2012, 44, 614–623. [Google Scholar] [CrossRef]
- Lance, J. Symposium Synopsis. In Spasticity: Disordered Motor Control Chicago; Felman, R.G., Young, R.R., Koella, W.P., Eds.; Book Medic; Symposia Specialists: Sydney, NSW, Australia, 1980; pp. 485–494. [Google Scholar]
- Gracies, J.M. Pathophysiology of Spastic Paresis. II: Emergence of Muscle Overactivity. Muscle Nerve 2005, 31, 552–571. [Google Scholar] [CrossRef]
- Pandyan, A.D.; Gregoric, M.; Barnes, M.P.; Wood, D.; Van Wijck, F.; Burridge, J.; Hermens, H.; Johnson, G.R. Spasticity: Clinical Perceptions, Neurological Realities and Meaningful Measurement. Disabil. Rehabil. 2005, 27, 2–6. [Google Scholar] [CrossRef]
- Lv, Q.; Zhang, J.; Pan, Y.; Liu, X.; Miao, L.; Peng, J.; Song, L.; Zou, Y.; Chen, X. Somatosensory Deficits After Stroke: Insights from MRI Studies. Front. Neurol. 2022, 13, 891283. [Google Scholar] [CrossRef]
- Craig, A.D. Interoception: The Sense of the Physiological Condition of the Body. Curr. Opin. Neurobiol. 2003, 13, 500–505. [Google Scholar] [CrossRef]
- Khalsa, S.S.; Adolphs, R.; Cameron, O.G.; Critchley, H.D.; Davenport, P.W.; Feinstein, J.S.; Feusner, J.D.; Garfinkel, S.N.; Lane, R.D.; Mehling, W.E.; et al. Interoception and Mental Health: A Roadmap. Biol. Psychiatry Cogn. Neurosci. Neuroimaging 2018, 3, 501–513. [Google Scholar] [CrossRef]
- Critchley, H.D.; Garfinkel, S.N. Interoception and Emotion. Curr. Opin. Psychol. 2017, 17, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Wallman-Jones, A.; Perakakis, P.; Tsakiris, M.; Schmidt, M. Physical Activity and Interoceptive Processing: Theoretical Considerations for Future Research. Int. J. Psychophysiol. 2021, 166, 38–49. [Google Scholar] [CrossRef] [PubMed]
- Craig, B. Interoception and Emotion: A Neuroanatomical Perspective. In Handbook of Emotion, 3rd ed.; Guilford Press: New York, NY, USA, 2008; Volume 6, pp. 272–290. [Google Scholar]
- Quadt, L.; Critchley, H.D.; Garfinkel, S.N. The Neurobiology of Interoception in Health and Disease. Ann. N. Y Acad. Sci. 2018, 1428, 11–14. [Google Scholar] [CrossRef] [PubMed]
- Critchley, H.D. The Human Cortex Responds to an Interoceptive Challenge. Proc. Natl. Acad. Sci. USA 2004, 101, 6333–6334. [Google Scholar] [CrossRef] [PubMed]
- Bassolino, M.; Franza, M.; Guanziroli, E.; Sorrentino, G.; Canzoneri, E.; Colombo, M.; Crema, A.; Bertoni, T.; Mastria, G.; Vissani, M.; et al. Body and Peripersonal Space Representations in Chronic Stroke Patients with Upper Limb Motor Deficits. Brain Commun. 2022, 4, fcac179. [Google Scholar] [CrossRef]
- Baricich, A.; Picelli, A.; Molteni, F.; Guanziroli, E.; Santamato, A. Post-Stroke Spasticity as a Condition: A New Perspective on Patient Evaluation. Funct. Neurol. 2016, 31, 179. [Google Scholar] [CrossRef]
- Borsook, D. Neurological Diseases and Pain. Brain 2012, 135, 320. [Google Scholar] [CrossRef]
- Treister, A.K.; Hatch, M.N.; Cramer, S.C.; Chang, E.Y. Demystifying Post-Stroke Pain: From Etiology to Treatment. PM&R 2017, 9, 63. [Google Scholar] [CrossRef]
- World Health Organization. International Classification of Functioning, Disability and Health (ICF); International Classification; World Health Organisation: Geneva, Switzerland, 2001. [Google Scholar]
- Brashear, A.; Zafonte, R.; Corcoran, M.; Galvez-Jimenez, N.; Gracies, J.M.; Gordon, M.F.; McAfee, A.; Ruffing, K.; Thompson, B.; Williams, M.; et al. Inter- and Intrarater Reliability of the Ashworth Scale and the Disability Assessment Scale in Patients with Upper-Limb Poststroke Spasticity. Arch. Phys. Med. Rehabil. 2002, 83, 1349–1354. [Google Scholar] [CrossRef]
- Dijkers, M. Comparing Quantification of Pain Severity by Verbal Rating and Numeric Rating Scales. J. Spinal Cord. Med. 2010, 33, 232–242. [Google Scholar] [CrossRef]
- Quinn, T.J.; Langhorne, P.; Stott, D.J. Barthel Index for Stroke Trials. Stroke 2011, 42, 1146–1151. [Google Scholar] [CrossRef]
- Bergfeldt, U.; Sköld, C.; Julin, P. Short Form 36 Assessed Health-Related Quality of Life after Focal Spasticity Therapy. J. Rehabil. Med. 2009, 41, 279–281. [Google Scholar] [CrossRef]
- Chard, G. ICF Checklist, Version 2.1a, Clinician Form, International Classification of Functioning, Disability and Health; World Health Organisation: Geneva, Switzerland, 2003. [Google Scholar]
- Santamato, A.; Facciorusso, S.; Spina, S.; Cinone, N.; Avvantaggiato, C.; Santoro, L.; Ciritella, C.; Smania, N.; Picelli, A.; Gasperini, G.; et al. Discontinuation of Botulinum Neurotoxin Type-A Treatment during COVID-19 Pandemic: An Italian Survey in Post Stroke and Traumatic Brain Injury Patients Living with Spasticity. Eur. J. Phys. Rehabil. Med. 2021, 57, 424–433. [Google Scholar] [CrossRef]
- Cohen, J. Statistical Power Analysis for the Behavioural Science, 2nd ed.; Routledge: London, UK, 1988; ISBN 0805802835. [Google Scholar]
- Arroyo, R.; Massana, M.; Vila, C. Correlation between Spasticity and Quality of Life in Patients with Multiple Sclerosis: The CANDLE Study. Int. J. Neurosci. 2013, 123, 850–858. [Google Scholar] [CrossRef]
- Hoffman, D.L.; Sadosky, A.; Dukes, E.M.; Alvir, J. How Do Changes in Pain Severity Levels Correspond to Changes in Health Status and Function in Patients with Painful Diabetic Peripheral Neuropathy? Pain 2010, 149, 194–201. [Google Scholar] [CrossRef]
- Phadke, C.; Reid, S.; Sasaki, A.; Choi, K.; Peters, J.; Chapman, E.; Tri, A.; Ismail, F.; Boulias, C. ICF-WHO Model to Describe Goal Characteristics among Individuals with Spasticity Receiving Botulinum Injections. Arch. Phys. Med. Rehabil. 2016, 97, e42. [Google Scholar] [CrossRef]
- Schmitt, C.M.; Schoen, S. Interoception: A Multi-Sensory Foundation of Participation in Daily Life. Front. Neurosci. 2022, 16, 875200. [Google Scholar] [CrossRef]
- Hoeppli, M.E.; Nahman-Averbuch, H.; Hinkle, W.A.; Leon, E.; Peugh, J.; Lopez-Sola, M.; King, C.D.; Goldschneider, K.R.; Coghill, R.C. Dissociation between Individual Differences in Self-Reported Pain Intensity and Underlying FMRI Brain Activation. Nat. Commun. 2022, 13, 3569. [Google Scholar] [CrossRef]
- Chang, E.M.; Gillespie, E.F.; Shaverdian, N. Truthfulness in Patient-Reported Outcomes: Factors Affecting Patients’ Responses and Impact on Data Quality. Patient Relat. Outcome Meas. 2019, 10, 171. [Google Scholar] [CrossRef]
- Van der Willik, E.M.; Terwee, C.B.; Bos, W.J.W.; Hemmelder, M.H.; Jager, K.J.; Zoccali, C.; Dekker, F.W.; Meuleman, Y. Patient-Reported Outcome Measures (PROMs): Making Sense of Individual PROM Scores and Changes in PROM Scores over Time. Nephrology 2021, 26, 391–399. [Google Scholar] [CrossRef]
- Lubowitz, J.H.; Brand, J.C.; Rossi, M.J. Our Measure of Medical Research Should Be Appreciable Benefit to the Patient. Arthroscopy 2019, 35, 1943–1944. [Google Scholar] [CrossRef]
- Coghill, R.C.; McHaffie, J.G.; Yen, Y.F. Neural Correlates of Interindividual Differences in the Subjective Experience of Pain. Proc. Natl. Acad. Sci. USA 2003, 100, 8538. [Google Scholar] [CrossRef]
- Orbai, A.M.; Smith, K.C.; Bartlett, S.J.; De Leon, E.; Bingham, C.O. “Stiffness Has Different Meanings, I Think, to Everyone”. Examining Stiffness from the Perspective of People Living with Rheumatoid. Arthritis Care Res. 2014, 66, 1662. [Google Scholar] [CrossRef]
- Stanton, T.R.; Moseley, G.L.; Wong, A.Y.L.; Kawchuk, G.N. Feeling Stiffness in the Back: A Protective Perceptual Inference in Chronic Back Pain. Sci. Rep. 2017, 7, 9681. [Google Scholar] [CrossRef]
- Thakral, M.; Shi, L.; Shmerling, R.H.; Bean, J.F.; Leveille, S.G. A Stiff Price to Pay: Does Joint Stiffness Predict Disability in an Older Population? J. Am. Geriatr. Soc. 2014, 62, 1891. [Google Scholar] [CrossRef]
- Dijkerman, H.C.; de Haan, E.H.F. Somatosensory Processes Subserving Perception and Action. Behav. Brain Sci. 2007, 30, 189–201. [Google Scholar] [CrossRef]
- Riva, G. The Neuroscience of Body Memory: From the Self through the Space to the Others. Cortex 2018, 104, 241–260. [Google Scholar] [CrossRef]
- Luu, B.L.; Day, B.L.; Cole, J.D.; Fitzpatrick, R.C. The Fusimotor and Reafferent Origin of the Sense of Force and Weight. J. Physiol. 2011, 589, 3135–3147. [Google Scholar] [CrossRef] [PubMed]
- Saravanan, A.; Reagan, L.; Bai, J.; Booker, S.Q.; Starkweather, A. Pain Hygiene: A Content Analysis of Student Nurses’ Perspectives. Nurs. Forum 2022, 57, 1415–1423. [Google Scholar] [CrossRef] [PubMed]
- Matthews, D.; Cancino, E.E.; Falla, D.; Khatibi, A. Exploring Pain Interference with Motor Skill Learning in Humans: A Systematic Review. PLoS ONE 2022, 17, e0274403. [Google Scholar] [CrossRef] [PubMed]
- Turner-Stokes, L.; Jackson, D. Shoulder Pain after Stroke: A Review of the Evidence Base to Inform the Development of an Integrated Care Pathway. Clin. Rehabil. 2002, 16, 276–298. [Google Scholar] [CrossRef]
- Andringa, A.; Van De Port, I.; Meijer, J.W. Long-Term Use of a Static Hand-Wrist Orthosis in Chronic Stroke Patients: A Pilot Study. Stroke Res. Treat. 2013, 2013, 546093. [Google Scholar] [CrossRef] [PubMed]
- Erel, S.; Uygur, F.; Şimşek, I.E.; Yakut, Y. The Effects of Dynamic Ankle-Foot Orthoses in Chronic Stroke Patients at Three-Month Follow-up: A Randomized Controlled Trial. Clin. Rehabil. 2011, 25, 515–523. [Google Scholar] [CrossRef]
- Finnerup, N.B. Neuropathic Pain and Spasticity: Intricate Consequences of Spinal Cord Injury. Spinal Cord. 2017, 55, 1046–1050. [Google Scholar] [CrossRef]
- Esquenazi, A. Video Enhanced Visual Analysis of Hemiparetic Gait Muscle Selection: A Pilot Study. Toxicon 2023, 228, 107112. [Google Scholar] [CrossRef] [PubMed]
- Capra, N.F.; Ro, J.Y. Experimental Muscle Pain Produces Central Modulation of Proprioceptive Signals Arising from Jaw Muscle Spindles. Pain 2000, 86, 151–162. [Google Scholar] [CrossRef]
- Baierle, T.; Kromer, T.; Petermann, C.; Magosch, P.; Luomajoki, H. Balance Ability and Postural Stability among Patients with Painful Shoulder Disorders and Healthy Controls. BMC Musculoskelet. Disord. 2013, 14, 282. [Google Scholar] [CrossRef]
- Trompetto, C.; Marinelli, L.; Mori, L.; Puce, L.; Avanti, C.; Saretti, E.; Biasotti, G.; Amella, R.; Cotellessa, F.; Restivo, D.A.; et al. Effectiveness of Botulinum Toxin on Pain in Stroke Patients Suffering from Upper Limb Spastic Dystonia. Toxins 2022, 14, 39. [Google Scholar] [CrossRef] [PubMed]
- Conroy, S.K.; Brownlowe, K.B.; McAllister, T.W. Depression Comorbid with Stroke, Traumatic Brain Injury, Parkinson’s Disease, and Multiple Sclerosis: Diagnosis and Treatment. Focus J. Life Long Learn. Psychiatry 2020, 18, 150. [Google Scholar] [CrossRef]
- Suksasilp, C.; Garfinkel, S.N. Towards a Comprehensive Assessment of Interoception in a Multi-Dimensional Framework. Biol. Psychol. 2022, 168, 108262. [Google Scholar] [CrossRef] [PubMed]
- Scheydt, S.; Müller Staub, M.; Frauenfelder, F.; Nielsen, G.H.; Behrens, J.; Needham, I. Sensory Overload: A Concept Analysis. Int. J. Ment. Health Nurs. 2017, 26, 110–120. [Google Scholar] [CrossRef] [PubMed]
- Marzolla, M.C.; Thielen, H.; Hurks, P.; Borghans, L.; van Heugten, C. Qualitative Data on Triggers and Coping of Sensory Hypersensitivity in Acquired Brain Injury Patients: A Proposed Model. Neuropsychol. Rehabil. 2023. [Google Scholar] [CrossRef]
- Thielen, H.; Tuts, N.; Welkenhuyzen, L.; Huenges Wajer, I.M.C.; Lafosse, C.; Gillebert, C.R. Sensory Sensitivity after Acquired Brain Injury: A Systematic Review. J. Neuropsychol. 2023, 17, 1–31. [Google Scholar] [CrossRef]
- Bolognini, N.; Russo, C.; Edwards, D.J. The Sensory Side of Post-Stroke Motor Rehabilitation. Restor. Neurol. Neurosci. 2016, 34, 571–586. [Google Scholar] [CrossRef]
- Howell, D.R.; Oldham, J.R.; Brilliant, A.N.; Meehan, W.P. Trouble Falling Asleep After Concussion Is Associated with Higher Symptom Burden among Children and Adolescents. J. Child Neurol. 2019, 34, 256–261. [Google Scholar] [CrossRef] [PubMed]
- Alwawi, D.A.; Dean, E.; Heldstab, A.; Lawson, L.M.; Peltzer, J.; Dunn, W. A Qualitative Study of Stroke Survivors’ Experience of Sensory Changes. Can. J. Occup. Ther. 2020, 87, 298–306. [Google Scholar] [CrossRef] [PubMed]
- Bovend’Eerdt, T.J.H.; Botell, R.E.; Wade, D.T. Writing SMART Rehabilitation Goals and Achieving Goal Attainment Scaling: A Practical Guide. Clin. Rehabil. 2009, 23, 352–361. [Google Scholar] [CrossRef] [PubMed]

| Characteristics | n (151) |
|---|---|
| Gender | |
| Male, n (%) | 90 (59.6) |
| Female, n (%) | 61 (40.4) |
| Age (mean ± SD, years) | 58.42 ± 14.64 |
| Time since event (mean ± SD, years) | 7.81 ± 7.34 |
| Time since first injection (mean ± SD, years) | 3.07 ± 1.03 |
| Disease | |
| Ischemic stroke, n (%) | 75 (49.7) |
| Hemorrhagic stroke, n (%) | 48 (3.,8) |
| Traumatic brain injury, n (%) | 16 (10.6) |
| Paretic side | |
| Left, n (%) | 60 (39.7) |
| Right, n (%) | 80 (53) |
| Both, n (%) | 11 (7.3) |
| Affected limb | |
| upper limb, n (%) | 21 (13.9) |
| lower limb, n (%) | 16 (10.6) |
| Both, n (%) | 114 (75.5) |
| Unpleasant Sensations | ||||||
|---|---|---|---|---|---|---|
| Pain UL | Pain LL | Stiffness UL | Stiffness LL | Heaviness UL | Heaviness LL | |
| N (%) | N (%) | N (%) | N (%) | N (%) | N (%) | |
| none | 54 (35.8) | 58 (38.4) | 18 (11.9) | 16 (10.6) | 26 (17.2) | 23 (15.2) |
| mild | 21 (13.9) | 22 (14.6) | 12 (7.9) | 30 (19.9) | 23 (15.2) | 27 (17.9) |
| moderate | 33 (21.9) | 31 (20.5) | 45 (29.8) | 53 (35.1) | 34 (22.5) | 54 (35.8) |
| severe | 43 (28.5) | 40 (26.5) | 76 (50.3) | 52 (34.4) | 68 (45) | 47 (31.1) |
| Median score (min–max) | 4 (0–10) 6 (1–10) | 3 (0–10) 6 (1–10) | 7 (0–10) 7 (1–10) | 6 (0–10) 6 (1–10) | 6 (0–10) 7 (1–10) | 5 (0–10) 6 (1–10) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Facciorusso, S.; Spina, S.; Picelli, A.; Baricich, A.; Molteni, F.; Santamato, A. May Spasticity-Related Unpleasant Sensations Interfere with Daily Activities in People with Stroke and Traumatic Brain Injury? Secondary Analysis from the CORTOX Study. J. Clin. Med. 2024, 13, 1720. https://doi.org/10.3390/jcm13061720
Facciorusso S, Spina S, Picelli A, Baricich A, Molteni F, Santamato A. May Spasticity-Related Unpleasant Sensations Interfere with Daily Activities in People with Stroke and Traumatic Brain Injury? Secondary Analysis from the CORTOX Study. Journal of Clinical Medicine. 2024; 13(6):1720. https://doi.org/10.3390/jcm13061720
Chicago/Turabian StyleFacciorusso, Salvatore, Stefania Spina, Alessandro Picelli, Alessio Baricich, Franco Molteni, and Andrea Santamato. 2024. "May Spasticity-Related Unpleasant Sensations Interfere with Daily Activities in People with Stroke and Traumatic Brain Injury? Secondary Analysis from the CORTOX Study" Journal of Clinical Medicine 13, no. 6: 1720. https://doi.org/10.3390/jcm13061720
APA StyleFacciorusso, S., Spina, S., Picelli, A., Baricich, A., Molteni, F., & Santamato, A. (2024). May Spasticity-Related Unpleasant Sensations Interfere with Daily Activities in People with Stroke and Traumatic Brain Injury? Secondary Analysis from the CORTOX Study. Journal of Clinical Medicine, 13(6), 1720. https://doi.org/10.3390/jcm13061720








