The Impact of the COVID-19 Pandemic on Esophageal and Gastric Cancer Surgery in Germany: A Four-Year Retrospective Single-Center Study of 287 Patients
Abstract
1. Introduction
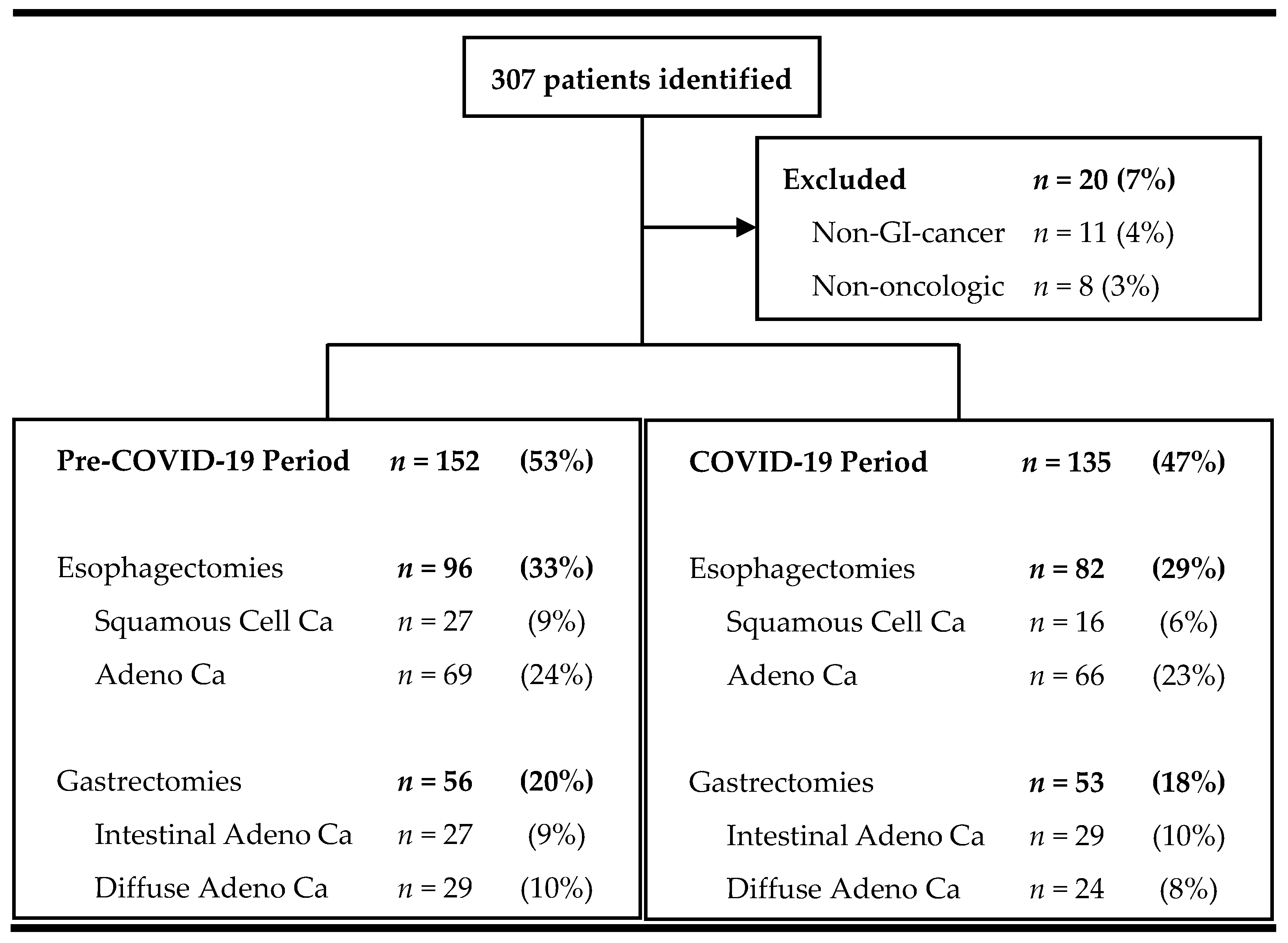
2. Materials and Methods
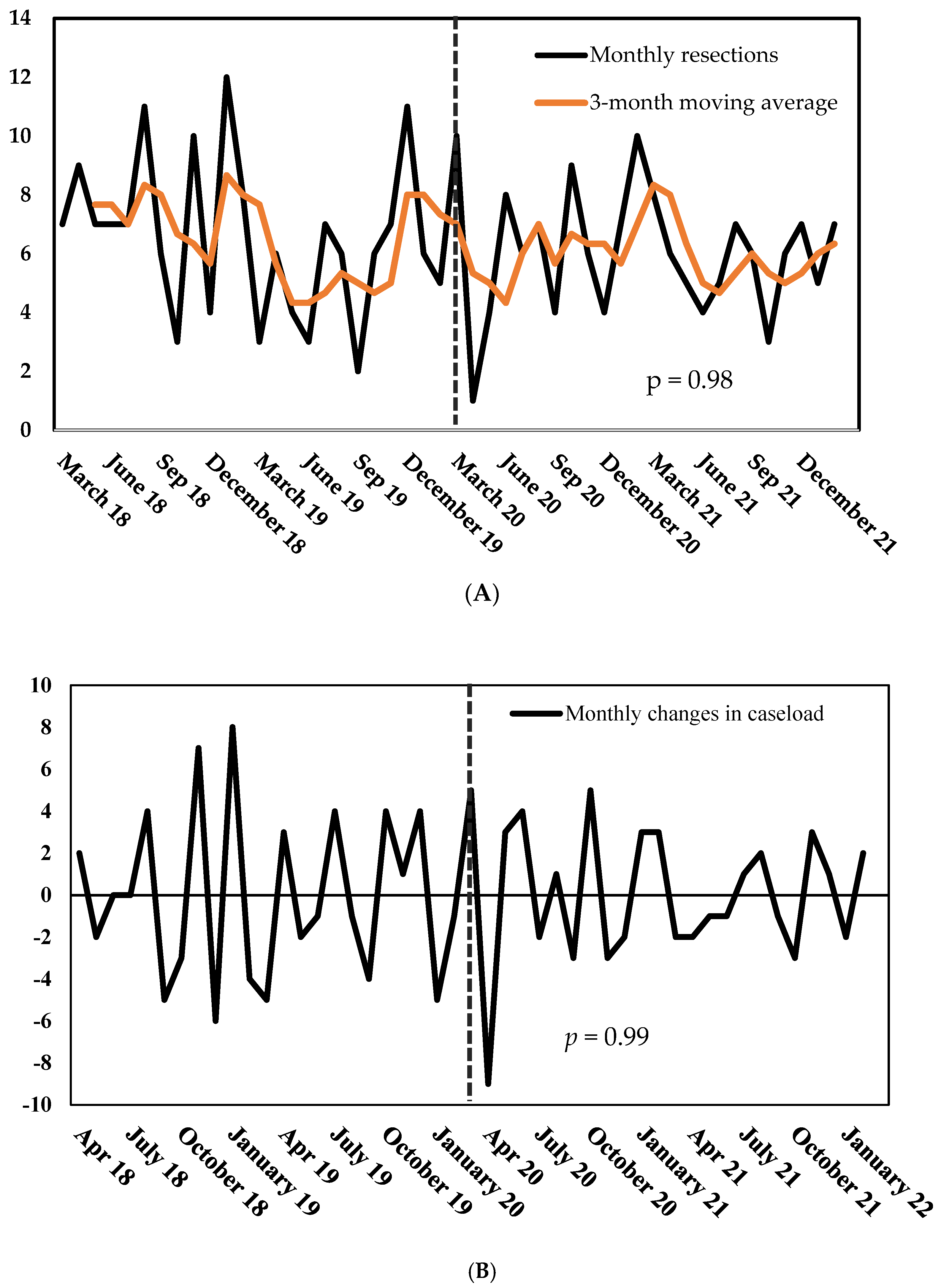
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- WHO Director-General’s Opening Remarks at the Media Briefing on COVID-19. Available online: https://www.who.int/director-general/speeches/detail/who-director-general-s-opening-remarks-at-the-media-briefing-on-covid-19---11-march-2020 (accessed on 25 May 2023).
- Robert Koch Institute: Ergänzung zum Nationalen Pandemieplan—COVID-19—Neuartige Coronaviruserkrankung. Available online: https://www.rki.de/DE/Content/InfAZ/N/Neuartiges_Coronavirus/Ergaenzung_Pandemieplan_Covid.pdf?__blob=publicationFile (accessed on 2 June 2023).
- Haldane, V.; De Foo, C.; Abdalla, S.M.; Jung, A.S.; Tan, M.; Wu, S.; Chua, A.; Verma, M.; Shrestha, P.; Singh, S.; et al. Health systems resilience in managing the COVID-19 pandemic: Lessons from 28 countries. Nat. Med. 2021, 27, 964–980. [Google Scholar] [CrossRef] [PubMed]
- Federal Ministry of the Interior (Germany). Available online: https://www.bbk.bund.de/SharedDocs/Downloads/DE/Gesundheit/KAEP/checkliste-pandemie-krankenhaus.pdf?__blob=publicationFile&v=4 (accessed on 14 May 2023).
- Johns Hopkins University Medicine: Mortality Analyses. Available online: https://coronavirus.jhu.edu/data/mortality (accessed on 14 May 2023).
- Riera, R.; Bagattini, Â.M.; Pacheco, R.L.; Pachito, D.V.; Roitberg, F.; Ilbawi, A. Delays and disruptions in cancer health care due to COVID-19 pandemic: Systematic review. JCO Glob. Oncol. 2021, 7, 311–323. [Google Scholar] [CrossRef] [PubMed]
- Hanna, T.P.; King, W.D.; Thibodeau, S.; Jalink, M.; Paulin, G.A.; Harvey-Jones, E.; O’Sullivan, D.E.; Booth, C.M.; Sullivan, R.; Aggarwal, A. Mortality due to cancer treatment delay: Systematic review and meta-analysis. BMJ 2020, 371, m4087. [Google Scholar] [CrossRef] [PubMed]
- Luketich, J.D.; Pennathur, A.; Awais, O.; Levy, R.M.; Keeley, S.; Shende, M.; Christie, N.A.; Weksler, B.; Landreneau, R.J.; Abbas, G.; et al. Outcomes after minimally invasive esophagectomy: Review of over 1000 patients. Ann. Surg. 2012, 256, 95. [Google Scholar] [CrossRef] [PubMed]
- Takeshita, H.; Ichikawa, D.; Komatsu, S.; Kubota, T.; Okamoto, K.; Shiozaki, A.; Fujiwara, H.; Otsuji, E. Surgical outcomes of gastrectomy for elderly patients with gastric cancer. World J. Surg. 2013, 37, 2891–2898. [Google Scholar] [CrossRef] [PubMed]
- Wahed, S.; Chmelo, J.; Navidi, M.; Hayes, N.; Phillips, A.W.; Immanuel, A. Delivering esophago-gastric cancer care during the COVID-19 pandemic in the United Kingdom: A surgical perspective. Dis. Esophagus 2020, 33, doaa091. [Google Scholar] [CrossRef] [PubMed]
- Dolan, D.P.; Swanson, S.J.; Lee, D.N.; Polhemus, E.; Kucukak, S.; Wiener, D.C.; Bueno, R.; Wee, J.O.; White, A. Esophagectomy for esophageal cancer performed during the early phase of the COVID-19 pandemic. Semin. Thorac. Cardiovasc. Surg. 2022, 34, 1075–1080. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.X.; He, C.Z.; Liu, Y.C.; Zhao, P.Y.; Xu, X.L.; Wang, Y.F.; Xia, S.Y.; Du, X.H. The impact of COVID-19 on gastric cancer surgery: A single-center retrospective study. BMC Surg. 2020, 20, 222. [Google Scholar] [CrossRef] [PubMed]
- Rebecchi, F.; Arolfo, S.; Ugliono, E.; Morino, M.; Asti, E.; Bonavina, L.; Borghi, F.; Coratti, A.; Cossu, A.; De Manzoni, G.; et al. Impact of COVID-19 outbreak on esophageal cancer surgery in Northern Italy: Lessons learned from a multicentric snapshot. Dis. Esophagus 2021, 34, doaa124. [Google Scholar] [CrossRef] [PubMed]
- Glasbey, J.; Ademuyiwa, A.; Adisa, A.; AlAmeer, E.; Arnaud, A.P.; Ayasra, F.; Azevedo, J.; Minaya-Bravo, A.; Costas-Chavarri, A.; Edwards, J.; et al. Effect of COVID-19 pandemic lockdowns on planned cancer surgery for 15 tumour types in 61 countries: An international, prospective, cohort study. Lancet Oncol. 2021, 22, 1507–1517. [Google Scholar] [CrossRef] [PubMed]
- Hale, T.; Angrist, N.; Goldszmidt, R.; Kira, B.; Petherick, A.; Phillips, T.; Webster, S.; Cameron-Blake, E.; Hallas, L.; Majumdar, S.; et al. A global panel database of pandemic policies (Oxford COVID-19 Government Response Tracker). Nat. Hum. Behav. 2021, 5, 529–538. [Google Scholar] [CrossRef] [PubMed]
- Wolters, U.; Wolf, T.; Stützer, H.; Schröder, T. ASA classification and perioperative variables as predictors of postoperative outcome. Br. J. Anaesth. 1996, 77, 217–222. [Google Scholar] [CrossRef] [PubMed]
- Clavien, P.A.; Barkun, J.; de Oliveira, M.L.; Vauthey, J.N.; Dindo, D.; Schulick, R.D.; de Santibañes, E.; Pekolj, J.; Slankamenac, K.; Bassi, C.; et al. The Clavien-Dindo classification of surgical complications: Five-year experience. Ann. Surg. 2009, 250, 187–196. [Google Scholar] [CrossRef] [PubMed]
- Liang, W.; Guan, W.; Chen, R.; Wang, W.; Li, J.; Xu, K.; Li, C.; Ai, Q.; Lu, W.; Liang, H.; et al. Cancer patients in SARS-CoV-2 infection: A nationwide analysis in China. Lancet Oncol. 2020, 21, 335–337. [Google Scholar] [CrossRef] [PubMed]
- Musche, V.; Bäuerle, A.; Steinbach, J.; Schweda, A.; Hetkamp, M.; Weismüller, B.; Kohler, H.; Beckmann, M.; Herrmann, K.; Tewes, M.; et al. COVID-19-related fear and health-related safety behavior in oncological patients. Front. Psychol. 2020, 11, 1984. [Google Scholar] [CrossRef] [PubMed]
- Gandjour, A. How many intensive care beds are justifiable for hospital pandemic preparedness? A cost-effectiveness analysis for COVID-19 in Germany. Appl. Health Econ. Health Policy 2021, 19, 181–190. [Google Scholar] [CrossRef] [PubMed]
- Federal Ministry of Health (Germany). Available online: https://www.bundesgesundheitsministerium.de/fileadmin/Dateien/3_Downloads/K/Krankenhausreform/5te_Stellungnahme_der_Reg-komm_Potenzialanalyse.pdf (accessed on 7 June 2023).
- Karagiannidis, C.; Kluge, S.; Riessen, R.; Krakau, M.; Bein, T.; Janssens, U. Impact of nursing staff shortage on intensive care medicine capacity in Germany. Med. Klin.-Intensivmed. Notfallmedizin 2019, 114, 327–333. [Google Scholar] [CrossRef] [PubMed]
- Schug, C.; Geiser, F.; Hiebel, N.; Beschoner, P.; Jerg-Bretzke, L.; Albus, C.; Weidner, K.; Morawa, E.; Erim, Y. Sick leave and intention to quit the job among nursing staff in German hospitals during the COVID-19 pandemic. Int. J. Environ. Res. Public Health 2022, 19, 1947. [Google Scholar] [CrossRef]

| Characteristic | COVID-19 | Control | p |
|---|---|---|---|
| N | 135 | 152 | |
| Age (years) | 63 ± 11 | 63 ± 12 | 0.972 |
| Male sex | 102 (76%) | 105 (70%) | 0.237 |
| BMI | 26 ± 6 | 26 ± 4 | 0.311 |
| Primary tumor | 0.715 | ||
| Esophagus, including AEG I, II | 82 (61%) | 96 (63%) | |
| Gastric, including AEG III | 53 (39%) | 56 (37%) | |
| ASA score, mean | 2.6 ± 0.5 | 2.6 ± 0.6 | 0.414 |
| ASA score categorization | 0.582 | ||
| 1 | 1 (1%) | 4 (3%) | |
| 2 | 50 (37%) | 59 (39%) | |
| 3 | 87 (60%) | 86 (57%) | |
| 4 | 3 (2%) | 2 (1%) | |
| Co-morbidities | |||
| Yes | 103 (76%) | 125 (82%) | 0.243 |
| Cirrhosis | 10 (7%) | 15 (10%) | 0.532 |
| Pulmonary disease | 16 (12%) | 15 (10%) | 0.704 |
| Diabetes | 22 (16%) | 14 (9%) | 0.077 |
| CNS disorders | 11 (8%) | 12 (8%) | 0.937 |
| Coronary artery disease | 14 (10%) | 10 (7%) | 0.289 |
| Heart failure | 9 (7%) | 10 (7%) | 0.976 |
| Heart valve disease | 6 (4%) | 7 (5%) | 0.948 |
| Hypertension | 73 (54%) | 82 (54%) | 0.983 |
| Peripheral artery disease | 4 (3%) | 3 (2%) | 0.588 |
| Coagulation disorders | 2 (2%) | 3 (2%) | 0.751 |
| Autoimmune disease | 1 (1%) | 1 (1%) | 0.933 |
| Chronic kidney disease | 0.035 * | ||
| 0 | 59 (44%) | 53 (35%) | |
| G1 | 12 (9%) | 33 (22%) | |
| G2 | 52 (39%) | 49 (32%) | |
| G3 | 9 (7%) | 15 (10%) | |
| G4 | 1 (1%) | 2 (1%) | |
| G5 | 1 (1%) | 0 |
| Characteristic | COVID-19 | Control | p |
|---|---|---|---|
| Esophageal | N = 82 | N = 96 | |
| Histology | 0.220 | ||
| Squamous cell carcinoma | 16 (20%) | 27 (28%) | |
| Adenocarcinoma | 66 (80%) | 69 (72%) | |
| Tumor stage | 0.541 | ||
| Early | 46 (56%) | 59 (62%) | |
| Advanced | 36 (44%) | 37 (39%) | |
| Neoadjuvant Therapy | 0.99 | ||
| (Radio)Chemotherapy | 75 (92%) | 87 (91%) | |
| None | 4 (5%) | 9 (9%) | |
| Unknown | 3 (3%) | - | |
| Cycles | 3.6 ± 1.6 | 3.7 ± 1.7 | 0.590 |
| Gastric | N = 53 | N = 56 | |
| Histology | 0.370 | ||
| Diffuse Adenocarcinoma | 24 (45%) | 29 (52%) | |
| Intestinal Adenocarcinoma | 29 (55%) | 27 (48%) | |
| Tumor stage | 0.391 | ||
| Early | 19 (36%) | 15 (27%) | |
| Advanced | 34 (64%) | 40 (71%) | |
| Unavailable | - | 1 (2%) | |
| Neoadjuvant Therapy | 0.92 | ||
| (Radio)Chemotherapy | 51 (96%) | 48 (86%) | |
| None | - | 1 (2%) | |
| Unknown | 2 (4%) | - | |
| Cycles | 4.3 ± 2.2 | 3.7 ± 1.8 | 0.471 |
| Characteristic | COVID-19 | Control | p |
|---|---|---|---|
| Esophagectomy | N = 81 | N = 96 | |
| Operating time (min) | 344 ± 72 | 415 ± 73 | <0.001 * |
| Procedure | |||
| Ivor–Lewis Esophagectomy | 81 (100%) | 96 (100%) | |
| Approach | 0.239 | ||
| Open | 2 (2%) | 1 (1%) | |
| Combined Laparotomy/Thoracoscopy | 17 (18%) | 14 (17%) | |
| Laparoscopy | 48 (50%) | 52 (63%) | |
| Robotic | 29 (30%) | 15 (18%) | |
| Gastrectomy | |||
| Operating time | 302 ± 100 | 316 ± 88 | 0.253 |
| Procedure | 0.163 | ||
| Total | 51 (96%) | 49 (88%) | |
| Subtotal | 2 (4%) | 7 (13%) | |
| Approach | 0.238 | ||
| Open | 23 (41%) | 31 (59%) | |
| Hand-assisted laparoscopic surgery | 28 (50%) | 17 (32%) | |
| Conversion to open | 2 (4%) | 3 (6%) | |
| Robotic | 3 (5%) | 2 (4%) |
| (A) | |||
| Characteristic | COVID-19 | Control | p |
| N = 98 | N = 82 | ||
| Procedural | |||
| Days between neoadjuvant therapy and surgery | 47 ± 18 | 45 ± 19 | 0.41 |
| Morbidity | |||
| Complications | |||
| Clavien–Dindo | 0.59 | ||
| I and II | 15 (15%) | 8 (10%) | |
| III | 29 (20%) | 26 (32%) | |
| IV | 19 (19%) | 18 (22%) | |
| V | 3 (3%) | 1 (1%) | |
| Clavien–Dindo ≥ 3 | 51 (52%) | 45 (55%) | 0.82 |
| ICU (days) | 4 (1–55) | 3 (1–97) | 0.65 |
| Length of stay (days) | 14 (8–110) | 17 (10–107) | 0.25 |
| Pneumonia | 13 (16%) | 25 (26%) | 0.01 * |
| Mortality | |||
| Death within primary stay | 3 (3%) | 2 (1%) | 1.00 |
| (B) | |||
| Characteristic | COVID-19 | Control | p |
| N = 50 | N = 55 | ||
| Procedural | |||
| Days between neoadjuvant therapy and surgery | 42 ± 15 | 42 ± 15 | 0.92 |
| Morbidity | |||
| Complications | |||
| Clavien–Dindo | 0.39 | ||
| I and II | 4 (8%) | 9 (16%) | |
| III | 5 (10%) | 4 (7%) | |
| IV | 5 (10%) | 4 (7%) | |
| V | 0 (0%) | 0 (0%) | |
| Clavien–Dindo ≥ 3 | 10 (20%) | 8 (15%) | 0.63 |
| ICU (days) | 2 (1–13) | 1 (0–7) | 0.07 |
| Length of stay (days) | 12 (7–21) | 12 (8–60) | 0.51 |
| Pneumonia | 3 (4%) | 8 (15%) | 0.07 |
| Mortality | |||
| Death within primary stay | 0 (0%) | 0 (0%) | 1.00 |
| Variable | Coefficient | Std. Error | t-Value | p-Value |
|---|---|---|---|---|
| Intercept | 2.13 | 0.347 | 6.15 | <0.01 * |
| COVID Period | −0.19 | 0.07 | −2.99 | <0.01 * |
| Age (y) | 0.01 | 0.003 | 1.99 | 0.048 * |
| BMI | 0.003 | 0.006 | 0.45 | 0.65 |
| ASA 2 | 0.20 | 0.25 | 0.80 | 0.422 |
| ASA 3 | 0.28 | 0.25 | 1.15 | 0.25 |
| ASA 4 | 1.20 | 0.36 | 3.32 | <0.01 * |
| CKD ¥ ≥ 3 | 0.114 | 0.11 | 1.02 | 0.31 |
| Clavien–Dindo ≥ 3 | 0.72 | 0.07 | 10.81 | <0.001 * |
| Gastrectomy | −0.01 | 0.08 | −0.02 | 0.986 |
| Variable | Coefficient | Std. Error | t-Value | p-Value |
|---|---|---|---|---|
| Intercept | −0.03 | 0.53 | −0.03 | 0.98 |
| COVID Period | 0.11 | 0.10 | 1.09 | 0.28 |
| Age (y) | 0.01 | 0.01 | 1.58 | 0.12 |
| BMI | 0.002 | 0.01 | 0.25 | 0.80 |
| ASA 2 | 0.30 | 0.38 | 0.80 | 0.43 |
| ASA 3 | 0.61 | 0.38 | 1.62 | 0.11 |
| ASA 4 | 1.95 | 0.55 | 3.53 | <0.01 * |
| CKD ¥ > 3 | 0.119 | 0.17 | 0.70 | 0.49 |
| Clavien–Dindo ≥ 3 | 0.27 | 0.13 | 2.08 | <0.04 * |
| Gastrectomy | −0.62 | 0.13 | −4.76 | <0.01 * |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ibach, M.; Winter, A.; Seika, P.; Ritschl, P.; Berndt, N.; Dobrindt, E.; Raakow, J.; Pratschke, J.; Denecke, C.; Maurer, M.M. The Impact of the COVID-19 Pandemic on Esophageal and Gastric Cancer Surgery in Germany: A Four-Year Retrospective Single-Center Study of 287 Patients. J. Clin. Med. 2024, 13, 1560. https://doi.org/10.3390/jcm13061560
Ibach M, Winter A, Seika P, Ritschl P, Berndt N, Dobrindt E, Raakow J, Pratschke J, Denecke C, Maurer MM. The Impact of the COVID-19 Pandemic on Esophageal and Gastric Cancer Surgery in Germany: A Four-Year Retrospective Single-Center Study of 287 Patients. Journal of Clinical Medicine. 2024; 13(6):1560. https://doi.org/10.3390/jcm13061560
Chicago/Turabian StyleIbach, Marius, Axel Winter, Philippa Seika, Paul Ritschl, Nadja Berndt, Eva Dobrindt, Jonas Raakow, Johann Pratschke, Christian Denecke, and Max Magnus Maurer. 2024. "The Impact of the COVID-19 Pandemic on Esophageal and Gastric Cancer Surgery in Germany: A Four-Year Retrospective Single-Center Study of 287 Patients" Journal of Clinical Medicine 13, no. 6: 1560. https://doi.org/10.3390/jcm13061560
APA StyleIbach, M., Winter, A., Seika, P., Ritschl, P., Berndt, N., Dobrindt, E., Raakow, J., Pratschke, J., Denecke, C., & Maurer, M. M. (2024). The Impact of the COVID-19 Pandemic on Esophageal and Gastric Cancer Surgery in Germany: A Four-Year Retrospective Single-Center Study of 287 Patients. Journal of Clinical Medicine, 13(6), 1560. https://doi.org/10.3390/jcm13061560






