A Randomized Clinical Trial Comparing Dubuisson Laparoscopic Lateral Suspension with Laparoscopic Sacropexy for Pelvic Organ Prolapse: Short-Term Results
Abstract
1. Introduction
2. Material and Methods
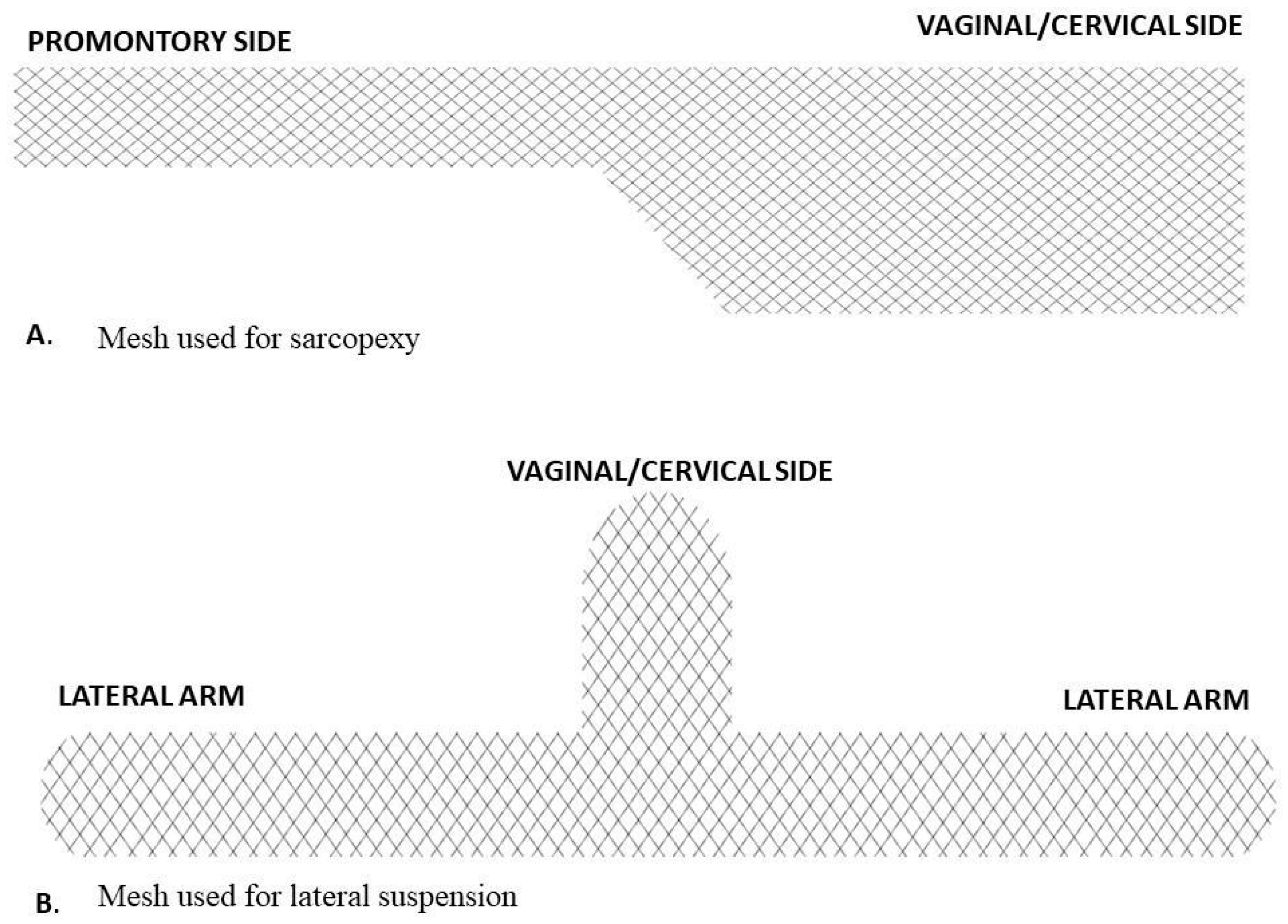
2.1. Surgical Technique
2.2. Power Calculation
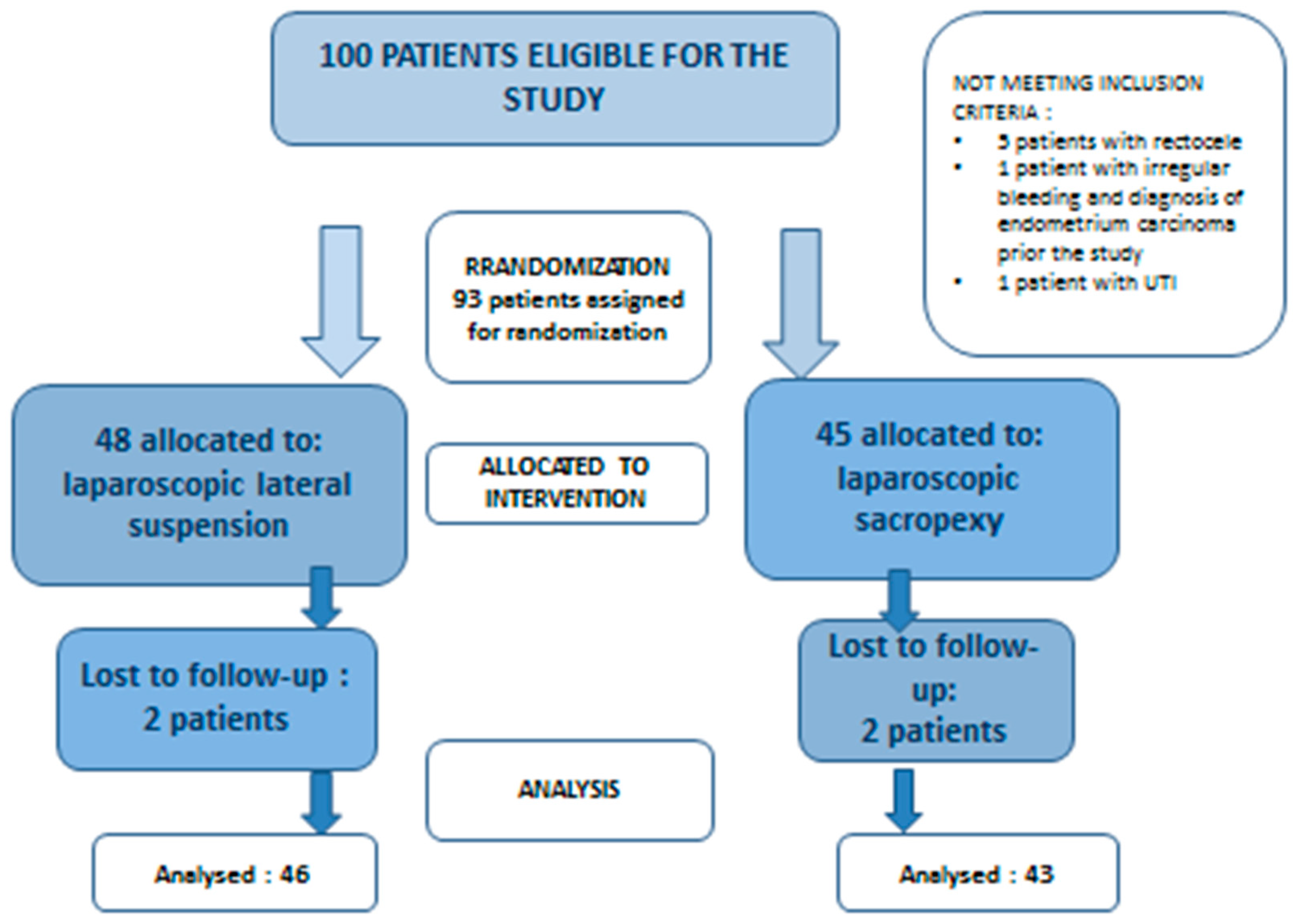
2.3. Randomization and Blinding
2.4. Patient Characteristics and Comparisons between Groups
2.5. Statistical Evaluation
3. Results
3.1. Evaluation of Anatomical and Functional Outcomes between Groups
3.2. Operation Time
3.3. Complications
3.4. Patient Satisfaction Evaluation
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Barber, M.D.; Maher, C. Epidemiology and outcome assessment of pelvic organ prolapse. Int. Urogynecol. J. 2013, 24, 1783–1790. [Google Scholar] [PubMed]
- Barber, M.D. Pelvic organ prolapse. BMJ 2016, 354, i3853. [Google Scholar] [CrossRef]
- Chow, D.; Rodríguez, L.V. Epidemiology and prevalence of pelvic organ prolapse. Curr. Opin. Urol. 2013, 23, 2938. [Google Scholar] [CrossRef] [PubMed]
- Ward, R.M.; Edwards, D.R.V.; Edwards, T.; Giri, A.; Jerome, R.N.; Wu, J.M. Genetic epidemiology of pelvic organ prolapse: A systematic review. Am. J. Obstet. Gynecol. 2014, 211, 326–335. [Google Scholar] [CrossRef]
- Mattsson, N.K.; Karjalainen, P.K.; Tolppanen, A.M.; Heikkinen, A.M.; Sintonen, H.; Härkki, P.; Nieminen, K.; Jalkanen, J. Pelvic organ prolapse surgery and quality of life—A nationwide cohort study. Am. J. Obstet. Gynecol. 2020, 222, 588.e1–588.e10. [Google Scholar]
- Balzarro, M.; Rubilotta, E.; Goss, C.; Costantini, E.; Artibani, W.; Sand, P. Counseling in urogynecology: A difficult task, or simply good surgeon–patient communication? Int. Urogynecol. J. 2018, 29, 943–948. [Google Scholar] [CrossRef]
- Li, C.; Gong, Y.; Wang, B. The efficacy of pelvic floor muscle training for pelvic organ prolapse: A systematic review and meta-analysis. Int. Urogynecol. J. 2016, 27, 981–992. [Google Scholar] [CrossRef]
- Hagen, S.; Stark, D.; Glazener, C.; Dickson, S.; Barry, S.; Elders, A.; Frawley, H.; Galea, M.P.; Logan, J.; McDonald, A.; et al. Individualised pelvic floor muscle training in women with pelvic organ prolapse (POPPY): A multicentre randomised controlled trial. Lancet 2014, 383, 796–806. [Google Scholar]
- Bugge, C.; Adams, E.J.; Gopinath, D.; Reid, F. Pessaries (mechanical devices) for pelvic organ prolapse in women. Cochrane Database Syst. Rev. 2013, 2, CD004010. [Google Scholar] [CrossRef] [PubMed]
- Bai, S.W.; Yoon, B.S.; Kwon, J.Y.; Shin, J.S.; Kim, S.K.; Park, K.H. Survey of the characteristics and satisfaction degree of the patients using a pessary. Int. Urogynecol. J. Pelvic Floor. Dysfunct. 2005, 16, 1826. [Google Scholar] [CrossRef] [PubMed]
- Bodner-Adler, B.; Bodner, K.; Stinglmeier, A. Prolapse surgery versus vaginal pessary in women with symptomatic pelvic organ prolapse: Which factors influence the choice of treatment? Arch. Gynecol. Obstet. 2019, 299, 773–777. [Google Scholar] [CrossRef] [PubMed]
- Szcześniewska, A.; Szpakowski, M.; Władziński, J.; Stetkiewicz, T.; Wilczyński, J.R. Zapomniana operacja Neugebauera-Le Forta. Pośrodkowe zamknięcie pochwy—Bezpieczna i skuteczna procedura zabiegowego leczenia wypadania narządu płciowego u starszych kobiet. Ginekol. Pol. 2015, 86, 198–202. [Google Scholar] [CrossRef]
- Intersdyscyplinarne Wytyczne Polskiego Towarzystwa Uroginekologicznego Odnośnie Diagnostyki I Leczenia Obniżenia Narządów Miednicy Mniejszej. Available online: https://ptug.pl/algorytmy-postepowania/intersdyscyplinarne-wytyczne-polskiego-towarzystwa-uroginekologicznego-odnosnie-diagnostyki-i-leczenia-obnizenia-narzadow-miednicy-mniejszej/ (accessed on 10 January 2019).
- ICS (International Continence Society). Pelvic Organ Prolapse. Available online: https://www.ics.org/standards (accessed on 31 January 2024).
- Dwyer, P.L. Choice of pelvic organ prolapse surgery: Vaginal or abdominal, native tissue or synthetic grafts, open abdominal versus laparoscopic or robotic. Int. Urogynecol. J. 2014, 25, 1151–1152. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Rogers, R.G.; Fashokun, T.B.; Eckler, K. Pelvic Organ Prolapse in Women: Epidemiology, Risk Factors, Clinical Manifestations, and Management; Rekomendacje Upto Date (luty 2020); Wolters Kluwer: Waltham, MA, USA, 2017. [Google Scholar]
- Szymanowski, P.; Szweda, H.; Szczepieniec, W.K.; Zarawski, M.; Malanowska, E.; Świś, E.; Jóźwik, M. Rola defektu apikalnego w patogenezie obniżenia narządów miednicy mniejszej: Cystocele z defektem apikalnym. Państw. Społecz. 2017, 4, 9–21. [Google Scholar] [CrossRef]
- Maher, C.; Feiner, B.; Baessler, K.; Christmann-Schmid, C.; Haya, N.; Brown, J.; Cochrane Gynaecology Fertility Group. Surgery for women with apical vaginal prolapse. Cochrane Database Syst. Rev. 2016, 10, CD012376. [Google Scholar] [CrossRef]
- Nygaard, I.; Brubaker, L.; Zyczynski, H.M.; Cundiff, G.; Richter, H.; Gantz, M.; Fine, P.; Menefee, S.; Ridgeway, B.; Visco, A.; et al. Long-term outcomes following abdominal sacrocolpopexy for pelvic organ prolapse. JAMA 2013, 309, 2016–2024, Erratum in JAMA 2013, 310, 1076. [Google Scholar] [CrossRef] [PubMed]
- Costantini, E.; Brubaker, L.; Cervigni, M.; Matthews, C.A.; O’Reilly, B.A.; Rizk, D.; Giannitsas, K.; Maher, C.F. Sacrocolpopexy for pelvic organ prolapse: Evidence-based review and recommendations. Eur. J. Obstet. Gynecol. Reprod. Biol. 2016, 205, 60–65. [Google Scholar] [CrossRef] [PubMed]
- Nosti, P.A.; Andy, U.U.; Kane, S.; White, D.E.; Harvie, H.S.; Lowenstein, L.; Gutman, R.E. Outcomes of abdominal and minimally invasive sacrocolpopexy: A retrospective cohort study. Female Pelvic Med. Reconstr. Surg. 2014, 20, 33–37. [Google Scholar] [CrossRef]
- Siddiqui, N.Y.; Grimes, C.L.; Casiano, E.R.; Abed, H.T.; Jeppson, P.C.; Olivera, C.K.; Sanses, T.V.; Steinberg, A.C.; South, M.M.; Balk, E.M.; et al. Mesh sacrocolpopexy compared with native tissue vaginal repair: A systematic review and meta-analysis. Obstet. Gynecol. 2015, 125, 44–55. [Google Scholar] [CrossRef]
- Coolen, A.L.W.; van Oudheusden, A.M.; Mol, B.W.J.; van Eijndhoven, H.W.; Roovers, J.P.W.; Bongers, M.Y. Laparoscopic sacrocolpopexy compared with open abdominal sacrocolpopexy for vault prolapse repair: A randomised controlled trial. Int. Urogynecol. J. 2017, 28, 1469–1479. [Google Scholar] [CrossRef]
- Szymczak, P.; Grzybowska, M.E.; Wydra, D.G. Comparison of laparoscopic techniques for apical organ prolapse repair—A systematic review of the literature. Neurourol. Urodyn. 2019, 38, 2031–2050. [Google Scholar] [CrossRef]
- Noé, K.G.; Schiermeier, S.; Alkatout, I.; Anapolski, M. Laparoscopic Pectopexy: A Prospective, Randomized, Comparative Clinical Trial of Standard Laparoscopic Sacral Colpocervicopexy with the New Laparoscopic Pectopexy—Postoperative Results and Intermediate-Term Follow-Up in a Pilot Study. J. Endourol. 2015, 29, 210–215. [Google Scholar] [CrossRef]
- Noé, G.K.; Schiermeier, S.; Papathemelis, T.; Fuellers, U.; Khudyakov, A.; Altmann, H.H.; Borowski, S.; Morawski, P.P.; Gantert, M.; De Vree, B.; et al. Prospective international multicenter pectopexy trial: Interim results and findings post surgery. Eur. J. Obstet. Gynecol. Reprod. Biol. 2020, 244, 81–86. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.H.; Yang, S.C.; Park, S.T.; Park, S.H.; Kim, H.B. Laparoscopic reconstructive surgery is superior to vaginal reconstruction in the pelvic organ prolapse. Int. J. Med. Sci. 2014, 10, 1082–1088. [Google Scholar] [CrossRef]
- Dubuisson, J.B.; Dubuisson, J. Comment je fais… le traitement cœlioscopique des prolapsus du dôme vaginal par colposuspension latérale. Gynecol. Obstet. Fertil. 2012, 40, 617–619. [Google Scholar] [CrossRef] [PubMed]
- Dubuisson, J.; Eperon, I.; Dällenbach, P.; Dubuisson, J.B. Laparoscopic repair of vaginal vault prolapse by lateral suspension with mesh. Arch. Gynecol. Obstet. 2013, 287, 307–312. [Google Scholar] [CrossRef]
- Cosma, S.; Menato, G.; Ceccaroni, M.; Marchino, G.L.; Petruzzelli, P.; Volpi, E.; Benedetto, C. Laparoscopic sacropexy and obstructed defecation syndrome: An anatomoclinical study. Int. Urogynecol. J. 2013, 24, 1623–1630. [Google Scholar] [CrossRef]
- Vieillefosse, S.; Thubert, T.; Dache, A.; Hermieu, J.F.; Deffieux, X. Satisfaction, quality of life and lumbar pain following laparoscopic sacrocolpopexy: Suture vs. tackers. Eur. J. Obstet. Gynecol. Reprod. Biol. 2015, 187, 51–56. [Google Scholar] [CrossRef]
- Haylen, B.T.; de Ridder, D.; Freeman, R.M.; Swift, S.E.; Berghmans, B.; Lee, J.; Monga, A.; Petri, E.; Rizk, D.E.; Sand, P.K.; et al. An International Urogynecological Association (IUGA)/International Continence Society (ICS) joint report on the terminology for female pelvic floor dysfunction. Neurourol. Urodyn. 2010, 29, 4–20. [Google Scholar] [CrossRef] [PubMed]
- Grzybowska, M.E.; Griffith, J.W.; Kenton, K.; Mueller, M.; Piaskowska-Cala, J.; Lewicky-Gaupp, C.; Wydra, D.; Bochenska, K. Validation of the Polish version of the Pelvic Floor Distress Inventory. Int. Urogynecol. J. 2019, 30, 101–105. [Google Scholar] [CrossRef]
- Surgical Mesh Implants—Regulatory Action on Surgical Mesh Products. Medsafe 2018. Available online: https://www.medsafe.govt.nz/hot/alerts/UrogynaecologicaSurgicalMeshImplants.asp (accessed on 31 January 2018).
- Maher, C.F.; Feiner, B.; DeCuyper, E.M.; Nichlos, C.J.; Hickey, K.V.; O’Rourke, P. Laparoscopic sacral colpopexy versus total vaginal mesh for vaginal vault prolapse: A randomized trial. Am. J. Obstet. Gynecol. 2011, 204, 360.e1–360.e7. [Google Scholar] [CrossRef] [PubMed]
- Mereu, L.; Tateo, S.; D’Alterio, M.N.; Russo, E.; Giannini, A.; Mannella, P.; Pertile, R.; Cai, T.; Simoncini, T. Laparoscopic lateral suspension with mesh for apical and anterior pelvic organ prolapse: A prospective double center study. Eur. J. Obstet. Gynecol. Reprod. Biol. 2020, 244, 16–20. [Google Scholar] [CrossRef]
- Dällenbach, P.; Alec, M.; Boulvain, M.; Shabanov, S. Outcomes of robotically assisted laparoscopic lateral suspension (RALLS) with mesh for anterior and apical prolapse. J. Robot. Surg. 2022, 16, 287–294. [Google Scholar] [CrossRef]
- Rubin, N.; Dubuisson, J.-B.; Gayet-Ageron, A.; Lange, S.; Eperon, I.; Dubuisson, J. Patient satisfaction after laparoscopic lateral suspension with mesh for pelvic organ prolapse: Outcome report of a continuous series of 417 patients. Int. Urogynecol. J. 2017, 28, 1685–1693. [Google Scholar] [CrossRef]
- Ruiz-Hernández, M.; López-Fando, L.; Sánchez-Guerrero, C.; Sánchez-González, Á.; Artiles-Medina, A.; Santiago-González, M.; Jiménez-Cidre, M.Á.; Burgos-Revilla, F.J. Combined laparoscopic surgery for the treatment of pelvic organ prolapse and recurrent urinary incontinence. Actas Urol. Esp. 2021, 45, 160–166, (In English and Spanish). [Google Scholar] [CrossRef] [PubMed]
- Deffieux, X.; Letouzey, V.; Savary, D.; Sentilhes, L.; Agostini, A.; Mares, P.; Pierre, F.; French College of Obstetrics and Gynecology (CNGOF). Prevention of complications related to the use of prosthetic meshes in prolapse surgery: Guidelines for clinical practice. Eur. J. Obstet. Gynecol. Reprod. Biol. 2012, 165, 170–180. [Google Scholar] [CrossRef] [PubMed]
- Pulatoğlu, Ç.; Yassa, M.; Turan, G.; Türkyılmaz, D.; Doğan, O. Vaginal axis on MRI after laparoscopic lateral mesh suspension surgery: A controlled study. Int. Urogynecol. J. 2021, 32, 851–858. [Google Scholar] [CrossRef] [PubMed]
- Roos, A.M.; Thakar, R.; Sultan, A.H.; de Leeuw, J.W.; Paulus, A.T. The impact of pelvic floor surgery on female sexual function: A mixed quantitative and qualitative study. BJOG 2014, 121, 92–101. [Google Scholar] [CrossRef]
- Dubuisson, J.B.; Dubuisson, J.; Puigventos, J. Optional Treatment of the Posterior Compartment and Techniques of Laparoscopic Lateral Suspension for Vaginal Vault Prolapse. In Laparoscopic Anatomy of the Pelvic Floor; Springer: Cham, Switzerland, 2020. [Google Scholar]
- Mulayim, B.; Sendag, F. Modified Laparoscopic Lateral Suspension: The Mulayim Technique. J. Minim. Invasive Gynecol. 2019, 26, 407–408. [Google Scholar] [CrossRef]
- Akbaba, E.; Sezgin, B. Modified laparoscopic lateral suspension with a five-arm mesh in pelvic organ prolapse surgery. BMC Women’s Health 2021, 21, 244. [Google Scholar] [CrossRef]
- Mowat, A.; Maher, D.; Baessler, K.; Christmann-Schmid, C.; Haya, N.; Maher, C. Surgery for women with posterior compartment prolapse. Cochrane Database Syst. Rev. 2018, 3, CD012975. [Google Scholar] [CrossRef]
- Dällenbach, P.; De Oliveira, S.S.; Marras, S.; Boulvain, M. Incidence and risk factors for mesh erosion after laparoscopic repair of pelvic organ prolapse by lateral suspension with mesh. Int. Urogynecol. J. 2016, 27, 1347–1355. [Google Scholar] [CrossRef]
- NICE Guidance. Sacrocolpopexy with Hysterectomy Using Mesh to Repair Uterine Prolapse. Available online: https://www.nice.org.uk/guidance/ipg577 (accessed on 22 March 2017).
- FDA. Urogynecologic Surgical Mesh Implants. 2019. Available online: https://www.fda.gov/medical-devices/implants-and-prosthetics/urogynecologic-surgical-mesh-implants (accessed on 12 February 2019).
- Sarlos, D.; Kots, L.; Ryu, G.; Schaer, G. Long-term follow-up of laparoscopic sacrocolpopexy. Int. Urogynecol. J. 2014, 25, 1207–1212. [Google Scholar] [CrossRef]
- Wong, V.; Guzman Rojas, R.; Shek, K.L.; Chou, D.; Moore, K.H.; Dietz, H.P. Laparoscopic sacrocolpopexy—How low does the mesh go? Ultrasound Obstet. Gynecol. 2013, 42, 230–234. [Google Scholar] [CrossRef] [PubMed]
- Baessler, K.; Christmann-Schmid, C.; Maher, C.; Haya, N.; Crawford, T.J.; Brown, J.; Cochrane Gynaecology Fertility Group. Surgery for women with pelvic organ prolapse with or without stress urinary incontinence. Cochrane Database Syst. Rev. 2018, 8, CD013108. [Google Scholar] [CrossRef]
- O’Sullivan, O.E.; Matthews, C.A.; O’Reilly, B.A. Sacrocolpopexy: Is there a consistent surgical technique? Int. Urogynecol. J. 2016, 27, 747–750. [Google Scholar] [CrossRef] [PubMed]
- Veit-Rubin, N.; Dubuisson, J.; Constantin, F.; Lange, S.; Eperon, I.; Gomel, V.; Dubuisson, J.B. Uterus preservation is superior to hysterectomy when performing laparoscopic lateral suspension with mesh. Int. Urogynecol. J. 2019, 30, 557–564. [Google Scholar] [CrossRef] [PubMed]
- Labanca, L.; Centini, G.; Lazzeri, L.; Afors, K.; Argay, I.M.; Habib, N.; Zupi, E. Sacrohysteropexy: A Way to Spare the Uterus. J. Minim. Invasive Gynecol. 2019, 27, 1254–1255. [Google Scholar] [CrossRef]
- Rahmanou, P.; White, B.; Price, N.; Jackson, S. Laparoscopic hysteropexy: 1 to 4-year follow-up of women postoperatively. Int. Urogynecol. J. 2014, 25, 131–138. [Google Scholar] [CrossRef]
- Korbly, N.B.; Kassis, N.C.; Good, M.M.; Richardson, M.L.; Book, N.M.; Yip, S.; Saguan, D.; Gross, C.; Evans, J.; Lopes, V.V.; et al. Patient preferences for uterine preservation and hysterectomy in women with pelvic organ prolapse. Am. J. Obstet. Gynecol. 2013, 209, 470.e1–470.e6. [Google Scholar] [CrossRef]
- Illiano, E.; Zucchi, A.; Giannitsas, K.; Carbone, A.; Pastore, A.L.; Costantini, E. Uterus preserving prolapse repair: How long does it last? Urol. Int. 2019, 102, 319–325. [Google Scholar] [CrossRef]
- Malanowska, E.; Starczewski, A.; Bielewicz, W.; Balzarro, M. Assessment of Overactive Bladder after Laparoscopic Lateral Suspension for Pelvic Organ Prolapse. Biomed. Res. Int. 2019, 2019, 9051963. [Google Scholar] [CrossRef]
- Welch, E.K.; Dengler, K.L.; Guirguis, M.; Strauchon, C.; Olsen, C.; Von Pechmann, W. Risk factors of lower urinary tract injury with laparoscopic sacrocolpopexy. AJOG Glob. Rep. 2021, 2, 100035. [Google Scholar] [CrossRef]
- Dubuisson, J.B.; Yaron, M.; Wenger, J.M.; Jacob, S. Treatment of genital prolapse by laparoscopic lateral suspension using mesh: A series of 73 patients. J. Minim. Invasive Gynecol. 2008, 15, 49–55. [Google Scholar] [CrossRef]
- Vandendriessche, D.; Giraudet, G.; Lucot, J.P.; Béhal, H.; Cosson, M. Impact of laparoscopic sacrocolpopexy learning curve on operative time, perioperative complications and short term results. Eur. J. Obstet. Gynecol. Reprod. Biol. 2015, 191, 84–89. [Google Scholar] [CrossRef] [PubMed]
- Hoshino, K.; Yoshimura, K.; Nishimura, K.; Hachisuga, T. How to reduce the operative time of laparoscopic sacrocolpopexy? Gynecol. Minim. Invasive Ther. 2017, 6, 17–19. [Google Scholar] [CrossRef] [PubMed]
- Fink, K.; Shachar, I.B.; Braun, N.M. Uterine preservation for advanced pelvic organ prolapse repair: Anatomical results and patient satisfaction. Int. Braz. J. Urol. 2016, 42, 773–778. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.; Nie, J.; Zeng, W.; Lai, Y.; Chen, X.; Yu, Y. A modified laparoscopic lateral suspension with mesh for apical and anterior pelvic organ prolapse: A retrospective cohort study. Videosurg. Other Miniinvasive Tech./Wideochir. Inne Tech. Małoinwazyjne 2023, 18, 690–697. [Google Scholar] [CrossRef] [PubMed]
- Yassa, M.; Tug, N. Uterus-preserving Laparoscopic Lateral Suspension with Mesh Operation in Pelvic Organ Prolapse: Initial Experience in a Single Tertiary Center with a Median 24-Month Follow-up. Geburtshilfe Frauenheilkd. 2019, 79, 983–992. [Google Scholar] [CrossRef] [PubMed]
- Dällenbach, P. Laparoscopic Lateral Suspension (LLS) for the Treatment of Apical Prolapse: A New Gold Standard? Front. Surg. 2022, 9, 898392. [Google Scholar] [CrossRef]
- Russo, E.; Montt Guevara, M.M.; Sacinti, K.G.; Misasi, G.; Falcone, M.; Morganti, R.; Mereu, L.; Dalprà, F.; Tateo, S.; Simoncini, T. Minimal Invasive Abdominal Sacral Colpopexy and Abdominal Lateral Suspension: A Prospective, Open-Label, Multicenter, Non-Inferiority Trial. J. Clin. Med. 2023, 12, 2926. [Google Scholar] [CrossRef] [PubMed]
| Demographics | LSC n = 43 | LLS n = 46 | p Value |
|---|---|---|---|
| Age (years), mean (±SD) | 58.14 (±8.28) | 59.49 (±8.84) | p = 0.467 |
| Age (years) of last period, mean (±SD) | 50.44 (±3.76) | 50.21 (±3.07) | p = 0.754 |
| Number of patients after menopause (%) | 41 (95.35) | 42 (93.02) | p = 0.649 |
| Number of vaginal deliveries, mean (±SD) | 2.26 (±1.03) | 2.42 (±1.18) | p = 0.497 |
| Child’s birth weight, mean (±SD) | 3743.72 (±402.07 g) | 3625.81 (±345.82 g) | p = 0.149 |
| Age at delivery, mean (±SD) | 24.65 (±3.62) | 24.37 (±4.39) | p = 0.749 |
| BMI, mean (±SD) | 26.59 (±3.73) | 25.98 (±3.71) | p = 0.453 |
| POP-Q before LSC | POP-Q after LSC | POP-Q before LLS | POP-Q after LLS | ||||
|---|---|---|---|---|---|---|---|
| C | N% | C | N% | C | N% | C | N% |
| III | 8 (36.6) | I | 17 (77.27) | III | 5 (25) | I | 16 (80) |
| III | 10 (45.45) | I | 1 (4.55) | III | 11 (55) | I | 2 (10) |
| III | 4 (18.18) | III | 4 (18.18) | III | 4 (20) | III | 2 (10) |
| TOTAL: 22 (100) | 22 (100) | 20 (100) | 20 (100) | ||||
| POP-Q before LSC | POP-Q after LSC | POP-Q before LLS | POP-Q after LLS | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| A | C | N% | A | C | N% | A | C | N% | A | C | N% |
| III | II | 16 (72.72) | I | I | 12 (57.14) | III | II | 17 (65.38) | I | I | 14 (53.84) |
| II | I | 3 (14.28) | II | I | 2 (7.69) | ||||||
| III | I | 1 (4.76) | III | III | 1 (3.84) | ||||||
| II | II | 5 (22.72) | I | I | 5 (23.8) | II | II | 9 (34.61) | I | I | 7 (26.92) |
| I | III | 1 (3.84) | |||||||||
| II | II | 1 (3.84) | |||||||||
| TOTAL | 21 (100) | 2I (100) | 26 (100) | 26 (100) |
| LLS/LSC | |||||
|---|---|---|---|---|---|
| Postoperatively | Aa | Ba | Ap | Bp | C |
| Mean | 1.50 | 1.39 | 1.59 | 1.98 | 5.36 |
| SD | 1.18 | 1.49 | 0.94 | 1.74 | 2.70 |
| Chi-square | p > 0.005 | p > 0.005 | p > 0.005 | p > 0.005 | p > 0.005 |
| Procedure | Preoperative (Mean) | Postoperative (Mean) | p | p Sacropexy/Lateral Suspension | |
|---|---|---|---|---|---|
| POPDI6 | LSC | 52.91 | 6.69 | <0.005 | p = 0.460 |
| LLS | 52.63 | 4.44 | <0.005 | ||
| CRADI8 | LSC | 8.94 | 2.76 | <0.005 | p = 0.012 |
| LLS | 11.13 | 6.65 | 0.043 | ||
| UDI6 | LSC | 44.96 | 10.66 | <0.005 | p = 0.077 |
| LLS | 38.59 | 5.34 | <0.005 | ||
| PFDI-20 | LSC | 106.81 | 20.11 | <0.005 | p = 0.491 |
| LLS | 102.02 | 16.24 | <0.005 |
| Symptoms | LSC n (%) 43 | p | LLS n (%) 46 | p | P LLS/LSC | ||
|---|---|---|---|---|---|---|---|
| Preoperative | Postoperative | Preoperative | Postoperative | ||||
| Bulging | 43 (100) | 11 (25.58) | p < 0.005 | 42 (91.30) | 4 (8.7) | p < 0.005 | p > 0.005 |
| Urinary urgency (wet) | 24 (55.81) | 7 (16.28) | p < 0.005 | 26 (56.52) | 9 (19.56) | p < 0.005 | p > 0.005 |
| Urinary frequency | 24 (55.81) | 7 (16.28) | p < 0.005 | 29 (63.04) | 10 (21.73) | p < 0.005 | p > 0.005 |
| OAB | 16 (37.21) | 4 (9.30) | p = 0.05 | 23 (50) | 6 (13.04) | p = 0.005 | p > 0.005 |
| UI | 11 (25.58) | 7 (16.27) | p = 0.78 | 6 (13.04) | 2 (4.35) | p = 0.08 | p > 0.005 |
| Constipation | 27 (62.79) | 12 (27.90) | p = 0,02 | 27 (58.69) | 22 (47.82) | p = 0.187 | p > 0.005 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Malanowska-Jarema, E.; Starczewski, A.; Melnyk, M.; Oliveira, D.; Balzarro, M.; Rubillota, E. A Randomized Clinical Trial Comparing Dubuisson Laparoscopic Lateral Suspension with Laparoscopic Sacropexy for Pelvic Organ Prolapse: Short-Term Results. J. Clin. Med. 2024, 13, 1348. https://doi.org/10.3390/jcm13051348
Malanowska-Jarema E, Starczewski A, Melnyk M, Oliveira D, Balzarro M, Rubillota E. A Randomized Clinical Trial Comparing Dubuisson Laparoscopic Lateral Suspension with Laparoscopic Sacropexy for Pelvic Organ Prolapse: Short-Term Results. Journal of Clinical Medicine. 2024; 13(5):1348. https://doi.org/10.3390/jcm13051348
Chicago/Turabian StyleMalanowska-Jarema, Ewelina, Andrzej Starczewski, Mariia Melnyk, Dulce Oliveira, Matteo Balzarro, and Emanuel Rubillota. 2024. "A Randomized Clinical Trial Comparing Dubuisson Laparoscopic Lateral Suspension with Laparoscopic Sacropexy for Pelvic Organ Prolapse: Short-Term Results" Journal of Clinical Medicine 13, no. 5: 1348. https://doi.org/10.3390/jcm13051348
APA StyleMalanowska-Jarema, E., Starczewski, A., Melnyk, M., Oliveira, D., Balzarro, M., & Rubillota, E. (2024). A Randomized Clinical Trial Comparing Dubuisson Laparoscopic Lateral Suspension with Laparoscopic Sacropexy for Pelvic Organ Prolapse: Short-Term Results. Journal of Clinical Medicine, 13(5), 1348. https://doi.org/10.3390/jcm13051348







