Patient Experiences and Challenges in the Management of Autoinflammatory Diseases—Data from the International FMF & AID Global Association Survey
Abstract
1. Introduction
2. Materials and Methods
2.1. Description of the Survey
2.2. Statistical Analysis
3. Results
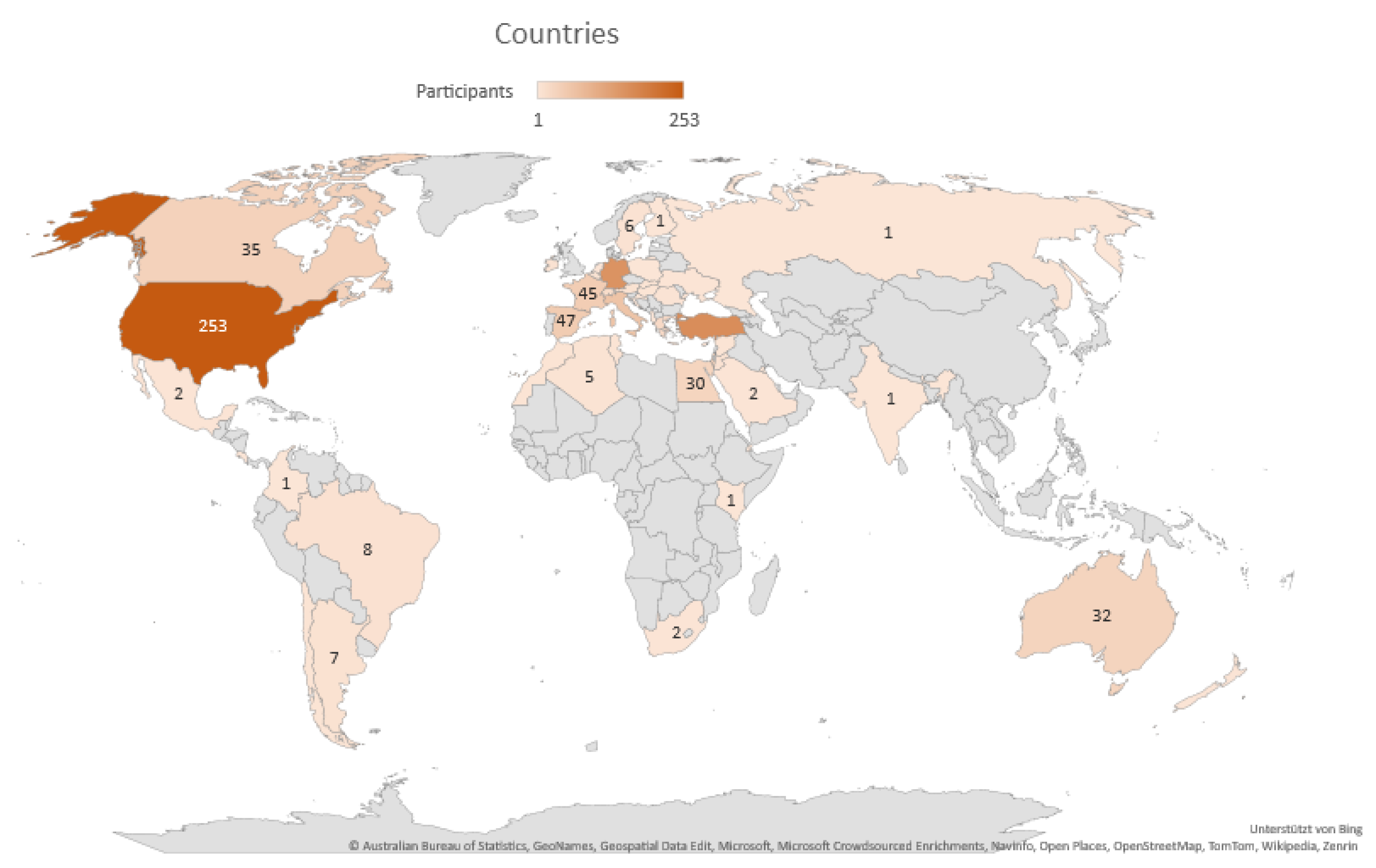
3.1. Demographic Characteristics
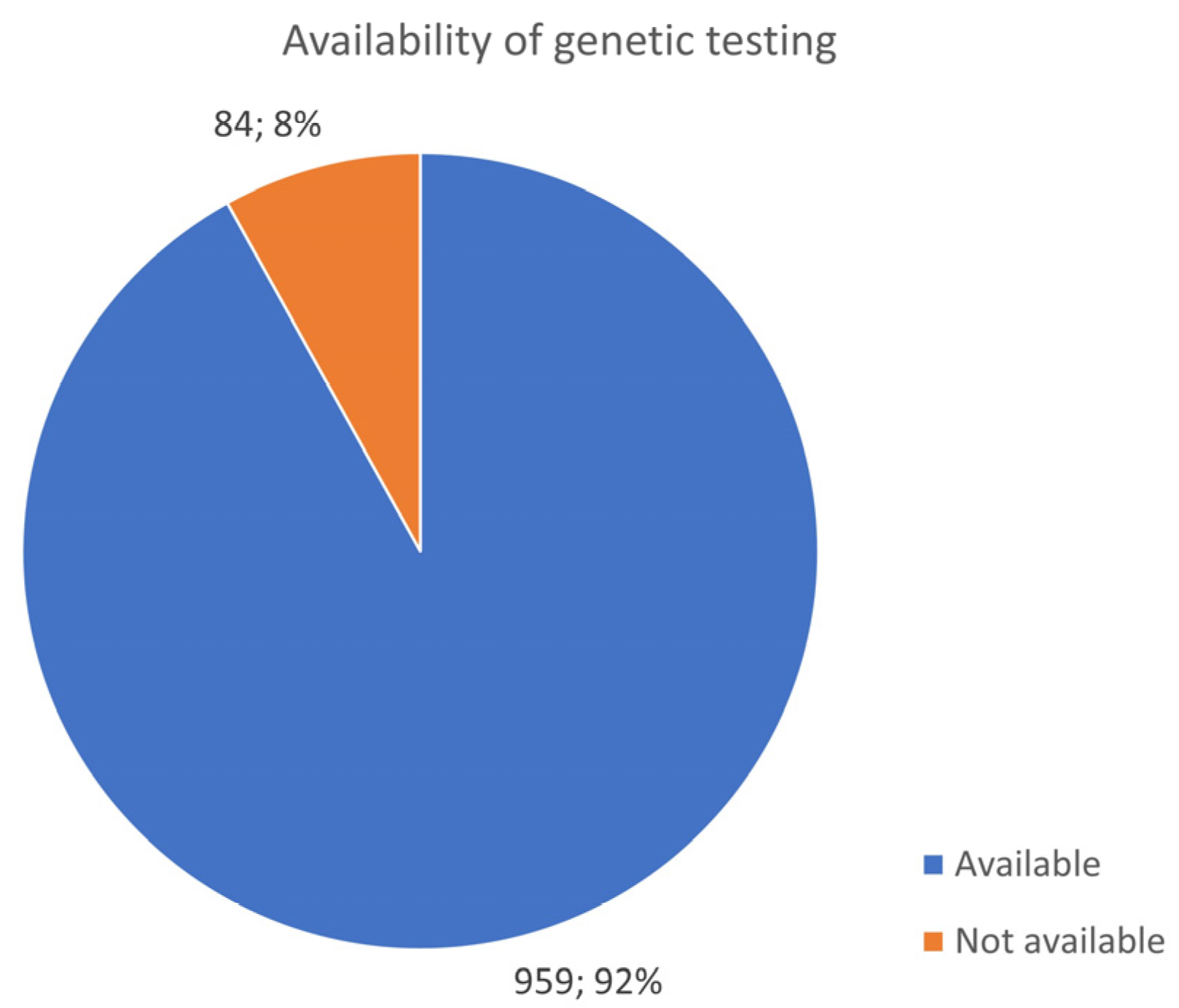
3.2. Genetic Testing
3.3. Laboratory Changes
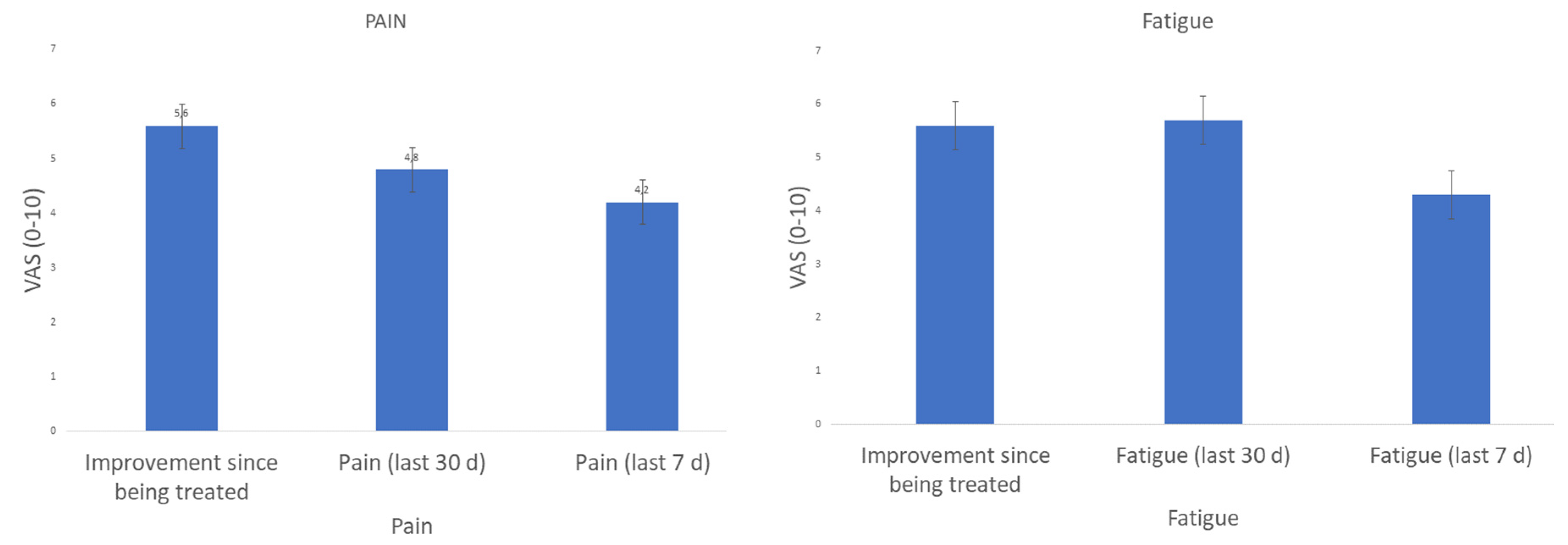
3.4. Disease Impact on Pain and Fatigue
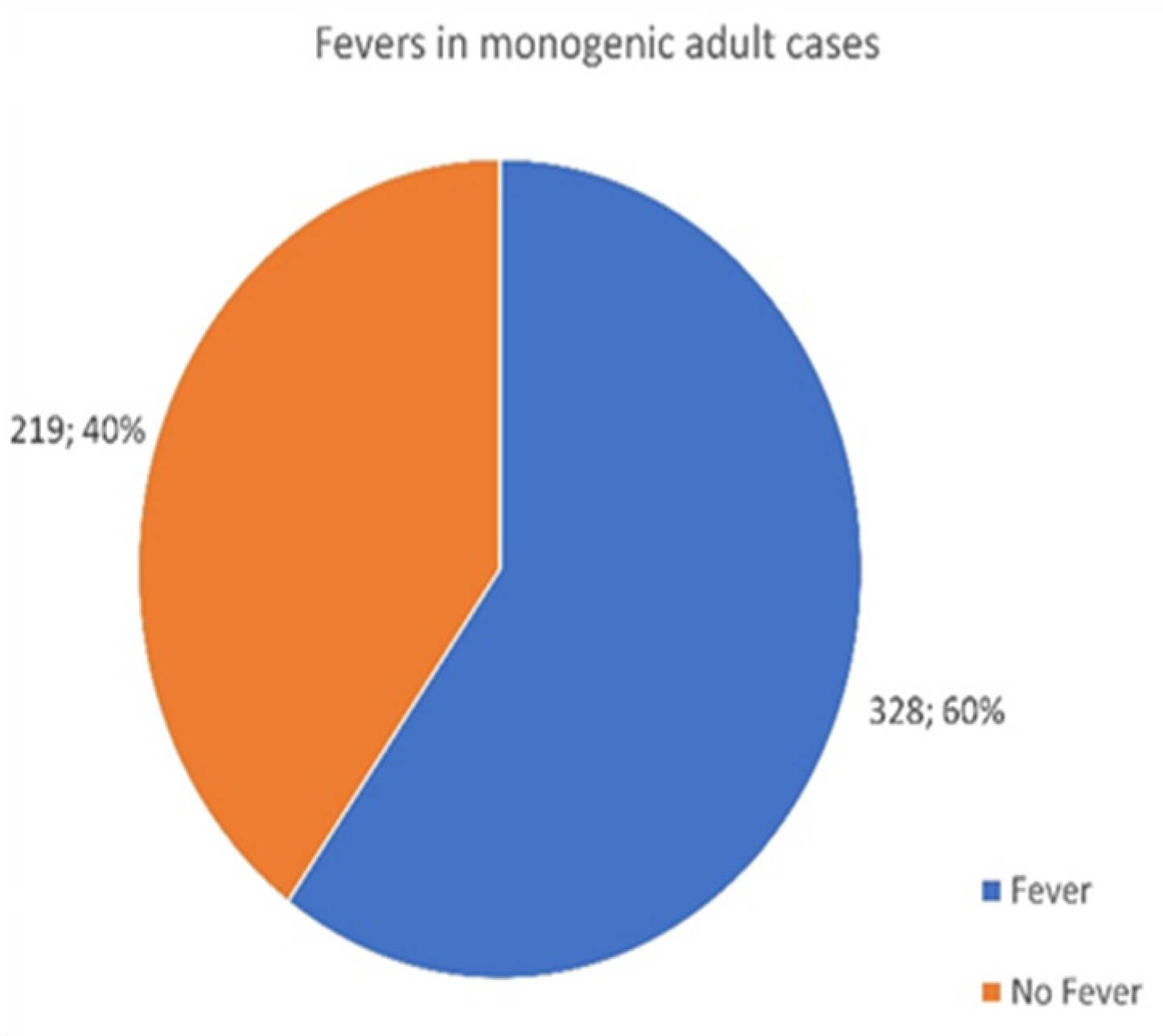
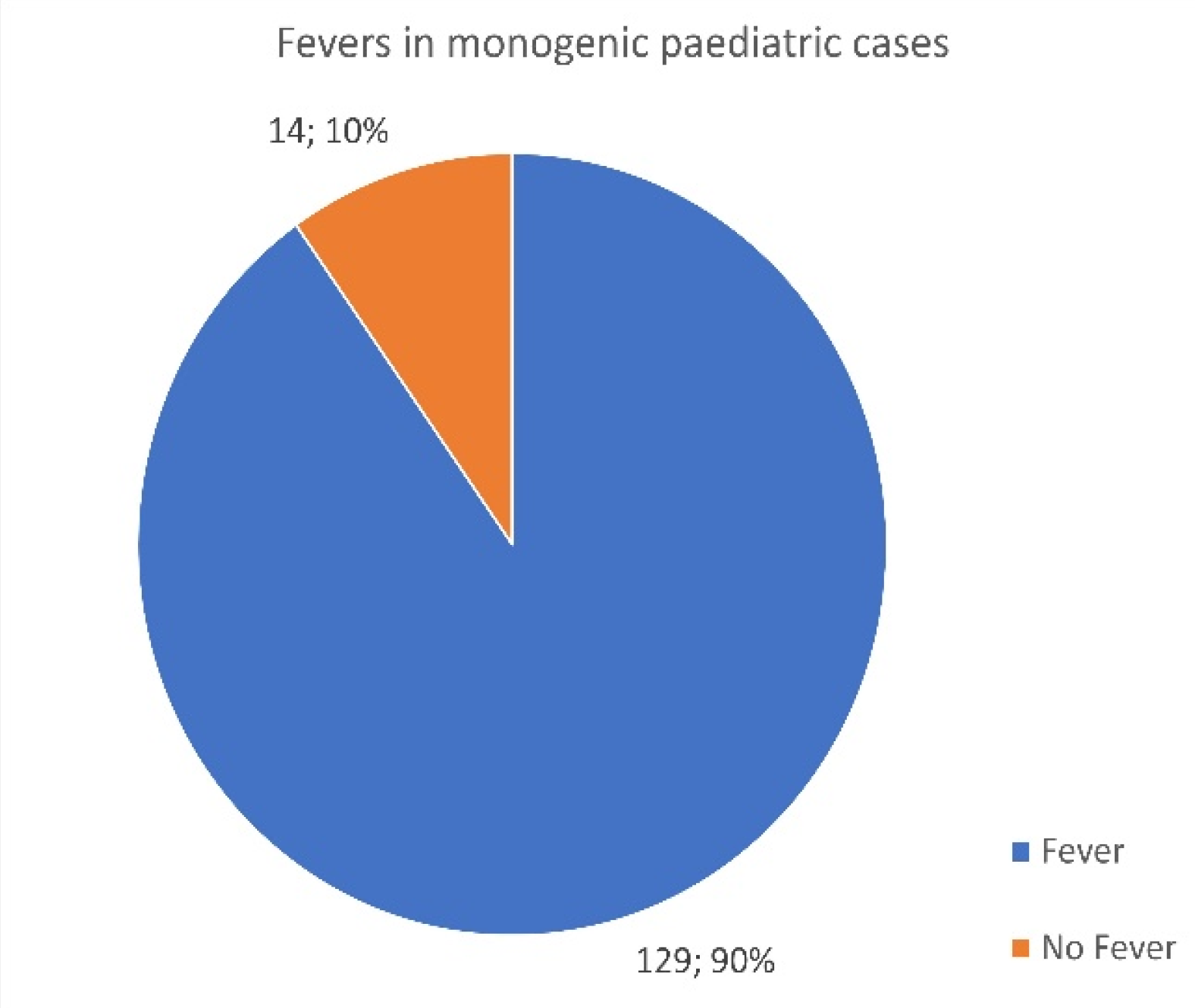
3.5. Monogenic Patients Presenting with a Fever
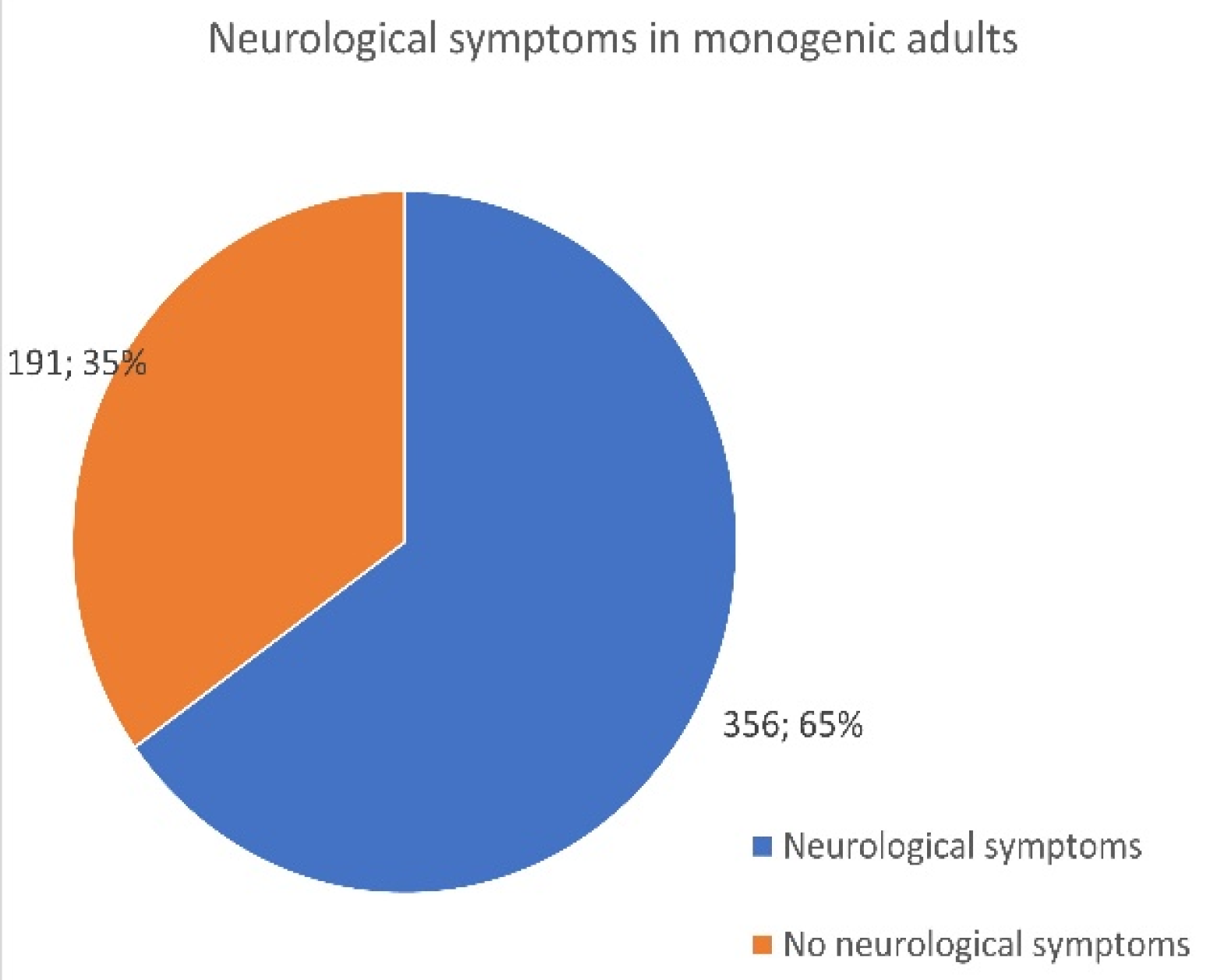
3.6. Monogenic Patients Presenting with Neurological Symptoms
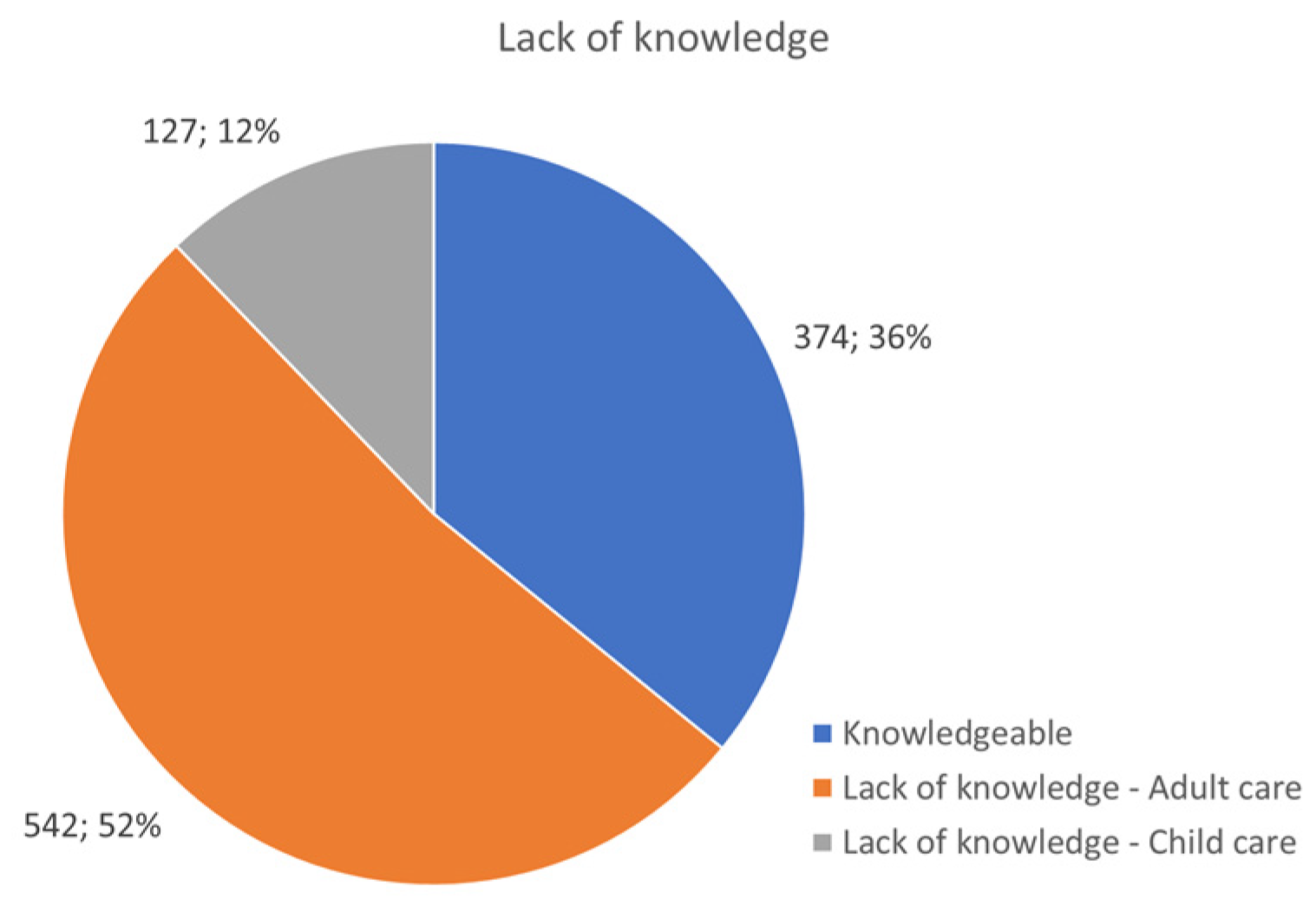
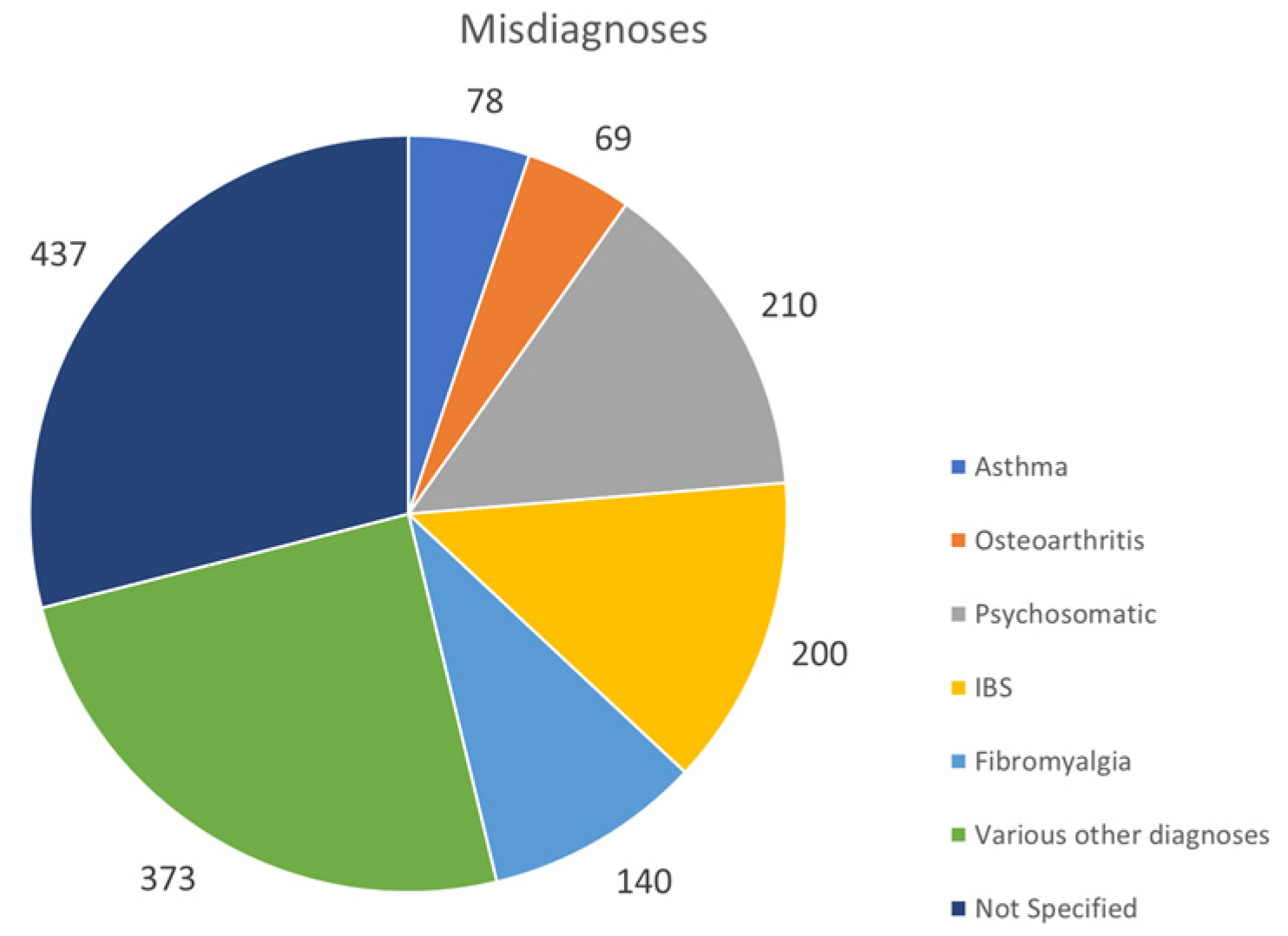
3.7. Issues in Diagnosis and Disease Management
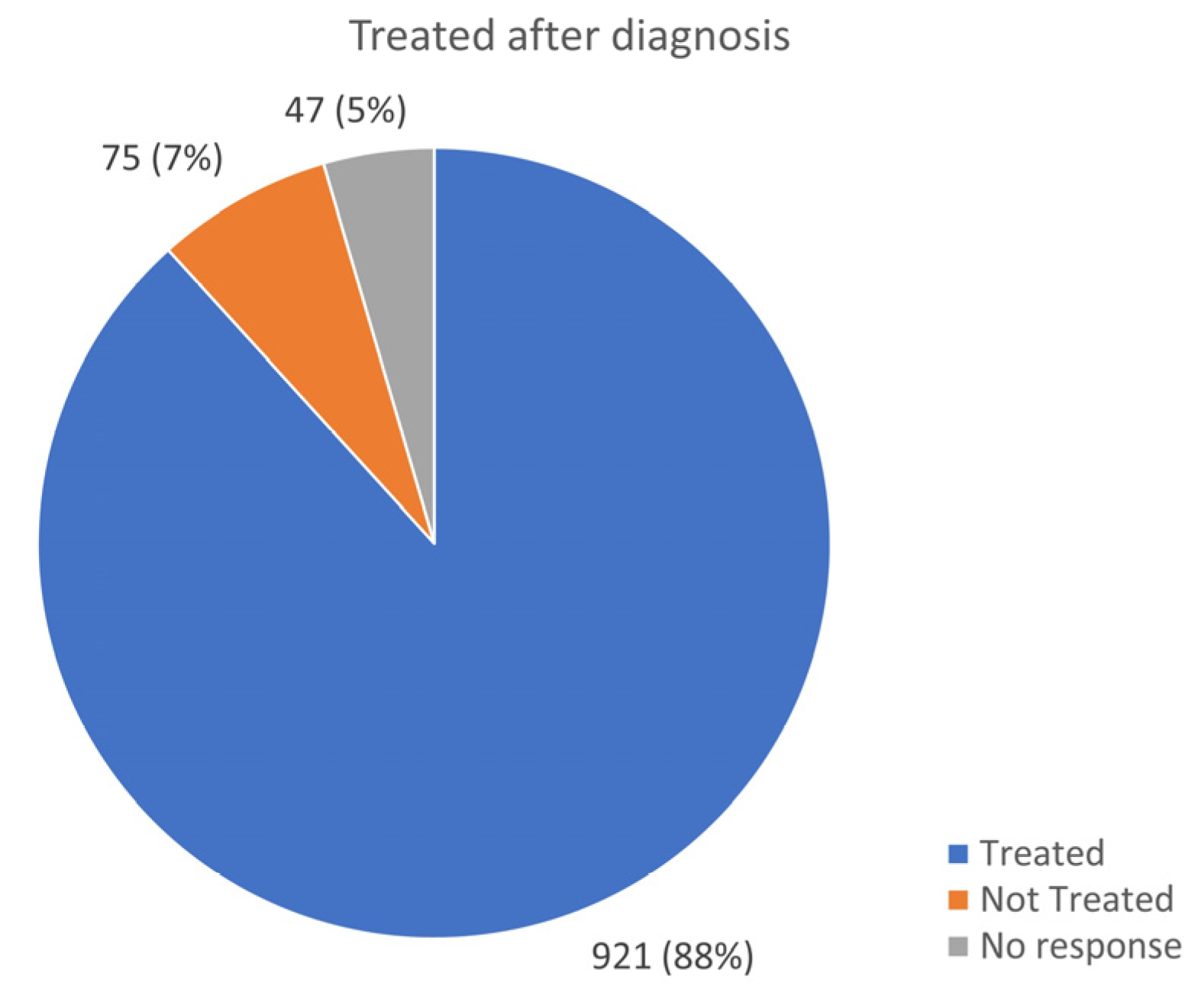
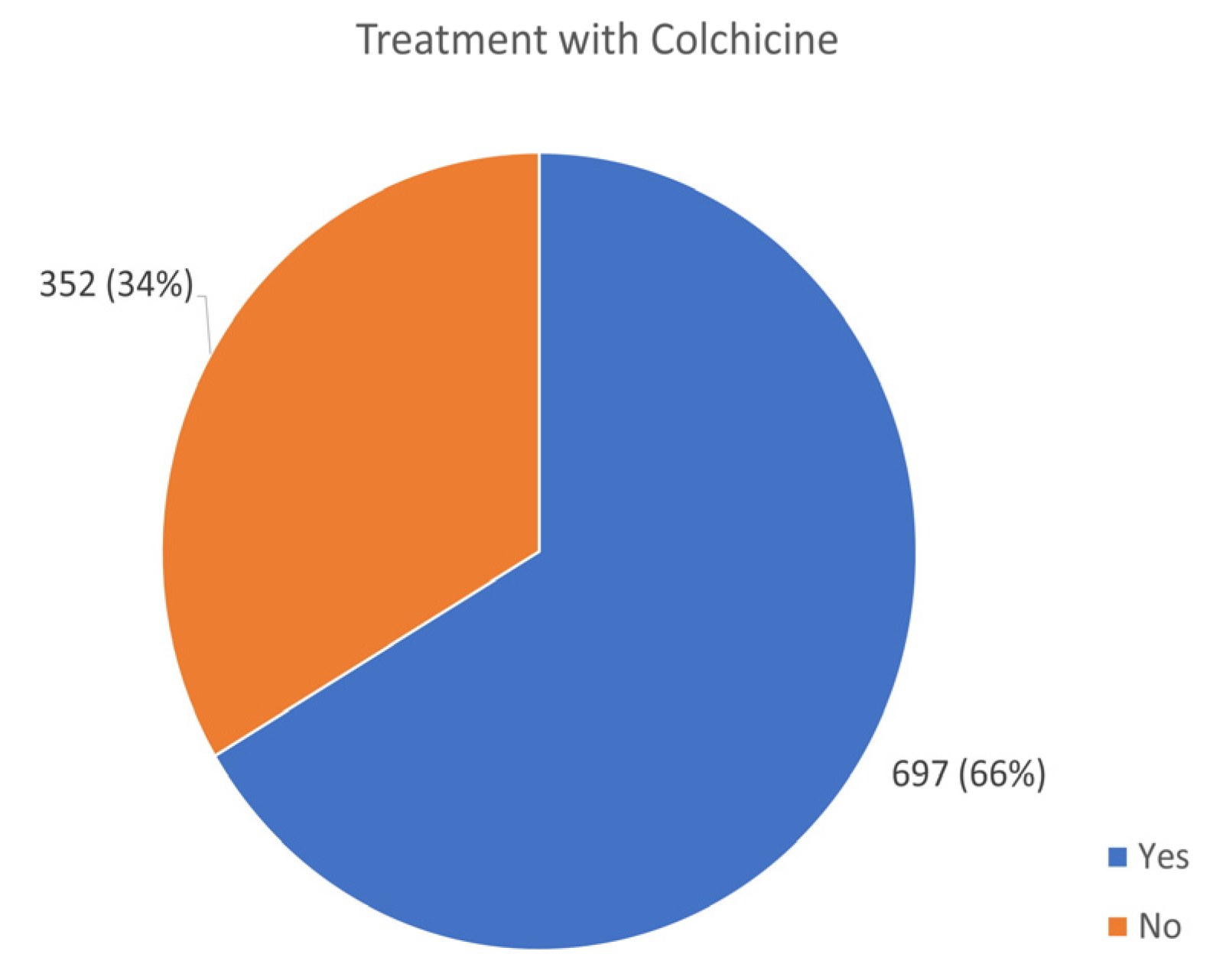
3.8. Medical Treatment in AID
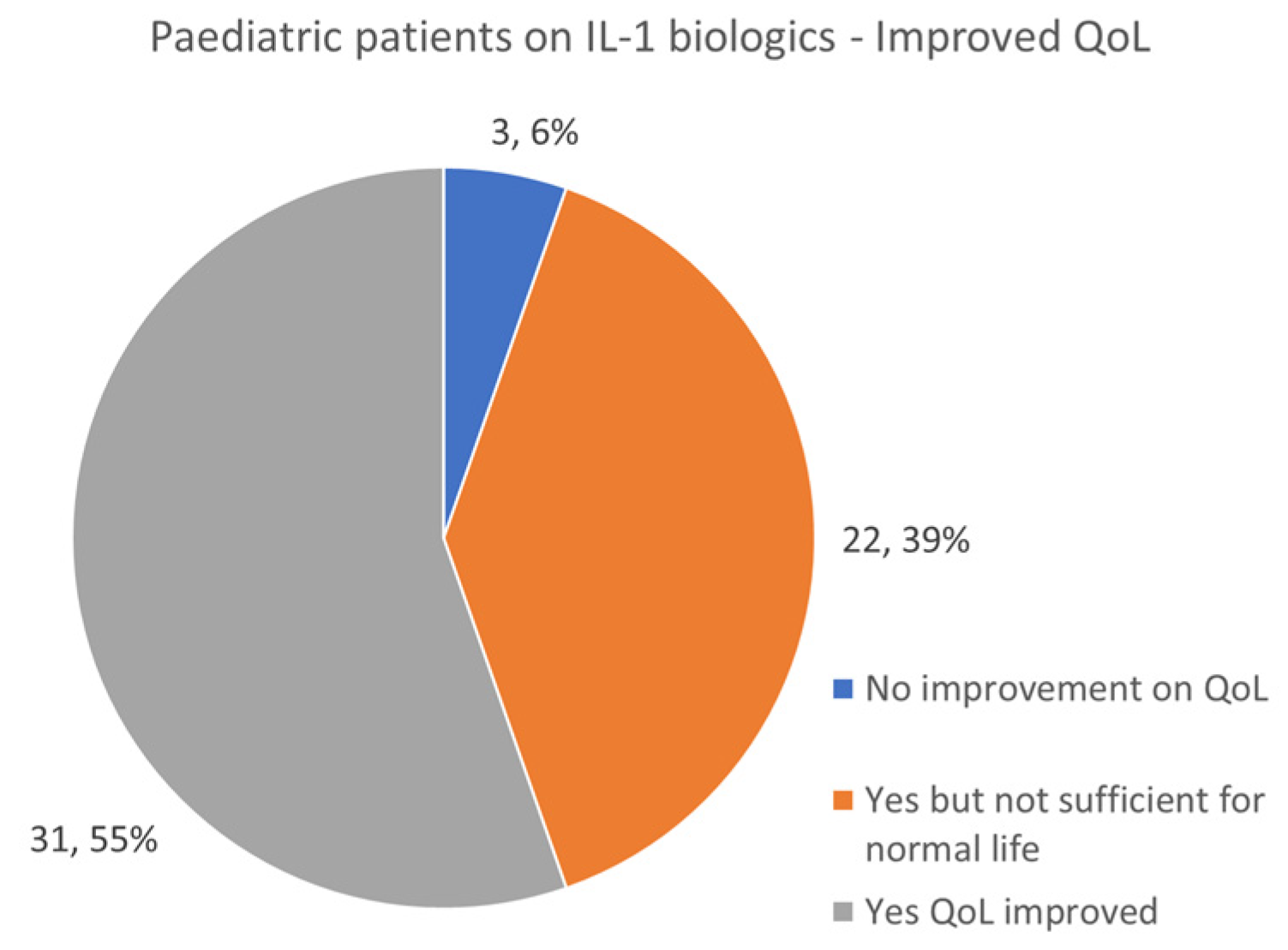
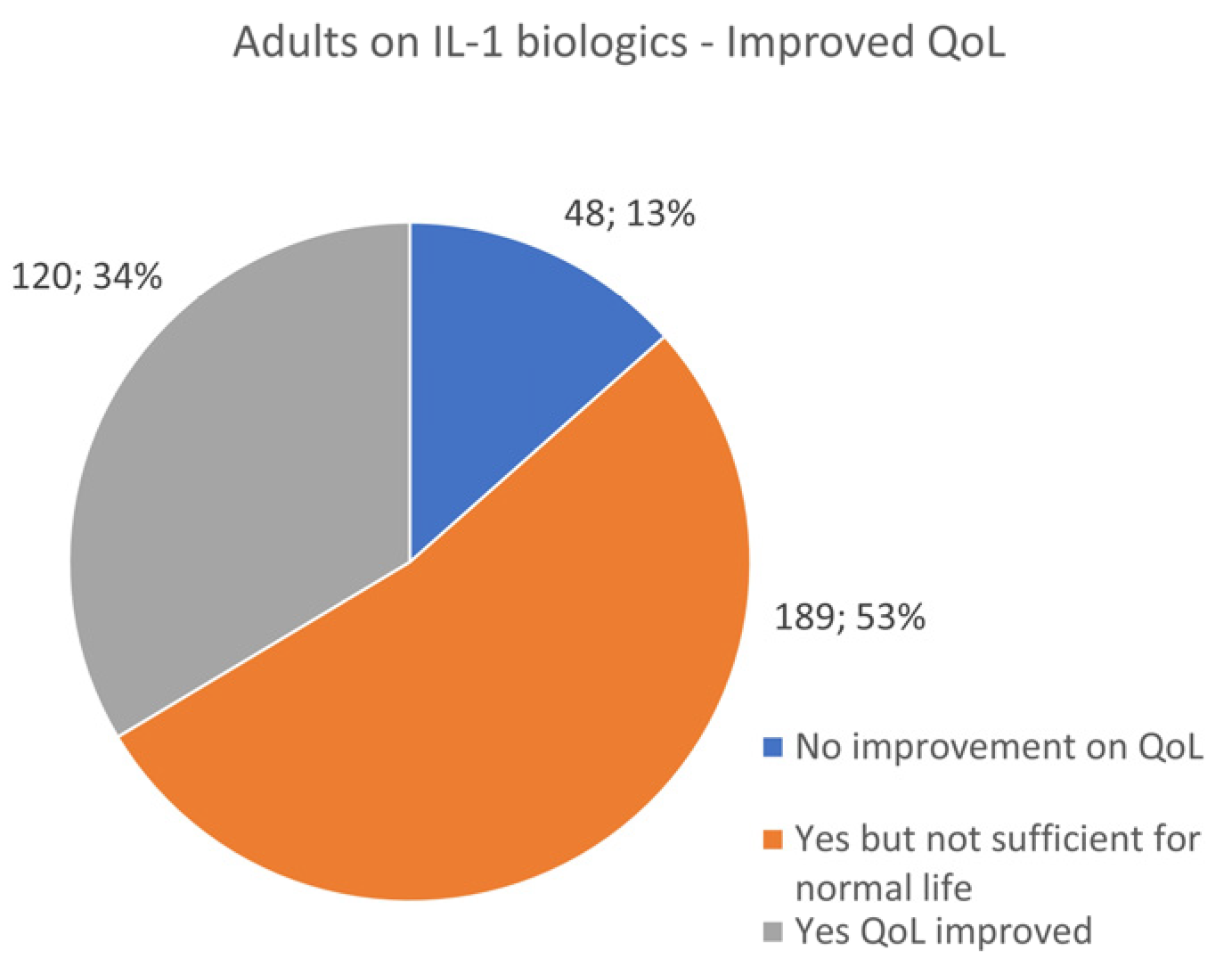
3.9. IL-1 Biologic Use in Pediatrics and Adults—Quality of Life Improvement
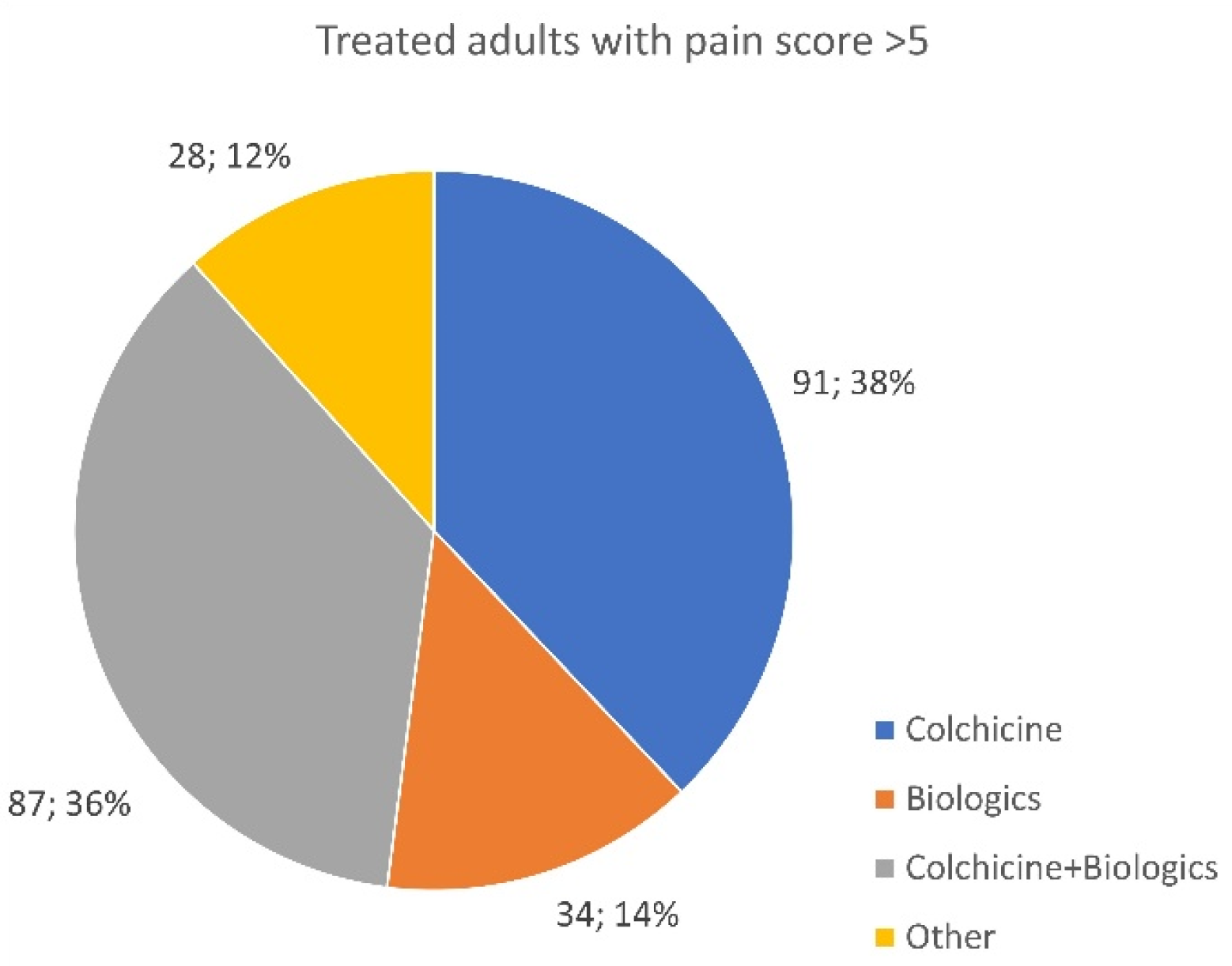
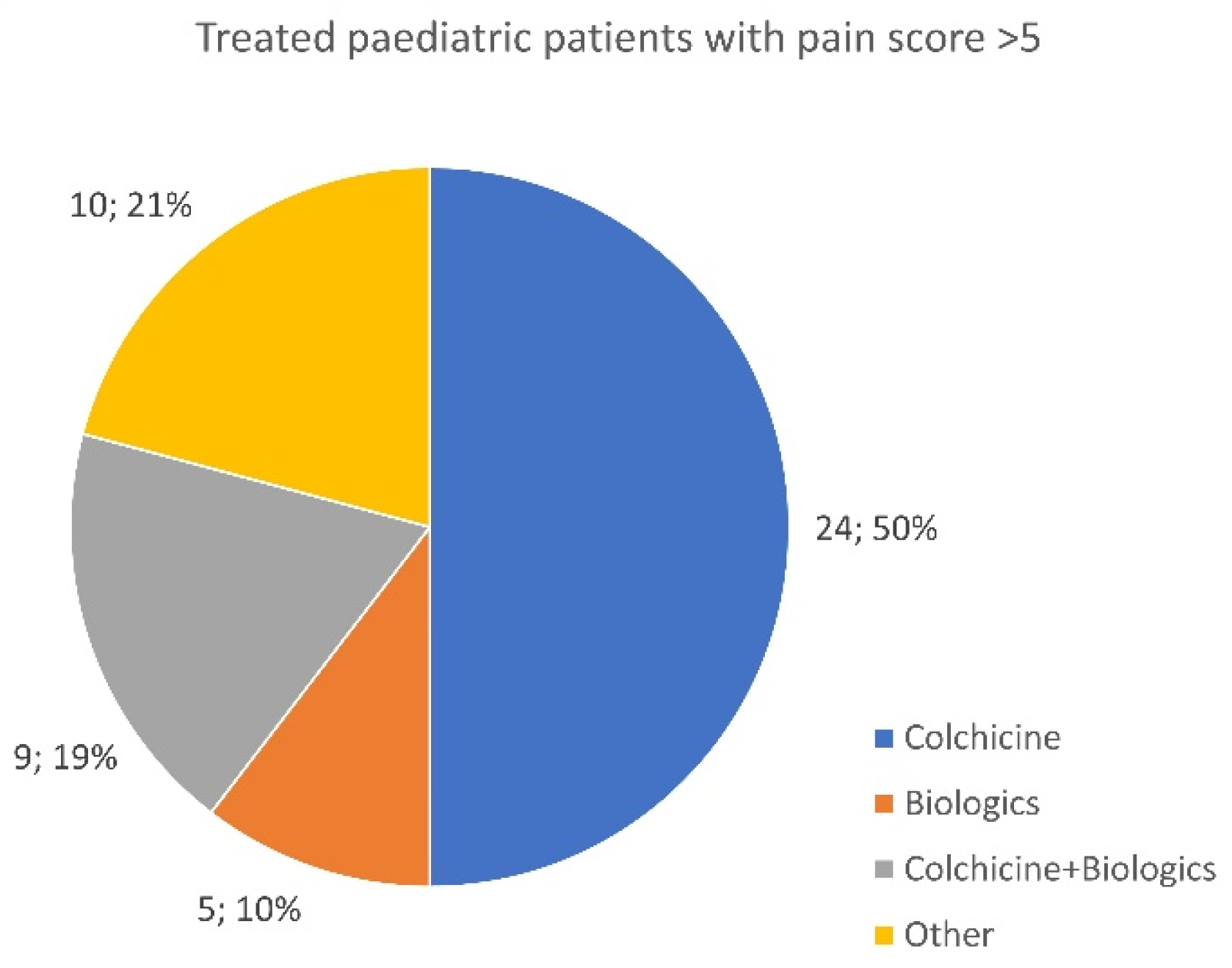
3.10. Treated Patients Recording a 5+ Pain Score
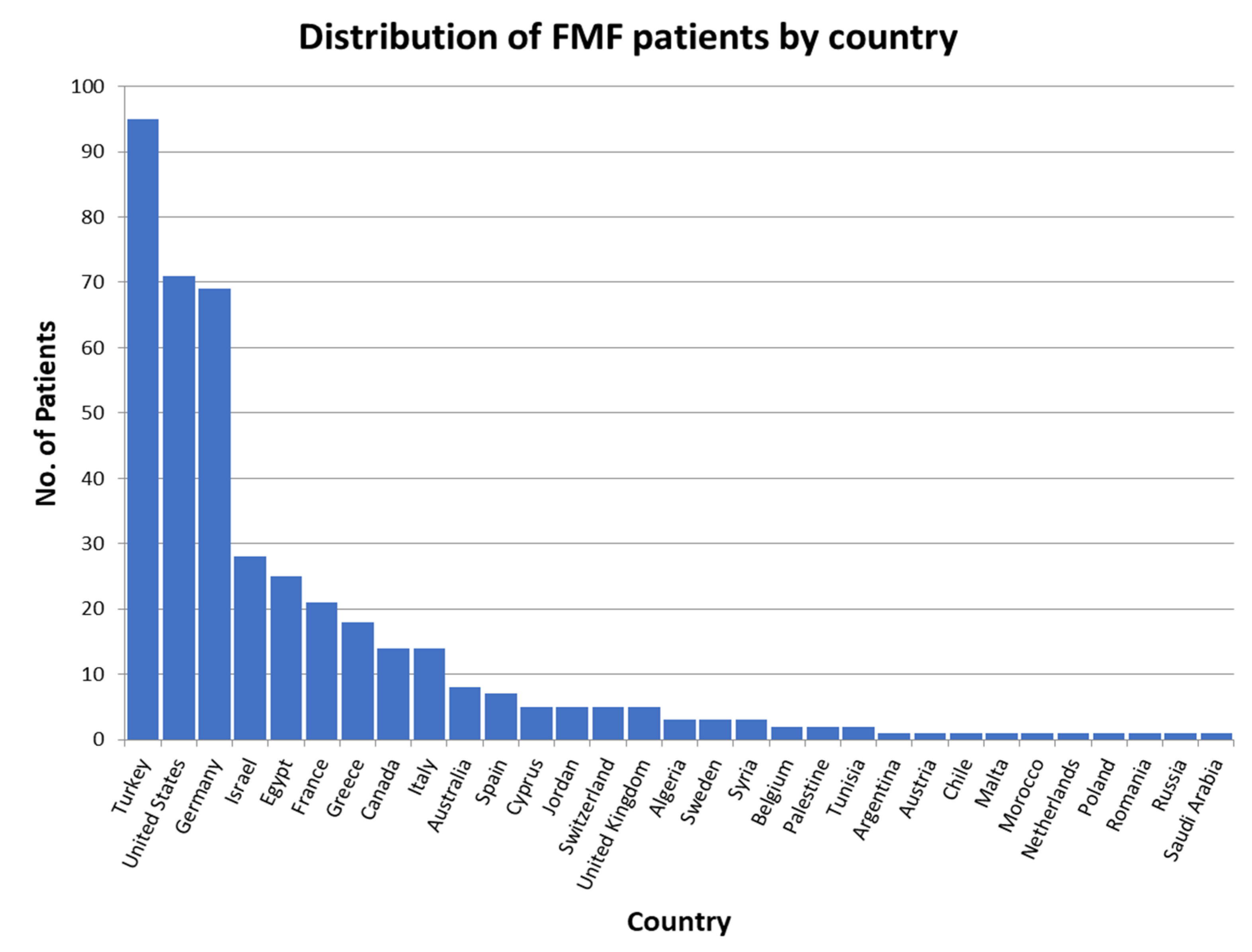
3.11. Global Distribution of Familial Mediterranean Fever Cases
3.12. FMF Patients QoL Improvement after Treatment
3.13. Global Distribution of Behçet’s Cases
3.14. Improving the Quality of Life of Behcet Patients after Treatment
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- McDermott, M.F.; Aksentijevich, I.; Galon, J.; McDermott, E.M.; Ogunkolade, B.; Centola, M.; Mansfield, E.; Gadina, M.; Karenko, L.; Pettersson, T.; et al. Germline mutations in the extracellular domains of the 55 kDa TNF receptor, TNFR1, define a family of dominantly inherited autoinflammatory syndromes. Cell 1999, 97, 133–144. [Google Scholar] [CrossRef] [PubMed]
- Booty, M.G.; Chae, J.J.; Masters, S.L.; Remmers, E.F.; Barham, B.; Le, J.M.; Barron, K.S.; Holland, S.M.; Kastner, D.L.; Aksentijevich, I. Familial Mediterranean fever with a single MEFV mutation: Where is the second hit? Arthritis Rheum. 2009, 60, 1851–1861. [Google Scholar] [CrossRef]
- Alpsoy, E.; Leccese, P.; Emmi, G.; Ohno, S. Treatment of Behçet’s Disease: An Algorithmic Multidisciplinary Approach. Front. Med. 2021, 8, 624795. [Google Scholar] [CrossRef]
- Marek-Yagel, D.; Berkun, Y.; Padeh, S.; Abu, A.; Reznik-Wolf, H.; Livneh, A.; Pras, M.; Pras, E. Clinical disease among patients heterozygous for familial mediterranean fever. Arthritis Rheum. 2009, 60, 1862–1866. [Google Scholar] [CrossRef]
- Gedik, K.C.; Lamot, L.; Romano, M.; Demirkaya, E.; Piskin, D.; Torreggiani, S.; Adang, L.A.; Armangue, T.; Barchus, K.; Cordova, D.R.; et al. The 2021 European Alliance of Associations for Rheumatology/American College of Rheumatology points to consider for diagnosis and management of autoinflammatory type I interferonopathies: CANDLE/PRAAS, SAVI and AGS. Ann. Rheum. Dis. 2022, 81, 601–613. [Google Scholar] [CrossRef] [PubMed]
- Yeh, T.C.; Chen, W.S.; Chang, Y.S.; Lin, Y.C.; Huang, Y.H.; Tsai, C.Y.; Chen, J.H.; Chang, C.C. Risk of obstructive sleep apnea in patients with Sjögren syndrome and Behçet’s disease: A nationwide, population-based cohort study. Sleep Breath 2020, 24, 1199–1205. [Google Scholar] [CrossRef] [PubMed]
- Ozen, S.; Demirkaya, E.; Erer, B.; Livneh, A.; Ben-Chetrit, E.; Giancane, G.; Ozdogan, H.; Abu, I.; Gattorno, M.; Hawkins, P.N.; et al. EULAR recommendations for the management of familial Mediterranean fever. Ann. Rheum. Dis. 2016, 75, 644–651. [Google Scholar] [CrossRef]
- Ozen, S.; Kone-Paut, I.; Gül, A. Colchicine resistance and intolerance in familial mediterranean fever: Definition, causes, and alternative treatments. Semin. Arthritis Rheum. 2017, 47, 115–120. [Google Scholar] [CrossRef]
- Baglan, E.; Ozdel, S.; Bulbul, M. Do all colchicine preparations have the same effectiveness in patients with familial Mediterranean fever? Mod. Rheumatol. 2021, 31, 481–484. [Google Scholar] [CrossRef]
- Emmungil, H.; Ilgen, U.; Turan, S.; Yaman, S.; Küçükşahin, O. Different pharmaceutical preparations of colchicine for Familial Mediterranean Fever: Are they the same? Rheumatol. Int. 2020, 40, 129–135. [Google Scholar] [CrossRef]
- Tufan, A.; Babaoglu, M.O.; Akdogan, A.; Yasar, U.; Calguneri, M.; Kalyoncu, U.; Karadag, O.; Hayran, M.; Ertenli, A.I.; Bozkurt, A.; et al. Association of drug transporter gene ABCB1 (MDR1) 3435C to T polymorphism with colchicine response in familial Mediterranean fever. J. Rheumatol. 2007, 34, 1540–1544. [Google Scholar] [PubMed]
- Broderick, L.; Hoffman, H.M. IL-1 and autoinflammatory disease: Biology, pathogenesis and therapeutic targeting. Nat. Rev. Rheumatol. 2022, 18, 448–463. [Google Scholar] [CrossRef] [PubMed]
- Vana, D.R. Canakinumab (ILARIS®) in cryopyrin-associated periodic syndrome (CAPS) patients below 2 years of age: Review of four cases and literature. Curr. Pediatr. Res. 2017, 21, 646–651. [Google Scholar]
- Sibley, C.H.; Chioato, A.; Felix, S.; Colin, L.; Chakraborty, A.; Plass, N.; Rodriguez-Smith, J.; Brewer, C.; King, K.; Zalewski, C.; et al. A 24-month open-label study of canakinumab in neonatal-onset multisystem inflammatory disease. Ann. Rheum. Dis. 2015, 74, 1714–1719. [Google Scholar] [CrossRef]
- Fingerhutová, Š.; Jančová, E.; Doležalová, P. Anakinra in Paediatric Rheumatology and Periodic Fever Clinics: Is the Higher Dose Safe? Front. Pediatr. 2022, 10, 823847. [Google Scholar] [CrossRef]
- Savic, S.; Coe, J.; Laws, P. Autoinflammation: Interferonopathies and Other Autoinflammatory Diseases. J. Investig. Dermatol. 2022, 142, 781–792. [Google Scholar] [CrossRef]
- Tangye, S.G.; Al-Herz, W.; Bousfiha, A.; Cunningham-Rundles, C.; Franco, J.L.; Holland, S.M.; Klein, C.; Morio, T.; Oksenhendler, E.; Picard, C.; et al. Human Inborn Errors of Immunity: 2022 Update on the Classification from the International Union of Immunological Societies Expert Committee. J. Clin. Immunol. 2022, 42, 1473–1507. [Google Scholar] [CrossRef]
- Poulter, J.A.; Savic, S. Genetics of somatic auto-inflammatory disorders. Semin. Hematol. 2021, 58, 212–217. [Google Scholar] [CrossRef]
- Sharma, P.; Jain, A.; Scaria, V. Genetic Landscape of Rare Autoinflammatory Disease Variants in Qatar and Middle Eastern Populations Through the Integration of Whole-Genome and Exome Datasets. Front. Genet. 2021, 12, 631340. [Google Scholar] [CrossRef] [PubMed]
- Hammam, N.; Li, J.; Evans, M.; Kay, J.L.; Izadi, Z.; Anastasiou, C.; Gianfrancesco, M.A.; Yazdany, J.; Schmajuk, G. Epidemiology and treatment of Behçet’s disease in the USA: Insights from the Rheumatology Informatics System for Effectiveness (RISE) Registry with a comparison with other published cohorts from endemic regions. Arthritis Res. Ther. 2021, 23, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Yepiskoposyan, L.; Harutyunyan, A. Population genetics of familial Mediterranean fever: A review. Eur. J. Hum. Genet. 2007, 15, 911–916. [Google Scholar] [CrossRef] [PubMed]
- Krainer, J.; Siebenhandl, S.; Weinhäusel, A. Systemic autoinflammatory diseases. J. Autoimmun. 2020, 109, 102421. [Google Scholar] [CrossRef] [PubMed]
- Shen, X.; Song, S.; Li, C.; Zhang, J. Synonymous mutations in representative yeast genes are mostly strongly non-neutral. Nature 2022, 606, 725–731. [Google Scholar] [CrossRef] [PubMed]
- Arpacı, A.; Doğan, S.; Erdoğan, H.F.; El, Ç.; Cura, S.E. Presentation of a new mutation in FMF and evaluating the frequency of distribution of the MEFV gene mutation in our region with clinical findings. Mol. Biol. Rep. 2021, 48, 2025–2033. [Google Scholar] [CrossRef]
- Çankaya, T.; Bora, E.; Bayram, M.T.; Ülgenalp, A.; Kavukçu, S.; Türkmen, M.A.; Soylu, A. Clinical Significance of R202Q Alteration of MEFV Gene in Children With Familial Mediterranean Fever. Arch. Rheumatol. 2015, 30, 51–56. [Google Scholar] [CrossRef]
- Faulk, T.; Carroll, M.B. Case Report: A Patient Helps Diagnose Familial Mediterranean Fever. The Rheumatologist, 16 August 2019. [Google Scholar]
- Topaloglu, R.; Ozaltin, F.; Yilmaz, E.; Ozen, S.; Balci, B.; Besbas, N.; Bakkaloglu, A. E148Q is a disease-causing MEFV mutation: A phenotypic evaluation in patients with familial Mediterranean fever. Ann. Rheum. Dis. 2005, 64, 750–752. [Google Scholar] [CrossRef]
- El Roz, A.; Ghssein, G.; Khalaf, B.; Fardoun, T.; Ibrahim, J.-N. Spectrum of MEFV Variants and Genotypes among Clinically Diagnosed FMF Patients from Southern Lebanon. Med. Sci. 2020, 8, 35. [Google Scholar] [CrossRef]
- Sohar, E.; Gafni, J.; Pras, M.; Heller, H. Familial Mediterranean fever. A survey of 470 cases and review of the literature. Am. J. Med. 1967, 43, 227–253. [Google Scholar] [CrossRef]
- Yoshida, S.; Sumichika, Y.; Saito, K.; Matsumoto, H.; Temmoku, J.; Fujita, Y.; Matsuoka, N.; Asano, T.; Sato, S.; Migita, K. Effectiveness of Colchicine or Canakinumab in Japanese Patients with Familial Mediterranean Fever: A Single-Center Study. J. Clin. Med. 2023, 12, 6272. [Google Scholar] [CrossRef] [PubMed]
- Özen, S.; Batu, E.D.; Demir, S. Familial Mediterranean Fever: Recent Developments in Pathogenesis and New Recommendations for Management. Front. Immunol. 2017, 8, 253. [Google Scholar] [CrossRef] [PubMed]
- Ozdogan, H.; Ugurlu, S. Familial Mediterranean Fever. La Presse Médicale 2019, 48, e61–e76. [Google Scholar] [CrossRef] [PubMed]
- Kuemmerle-Deschner, J.B.; Verma, D.; Endres, T.; Broderick, L.; de Jesus, A.A.; Hofer, F.; Blank, N.; Krause, K.; Rietschel, C.; Horneff, G.; et al. Clinical and Molecular Phenotypes of Low-Penetrance Variants of NLRP3: Diagnostic and Therapeutic Challenges. Arthritis Rheumatol. 2017, 69, 2233–2240. [Google Scholar] [CrossRef] [PubMed]
- Verma, D.; Särndahl, E.; Andersson, H.; Eriksson, P.; Fredrikson, M.; Jönsson, J.-I.; Lerm, M.; Söderkvist, P. The Q705K polymorphism in NLRP3 is a gain-of-function alteration leading to excessive interleukin-1β and IL-18 production. PLoS ONE 2012, 7, e34977. [Google Scholar] [CrossRef] [PubMed]
- Theodoropoulou, K.; Wittkowski, H.; Busso, N.; Von Scheven-Gête, A.; Moix, I.; Vanoni, F.; Hengten, V.; Horneff, G.; Haas, J.P.; Fischer, N.; et al. Increased Prevalence of NLRP3 Q703K Variant Among Patients With Autoinflammatory Diseases: An In-ternational Multicentric Study. Front. Immunol. 2020, 11, 877. [Google Scholar] [CrossRef] [PubMed]
- von Mühlenen, I.; Gabay, C.; Finckh, A.; Kuemmerle-Deschner, J. NLRP3 Q703K and TNFRSF1A R92Q mutations in a patient with auto inflammatory disease. Pediatr. Rheumatol. 2015, 13, P47. [Google Scholar] [CrossRef][Green Version]
- E Saper, V.; Ombrello, M.J.; Tremoulet, A.H.; Montero-Martin, G.; Prahalad, S.; Canna, S.; Shimizu, C.; Deutsch, G.; Tan, S.Y.; Remmers, E.F.; et al. Severe delayed hypersensitivity reactions to IL-1 and IL-6 inhibitors link to common HLA-DRB1*15 alleles. Ann. Rheum. Dis. 2022, 81, 406–415. [Google Scholar] [CrossRef]
- Sezgin, E.; Kaplan, E. Diverse selection pressures shaping the genetic architecture of behçet disease susceptibility. Front. Genet. 2022, 13, 983646. [Google Scholar] [CrossRef]
- Sazzini, M.; Garagnani, P.; Sarno, S.; De Fanti, S.; Lazzano, T.; Yang Yao, D.; Boattini, A.; Pazzola, G.; Maramotti, S.; Boiardi, L.; et al. Tracing Behçet’s disease origins along the Silk Road: An anthropological evolu-tionary genetics perspective. Clin. Exp. Rheumatol. 2015, 33 (Suppl. 94), S60–S66. [Google Scholar]
- Touitou, I.; Lesage, S.; McDermott, M.; Cuisset, L.; Hoffman, H.; Dode, C.; Shoham, N.; Aganna, E.; Hugot, J.P.; Wise, C.; et al. Infevers: An evolving mutation database for auto-inflammatory syndromes. Hum Mutat. 2004, 24, 194–198. [Google Scholar] [CrossRef]
- Price, T.J.; Gold, M.S. From Mechanism to Cure: Renewing the Goal to Eliminate the Disease of Pain. Pain Med. 2018, 19, 1525–1549. [Google Scholar] [CrossRef]
- Moens, M.; Jansen, J.; De Smedt, A.; Roulaud, M.; Billot, M.; Laton, J.; Rigoard, P.; Goudman, L. Acceptance and Commitment Therapy to Increase Resilience in Chronic Pain Patients: A Clinical Guideline. Medicina 2022, 58, 499. [Google Scholar] [CrossRef] [PubMed]
- Totsch, S.K.; Sorge, R.E. Immune System Involvement in Specific Pain Conditions. Mol. Pain 2017, 13, 1744806917724559. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.C.; Meckfessel, M.H. Autoinflammatory disorders, pain, and neural regulation of inflammation. Dermatol. Clin. 2013, 31, 461–470. [Google Scholar] [CrossRef] [PubMed]
- de Moraes, M.P.M.; Nascimento, R.R.N.R.D.; Abrantes, F.F.; Pedroso, J.L.; Perazzio, S.F.; Barsottini, O.G.P. What General Neurologists Should Know about Autoinflammatory Syndromes? Brain Sci. 2023, 13, 1351. [Google Scholar] [CrossRef] [PubMed]
- Gok, I.; Huseyinoglu, N.; Ilhan, D. Genetic polymorphisms variants in interleukin-6 and interleukin- 1beta patients with obstructive sleep apnea syndrome in East Northern Turkey. Med. Glas. 2015, 12, 216–222. [Google Scholar] [CrossRef]
- Renson, T.; Hamiwka, L.; Benseler, S. Central nervous system manifestations of monogenic autoinflammatory disorders and the neurotropic features of SARS-CoV-2: Drawing the parallels. Front. Pediatr. 2022, 10, 931179. [Google Scholar] [CrossRef] [PubMed]
- Uccelli, A.; Gattorno, M. Neurological manifestations in autoinflammatory diseases. Clin. Exp. Rheumatol. 2018, 36 (Suppl. 110), 61–67. [Google Scholar]
- Pisanti, S.; Rimondi, E.; Pozza, E.; Melloni, E.; Zauli, E.; Bifulco, M.; Martinelli, R.; Marcuzzi, A. Prenylation Defects and Oxidative Stress Trigger the Main Consequences of Neuroinflammation Linked to Mevalonate Pathway Deregulation. Int. J. Environ. Res. Public Heal. 2022, 19, 9061. [Google Scholar] [CrossRef]
- Ben-Chetrit, E.; Gattorno, M.; Gul, A.; Kastner, D.L.; Lachmann, H.J.; Touitou, I.; Ruperto, N. Consensus proposal for taxonomy and definition of the autoinflammatory diseases (AIDs): A Delphi study. Ann. Rheum. Dis. 2018, 77, 1558–1565. [Google Scholar] [CrossRef]
- Senusi, A.; Seoudi, N.; Bergmeier, L.A.; Fortune, F. Genital ulcer severity score and genital health quality of life in Behçet’s disease. Orphanet. J. Rare Dis. 2015, 10, 117. [Google Scholar] [CrossRef]
- Irwin, M.R. Sleep and inflammation: Partners in sickness and in health. Nat. Rev. Immunol. 2019, 19, 702–715. [Google Scholar] [CrossRef] [PubMed]
- Jewett, K.A.; Krueger, J.M. Humoral sleep regulation; interleukin-1 and tumor necrosis factor. Vitam. Horm. 2012, 89, 241–257. [Google Scholar] [CrossRef] [PubMed]
- American Autoimmune Related Diseases Association (AARDA). Profound, Debiliating Fatigue Found to Be a Major Issue for Autoimmune Disease Patients in New National Survey. ScienceDaily, 23 March 2015.
- Nikolaus, S.; Bode, C.; Taal, E.; van de Laar, M.A.F.J. Fatigue and factors related to fatigue in rheumatoid arthritis: A systematic review. Arthritis Care Res. 2013, 65, 1128–1146. [Google Scholar] [CrossRef] [PubMed]
- Bergman, M.J.; Shahouri, S.S.; Shaver, T.S.; Anderson, J.D.; Weidensaul, D.N.; Busch, R.E.; Wang, S.; Wolfe, F. Is fatigue an inflammatory variable in rheumatoid arthritis (RA)? Analyses of fatigue in RA, osteoarthritis, and fibromyalgia. J. Rheumatol. 2009, 36, 2788–2794. [Google Scholar] [CrossRef] [PubMed]
- Pollard, L.C.; Choy, E.H.; Gonzalez, J.; Khoshaba, B.; Scott, D.L. Fatigue in rheumatoid arthritis reflects pain, not disease activity. Rheumatology 2006, 45, 885–889. [Google Scholar] [CrossRef]
- van Dartel, S.A.; Repping-Wuts, J.W.; van Hoogmoed, D.; Bleijenberg, G.; van Riel, P.L.; Fransen, J. Association between fatigue and pain in rheumatoid arthritis: Does pain precede fatigue or does fatigue precede pain? Arthritis Care Res. 2013, 65, 862–869. [Google Scholar] [CrossRef]
- Dantzer, R.; Heijnen, C.J.; Kavelaars, A.; Laye, S.; Capuron, L. The neuroimmune basis of fatigue. Trends Neurosci. 2013, 37, 39–46. [Google Scholar] [CrossRef]
- Morris, G.; Berk, M.; Walder, K.; Maes, M. Central pathways causing fatigue in neuro-inflammatory and autoimmune illnesses. BMC Med. 2015, 13, 1–23. [Google Scholar] [CrossRef]
- Zielinski, M.R.; McKenna, J.T.; McCarley, R.W. Functions and Mechanisms of Sleep. AIMS Neurosci. 2016, 3, 67–104. [Google Scholar] [CrossRef]
- Kelesoglu, F.M.; Aygun, E.; Okumus, N.K.; Ersoy, A.; Karapınar, E.; Saglam, N.; Aydın, N.G.; Senay, B.B.; Gonultas, S.; Sarisik, E.; et al. Evaluation of subclinical inflammation in familial Mediterranean fever patients: Relations with mutation types and attack status: A retrospective study. Clin. Rheumatol. 2016, 35, 2757–2763. [Google Scholar] [CrossRef]





Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rech, J.; Schett, G.; Tufan, A.; Kuemmerle-Deschner, J.B.; Özen, S.; Tascilar, K.; Geck, L.; Krickau, T.; Cohen, E.; Welzel, T.; et al. Patient Experiences and Challenges in the Management of Autoinflammatory Diseases—Data from the International FMF & AID Global Association Survey. J. Clin. Med. 2024, 13, 1199. https://doi.org/10.3390/jcm13051199
Rech J, Schett G, Tufan A, Kuemmerle-Deschner JB, Özen S, Tascilar K, Geck L, Krickau T, Cohen E, Welzel T, et al. Patient Experiences and Challenges in the Management of Autoinflammatory Diseases—Data from the International FMF & AID Global Association Survey. Journal of Clinical Medicine. 2024; 13(5):1199. https://doi.org/10.3390/jcm13051199
Chicago/Turabian StyleRech, Jürgen, Georg Schett, Abdurrahman Tufan, Jasmin B. Kuemmerle-Deschner, Seza Özen, Koray Tascilar, Leonie Geck, Tobias Krickau, Ellen Cohen, Tatjana Welzel, and et al. 2024. "Patient Experiences and Challenges in the Management of Autoinflammatory Diseases—Data from the International FMF & AID Global Association Survey" Journal of Clinical Medicine 13, no. 5: 1199. https://doi.org/10.3390/jcm13051199
APA StyleRech, J., Schett, G., Tufan, A., Kuemmerle-Deschner, J. B., Özen, S., Tascilar, K., Geck, L., Krickau, T., Cohen, E., Welzel, T., Kuehn, M., & Vetterli, M. (2024). Patient Experiences and Challenges in the Management of Autoinflammatory Diseases—Data from the International FMF & AID Global Association Survey. Journal of Clinical Medicine, 13(5), 1199. https://doi.org/10.3390/jcm13051199






