Evaluation of Mesh Closure of Laparotomy and Extraction Incisions in Open and Laparoscopic Colorectal Surgery: A Systematic Review and Meta-Analysis †
Abstract
1. Introduction
2. Materials and Methods
- Randomised controlled trials (RCTs) or comparative studies.
- Studies including patients of all age groups and of any gender.
- Studies including patients who underwent colorectal surgery, either for benign or malignant conditions.
- Studies including patients who underwent laparotomy or laparoscopy with EI performed.
- Studies comparing mesh closure of laparotomy or EI versus conventional suture closure.
- Studies reporting IH and/or evisceration (wound dehiscence) as primary outcome.
- Non-comparative studies.
- Studies of mixed surgeries, not only colorectal.
- Case series, case reports, and letters.
2.1. Search Strategy
2.2. Study Selection and Data Extraction
2.3. Outcome Measures
2.4. Risk of Bias
2.5. Statistical Analysis
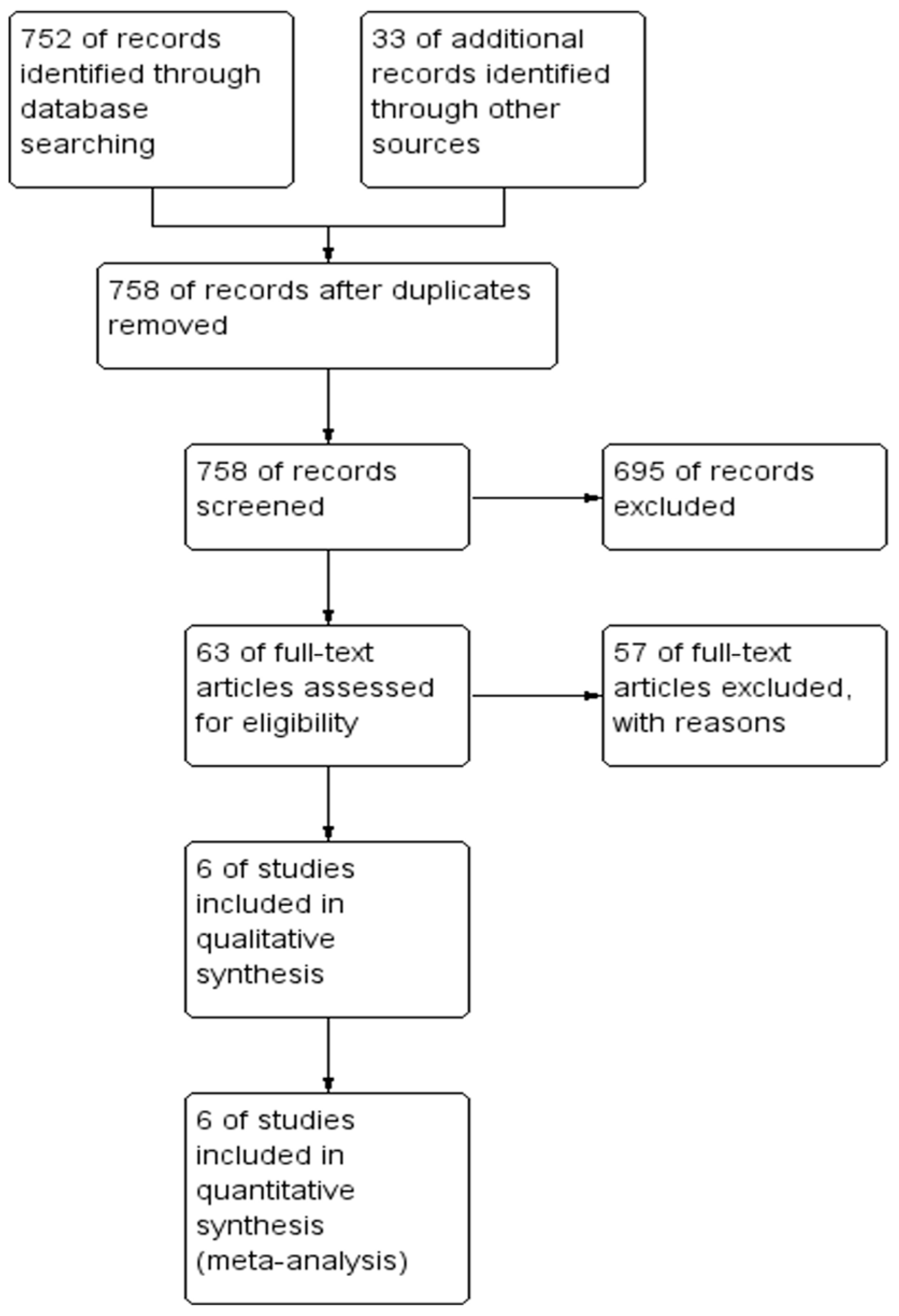
3. Results
3.1. Methodological Appraisal
3.2. Primary Outcomes
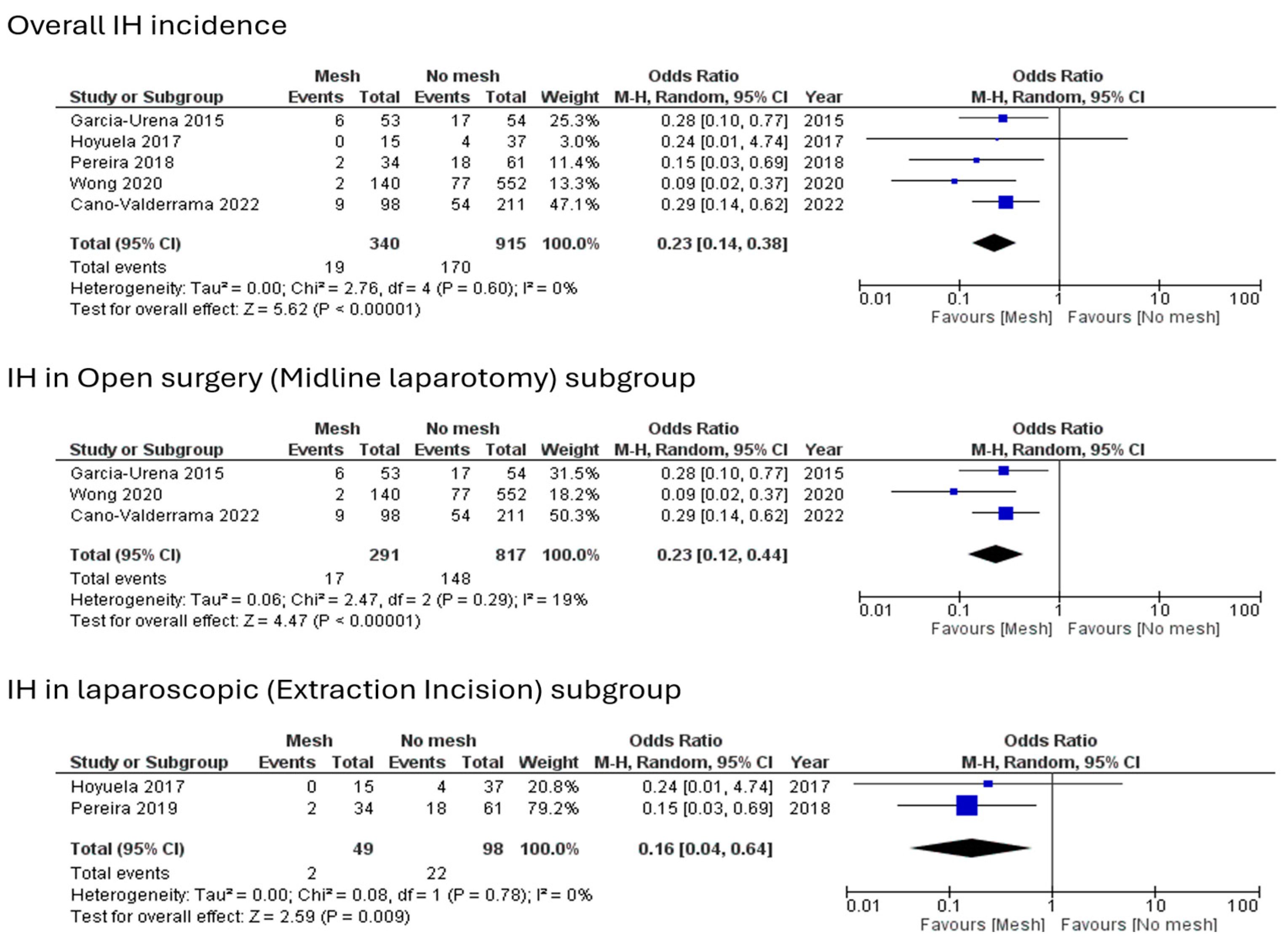
3.2.1. Incidence of Incisional Hernia
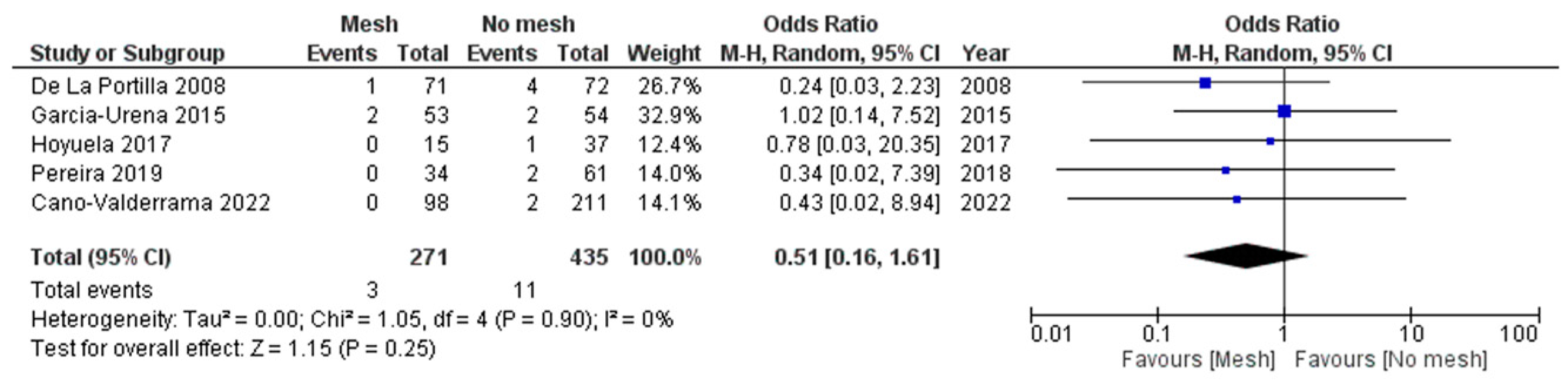
3.2.2. Evisceration Rate
3.3. Secondary Outcomes
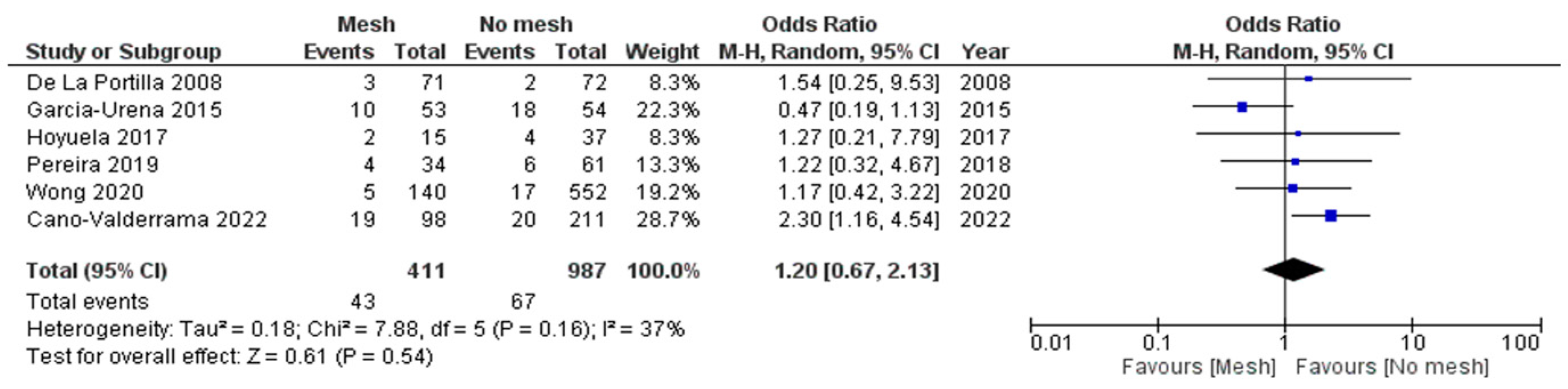
3.3.1. Surgical Site Infection (SSI)
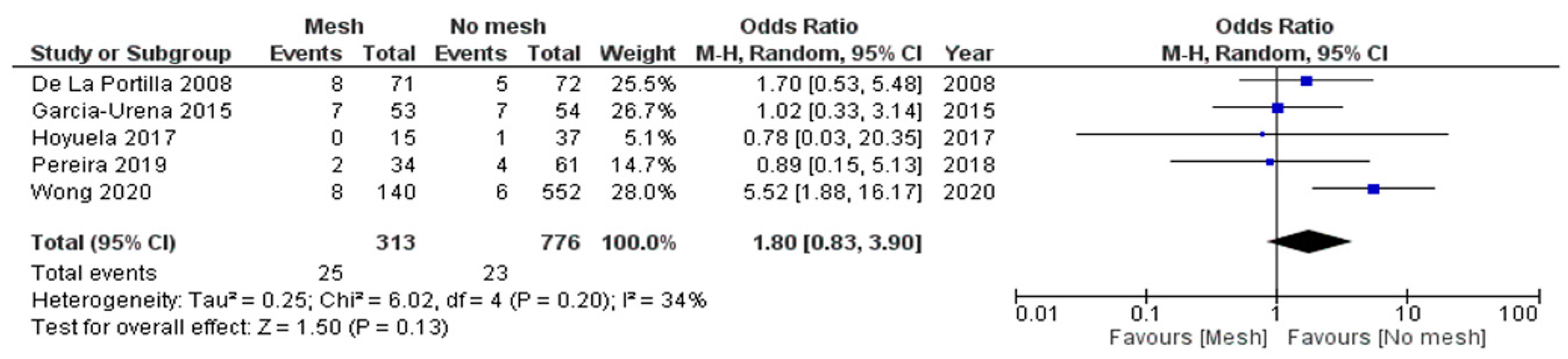
3.3.2. Seroma Formation
3.3.3. Length of Hospital Stay (LOS)
3.4. Sensitivity Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Diener, M.K.; Voss, S.; Jensen, K.; Büchler, M.W.; Seiler, C.M. Elective midline laparotomy closure: The INLINE systematic review and meta-analysis. Ann. Surg. 2010, 251, 843–856. [Google Scholar] [CrossRef] [PubMed]
- Moussavian, M.R.; Schuld, J.; Dauer, D.; Justinger, C.; Kollmar, O.; Schilling, M.K.; Richter, S. Long term follow up for incisional hernia after severe secondary peritonitis—Incidence and risk factors. Am. J. Surg. 2010, 200, 229–234. [Google Scholar] [CrossRef] [PubMed]
- Köckerling, F. Rcurrent Incisional Hernia Repair—An Overview. Front. Surg. 2019, 6, 26. [Google Scholar] [CrossRef]
- El-Khadrawy, O.; Moussa, G.; Mansour, O.; Hashish, M. Prophylactic prosthetic reinforcement of midline abdominal incisions in high-risk patients. Hernia 2009, 13, 267–274. [Google Scholar] [CrossRef]
- García-Ureña, M.Á.; López-Monclús, J.; Hernando, L.A.B.; Montes, D.M.; de Lersundi, A.R.V.; Pavón, C.C.; Ceinos, C.J.; Quindós, P.L. Randomized controlled trial of the use of a large-pore polypropylene mesh to prevent incisional hernia in colorectal surgery. Ann. Surg. 2015, 261, 876–881. [Google Scholar] [CrossRef]
- Cano-Valderrama, O.; Sanz-López, R.; Domínguez-Serrano, I.; Dziakova, J.; Catalán, V.; Rojo, M.; García-Alonso, M.; Mugüerza, J.M.; Torres, A.J. Extraction-site incisional hernia after laparoscopic colorectal surgery: Should we carry out a study about prophylactic mesh closure? Surg. Endosc. 2020, 34, 4048–4052. [Google Scholar] [CrossRef]
- van Ramshorst, G.H.; Eker, H.H.; Hop, W.C.; Jeekel, J.; Lange, J.F. Impact of incisional hernia on health-related quality of life and body image: A prospective cohort study. Am. J. Surg. 2012, 204, 144–150. [Google Scholar] [CrossRef]
- Gillion, J.F.; Sanders, D.; Miserez, M.; Muysoms, F. The economic burden of incisional ventral hernia repair: A multicentric cost analysis. Hernia 2016, 20, 819–830. [Google Scholar] [CrossRef]
- Harries, R.L.; Torkington, J. Stomal Closure: Strategies to Prevent Incisional Hernia. Front. Surg. 2018, 5, 28. [Google Scholar] [CrossRef]
- Hoffman, R.D.; Danos, D.M.; Lau, F.H. National health disparities in incisional hernia repair outcomes: An analysis of the Healthcare Cost and Utilization Project National Inpatient Sample (HCUP-NIS) 2012–2014. Surgery 2021, 169, 1393–1399. [Google Scholar] [CrossRef]
- Pawlak, M.; Tulloh, B.; de Beaux, A. Current trends in hernia surgery in NHS England. Ann. R. Coll. Surg. Engl. 2020, 102, 25–27. [Google Scholar] [CrossRef] [PubMed]
- Muysoms, F.E.; Miserez, M.; Berrevoet, F.; Campanelli, G.; Champault, G.G.; Chelala, E.; Dietz, U.A.; Eker, H.H.; El Nakadi, I.; Hauters, P.; et al. Classification of primary and incisional abdominal wall hernias. Hernia 2009, 13, 407–414. [Google Scholar] [CrossRef] [PubMed]
- Alnassar, S.; Bawahab, M.; Abdoh, A.; Guzman, R.; Al Tuwaijiri, T.; Louridas, G. Incisional hernia postrepair of abdominal aortic occlusive and aneurysmal disease: Five-year incidence. Vascular 2012, 20, 273–277. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.; Green, S. (Eds.) Cochrane Handbook for Systematic Reviews of Interventions; Version 5.1. 0; The Cochrane Collaboration, 2011. Available online: https://handbook-5-1.cochrane.org/ (accessed on 28 September 2024).
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. Int. J. Surg. 2010, 8, 336–341. [Google Scholar] [CrossRef]
- Albendary, M.; Issa, M.; Mohamedahmed, A.Y. WP1.14—Evaluation of mesh closure of laparotomy and extraction incisions in open and laparoscopic colorectal surgery: A systematic review and meta-analysis. In Proceedings of the ASGBI Conference, Belfast, UK, 8–10 May 2024. [Google Scholar]
- Higgins, J.P.; Altman, D.G.; Gøtzsche, P.C.; Jüni, P.; Moher, D.; Oxman, A.D.; Savović, J.; Schulz, K.F.; Weeks, L.; Sterne, J.A. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. Bmj 2011, 343, d5928. [Google Scholar] [CrossRef]
- Wells, G.A.; Shea, B.; O’Connell, D.; Peterson, J.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Nonrandomised Studies in Meta-Analyses. ScienceOpen 2000. Available online: https://www.scienceopen.com/document?vid=54b48470-4655-4081-b5d4-e8ebe8d1792e (accessed on 28 September 2024).
- Hozo, S.P.; Djulbegovic, B.; Hozo, I. Estimating the mean and variance from the median, range, and the size of a sample. BMC Med. Res. Methodol. 2005, 5, 13. [Google Scholar] [CrossRef]
- de la Portilla, F.; Flikier, B.; Espinosa, E.; Utrera, A.; Rada, R.; Vega, J.; Cisneros, N.; Maldonado, V.H. Randomised study of the use resorbable mesh for the prevention of evisceration in colorectal surgery. Cir. Esp. 2008, 83, 12–17. [Google Scholar] [CrossRef]
- Hoyuela, C.; Juvany, M.; Trias, M.; Ardid, J.; Martrat, A. Incisional hernia prevention using a cyanoacrilate-fixed retrofascial mesh. Cir. Esp. 2018, 96, 35–40. [Google Scholar] [CrossRef]
- Pereira, J.A.; Pera, M.; López-Cano, M.; Pascual, M.; Alonso, S.; Salvans, S.; Jiménez-Toscano, M.; González-Martín, A.; Grande-Posa, L. Hernias at the extraction incision after laparoscopic colon and rectal resection: Influence of incision location and use of prophylactic mesh. Cir. Esp. 2019, 97, 20–26. [Google Scholar] [CrossRef]
- Wong, J.; Jones, J.; Ananthapadmanabhan, S.; Meagher, A.P. Abdominal wall closure with prophylactic mesh in colorectal operations. ANZ J. Surg. 2020, 90, 564–568. [Google Scholar] [CrossRef] [PubMed]
- Cano-Valderrama, O.; García-Alonso, M.; Sanz-Ortega, G.; Rojo, M.; Catalán, V.; Domínguez-Serrano, I.; Dziakova, J.; Sanz-López, R.; Torres, A.J. Is prophylactic mesh closure effective to decrease the incidence of incisional hernia after laparotomy in colorectal surgery? Acta Chir. Belg. 2022, 122, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Malviya, A.; Patel, A.; Bhardwaj, G.; Bulchandani, H.P.; Saini, V. A comprehensive study on the incidence and management of incisional hernia. Int. Surg. J. 2017, 4, 2303–2307. [Google Scholar] [CrossRef]
- Holihan, J.L.; Alawadi, Z.; Martindale, R.G.; Roth, J.S.; Wray, C.J.; Ko, T.C.; Kao, L.S.; Liang, M.K. Adverse events after ventral hernia repair: The vicious cycle of complications. J. Am. Coll. Surg. 2015, 221, 478–485. [Google Scholar] [CrossRef] [PubMed]
- Howard, R.; Thompson, M.; Fan, Z.; Englesbe, M.; Dimick, J.B.; Telem, D.A. Costs Associated with Modifiable Risk Factors in Ventral and Incisional Hernia Repair. JAMA Netw. Open 2019, 2, e191633. [Google Scholar] [CrossRef]
- Söderbäck, H.; Gunnarsson, U.; Hellman, P.; Sandblom, G. Incisional hernia after surgery for colorectal cancer: A population-based register study. Int. J. Color. Dis. 2018, 33, 1411–1417. [Google Scholar] [CrossRef]
- Jenkins, T. The burst abdominal wound: A mechanical approach. J. Br. Surg. 1976, 63, 873–876. [Google Scholar] [CrossRef]
- Deerenberg, E.B.; Harlaar, J.J.; Steyerberg, E.W.; Lont, H.E.; van Doorn, H.C.; Heisterkamp, J.; Wijnhoven, B.P.; Schouten, W.R.; Cense, H.A.; Stockmann, H.B. Small bites versus large bites for closure of abdominal midline incisions (STITCH): A double-blind, multicentre, randomised controlled trial. Lancet 2015, 386, 1254–1260. [Google Scholar] [CrossRef]
- Hernández-Granados, P.; López-Cano, M.; Morales-Conde, S.; Muysoms, F.; García-Alamino, J.; Pereira-Rodríguez, J.A. Incisional hernia prevention and use of mesh. A narrative review. Cirugía Española 2018, 96, 76–87. [Google Scholar] [CrossRef]
- Jairam, A.P.; Timmermans, L.; Eker, H.H.; Pierik, R.E.; van Klaveren, D.; Steyerberg, E.W.; Timman, R.; van der Ham, A.C.; Dawson, I.; Charbon, J.A. Prevention of incisional hernia with prophylactic onlay and sublay mesh reinforcement versus primary suture only in midline laparotomies (PRIMA): 2-year follow-up of a multicentre, double-blind, randomised controlled trial. Lancet 2017, 390, 567–576. [Google Scholar] [CrossRef]
- Durbin, B.; Spencer, A.; Briese, A.; Edgerton, C.; Hope, W.W. If Evidence is in Favor of Incisional Hernia Prevention with Mesh, why is it not Implemented? J. Abdom. Wall Surg. 2023, 2, 11000. [Google Scholar] [CrossRef] [PubMed]
- Bali, C.; Papakostas, J.; Georgiou, G.; Kouvelos, G.; Avgos, S.; Arnaoutoglou, E.; Papadopoulos, G.; Matsagkas, M. A comparative study of sutured versus bovine pericardium mesh abdominal closure after open abdominal aortic aneurysm repair. Hernia 2015, 19, 267–271. [Google Scholar] [CrossRef] [PubMed]
- Manzini, G.; Henne-Bruns, D.; Kremer, M. Severe complications after mesh migration following abdominal hernial repair: Report of two cases and review of literature. GMS Interdiscip. Plast. Reconstr. Surg. DGPW 2019, 8, Doc09. [Google Scholar] [PubMed]
- Depuydt, M.; Allaeys, M.; De Carvalho, L.A.; Vanlander, A.; Berrevoet, F. Prophylactic mesh after midline laparotomy: Evidence is out there, but why do surgeons hesitate? World J. Surg. 2021, 45, 1349–1361. [Google Scholar] [CrossRef]
- Tsirline, V.B.; Klima, D.A.; Belyansky, I.; Kercher, K.W. Chronic Pain After Ventral Hernia Repair; Springer: Berlin/Heidelberg, Germany, 2013. [Google Scholar]
- Jairam, A.P.; López-Cano, M.; García-Alamino, J.M.; Pereira, J.; Timmermans, L.; Jeekel, J.; Lange, J.; Muysoms, F. Prevention of incisional hernia after midline laparotomy with prophylactic mesh reinforcement: A meta-analysis and trial sequential analysis. BJS Open 2020, 4, 357–368. [Google Scholar] [CrossRef] [PubMed]
- Tansawet, A.; Numthavaj, P.; Techapongsatorn, S.; Wilasrusmee, C.; Attia, J.; Thakkinstian, A. Mesh position for hernia prophylaxis after midline laparotomy: A systematic review and network meta-analysis of randomized clinical trials. Int. J. Surg. 2020, 83, 144–151. [Google Scholar] [CrossRef]
- Caro-Tarrago, A.; Olona Casas, C.; Jimenez Salido, A.; Duque Guilera, E.; Moreno Fernandez, F.; Vicente Guillen, V. Prevention of incisional hernia in midline laparotomy with an onlay mesh: A randomized clinical trial. World J. Surg. 2014, 38, 2223–2230. [Google Scholar] [CrossRef]
- Caro-Tarrago, A.; Olona, C.; Millán, M.; Olona, M.; Espina, B.; Jorba, R. Long-term results of a prospective randomized trial of midline laparotomy closure with onlay mesh. Hernia 2019, 23, 335–340. [Google Scholar] [CrossRef]
- Borab, Z.M.; Shakir, S.; Lanni, M.A.; Tecce, M.G.; MacDonald, J.; Hope, W.W.; Fischer, J.P. Does prophylactic mesh placement in elective, midline laparotomy reduce the incidence of incisional hernia? A systematic review and meta-analysis. Surgery 2017, 161, 1149–1163. [Google Scholar] [CrossRef] [PubMed]
- Burns, F.A.; Heywood, E.; Challand, C.; Lee, M.J. Is there a role for prophylactic mesh in abdominal wall closure after emergency laparotomy? A systematic review and meta-analysis. Hernia 2020, 24, 441–447. [Google Scholar] [CrossRef]
- Mingoli, A.; Puggioni, A.; Sgarzini, G.; Luciani, G.; Corzani, F.; Ciccarone, F.; Baldassarre, E.; Modini, C. Incidence of incisional hernia following emergency abdominal surgery. Ital. J. Gastroenterol. Hepatol. 1999, 31, 449–453. [Google Scholar] [PubMed]
- Albendary, M.; Mohamedahmed, A.Y.Y.; Alamin, A.; Rout, S.; George, A.; Zaman, S. Efficacy and safety of mesh closure in preventing wound failure following emergency laparotomy: A systematic review and meta-analysis. Langenbecks Arch. Surg. 2022, 407, 1333–1344. [Google Scholar] [CrossRef] [PubMed]
- Johnson, E.E.; Simpson, A.N.; Harvey, J.B.; Lockett, M.A.; Byrne, K.T.; Simpson, K.N. Trends in bariatric surgery, 2002–2012: Do changes parallel the obesity trend? Surg. Obes. Relat. Dis. 2016, 12, 398–404. [Google Scholar] [CrossRef] [PubMed]
- Dasari, M.; Wessel, C.B.; Hamad, G.G. Prophylactic mesh placement for prevention of incisional hernia after open bariatric surgery: A systematic review and meta-analysis. Am. J. Surg. 2016, 212, 615–622.e1. [Google Scholar] [CrossRef]
- Fassiadis, N.; Roidl, M.; Hennig, M.; South, L.; Andrews, S. Randomized clinical trial of vertical or transverse laparotomy for abdominal aortic aneurysm repair. J. Br. Surg. 2005, 92, 1208–1211. [Google Scholar] [CrossRef]
- Muysoms, F.E.; Detry, O.; Vierendeels, T.; Huyghe, M.; Miserez, M.; Ruppert, M.; Tollens, T.; Defraigne, J.O.; Berrevoet, F. Prevention of Incisional Hernias by Prophylactic Mesh-augmented Reinforcement of Midline Laparotomies for Abdominal Aortic Aneurysm Treatment: A Randomized Controlled Trial. Ann. Surg. 2016, 263, 638–645. [Google Scholar] [CrossRef]
- Murray, B.W.; Cipher, D.J.; Pham, T.; Anthony, T. The impact of surgical site infection on the development of incisional hernia and small bowel obstruction in colorectal surgery. Am. J. Surg. 2011, 202, 558–560. [Google Scholar] [CrossRef]
- Guzmán-Valdivia, G. Incisional hernia at the site of a stoma. Hernia 2008, 12, 471–474. [Google Scholar] [CrossRef]
- Bhangu, A.; Nepogodiev, D.; Ives, N.; Magill, L.; Glasbey, J.; Forde, C.; Bisgaard, T.; Handley, K.; Mehta, S.; Morton, D. Prophylactic biological mesh reinforcement versus standard closure of stoma site (ROCSS): A multicentre, randomised controlled trial. Lancet 2020, 395, 417–426. [Google Scholar] [CrossRef]
- Mohamedahmed, A.Y.Y.; Stonelake, S.; Zaman, S.; Hajibandeh, S. Closure of stoma site with or without prophylactic mesh reinforcement: A systematic review and meta-analysis. Int. J. Color. Dis. 2020, 35, 1477–1488. [Google Scholar] [CrossRef]
- Baucom, R.B.; Ousley, J.; Feurer, I.D.; Beveridge, G.B.; Pierce, R.A.; Holzman, M.D.; Sharp, K.W.; Poulose, B.K. Patient reported outcomes after incisional hernia repair-establishing the ventral hernia recurrence inventory. Am. J. Surg. 2016, 212, 81–88. [Google Scholar] [CrossRef] [PubMed]


| Study/Design/Country | Population | Inclusion and Exclusion Criteria | Surgical Technique and Type of Mesh | Follow-Up Months | Primary Outcome | IH Assessment |
|---|---|---|---|---|---|---|
| De La Portilla 2008 [20] RCT Spain | No-mesh: 72 Mesh: 71 Cancer diagnosis: 80 (56%) | Inclusion criteria: All laparotomy incisions and mixed emergency and elective colorectal surgeries. Exclusion criteria: <18-year-old. | No-mesh: Mass closure with continuous Poly-p-dioxanone suture. Mesh: Same technique plus Supra-aponeurotic Polyglycolic mesh | 1 | Evisceration (Midline laparotomy) No-mesh group: 4/72 (5.6%) Mesh group: 1 (1.4%) | Clinical examination |
| Garcia-Urena 2015 [5] RCT Spain | No-mesh: 54 Mesh: 53 Cancer diagnosis: No-mesh 39/54 (72%) Mesh 45/53 (85%) | Inclusion criteria: Midline laparotomy incision, mixed emergency and elective colorectal surgeries. Exclusion criteria: Previous IH, haemodynamically unstable, carcinomatosis. | No-mesh: continuous with slowly absorbable suture poly-4 hydroxybutyrate. Mesh: Same above technique plus onlay large-pore polypropylene mesh | 24 | IH (Midline laparotomy) No-mesh group: 17/54 (31.5%) Mesh group: 6/53 (11.3%) | CT scan |
| Hoyuela 2018 [21] Prospect-ive cohort Spain | No-mesh: 37 Mesh: 15 Cancer diagnosis: 100% | Inclusion criteria: Vertical or transverse assistance incision for elective laparoscopic-assisted oncological resections, BMI > 25. Exclusion criteria: Emergency cases, conversion to open surgery, previous abdominal wall mesh. | No-mesh: Absorbable suture for peritoneum and continuous, slowly absorbable suture for the sheath. Mesh: Same above technique plus Retro-fascial pre-muscular sublay polypropylene mesh | 22.3 | IH (Extraction incision): No-mesh: 4/37 (10.8%) Mesh: 0/15 (0%) Evisceration (Extraction incision): No-mesh: 1/37 (2.7%) Mesh: 0/15 (0%) | Clinical examination + CT scan |
| Pereira 2019 [22] Retrospe-ctive cohort Spain | No-mesh: 61 Mesh: 34 Cancer diagnosis: 100% | Inclusion criteria: Elective Laparoscopic colorectal cancer surgery. Exclusion criteria: Previous open surgery, conversion to open surgery. | No-mesh: Continuous PDS loop. Mesh: onlay mesh of polyvinylidene fluoride. | 13 | IH (Extraction incision): No-mesh: 18/61 (29.5%) Mesh: 2/34 (5.9%) Evisceration (Extraction incision): No-mesh: 2/61 (3.3%) Mesh: 0/34 (0%) | Clinical examination ± CT scan |
| Wong 2020 [23] Retrospect-ive cohort Australia | No-mesh: 552 Mesh: 140 Cancer diagnosis: 486/662 (73%) | Inclusion criteria: Midline laparotomy incision, mixed emergency and elective colorectal surgeries. | No-mesh: Continuous with 1 Nylon suture, 1cm bite/gap. Mesh: Same technique plus onlay Polypropylene mesh | 33 | IH (Midline laparotomy) No-mesh: 77/553 (13.9%) Mesh: 2/140 (1.4%) | Not mentioned |
| Cano-Valderra-ma 2022 [24] Retrospect-ive cohort Spain | No-mesh: 211 Mesh: 98 Cancer diagnosis: No-mesh 179/211 (85%) Mesh 75/98 (77%) | Inclusion criteria: Elective laparotomy for colorectal surgery. Exclusion criteria: Laparoscopic surgery, follow up < 1 month. | No-mesh: Single-layer running suture. Mesh: Same above technique plus polypropylene mesh (Sublay in 9 cases and onlay in 79 cases) | 22 | IH (Midline laparotomy) No-mesh: 54/211 (25.6%) Mesh: 9/98 (9.2%) | CT scan ± Clinical examination |
| Mesh Closure | No-mesh Closure | Odds Ratio (OR) | 95% CI | p-Value | |
|---|---|---|---|---|---|
| Primary outcomes: | |||||
| Incisional hernia (Midline Laparotomy) | 17/291 (5.8%) | 148/817 (18%) | 0.23 | 0.12-0.44 | * <0.00001 |
| Incisional hernia (Extraction site) | 2/49 (4%) | 22/98 (22.4%) | 0.16 | 0.04-0.64 | * 0.009 |
| Evisceration | 3/271 (1.1%) | 11/435 (2.5%) | 0.51 | 0.61-1.61 | 0.25 |
| Secondary outcomes: | |||||
| Surgical site infections | 43/411 (10.5%) | 67/987 (6.8%) | 1.20 | 0.67–2.13 | 0.54 |
| Seroma formation | 25/313 (8%) | 23/776 (3%) | 1.80 | 0.83–3.90 | 0.13 |
| Length of hospital stay | Mean difference = −0.54 | −2.73–1.64 | 0.63 | ||
| Study | Age [mean ± SD or Median (Range)] (years) | Gender Male: Female | BMI (Average/Mean ± SD) | Comorbidities |
|---|---|---|---|---|
| De La Portilla 2008 [20] | Mesh: 66.2 ± 12.53 No-mesh: 63.1 ± 15.6 | Mesh: 41:30 No-mesh: 39:33 | NA | Mesh: DM = 7, Steroids = 2, Respiratory disease = 5, Obesity = 2 No-mesh: DM = 4, Steroids = 4, Respiratory disease = 4, Obesity = 0 |
| Garcia-Urena 2015 [5] | Mesh:65.6 ± 13.3 No-mesh: 61.46 ± 15.6 | Mesh: 31:22 No-mesh: 33:21 | Mesh: 24 No-mesh: 22 | Mesh: DM = 18, Immuno-compromised = 6, Smoking = 5 No-mesh: DM = 9, Immuno-compromised = 5, Smoking = 9 |
| Hoyuela 2018 [21] | Mesh: 76.4 ± 11 No-mesh: 71 ± 11 | Mesh: 10:5 No-mesh: 23:14 | Mesh: 27.8 ± 2 No-mesh: 28.9 ± 2 | Mesh: DM = 4, Respiratory disease = 2 No-mesh: DM = 10, respiratory disease = 4 |
| Pereira 2019 [22] | Mesh: 72.4 ± 10.9 No-mesh: 69.3 ± 12.5 | Mesh: 17:17 No-mesh: 40:21 | Mesh: 30.2 ± 5.6 No-mesh: 26.8 ± 4.4 | Mesh: DM = 10, COPD = 10, Obesity = 19, Immuno-compromised = 3 No-mesh: DM = 13, COPD = 7, Obesity = 7, Immuno-compromised = 2 |
| Wong 2020 [23] | All patients: 65 (20–96) | All patients: 55.5%:44.5% | NA | NA |
| Cano-Valderrama 2022 [24] | Mesh: 73.4 ± 11.3 No-mesh: 69.6 ± 11.9 | Mesh: 53:45 No-mesh: 113:98 | NA | Mesh: DM = 33, COPD = 8, Obesity = 27, Steroids = 4, Malnutrition = 9 No-mesh: DM = 41, COPD = 18, Obesity = 52, Steroids = 8 |
| Study | Hoyuela, 2017 [21] | Pereira, 2019 [22] | Wong, 2020 [23] | Cano-Valderrama, 2022 [24] |
|---|---|---|---|---|
| Representativeness of the exposed cohort | * | * | * | * |
| Selection of the non-exposed cohort | * | * | * | * |
| Ascertainment of exposure | * | * | * | * |
| Demonstration that outcome of interest was not present at start of study | * | * | * | * |
| Comparability of cohorts based on the design or analysis controlled for confounders | * | |||
| Assessment of outcome | * | * | * | * |
| Was follow-up long enough for outcomes to occur | * | * | * | |
| Adequacy of follow-up of cohorts | * | * | * | * |
| Total | 8 | 6 | 7 | 7 |
| Studies | Midline EI | Lateral Transverse | Low Transverse (Suprapubic) | EI Size (cm) | ||||
|---|---|---|---|---|---|---|---|---|
| Mesh | No-Mesh | Mesh | No-Mesh | Mesh | No-Mesh | |||
| Hoyuela 2018 [21] | Number | 3 | 7 | 4 | 11 | 8 | 19 | Mesh: 5.8 ± 1 |
| IH incidence | 0 | 3 (43%) | 0 | 1 (9%) | Not performed | No-mesh: 6.2 ± 2 | ||
| Pereira 2019 [22] | Number | 34 | 61 | Not performed | 87 | Not reported | ||
| IH incidence | 2 (5.9%) | 18 (29.5%) | 3 (3.4%) | |||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Albendary, M.; Mohamedahmed, A.Y.; Mohamedahmed, M.Y.; Ihedioha, U.; Rout, S.; Van Der Avoirt, A. Evaluation of Mesh Closure of Laparotomy and Extraction Incisions in Open and Laparoscopic Colorectal Surgery: A Systematic Review and Meta-Analysis. J. Clin. Med. 2024, 13, 6980. https://doi.org/10.3390/jcm13226980
Albendary M, Mohamedahmed AY, Mohamedahmed MY, Ihedioha U, Rout S, Van Der Avoirt A. Evaluation of Mesh Closure of Laparotomy and Extraction Incisions in Open and Laparoscopic Colorectal Surgery: A Systematic Review and Meta-Analysis. Journal of Clinical Medicine. 2024; 13(22):6980. https://doi.org/10.3390/jcm13226980
Chicago/Turabian StyleAlbendary, Mohamed, Ali Yasen Mohamedahmed, Marwa Yassin Mohamedahmed, Ugochukwu Ihedioha, Shantanu Rout, and Anouk Van Der Avoirt. 2024. "Evaluation of Mesh Closure of Laparotomy and Extraction Incisions in Open and Laparoscopic Colorectal Surgery: A Systematic Review and Meta-Analysis" Journal of Clinical Medicine 13, no. 22: 6980. https://doi.org/10.3390/jcm13226980
APA StyleAlbendary, M., Mohamedahmed, A. Y., Mohamedahmed, M. Y., Ihedioha, U., Rout, S., & Van Der Avoirt, A. (2024). Evaluation of Mesh Closure of Laparotomy and Extraction Incisions in Open and Laparoscopic Colorectal Surgery: A Systematic Review and Meta-Analysis. Journal of Clinical Medicine, 13(22), 6980. https://doi.org/10.3390/jcm13226980





