Clinical Impacts of Pseudomonas aeruginosa Isolation in Patients with Bronchiectasis: Findings from KMBARC Registry
Abstract
1. Introduction
2. Materials and Methods
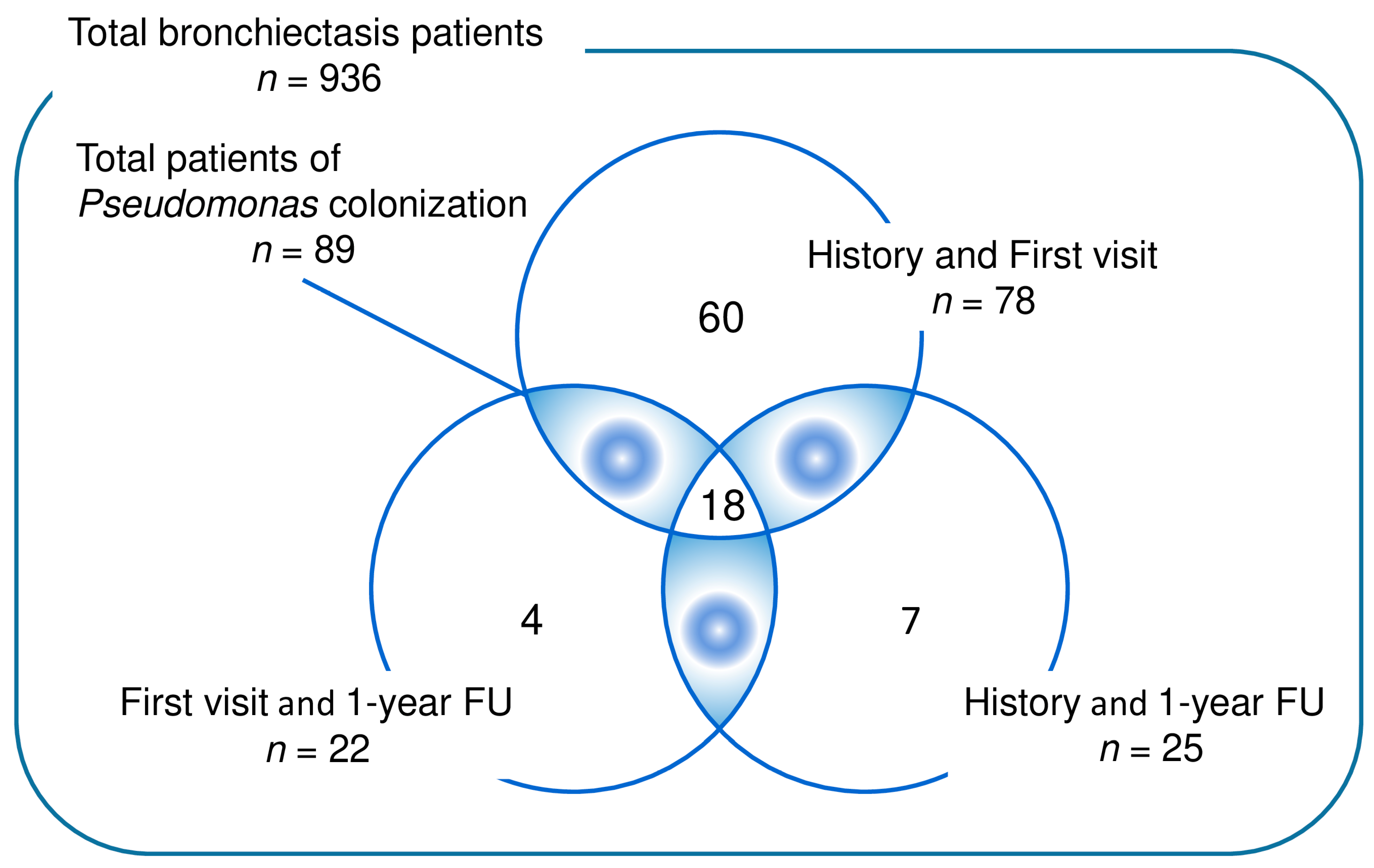
2.1. Study Population
2.2. Data Collection
2.3. Definition of Terminology and Outcomes
2.4. Statistical Analysis
3. Results
3.1. Baseline Characteristics of the Population
3.2. Clinical Characteristics Related to Bronchiectasis
3.3. Clinical Outcomes Based on Pseudomonas Colonization
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- McShane, P.J.; Naureckas, E.T.; Tino, G.; Strek, M.E. Non-cystic fibrosis bronchiectasis. Am. J. Respir. Crit. Care Med. 2013, 188, 647–656. [Google Scholar] [CrossRef]
- Kelly, M.G.; Murphy, S.; Elborn, J.S. Bronchiectasis in secondary care: A comprehensive profile of a neglected disease. Eur. J. Intern. Med. 2003, 14, 488–492. [Google Scholar] [CrossRef] [PubMed]
- Miszkiel, K.A.; Wells, A.U.; Rubens, M.B.; Cole, P.J.; Hansell, D.M. Effects of airway infection by Pseudomonas aeruginosa: A computed tomographic study. Thorax 1997, 52, 260–264. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ho, P.L.; Chan, K.N.; Ip, M.S.; Lam, W.K.; Ho, C.S.; Yuen, K.Y.; Tsang, K.W. The effect of Pseudomonas aeruginosa infection on clinical parameters in steady-state bronchiectasis. Chest 1998, 114, 1594–1598. [Google Scholar] [CrossRef] [PubMed]
- Vidaillac, C.; Chotirmall, S.H. Pseudomonas aeruginosa in bronchiectasis: Infection, inflammation, and therapies. Expert Rev. Respir. Med. 2021, 15, 649–662. [Google Scholar] [CrossRef] [PubMed]
- Chai, Y.H.; Xu, J.F. How does Pseudomonas aeruginosa affect the progression of bronchiectasis? Clin. Microbiol. Infect. 2020, 26, 313–318. [Google Scholar] [CrossRef]
- Martinez-García, M.A.; Oscullo, G.; Posadas, T.; Zaldivar, E.; Villa, C.; Dobarganes, Y.; Girón, R.; Olveira, C.; Maíz, L.; García-Clemente, M.; et al. Pseudomonas aeruginosa and lung function decline in patients with bronchiectasis. Clin. Microbiol. Infect. 2021, 27, 428–434. [Google Scholar] [CrossRef]
- McDonnell, M.J.; Jary, H.R.; Perry, A.; MacFarlane, J.G.; Hester, K.L.M.; Small, T.; Molyneux, C.; Perry, J.D.; Walton, K.E.; De Soyza, A. Non cystic fibrosis bronchiectasis: A longitudinal retrospective observational cohort study of Pseudomonas persistence and resistance. Respir. Med. 2015, 109, 716–726. [Google Scholar] [CrossRef]
- King, P.T.; Holdsworth, S.R.; Freezer, N.J.; Villanueva, E.; Holmes, P.W. Microbiologic follow-up study in adult bronchiectasis. Respir. Med. 2007, 101, 1633–1638. [Google Scholar] [CrossRef]
- Chawla, K.; Vishwanath, S.; Manu, M.K.; Lazer, B. Influence of Pseudomonas aeruginosa on exacerbation in patients with bronchiectasis. J. Glob. Infect. Dis. 2015, 7, 18–22. [Google Scholar] [CrossRef]
- Rogers, G.B.; Zain, N.M.M.; Bruce, K.D.; Burr, L.D.; Chen, A.C.; Rivett, D.W.; McGuckin, M.A.; Serisier, D.J. A novel microbiota stratification system predicts future exacerbations in bronchiectasis. Ann. Am. Thorac. Soc. 2014, 11, 496–503. [Google Scholar] [CrossRef]
- Pieters, A.; Bakker, M.; Hoek, R.A.S.; Altenburg, J.; van Westreenen, M.; Aerts, J.G.J.V.; van der Eerden, M.M. Predicting factors for chronic colonization of Pseudomonas aeruginosa in bronchiectasis. Eur. J. Clin. Microbiol. Infect. Dis. 2019, 38, 2299–2304. [Google Scholar] [CrossRef] [PubMed]
- Pasteur, M.C.; Helliwell, S.M.; Houghton, S.J.; Webb, S.C.; Foweraker, J.E.; Coulden, R.A.; Flower, C.D.; Bilton, D.; Keogan, M.T. An investigation into causative factors in patients with bronchiectasis. Am. J. Respir. Crit. Care Med. 2000, 162, 1277–1284. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Choi, H.; Sim, Y.S.; Park, S.; Kim, W.J.; Yoo, K.H.; Lee, S.J.; Kim, T.H.; Yang, B.; Jeong, I.; et al. KMBARC registry: Protocol for a multicentre observational cohort study on non-cystic fibrosis bronchiectasis in Korea. BMJ Open 2020, 10, e034090. [Google Scholar] [CrossRef]
- Lee, H.; Choi, H.; Chalmers, J.D.; Dhar, R.; Nguyen, T.Q.; Visser, S.K.; Morgan, L.C.; Oh, Y.M. Characteristics of bronchiectasis in Korea: First data from the Korean Multicentre Bronchiectasis Audit and Research Collaboration registry and comparison with other international registries. Respirology 2021, 26, 619–621. [Google Scholar] [CrossRef] [PubMed]
- Chalmers, J.D.; Aliberti, S.; Polverino, E.; Vendrell, M.; Crichton, M.; Loebinger, M.; Dimakou, K.; Clifton, I.; van der Eerden, M.; Rohde, G.; et al. The EMBARC European Bronchiectasis Registry: Protocol for an international observational study. ERJ Open Res. 2016, 2, 00081–02015. [Google Scholar] [CrossRef]
- Spinou, A.; Siegert, R.J.; Guan, W.J.; Patel, A.S.; Gosker, H.R.; Lee, K.K.; Elston, C.; Loebinger, M.R.; Wilson, R.; Garrod, R.; et al. The development and validation of the Bronchiectasis Health Questionnaire. Eur. Respir. J. 2017, 49, 1601532. [Google Scholar] [CrossRef]
- Chalmers, J.D.; Goeminne, P.; Aliberti, S.; McDonnell, M.J.; Lonni, S.; Davidson, J.; Poppelwell, L.; Salih, W.; Pesci, A.; Dupont, L.J.; et al. The bronchiectasis severity index. An international derivation and validation study. Am. J. Respir. Crit. Care Med. 2014, 189, 576–585. [Google Scholar] [CrossRef] [PubMed]
- Martínez-García, M.Á.; de Gracia, J.; Vendrell Relat, M.; Girón, R.M.; Máiz Carro, L.; de la Rosa Carrillo, D.; Olveira, C. Multidimensional approach to non-cystic fibrosis bronchiectasis: The FACED score. Eur. Respir. J. 2014, 43, 1357–1367. [Google Scholar] [CrossRef]
- Hill, A.T.; Haworth, C.S.; Aliberti, S.; Barker, A.; Blasi, F.; Boersma, W.; Chalmers, J.D.; De Soyza, A.; Dimakou, K.; Elborn, J.S.; et al. Pulmonary exacerbation in adults with bronchiectasis: A consensus definition for clinical research. Eur. Respir. J. 2017, 49, 1700051. [Google Scholar] [CrossRef]
- Finch, S.; McDonnell, M.J.; Abo-Leyah, H.; Aliberti, S.; Chalmers, J.D. A comprehensive analysis of the impact of Pseudomonas aeruginosa colonization on prognosis in adult bronchiectasis. Ann. Am. Thorac. Soc. 2015, 12, 1602–1611. [Google Scholar] [CrossRef] [PubMed]
- Wilson, C.B.; Jones, P.W.; O’Leary, C.J.; Hansell, D.M.; Cole, P.J.; Wilson, R. Effect of sputum bacteriology on the quality of life of patients with bronchiectasis. Eur. Respir. J. 1997, 10, 1754–1760. [Google Scholar] [CrossRef] [PubMed]
- Davies, G.; Wells, A.U.; Doffman, S.; Watanabe, S.; Wilson, R. The effect of Pseudomonas aeruginosa on pulmonary function in patients with bronchiectasis. Eur. Respir. J. 2006, 28, 974–979. [Google Scholar] [CrossRef]
- Emerson, J.; Rosenfeld, M.; McNamara, S.; Ramsey, B.; Gibson, R.L. Pseudomonas aeruginosa and other predictors of mortality and morbidity in young children with cystic fibrosis. Pediatr. Pulmonol. 2002, 34, 91–100. [Google Scholar] [CrossRef]
- Guan, W.-J.; Gao, Y.-H.; Xu, G.; Lin, Z.-Y.; Tang, Y.; Li, H.-M.; Li, Z.-M.; Zheng, J.-P.; Chen, R.-C.; Zhong, N.-S. Effect of airway Pseudomonas aeruginosa isolation and infection on steady-state bronchiectasis in Guangzhou, China. J. Thorac. Dis. 2015, 7, 625–636. [Google Scholar] [CrossRef]
- Tsang, K.W.; Chan, K.; Ho, P.; Zheng, L.; Ooi, G.C.; Ho, J.C.; Lam, W. Sputum elastase in steady-state bronchiectasis. Chest 2000, 117, 420–426. [Google Scholar] [CrossRef]
- Martínez-García, M.A.; Soler-Cataluña, J.J.; Perpiñá-Tordera, M.; Román-Sánchez, P.; Soriano, J. Factors associated with lung function decline in adult patients with stable non-cystic fibrosis bronchiectasis. Chest 2007, 132, 1565–1572. [Google Scholar] [CrossRef] [PubMed]
- Guan, W.J.; Gao, Y.H.; Xu, G.; Lin, Z.Y.; Tang, Y.; Li, H.M.; Lin, Z.M.; Zheng, J.P.; Chen, R.C.; Zhong, N.S. Characterization of lung function impairment in adults with bronchiectasis. PLoS ONE 2014, 9, e113373. [Google Scholar] [CrossRef]
- Kwok, W.C.; Ho, J.C.M.; Tam, T.C.C.; Ip, M.S.M.; Lam, D.C.L. Risk factors for Pseudomonas aeruginosa colonization in non-cystic fibrosis bronchiectasis and clinical implications. Respir. Res. 2021, 22, 132. [Google Scholar] [CrossRef]
- Gao, Y.H.; Guan, W.J.; Zhu, Y.N.; Chen, R.C.; Zhang, G.J. Antibiotic-resistant Pseudomonas aeruginosa infection in patients with bronchiectasis: Prevalence, risk factors and prognostic implications. Int. J. Chronic Obstr. Pulm. Dis. 2018, 13, 237–246. [Google Scholar] [CrossRef]
- Chalmers, J.D.; Polverino, E.; Crichton, M.L.; Ringshausen, F.C.; De Soyza, A.; Vendrell, M.; Burgel, P.R.; Haworth, C.S.; Loebinger, M.R.; Dimakou, K.; et al. Bronchiectasis in Europe: Data on disease characteristics from the European Bronchiectasis registry (EMBARC). Lancet Respir. Med. 2023, 11, 637–649. [Google Scholar] [CrossRef] [PubMed]
- Luppo, A.; Rached, S.Z.; Athanazio, R.A.; Stelmach, R.; Corso, S.D. In-person and online application of the Bronchiectasis Health Questionnaire: Are they interchangeable? J. Bras. Pneumol. 2022, 48, e20220075. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.K.; Lee, H.; Kim, S.H.; Choi, H.; Lee, J.H.; Lee, J.S.; Lee, S.W.; Oh, Y.M. Validation of the Korean version of the bronchiectasis health questionnaire. Tuberc. Respir. Dis. 2020, 83, 228–233. [Google Scholar] [CrossRef] [PubMed]
- Goeminne, P.C.; Nawrot, T.S.; Ruttens, D.; Seys, S.; Dupont, L.J. Mortality in non-cystic fibrosis bronchiectasis: A prospective cohort analysis. Respir. Med. 2014, 108, 287–296. [Google Scholar] [CrossRef]
- Hester, K.L.; Macfarlane, J.G.; Tedd, H.; Jary, H.; McAlinden, P.; Rostron, L.; Small, T.; Newton, J.L.; De Soyza, A. Fatigue in bronchiectasis. QJM 2012, 105, 235–240. [Google Scholar] [CrossRef]
| Variables | Total n = 445 | Pseudomonas Group n = 89 | Non-Pseudomonas Group n = 356 | p-Value |
|---|---|---|---|---|
| Age | 64.4 ± 9.4 | 63.5 ± 9.8 | 64.6 ± 9.3 | 0.358 |
| Sex, Female, n (%) | 289 (64.9) | 56 (62.9) | 233 (65.5) | 0.655 |
| BMI | 22.9 ± 3.5 | 23.0 ± 3.5 | 23.0 ± 3.5 | 0.120 |
| Median duration between BE diagnosed to enrollment, (IQR) | 3 (1–6) | 3 (1–6) | 3 (1–6) | 0.011 |
| Smoking history, n (%) | 0.469 | |||
| Current smoker | 14 (3.2) | 1 (1.1) | 13 (3.7) | |
| Former smoker | 111 (25.1) | 23 (25.8) | 88 (24.9) | |
| Never smoker | 318 (71.8) | 65 (73.0) | 253 (71.5) | |
| Pack-years | 3.2 (1.3) | 2.9 (1.2) | 3.3 (1.3) | 0.233 |
| Past medical history, n (%) | ||||
| COPD | 152 (34.4) | 39 (43.8) | 113 (32.0) | 0.036 |
| Asthma | 102 (23.0) | 14 (15.7) | 88 (24.9) | 0.069 |
| History of TB | 148 (36.1) | 35 (39.3) | 123 (35.2) | 0.474 |
| Rhinosinusitis | 28 (6.3) | 9 (10.1) | 19 (5.4) | 0.100 |
| Cardiovascular disease | 131 (29.4) | 24 (27.0) | 107 (30.1) | 0.567 |
| Pulmonary hypertension | 15 (3.5) | 1 (1.2) | 14 (4.1) | 0.323 |
| Stroke | 11 (2.6) | 1 (1.2) | 10 (2.9) | 0.700 |
| Diabetes mellitus | 58 (13.2) | 5 (6.8) | 52 (14.7) | 0.049 |
| Osteoporosis | 48 (10.8)6 | 6 (6.7) | 42 (11.8) | 0.167 |
| Chronic renal failure | 9 (2.1) | 0 (0.0) | 9 (2.6) | 0.131 |
| Cancer, total | 40 (9.0) | 4 (4.5) | 36 (10.2) | 0.095 |
| Depression | 19 (4.3) | 2 (2.3) | 17 (4.8) | 0.390 |
| Anxiety | 13 (2.9) | 2 (2.3) | 11 (3.1) | 0.999 |
| Radiologic severity of the bronchiectasis | ||||
| Total number of involved lobes, mean (SD) | 2.4 (1.6) | 2.7 (1.5) | 2.3 (1.6) | 0.021 |
| Patients with relevant lobe involvement, n (%) | ||||
| RUL | 175 (39.3) | 43 (48.3) | 132 (37.1) | 0.052 |
| RML | 257 (57.8) | 53 (59.6) | 204 (57.3) | 0.701 |
| RLL | 248 (55.7) | 57 (64.0) | 191 (53.7) | 0.077 |
| LUL | 159 (35.7) | 33 (37.1) | 126 (35.4) | 0.767 |
| Lingula | 224 (50.3) | 57 (64.0) | 167 (46.9) | 0.004 |
| LLL | 319 (71.7) | 74 (83.2) | 245 (68.8) | 0.007 |
| Cystic bronchiectasis, n (%) | 200 (44.9) | 53 (59.6) | 147 (41.3) | 0.002 |
| Laboratory findings | ||||
| WBC (103/uL) | 9.6 (3.9) (n = 65) | 10.3 (3.2) (n = 14) | 9.4 (4.0) (n = 51) | 0.437 |
| Neutrophil (%) | 70.5 (10.8) (n = 68) | 73.3 (7.2) (n = 16) | 69.7 (11.6) (n = 52) | 0.146 |
| Lymphocyte (%) | 20.8 (9.4) (n = 68) | 18.4 (6.4) (n = 16) | 21.6 (10.1) (n = 52) | 0.141 |
| Monocyte (%) | 6.3 (2.1) (n = 68) | 6.6 (1.3) (n = 16) | 6.2 (2.3) (n = 52) | 0.454 |
| Eosinophil (%) | 1.5 (1.2) (n = 68) | 1.0 (1.1) (n = 16) | 1.6(1.3) (n = 52) | 0.117 |
| Hemoglobin (g/dL) | 12.6 (1.5) (n = 65) | 12.9 (2.0) (n = 14) | 12.5(1.4) (n = 51) | 0.456 |
| Platelet (103/uL) | 283.1(83.0) (n = 65) | 303.1(106.3) (n = 14) | 277.6(75.8) (n = 51) | 0.314 |
| Total bilirubin (mg/dL) | 0.50 (0.03) (n = 70) | 0.50 (0.05) (n = 16) | 0.5 (0.04) (n = 54) | 0.946 |
| Serum albumin (g/dL) | 4.0(0.5) (n = 46) | 4.1 (0.5) (n = 10) | 4.0 (0.5) (n = 36) | 0.665 |
| BUN (mg/dL) | 15.2 (5.8) (n = 70) | 14.4 (3.8) (n = 16) | 15.4 (6.3) (n = 54) | 0.532 |
| Creatinine (mt/dL) | 0.75 (0.24) (n = 70) | 0.80 (0.19) (n = 16) | 0.73(0.25) (n = 54) | 0.318 |
| Past medication history | ||||
| Long term macrolide use (≥1 month) | 20 (6.7) | 8 (11.3) | 12 (5.3) | 0.081 |
| Inhaled corticosteroid use * | 78 (17.5) | 11 (12.4) | 67 (18.8) | 0.152 |
| Long term use of bronchodilators † | 218 (66.1) | 51 (71.8) | 167 (64.5) | 0.246 |
| Variables | Patients Obtained Microbiologic Samples from the Airway in Medically Stable Status n = 341 |
|---|---|
| Patients from whose respiratory secretion isolated microorganism, n (%) * | 159 (46.6) |
| Microorganism, n (%) # | |
| Pseudomonas aeruginosa | 92/159 (57.9) |
| Streptococcus viridans | 23/159 (14.5) |
| Klebsiella pneumonia | 10/159 (6.3) |
| Staphylococcus aureus | 10/159 (6.3) |
| Hemophilus influenzae | 7/159 (4.4) |
| Escherichia coli | 7/159 (4.4) |
| Enterobacter cloacae | 4/159 (2.5) |
| Streptococcus pneumonia | 3/159 (1.9) |
| Moraxella catarrhalis | 2/159 (1.2) |
| Acinetobacter baumannii | 1/159 (0.6) |
| Others § | 2/159 (1.2) |
| Source of microbiology. n (%) † | |
| Sputum | 269 (95.7%) |
| Induced sputum | 10 (2.6%) |
| Total n = 445 | Pseudomonas Group n = 89 | Non-Pseudomonas Group n = 356 | p-Value | |
|---|---|---|---|---|
| Lung function, mean (SD) | ||||
| Pre BD FEV1, absolute [L] | 1.59 (0.58) | 1.35 (0.49) | 1.66 (0.59) | <0.001 |
| Pre BD FEV1, % predicted | 64.2 (19.0) | 56.5 (18.1) | 66.0 (18.8) | <0.001 |
| Pre BD FVC, absolute [L] | 2.51 (0.76) | 2.24 (0.72) | 2.57 (0.75) | <0.001 |
| Pre BD FVC, % predicted | 73.2 (15.7) | 65.7 (15.5) | 74.8 (15.3) | <0.001 |
| Post BD FEV1, absolute [L] | 1.71 (0.58) | 1.50 (0.57) | 1.76 (0.58) | 0.001 |
| Post BD FEV1, % predicted | 66.6 (18.8) | 58.5 (17.6) | 68.5 (18.6) | <0.001 |
| Post BD FVC, absolute [L] | 2.55 (0.75) | 2.26 (0.72) | 2.61 (0.75) | <0.001 |
| Post BD FVC, % predicted | 73.9 (15.4) | 66.3 (15.0) | 75.6 (15.0) | <0.001 |
| Respiratory symptom | ||||
| mMRC, mean (SD) | 1.00 (0.84) | 1.12 (0.99) | 0.07 (0.80) | 0.165 |
| Patient with each mMRC, n (%) | 0.175 | |||
| mMRC grade ≥ 2 | 84 (19.0) | 22 (24.7) | 62 (17.5) | 0.121 |
| mMRC grade ≥ 3 | 28 (6.3) | 9 (10.1) | 19 (5.4) | 0.100 |
| mMRC grade 4 | 5 (1.1) | 3 (3.4) | 2 (0.56) | 0.058 |
| Sputum color | 0.242 | |||
| Mucus | 283 (65.2) | 59 (66.3) | 224 (64.9) | |
| Mucopurulent | 112 (25.8) | 18 (20.2) | 94 (27.3) | |
| Purulent | 34 (7.8) | 10 (11.2) | 24 (7.0) | |
| Very Purulent | 5 (1.2) | 2 (2.3) | 3 (0.9) | |
| Sputum volume, cc/day mean (SD) | 26.2 (25.0) | 21.0 (22.3) | 29.2 (26.1) | 0.145 |
| h/o major hemoptysis, n (%) | 29 (10.3) | 9 (14.1) | 20 (9.1) | 0.253 |
| Acute exacerbation history | ||||
| Presence of AE, n (%) | 209 (54.3) | 46 (54.8) | 163 (54.2) | 0.921 |
| Number of AE, mean (SD) | 1.5 (2.3) | 1.6 (2.2) | 1.5 (2.4) | 0.794 |
| More than 2 annual exacerbations, n (%) | 129 (33.5) | 30 (35.7) | 99 (32.9) | 0.628 |
| Requiring antibiotics, mean (SD) | 1.7 (2.1) | 2.3 (2.5) | 1.5 (2.0) | 0.096 |
| Requiring ER visit, n (%) | 31 (7.0) | 9 (10.1) | 22 (6.2) | 0.198 |
| Requiring hospital admission, n (%) | 81 (18.3) | 18 (20.2) | 63 (17.8) | 0.596 |
| Hospital admission due to bronchiectasis, n (%) | 118 (26.6) | 30 (33.7) | 88 (24.9) | 0.091 |
| BHQ score, mean (SD) | 63.7 (10.7) | 61.3 (11.0) | 64.3 (10.6) | 0.018 |
| FACED score, mean (SD) | 2.1 (1.7) | 3.1 (1.6) | 1.9 (1.6) | <0.001 |
| BSI score, mean (SD) | 7.4 (3.9) | 10.5 (3.4) | 6.6 (3.6) | <0.001 |
| Total n = 445 | Pseudomonas Group n = 89 | Non-Pseudomonas Group n = 356 | p-Value | |
|---|---|---|---|---|
| Risk of exacerbation, crude odds ratio (CI) | 1.78 (1.07–2.97) | Ref | 0.027 | |
| Risk of exacerbation, adjusted odds ratio (CI) * | 1.66 (0.98–2.82) | Ref | 0.058 | |
| Risk of exacerbation of ED visit, crude odds ratio (CI) | 1.95 (0.58–6.57) | Ref | 0.283 | |
| Risk of exacerbation of ED visit, adjusted odds ratio (CI) * | 1.64 (0.46–5.83) | Ref | 0.441 | |
| Risk of admission due to bronchiectasis, crude odds ratio (CI) | 2.29 (1.18–4.43) | Ref | 0.014 | |
| Risk of admission due to bronchiectasis, adjusted adds ratio (CI) * | 2.19 (1.08–4.42) | Ref | 0.028 | |
| Risk of hemoptysis, crude odds ratio (CI) | 1.48 (0.82–2.64) | Ref | 0.191 | |
| Risk of hemoptysis, adjusted adds ratio (CI) * | 1.54 (0.85–2.81) | Ref | 0.153 | |
| Risk of antibiotics treatment, crude odds ratio (CI) | 3.08 (1.22–7.80) | Ref | 0.018 | |
| Risk of antibiotics treatment, adjusted adds ratio (CI) * | 3.04 (1.17–7.91) | Ref | 0.022 | |
| BHQ score * | 66.0 (11.7) (n = 236) | 60.5 (11.7) (n = 54) | 67.7 (11.2) (n = 182) | <0.001 |
| FACED score * | 2.1 (1.6) (n = 37) | 3.0 (2.2) (n = 8) | 1.9 (1.4) (n = 29) | 0.080 |
| BSI score * | 7.6 (4.3) (n = 37) | 8.8 (4.9) (n = 8) | 7.2 (4.1) (n = 29) | 0.345 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Song, J.; Sin, S.; Kang, H.-R.; Oh, Y.-M.; Jeong, I., on behalf of the KMBARC. Clinical Impacts of Pseudomonas aeruginosa Isolation in Patients with Bronchiectasis: Findings from KMBARC Registry. J. Clin. Med. 2024, 13, 5011. https://doi.org/10.3390/jcm13175011
Song J, Sin S, Kang H-R, Oh Y-M, Jeong I on behalf of the KMBARC. Clinical Impacts of Pseudomonas aeruginosa Isolation in Patients with Bronchiectasis: Findings from KMBARC Registry. Journal of Clinical Medicine. 2024; 13(17):5011. https://doi.org/10.3390/jcm13175011
Chicago/Turabian StyleSong, Jinhwa, Sooim Sin, Hye-Rin Kang, Yeon-Mok Oh, and Ina Jeong on behalf of the KMBARC. 2024. "Clinical Impacts of Pseudomonas aeruginosa Isolation in Patients with Bronchiectasis: Findings from KMBARC Registry" Journal of Clinical Medicine 13, no. 17: 5011. https://doi.org/10.3390/jcm13175011
APA StyleSong, J., Sin, S., Kang, H.-R., Oh, Y.-M., & Jeong, I., on behalf of the KMBARC. (2024). Clinical Impacts of Pseudomonas aeruginosa Isolation in Patients with Bronchiectasis: Findings from KMBARC Registry. Journal of Clinical Medicine, 13(17), 5011. https://doi.org/10.3390/jcm13175011







