Standardized Solutions of Catecholamines in Intensive Care Medicine: Application, Safety and Economic Aspects
Abstract
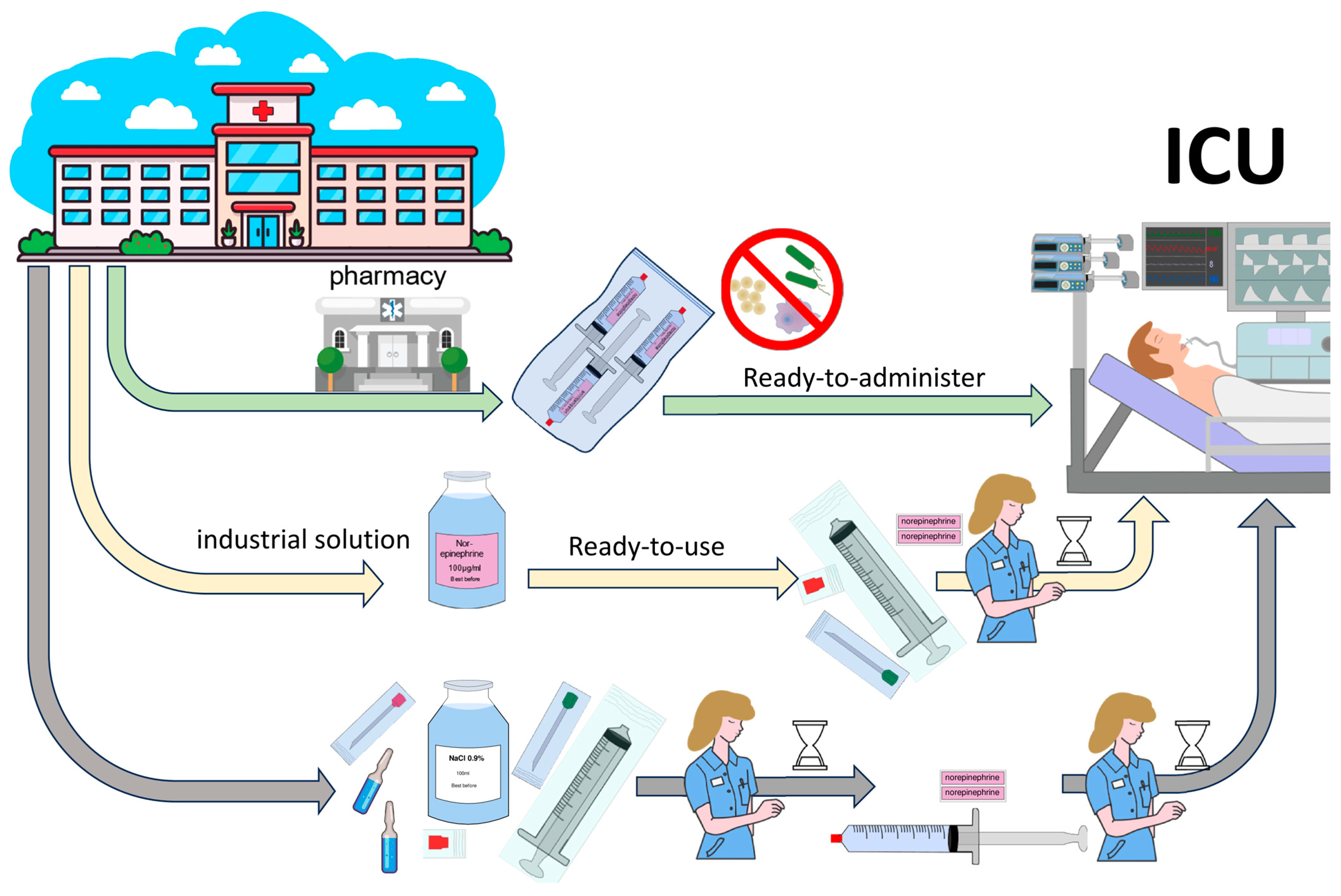
1. Introduction
2. Materials and Methods
2.1. Patient Population and Data Collection
2.2. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Starke, K. History of catecholamine research. In History of Allergy; Karger Publishers: Basel, Switzerland, 2014; Volume 100, pp. 288–301. [Google Scholar]
- Braun, B. Meilensteine in der Infusionstherapie. Available online: https://www.bbraun.de/de/ueber-uns/unternehmen/zahlen-und-fakten/geschichte/infusionstherapie.html (accessed on 18 April 2023).
- Johnston, A.; Steiner, L.; O’Connell, M.; Chatfield, D.; Gupta, A.; Menon, D. Catecholamine pharmacokinetics and pharmacodynamics in critical illness. Crit. Care 2003, 7, P046. [Google Scholar] [CrossRef]
- Lehtonen, L.A.; Antila, S.; Pentik??Inen, P.J. Pharmacokinetics and Pharmacodynamics of Intravenous Inotropic Agents. Clin. Pharmacokinet. 2004, 43, 187–203. [Google Scholar] [CrossRef] [PubMed]
- Mohr, A.; Bodenstein, M.; Sagoschen, I. Verordnung und Applikation von Dauerinfusionen in Standardkonzentrationen bei erwachsenen Intensivpatienten. Ein Meilenstein zur Verbesserung der Arzneimitteltherapiesicherheit. Anasthesiol Intensiv. 2021, 62, 314–322. [Google Scholar]
- National Coordinating Council for Medication Error Reporting and Prevention. NCC MERP Index for Categorizing Medication Errors. Available online: https://www.nccmerp.org/sites/default/files/index-color-2021-draft-change-10-2022.pdf (accessed on 30 June 2023).
- Cantor, N.; Durr, K.M.; McNeill, K.; Thompson, L.H.; Fernando, S.M.; Tanuseputro, P.; Kyeremanteng, K. Increased Mortality and Costs Associated with Adverse Events in Intensive Care Unit Patients. J. Intensive Care Med. 2022, 37, 1075–1081. [Google Scholar] [CrossRef]
- Ahgren, B. Is it better to be big?: The reconfiguration of 21st century hospitals: Responses to a hospital merger in Sweden. Health Policy 2008, 87, 92–99. [Google Scholar] [CrossRef] [PubMed]
- Rossbarnett, J. Rationalising Hospital Services: Reflections on Hospital Restructuring and its Impacts in New Zealand. New Zealand Geogr. 2000, 56, 5–21. [Google Scholar] [CrossRef]
- Marino, D.; Quattrone, G. A proposal for a new index to evaluate hospital resource allocation: The case of Italian NHS rationalisation. Eur. Res. Manag. Bus. Econ. 2019, 25, 23–29. [Google Scholar] [CrossRef]
- Anderson, S. Making Medicines: A Brief History of Pharmacy and Pharmaceuticals; Pharmaceutical Press: London, UK, 2005. [Google Scholar]
- Kolesar, J.M.; Vermeulen, L.C. Precision medicine: Opportunities for health-system pharmacists. Am. J. Health-Syst. Pharm. 2021, 78, 999–1003. [Google Scholar] [CrossRef]
- Rockville, M. Proceedings of a summit on preventing patient harm and death from iv medication errors. Am. J. Health-Syst. Pharm. 2008, 65, 2367–2379. [Google Scholar] [CrossRef]
- Lisha, J.; Annalakshmi, V.; Maria, J.; Padmini, D. Adverse drug reactions in critical care settings: A systematic review. Curr. Drug Saf. 2017, 12, 147–161. [Google Scholar] [CrossRef]
- Zeiter, B.; Krämer, I. Applikation standardisierter Injektionen und Infusionen mit „intelligenten“ Pumpen—Eine Maßnahme zur Optimierung der Arzneimitteltherapiesicherheit auf Intensivstationen. Krankenhauspharmazie 2011, 32, 28. [Google Scholar]
- Titiesari, Y.D.; Barton, G.; Borthwick, M.; Keeling, S.; Keeling, P. Infusion medication concentrations in UK’s critical care areas: Are the Intensive Care Society’s recommendations being used? J. Intensive Care Soc. 2017, 18, 30–35. [Google Scholar] [CrossRef] [PubMed]
- Bertsche, T.; Mayer, Y.; Stahl, R.; Hoppe-Tichy, T.; Encke, J.; Haefeli, W.E. Prevention of intravenous drug incompatibilities in an intensive care unit. Am. J. Health-Syst. Pharm. 2008, 65, 1834–1840. [Google Scholar] [CrossRef] [PubMed]
- Nemec, K.; Kopelent-Frank, H.; Greif, R. Standardization of infusion solutions to reduce the risk of incompatibility. Am. J. Health-Syst. Pharm. 2008, 65, 1648–1654. [Google Scholar] [CrossRef] [PubMed]
- Hilgarth, H.; Waydhas, C.; Dörje, F.; Sommer, J.; Kluge, S.; Ittner, K.P. Arzneimitteltherapiesicherheit gefördert durch die interprofessionelle Zusammenarbeit von Arzt und Apotheker auf Intensivstationen in Deutschland. Med. Klin. Intensivmed. Notfallmed. 2023, 118, 141–148. [Google Scholar] [CrossRef]
- Rickham, P.P. Human experimentation. Code of ethics of the world medical association. Declaration of Helsinki. Br. Med. J. 1964, 2, 177. [Google Scholar] [CrossRef]
- Jentzer, J.C.; Coons, J.C.; Link, C.B.; Schmidhofer, M. Pharmacotherapy update on the use of vasopressors and inotropes in the intensive care unit. J. Cardiovasc. Pharmacol. Ther. 2015, 20, 249–260. [Google Scholar] [CrossRef]
- Hamzaoui, O.; Scheeren, T.W.L.; Teboul, J.-L. Norepinephrine in septic shock: When and how much? Curr. Opin. Crit. Care 2017, 23, 342–347. [Google Scholar] [CrossRef]
- Evans, L.; Rhodes, A.; Alhazzani, W.; Antonelli, M.; Coopersmith, C.M.; French, C.; Machado, F.R.; Mcintyre, L.; Ostermann, M.; Prescott, H.C.; et al. Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock 2021. Crit. Care Med. 2021, 49, e1063–e1143. [Google Scholar] [CrossRef]
- Kimia, H.; Um, K.J.; Belley-Cote, E.P.; Waleed, A.; Farley, C.; Fernando, S.M.; Kirsten, F.; Grey, D.; Edita, H.; Herridge, M. Canadian Critical Care Society clinical practice guideline: The use of vasopressin and vasopressin analogues in critically ill adults with distributive shock. Can. J. Anesth. 2020, 67, 369–376. [Google Scholar] [CrossRef]
- Zhu, Y.; Yin, H.; Zhang, R.; Ye, X.; Wei, J. The effect of dobutamine in sepsis: A propensity score matched analysis. BMC Infect. Dis. 2021, 21, 1151. [Google Scholar] [CrossRef]
- Griffin, E.; Tweedie, O.; Kirk-Bayley, J. An Economic Analysis of Critical Care Nurse Resourcing Following the Uptake of Ready-to-Administer Noradrenaline for Hypotensive Shock in Adults in England. Adv. Ther. 2022, 39, 727–737. [Google Scholar] [CrossRef]
- Vonberg, R.P.; Gastmeier, P. Hospital-acquired infections related to contaminated substances. J. Hosp. Infect. 2007, 65, 15–23. [Google Scholar] [CrossRef]
- Larmené-Beld, K.H.M.; Frijlink, H.W.; Taxis, K. A systematic review and meta-analysis of microbial contamination of parenteral medication prepared in a clinical versus pharmacy environment. Eur. J. Clin. Pharmacol. 2019, 75, 609–617. [Google Scholar] [CrossRef]
- Brun, P.L.; Klovrzova, S. Safe aseptic preparation in the pharmacy and on the ward. Eur. J. Hosp. Pharm. Sci. Pract. 2012, 19, 474–476. [Google Scholar] [CrossRef]
- Boom, F.A.; Ris, J.M.; Veenbaas, T.; Le Brun, P.P.H.; Touw, D. Reducing the risk of non-sterility of aseptic handling in hospital pharmacies, part A: Risk assessment. Eur. J. Hosp. Pharm. 2022, 29, 151–156. [Google Scholar] [CrossRef]
- International, B.; Baxter Announces U.S. Approval and Launch of Ready-To-Use Cardiovascular Medicine Norepinephrine in Premix Formulation. Available online: https://www.baxter.com/baxter-newsroom/baxter-announces-us-approval-and-launch-ready-use-cardiovascular-medicine (accessed on 18 April 2023).
- Werumeus Buning, A.; Geersing, T.H.; Crul, M. The assessment of environmental and external cross-contamination in preparing ready-to-administer cytotoxic drugs: A comparison between a robotic system and conventional manual production. Int. J. Pharm. Pract. 2019, 28, 66–74. [Google Scholar] [CrossRef]
- Malik, P.; Rangel, M.; VonBriesen, T. Why the Utilization of Ready-to-Administer Syringes During High-Stress Situations Is More Important Than Ever. J. Infus. Nurs. 2022, 45, 27. [Google Scholar] [CrossRef]
- Ferris, N.H.; Crisp, C.B.; Hoyt, D.B.; Ting, J.; Lee, M.P. Analysis of workload and staffing requirements for a critical-care satellite pharmacy. Am. J. Hosp. Pharm. 1990, 47, 2473–2478. [Google Scholar] [CrossRef]
- Debaveye, Y.A.; Van den Berghe, G.H. Is There Still a Place for Dopamine in the Modern Intensive Care Unit? Anesth. Analg. 2004, 98, 461–468. [Google Scholar] [CrossRef]
- Pauly, M.V.; Burns, L.R. Price transparency for medical devices. Health Aff. 2008, 27, 1544–1553. [Google Scholar] [CrossRef] [PubMed]
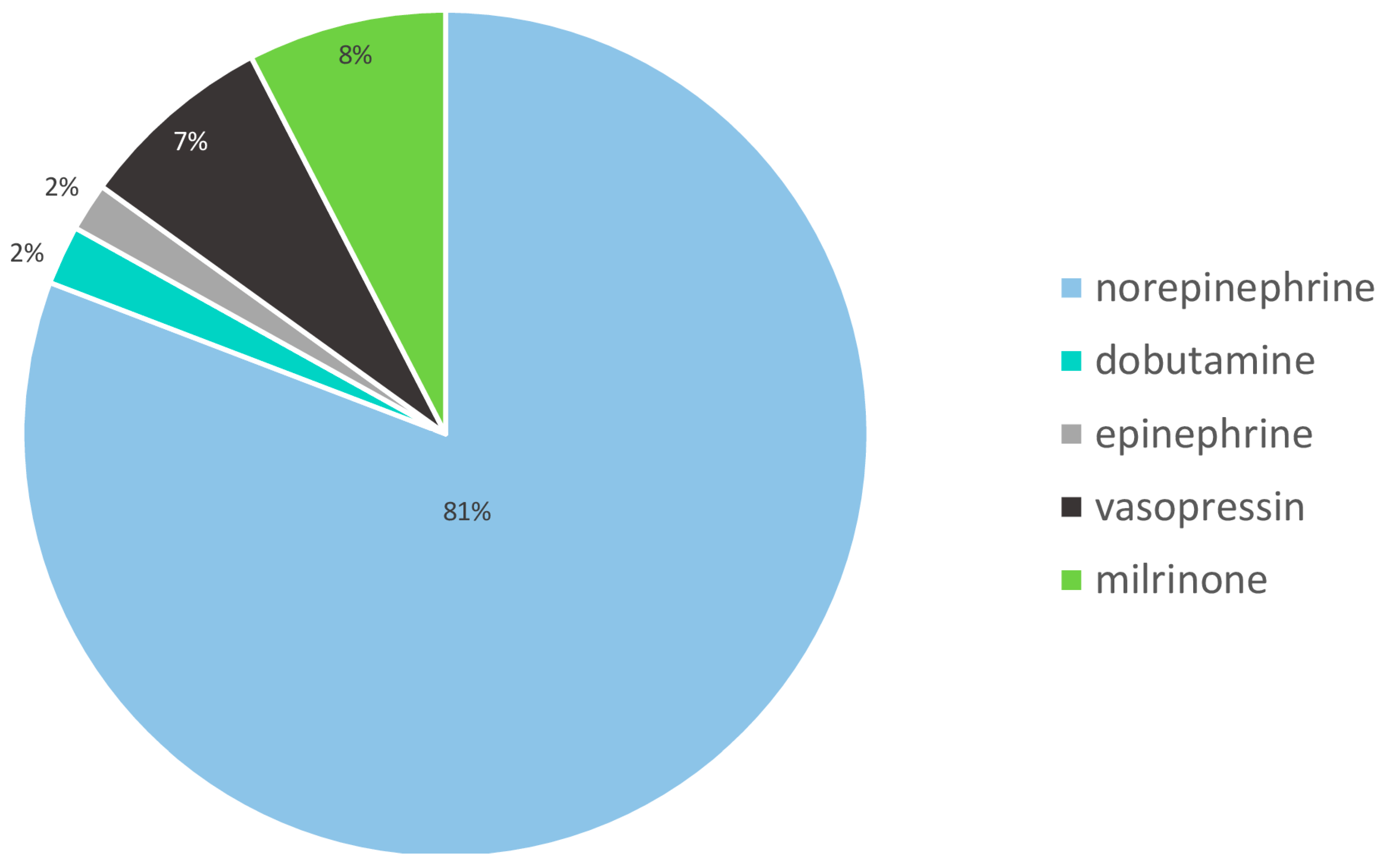
| Anaesthesiological–Surgical ICU | Internal Medicine ICU | Neurology–Neurosurgical ICU | Overall | |
|---|---|---|---|---|
| Patients [n] | 10,912 (40.3%) | 9833 (36.3%) | 6347 (23.4%) | 27,092 |
| Norepinephrine [50 mL; 100 µg/mL] | 39,093 (50.1%) | 22,436 (28.7%) | 16,562 (21.2%) | 78,091 |
| Epinephrine [50 mL; 100 µg/mL] | 1303 (73.2%) | 390 (21.9%) | 86 (4.8%) | 1179 |
| Vasopressin [40 mL; 0.5 IU/mL] | 4263 (58.9%) | 2176 (30.1%) | 795 (11.0%) | 7234 |
| Dobutamine [50 mL; 5 mg/mL] | 297 (13.6%) | 1878 (85.8%) | 13 (0.6%) | 2188 |
| Milrinon [50 mL; 0.2 mg/mL] | 6354 (86.7%) | 830 (11.3%) | 147 (2.0%) | 7331 |
| Overall | 51,310 | 27,710 | 17,630 | 96,623 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Flinspach, A.N.; Mohr, A.; Wehrle, J.; Zacharowski, K.; Neef, V.; Raimann, F.J. Standardized Solutions of Catecholamines in Intensive Care Medicine: Application, Safety and Economic Aspects. J. Clin. Med. 2024, 13, 3070. https://doi.org/10.3390/jcm13113070
Flinspach AN, Mohr A, Wehrle J, Zacharowski K, Neef V, Raimann FJ. Standardized Solutions of Catecholamines in Intensive Care Medicine: Application, Safety and Economic Aspects. Journal of Clinical Medicine. 2024; 13(11):3070. https://doi.org/10.3390/jcm13113070
Chicago/Turabian StyleFlinspach, Armin Niklas, André Mohr, Jahn Wehrle, Kai Zacharowski, Vanessa Neef, and Florian Jürgen Raimann. 2024. "Standardized Solutions of Catecholamines in Intensive Care Medicine: Application, Safety and Economic Aspects" Journal of Clinical Medicine 13, no. 11: 3070. https://doi.org/10.3390/jcm13113070
APA StyleFlinspach, A. N., Mohr, A., Wehrle, J., Zacharowski, K., Neef, V., & Raimann, F. J. (2024). Standardized Solutions of Catecholamines in Intensive Care Medicine: Application, Safety and Economic Aspects. Journal of Clinical Medicine, 13(11), 3070. https://doi.org/10.3390/jcm13113070






