Ventilators, Settings, Autotitration Algorithms
Abstract
1. Introduction
2. Overview of Ventilatory Modes and Settings
2.1. Volume-Limited or Volumetric Mode
- The tidal volume (VT) or volume delivered in each ventilatory cycle. In NIV, this is usually set at around 8–10 mL/kg of ideal patient weight to overcome the effect of potential leaks.
- The basal respiratory rate (RR), is usually programmed 2–4 cycles below the patient’s spontaneous rate.
- Inspiratory time (Ti). It should be noted that cycling (transition from inspiration to expiration) is always based on a time criterion in volumetric modes. A shorter Ti is usually used in patients with obstructive lung mechanics and a longer Ti in restrictive ones.
- The shape of the flow waveform, which can be constant (flow is the same throughout the inspiratory cycle) or decelerating, more physiological, or with a higher flow at the beginning of inspiration.
- The level of positive end-expiratory pressure (PEEP).
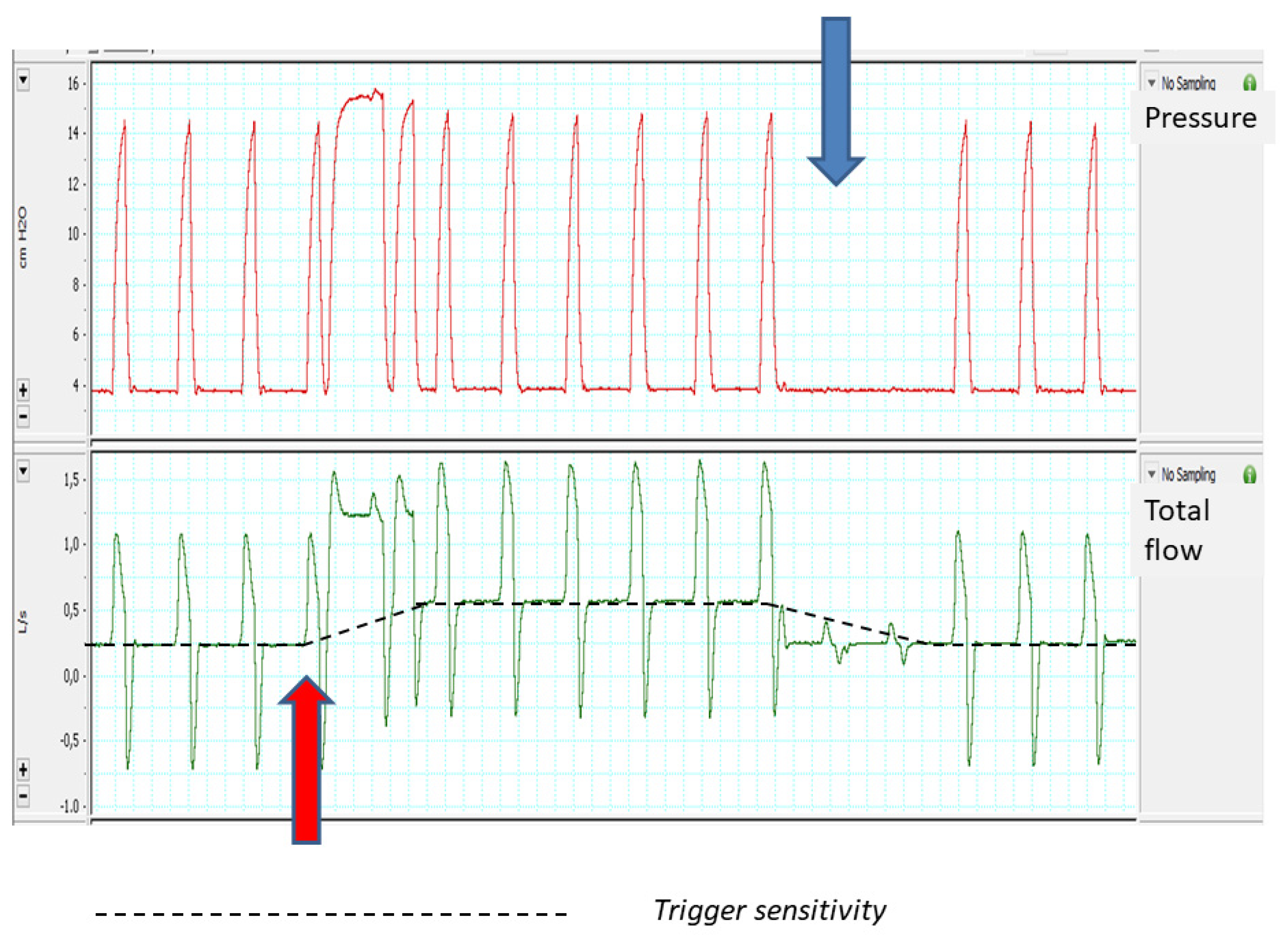
- The trigger sensitivity.
2.2. Pressure-Limited or Barometric Mode
2.2.1. IPAP and EPAP Levels
2.2.2. Backup Respiratory Rate (BURR)
2.2.3. Trigger Sensitivity
2.2.4. Pressurization Ramp (“Rise Time”)
2.2.5. Cycling to Expiration
2.2.6. Maximum and Minimum Inspiratory Time
2.2.7. Rise Fall or Expiratory Ramp
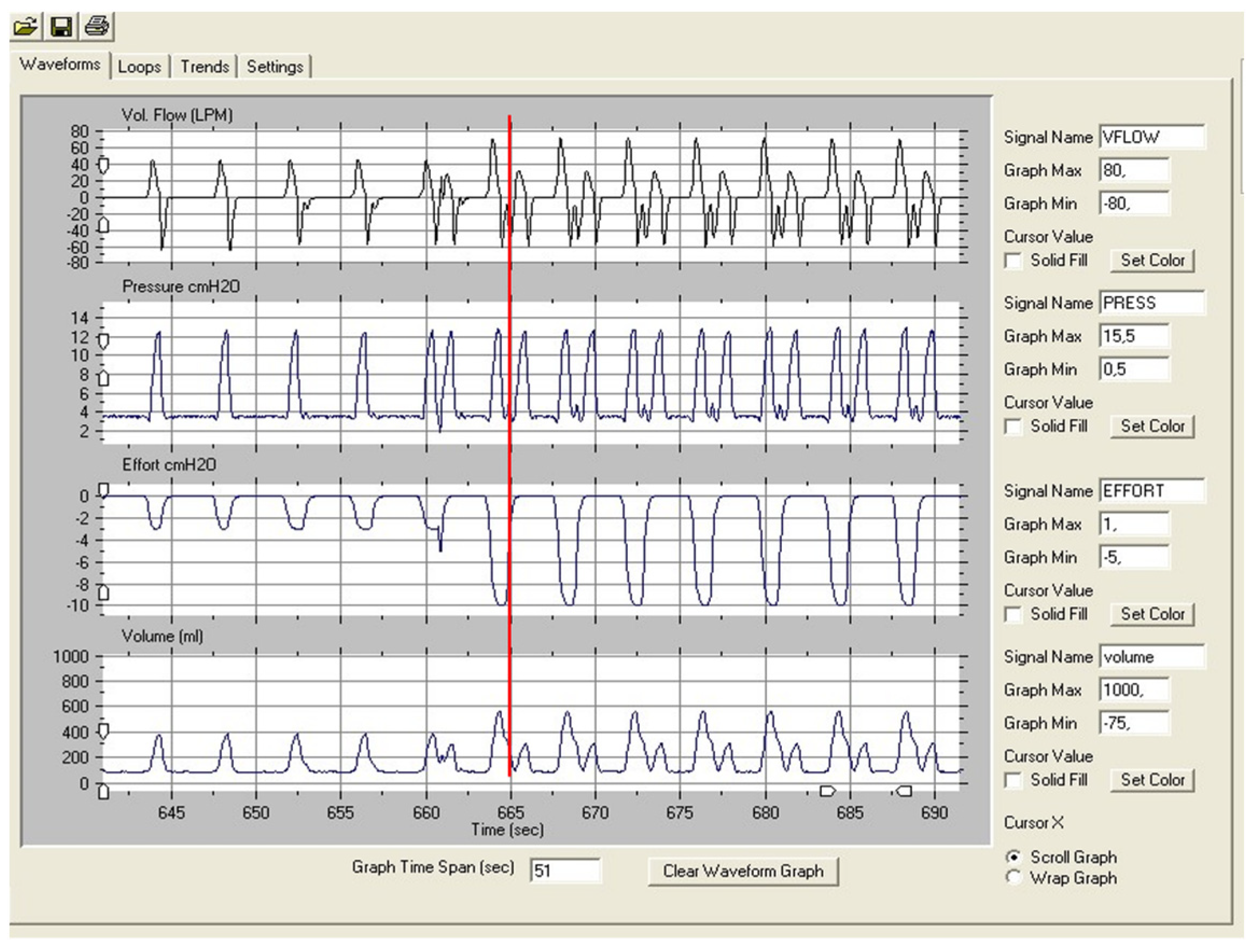
3. Autotitration Algorithms
3.1. Volume-Assured Pressure-Limited Modes
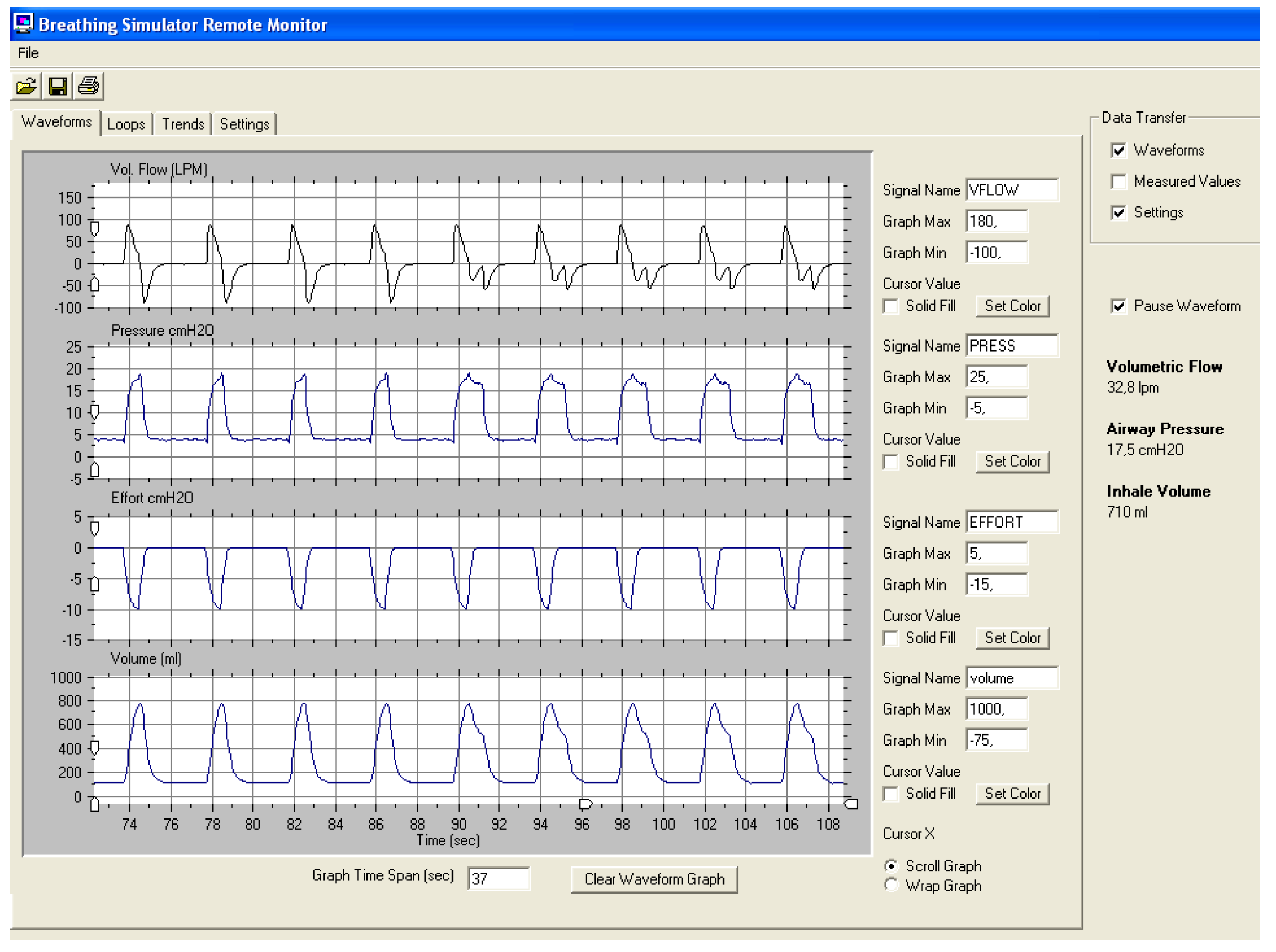
3.1.1. Average Volume-Assured Pressure Support (AVAPS, Phillips Respironics Murrysville, PA)
3.1.2. The “Intelligent Volume Assured Pressure Support” Mode (IVAPS, Resmed San Diego, CA)
3.2. Auto-EPAP Systems
3.2.1. Autotitration Algorithms to Maintain Upper Airway Patency
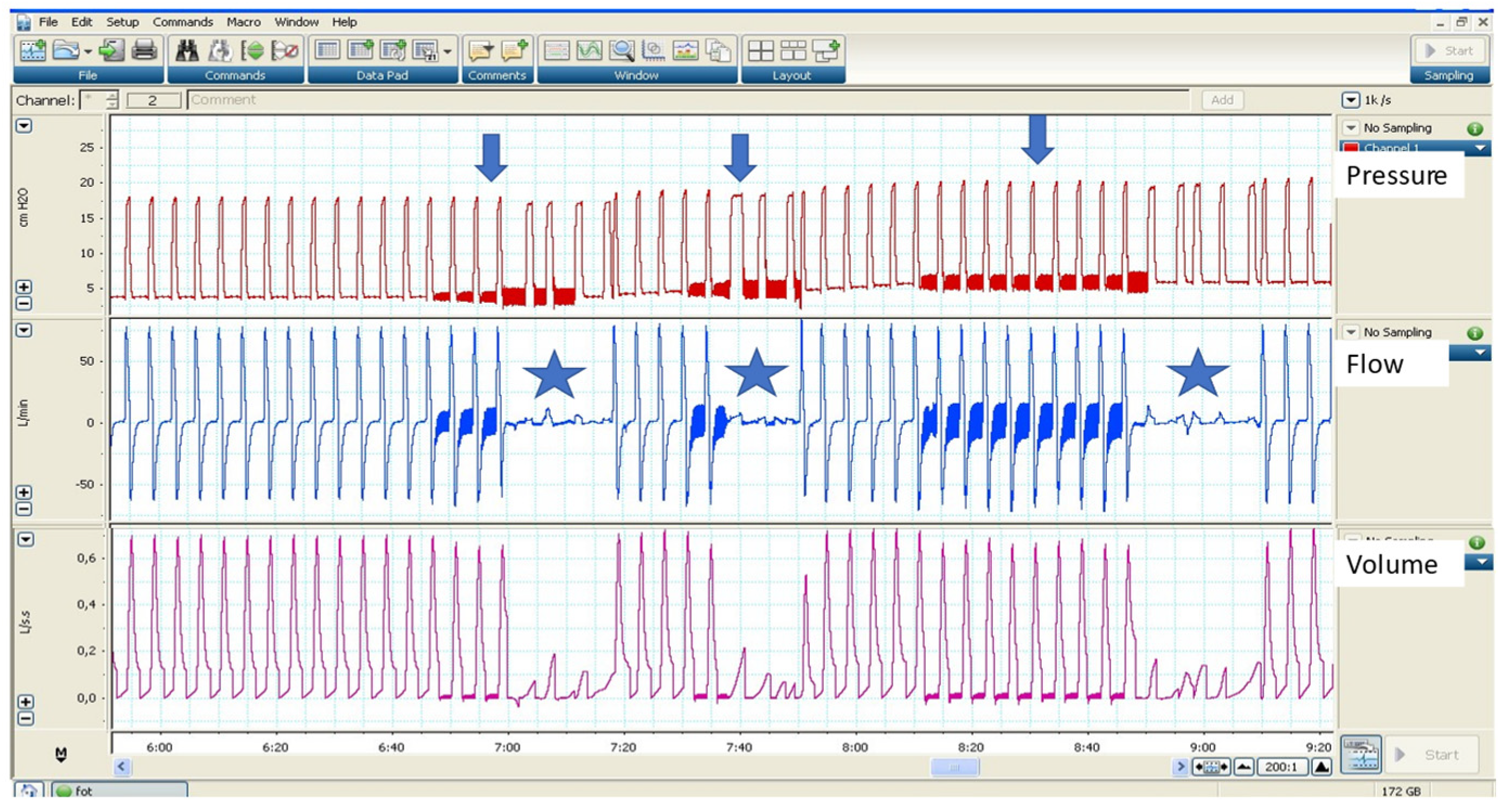
3.2.2. Autotitration Algorithms to Treat Expiratory Flow Limitation (EFL)
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lassen, H.C. A preliminary report on the 1952 epidemic of poliomyelitis in Copenhagen with special reference to the treatment of acute respiratory insufficiency. Lancet 1953, 26, 137–141. [Google Scholar] [CrossRef] [PubMed]
- Bach, J.R.; Alba, A.; Mosher, R.; Delaubier, A. Intermittent positive pressure ventilation via nasal access in the management of respiratory insufficiency. Chest 1987, 92, 168–170. [Google Scholar] [CrossRef] [PubMed]
- Bach, J.R.; Alba, A.S.; Shin, D. Management alternatives for post-polio respiratory insufficiency. Assisted ventilation by nasal or oral-nasal interface. Am. J. Phys. Med. Rehabil. 1989, 68, 264–271. [Google Scholar] [CrossRef]
- Melloni, B.; Mounier, L.; Laaban, J.-P.; Chambellan, A.; Foret, D.; Muir, J.-F. Home-Based Care Evolution in Chronic Respiratory Failure between 2001 and 2015 (Antadir Federation Observatory). Respir. Int. Rev. Thorac. Dis. 2018, 96, 446–454. [Google Scholar] [CrossRef] [PubMed]
- Scott, J.B. Ventilators for Noninvasive Ventilation in Adult Acute Care. Respir. Care 2019, 64, 712–722. [Google Scholar] [CrossRef]
- Scala, R.; Naldi, M. Ventilators for noninvasive ventilation to treat acute respiratory failure. Respir. Care 2008, 53, 1054–1080. [Google Scholar] [PubMed]
- Luján, M.; Peñuelas, Ó.; Cinesi Gómez, C.; García-Salido, A.; Moreno Hernando, J.; Romero Berrocal, A.; Gutiérrez Ibarluzea, I.; Masa Jiménez, J.F.; Mas, A.; Carratalá Perales, J.M.; et al. Summary of Recommendations and Key Points of the Consensus of Spanish Scientific Societies (SEPAR, SEMICYUC, SEMES; SECIP, SENEO, SEDAR, SENP) on the Use of Non-Invasive Ventilation and High-Flow Oxygen Therapy with Nasal Cannulas in Adult, Pediatric, and Neonatal Patients with Severe Acute Respiratory Failure. Arch. Bronconeumol. 2020, 57, 415–427. [Google Scholar] [CrossRef]
- Luján, M.; Sogo, A.; Monsó, E. Home mechanical ventilation monitoring software: Measure more or measure better? Arch. Bronconeumol. 2012, 48, 170–178. [Google Scholar] [CrossRef]
- Johnson, K.G. APAP, BPAP, CPAP, and New Modes of Positive Airway Pressure Therapy. Adv. Exp. Med. Biol. 2022, 1384, 297–330. [Google Scholar] [CrossRef] [PubMed]
- Gregoretti, C.; Navalesi, P.; Ghannadian, S.; Carlucci, A.; Pelosi, P. Choosing a ventilator for home mechanical ventilation. Breathe 2013, 9, 394–409. [Google Scholar] [CrossRef]
- Lloyd-Owen, S.J.; Donaldson, G.C.; Ambrosino, N.; Escarabill, J.; Farre, R.; Fauroux, B.; Robert, D.; Schoenhofer, B.; Simonds, A.K.; Wedzicha, J.A. Patterns of home mechanical ventilation use in Europe: Results from the Eurovent survey. Eur. Respir. J. 2005, 25, 1025–1031. [Google Scholar] [CrossRef] [PubMed]
- Georgopoulos, D.; Prinianakis, G.; Kondili, E. Bedside waveforms interpretation as a tool to identify patient-ventilator asynchronies. Intensive Care Med. 2006, 32, 34–47. [Google Scholar] [CrossRef]
- Rabec, C.; Rodenstein, D.; Leger, P.; Rouault, S.; Perrin, C.; Gonzalez-Bermejo, J. SomnoNIV group Ventilator modes and settings during non-invasive ventilation: Effects on respiratory events and implications for their identification. Thorax 2011, 66, 170–178. [Google Scholar] [CrossRef] [PubMed]
- Contal, O.; Adler, D.; Borel, J.-C.; Espa, F.; Perrig, S.; Rodenstein, D.; Pépin, J.-L.; Janssens, J.-P. Impact of different backup respiratory rates on the efficacy of noninvasive positive pressure ventilation in obesity hypoventilation syndrome: A randomized trial. Chest 2013, 143, 37–46. [Google Scholar] [CrossRef]
- Sinderby, C.; Navalesi, P.; Beck, J.; Skrobik, Y.; Comtois, N.; Friberg, S.; Gottfried, S.B.; Lindström, L. Neural control of mechanical ventilation in respiratory failure. Nat. Med. 1999, 5, 1433–1436. [Google Scholar] [CrossRef]
- Yonis, H.; Crognier, L.; Conil, J.-M.; Serres, I.; Rouget, A.; Virtos, M.; Cougot, P.; Minville, V.; Fourcade, O.; Georges, B. Patient-ventilator synchrony in Neurally Adjusted Ventilatory Assist (NAVA) and Pressure Support Ventilation (PSV): A prospective observational study. BMC Anesthesiol. 2015, 15, 117. [Google Scholar] [CrossRef]
- Goulet, R.; Hess, D.; Kacmarek, R.M. Pressure vs flow triggering during pressure support ventilation. Chest 1997, 111, 1649–1653. [Google Scholar] [CrossRef] [PubMed]
- Aslanian, P.; El Atrous, S.; Isabey, D.; Valente, E.; Corsi, D.; Harf, A.; Lemaire, F.; Brochard, L. Effects of flow triggering on breathing effort during partial ventilatory support. Am. J. Respir. Crit. Care Med. 1998, 157, 135–143. [Google Scholar] [CrossRef]
- Innovation and Technology|ResMed. Available online: https://www.resmed.com/ap/en/healthcare-professional/products/innovation-and-technology/ventilation-innovation-and-technology.html (accessed on 31 July 2020).
- Ferreira, J.C.; Chipman, D.W.; Hill, N.S.; Kacmarek, R.M. Bilevel vs ICU ventilators providing noninvasive ventilation: Effect of system leaks: A COPD lung model comparison. Chest 2009, 136, 448–456. [Google Scholar] [CrossRef]
- Carteaux, G.; Lyazidi, A.; Cordoba-Izquierdo, A.; Vignaux, L.; Jolliet, P.; Thille, A.W.; Richard, J.-C.M.; Brochard, L. Patient-ventilator asynchrony during noninvasive ventilation: A bench and clinical study. Chest 2012, 142, 367–376. [Google Scholar] [CrossRef]
- Ueno, Y.; Nakanishi, N.; Oto, J.; Imanaka, H.; Nishimura, M. A bench study of the effects of leak on ventilator performance during noninvasive ventilation. Respir. Care 2011, 56, 1758–1764. [Google Scholar] [CrossRef]
- Prinianakis, G.; Kondili, E.; Georgopoulos, D. Effects of the flow waveform method of triggering and cycling on patient-ventilator interaction during pressure support. Intensive Care Med. 2003, 29, 1950–1959. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Bermejo, J.; Janssens, J.-P.; Rabec, C.; Perrin, C.; Lofaso, F.; Langevin, B.; Carlucci, A.; Lujan, M. SomnoNIV group Framework for patient-ventilator asynchrony during long-term non-invasive ventilation. Thorax 2019, 74, 715–717. [Google Scholar] [CrossRef]
- Lalmolda, C.; Flórez, P.; Grimau, C.; Larrosa, R.; Corral, M.; Sayas, J.; Luján, M. A bench-to-bedside study about trigger asynchronies induced by the introduction of external gas into the non-invasive mechanical ventilation circuit. Sci. Rep. 2021, 11, 23814. [Google Scholar] [CrossRef]
- US Patent Application for A System and Method for Synchronization of Breathing in a Mechanical Ventilator Patent Application (Application #20160008559 Issued 14 January 2016)—Justia Patents Search. Available online: https://patents.justia.com/patent/20160008559 (accessed on 31 July 2020).
- Zhu, K.; Rabec, C.; Gonzalez-Bermejo, J.; Hardy, S.; Aouf, S.; Escourrou, P.; Roisman, G. Combined effects of leaks, respiratory system properties and upper airway patency on the performance of home ventilators: A bench study. BMC Pulm. Med. 2017, 17, 145. [Google Scholar] [CrossRef] [PubMed]
- Battisti, A.; Tassaux, D.; Janssens, J.-P.; Michotte, J.-B.; Jaber, S.; Jolliet, P. Performance characteristics of 10 home mechanical ventilators in pressure-support mode: A comparative bench study. Chest 2005, 127, 1784–1792. [Google Scholar] [CrossRef] [PubMed]
- Delgado, C.; Romero, J.E.; Puig, J.; Izquierdo, A.; Ferrando, C.; Belda, F.J.; Soro, M. Performance of the New Turbine Mid-Level Critical Care Ventilators. Respir. Care 2017, 62, 34–41. [Google Scholar] [CrossRef]
- Lalmolda, C.; Flórez, P.; Corral, M.; Hernández Voth, A.; Grimau, C.; Sayas, J.; Luján, M. Does the Efficacy of High Intensity Ventilation in Stable COPD Depend on the Ventilator Model? A Bench-to-Bedside Study. Int. J. Chron. Obstruct. Pulmon. Dis. 2022, 17, 155–164. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Bermejo, J.; Rabec, C.; Janssens, J.P.; Perrin, C.; Langevin, B.; Pepin, J.L.; Rodenstein, D. Noninvasive ventilation inefficacy due to technically incompatible ventilator settings. Intensive Care Med. 2013, 39, 1154–1156. [Google Scholar] [CrossRef]
- Amato, M.B.; Barbas, C.S.; Bonassa, J.; Saldiva, P.H.; Zin, W.A.; de Carvalho, C.R. Volume-assured pressure support ventilation (VAPSV). A new approach for reducing muscle workload during acute respiratory failure. Chest 1992, 102, 1225–1234. [Google Scholar] [CrossRef] [PubMed]
- Luján, M.; Lalmolda, C.; Ergan, B. Basic Concepts for Tidal Volume and Leakage Estimation in Non-Invasive Ventilation. Turk. Thorac. J. 2019, 20, 140–146. [Google Scholar] [CrossRef]
- Sogo, A.; Montanyà, J.; Monsó, E.; Blanch, L.; Pomares, X.; Lujàn, M. Effect of dynamic random leaks on the monitoring accuracy of home mechanical ventilators: A bench study. BMC Pulm. Med. 2013, 13, 75. [Google Scholar] [CrossRef]
- Luján, M.; Sogo, A.; Grimau, C.; Pomares, X.; Blanch, L.; Monsó, E. Influence of dynamic leaks in volume-targeted pressure support noninvasive ventilation: A bench study. Respir. Care 2015, 60, 191–200. [Google Scholar] [CrossRef]
- Briones Claudett, K.H.; Briones Claudett, M.; Chung Sang Wong, M.; Nuques Martinez, A.; Soto Espinoza, R.; Montalvo, M.; Esquinas Rodriguez, A.; Gonzalez Diaz, G.; Grunauer Andrade, M. Noninvasive mechanical ventilation with average volume assured pressure support (AVAPS) in patients with chronic obstructive pulmonary disease and hypercapnic encephalopathy. BMC Pulm. Med. 2013, 13, 12. [Google Scholar] [CrossRef] [PubMed]
- Magdy, D.M.; Metwally, A. Effect of average volume-assured pressure support treatment on health-related quality of life in COPD patients with chronic hypercapnic respiratory failure: A randomized trial. Respir. Res. 2020, 21, 64. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.A.; Du, Y.P.; Li, L.X.; Wu, F.F.; Hong, S.Q.; Tang, F.X.; Ye, Z.Q. Comparing the effects and compliance between volume-assured and pressure support non-invasive ventilation in patients with chronic respiratory failure. Clin. Respir. J. 2019, 13, 289–298. [Google Scholar] [CrossRef] [PubMed]
- Nilius, G.; Katamadze, N.; Domanski, U.; Schroeder, M.; Franke, K.-J. Non-invasive ventilation with intelligent volume-assured pressure support versus pressure-controlled ventilation: Effects on the respiratory event rate and sleep quality in COPD with chronic hypercapnia. Int. J. Chron. Obstruct. Pulmon. Dis. 2017, 12, 1039–1045. [Google Scholar] [CrossRef]
- Ekkernkamp, E.; Storre, J.H.; Windisch, W.; Dreher, M. Impact of intelligent volume-assured pressure support on sleep quality in stable hypercapnic chronic obstructive pulmonary disease patients: A randomized, crossover study. Respir. Int. Rev. Thorac. Dis. 2014, 88, 270–276. [Google Scholar] [CrossRef]
- Kelly, J.L.; Jaye, J.; Pickersgill, R.E.; Chatwin, M.; Morrell, M.J.; Simonds, A.K. Randomized trial of “intelligent” autotitrating ventilation versus standard pressure support non-invasive ventilation: Impact on adherence and physiological outcomes. Respirology 2014, 19, 596–603. [Google Scholar] [CrossRef]
- Oscroft, N.S.; Chadwick, R.; Davies, M.G.; Quinnell, T.G.; Smith, I.E. Volume assured versus pressure preset non-invasive ventilation for compensated ventilatory failure in COPD. Respir. Med. 2014, 108, 1508–1515. [Google Scholar] [CrossRef] [PubMed]
- Ergan, B.; Oczkowski, S.; Rochwerg, B.; Carlucci, A.; Chatwin, M.; Clini, E.; Elliott, M.; Gonzalez-Bermejo, J.; Hart, N.; Lujan, M.; et al. European Respiratory Society guidelines on long-term home non-invasive ventilation for management of COPD. Eur. Respir. J. 2019, 54, 1901003. [Google Scholar] [CrossRef]
- Patout, M.; Gagnadoux, F.; Rabec, C.; Trzepizur, W.; Georges, M.; Perrin, C.; Tamisier, R.; Pépin, J.-L.; Llontop, C.; Attali, V.; et al. AVAPS-AE versus ST mode: A randomized controlled trial in patients with obesity hypoventilation syndrome. Respirology 2020, 25, 1073–1081. [Google Scholar] [CrossRef]
- Orr, J.E.; Coleman, J.; Criner, G.J.; Sundar, K.M.; Tsai, S.C.; Benjafield, A.V.; Crocker, M.E.; Willes, L.; Malhotra, A.; Owens, R.L.; et al. Automatic EPAP intelligent volume-assured pressure support is effective in patients with chronic respiratory failure: A randomized trial. Respirology 2019, 24, 1204–1211. [Google Scholar] [CrossRef] [PubMed]
- Delorme, M.; Leroux, K.; Leotard, A.; Boussaid, G.; Prigent, H.; Louis, B.; Lofaso, F. Noninvasive Ventilation Automated Technologies: A Bench Evaluation of Device Responses to Sleep-Related Respiratory Events. Respir. Care 2023, 68, 18–30. [Google Scholar] [CrossRef] [PubMed]
- Fasquel, L.; Yazdani, P.; Zaugg, C.; Barras, A.; Michotte, J.-B.; Correvon, N.; Contal, O. Impact of Unintentional Air Leaks on Automatic Positive Airway Pressure Device Performance in Simulated Sleep Apnea Events. Respir. Care 2023, 68, 31–37. [Google Scholar] [CrossRef] [PubMed]
- Jounieaux, V.; Aubert, G.; Dury, M.; Delguste, P.; Rodenstein, D.O. Effects of nasal positive-pressure hyperventilation on the glottis in normal sleeping subjects. J. Appl. Physiol. 1995, 79, 186–193. [Google Scholar] [CrossRef] [PubMed]
- Jounieaux, V.; Rodenstein, D.O.; Mahjoub, Y. On Happy Hypoxia and on Sadly Ignored “Acute Vascular Distress Syndrome” in Patients with COVID-19. Am. J. Respir. Crit. Care Med. 2020, 202, 1598–1599. [Google Scholar] [CrossRef] [PubMed]
- Jounieaux, V.; Rodenstein, D.O. Glottic patency during noninvasive ventilation in patients with chronic obstructive pulmonary disease. Respir. Physiol. Neurobiol. 2019, 259, 178. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Bermejo, J.; Perrin, C.; Janssens, J.P.; Pepin, J.L.; Mroue, G.; Léger, P.; Langevin, B.; Rouault, S.; Rabec, C.; Rodenstein, D.; et al. Proposal for a systematic analysis of polygraphy or polysomnography for identifying and scoring abnormal events occurring during non-invasive ventilation. Thorax 2012, 67, 546–552. [Google Scholar] [CrossRef]
- Fry, D.L.; Ebert, R.V.; Stead, W.W.; Brown, C.C. The mechanics of pulmonary ventilation in normal subjects and in patients with emphysema. Am. J. Med. 1954, 16, 80–97. [Google Scholar] [CrossRef]
- Hyatt, R.E.; Schilder, D.P.; Fry, D.L. Relationship between maximum expiratory flow and degree of lung inflation. J. Appl. Physiol. 1958, 13, 331–336. [Google Scholar] [CrossRef] [PubMed]
- Hyatt, R.E. Expiratory flow limitation. J. Appl. Physiol. 1983, 55, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Dean, J.; Kolsum, U.; Hitchen, P.; Gupta, V.; Singh, D. Clinical characteristics of COPD patients with tidal expiratory flow limitation. Int. J. Chron. Obstruct. Pulmon. Dis. 2017, 12, 1503–1506. [Google Scholar] [CrossRef]
- Anderson, M.R.; Shashaty, M.G.S. Impact of Obesity in Critical Illness. Chest 2021, 160, 2135–2145. [Google Scholar] [CrossRef] [PubMed]
- Junhasavasdikul, D.; Telias, I.; Grieco, D.L.; Chen, L.; Gutierrez, C.M.; Piraino, T.; Brochard, L. Expiratory Flow Limitation During Mechanical Ventilation. Chest 2018, 154, 948–962. [Google Scholar] [CrossRef] [PubMed]
- Akita, T.; Shirai, T.; Mori, K.; Shimoda, Y.; Suzuki, T.; Hayashi, I.; Noguchi, R.; Mochizuki, E.; Sakurai, S.; Saigusa, M.; et al. Association of the forced oscillation technique with negative expiratory pressure in COPD. Respir. Physiol. Neurobiol. 2016, 220, 62–68. [Google Scholar] [CrossRef]
- Dellacà, R.L.; Rotger, M.; Aliverti, A.; Navajas, D.; Pedotti, A.; Farré, R. Noninvasive detection of expiratory flow limitation in COPD patients during nasal CPAP. Eur. Respir. J. 2006, 27, 983–991. [Google Scholar] [CrossRef] [PubMed]
- Dellacà, R.L.; Duffy, N.; Pompilio, P.P.; Aliverti, A.; Koulouris, N.G.; Pedotti, A.; Calverley, P.M.A. Expiratory flow limitation detected by forced oscillation and negative expiratory pressure. Eur. Respir. J. 2007, 29, 363–374. [Google Scholar] [CrossRef] [PubMed]
- Mead, J.; Whittenberger, J.L. Evaluation of airway interruption technique as a method for measuring pulmonary airflow resistance. J. Appl. Physiol. 1954, 6, 408–416. [Google Scholar] [CrossRef] [PubMed]
- Zannin, E.; Chakrabarti, B.; Govoni, L.; Pompilio, P.P.; Romano, R.; Calverley, P.M.A.; Dellacà, R.L. Detection of Expiratory Flow Limitation by Forced Oscillations during Noninvasive Ventilation. Am. J. Respir. Crit. Care Med. 2019, 200, 1063–1065. [Google Scholar] [CrossRef] [PubMed]
- Monteiro, M.B.; Berton, D.C.; Moreira, M.A.F.; Menna-Barreto, S.S.; Teixeira, P.J.Z. Effects of expiratory positive airway pressure on dynamic hyperinflation during exercise in patients with COPD. Respir. Care 2012, 57, 1405–1412. [Google Scholar] [CrossRef] [PubMed]
- Suh, E.-S.; Pompilio, P.; Mandal, S.; Hill, P.; Kaltsakas, G.; Murphy, P.B.; Romano, R.; Moxham, J.; Dellaca, R.; Hart, N. Autotitrating external positive end-expiratory airway pressure to abolish expiratory flow limitation during tidal breathing in patients with severe COPD: A physiological study. Eur. Respir. J. 2020, 56, 1902234. [Google Scholar] [CrossRef] [PubMed]
- Zannin, E.; Milesi, I.; Porta, R.; Cacciatore, S.; Barbano, L.; Trentin, R.; Fanfulla, F.; Vitacca, M.; Dellacà, R.L. Effect of nocturnal EPAP titration to abolish tidal expiratory flow limitation in COPD patients with chronic hypercapnia: A randomized, cross-over pilot study. Respir. Res. 2020, 21, 301. [Google Scholar] [CrossRef] [PubMed]
- McKenzie, J.; Nisha, P.; Cannon-Bailey, S.; Cain, C.; Kissel, M.; Stachel, J.; Proscyk, C.; Romano, R.; Hardy, B.; Calverley, P.M.A. Overnight variation in tidal expiratory flow limitation in COPD patients and its correction: An observational study. Respir. Res. 2021, 22, 319. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Luján, M.; Lalmolda, C. Ventilators, Settings, Autotitration Algorithms. J. Clin. Med. 2023, 12, 2942. https://doi.org/10.3390/jcm12082942
Luján M, Lalmolda C. Ventilators, Settings, Autotitration Algorithms. Journal of Clinical Medicine. 2023; 12(8):2942. https://doi.org/10.3390/jcm12082942
Chicago/Turabian StyleLuján, Manel, and Cristina Lalmolda. 2023. "Ventilators, Settings, Autotitration Algorithms" Journal of Clinical Medicine 12, no. 8: 2942. https://doi.org/10.3390/jcm12082942
APA StyleLuján, M., & Lalmolda, C. (2023). Ventilators, Settings, Autotitration Algorithms. Journal of Clinical Medicine, 12(8), 2942. https://doi.org/10.3390/jcm12082942




