A Systematic Review of Lung Autopsy Findings in Elderly Patients after SARS-CoV-2 Infection
Abstract
1. Introduction
2. Materials and Methods
2.1. Review Protocol
- -
- What are the most common autopsy features among elderly patients after COVID-19?
- -
- Are there any anatomical particularities associated with SARS-CoV-2 mortality?
2.2. Selection Process
2.3. Data Extraction and Quality Assessment
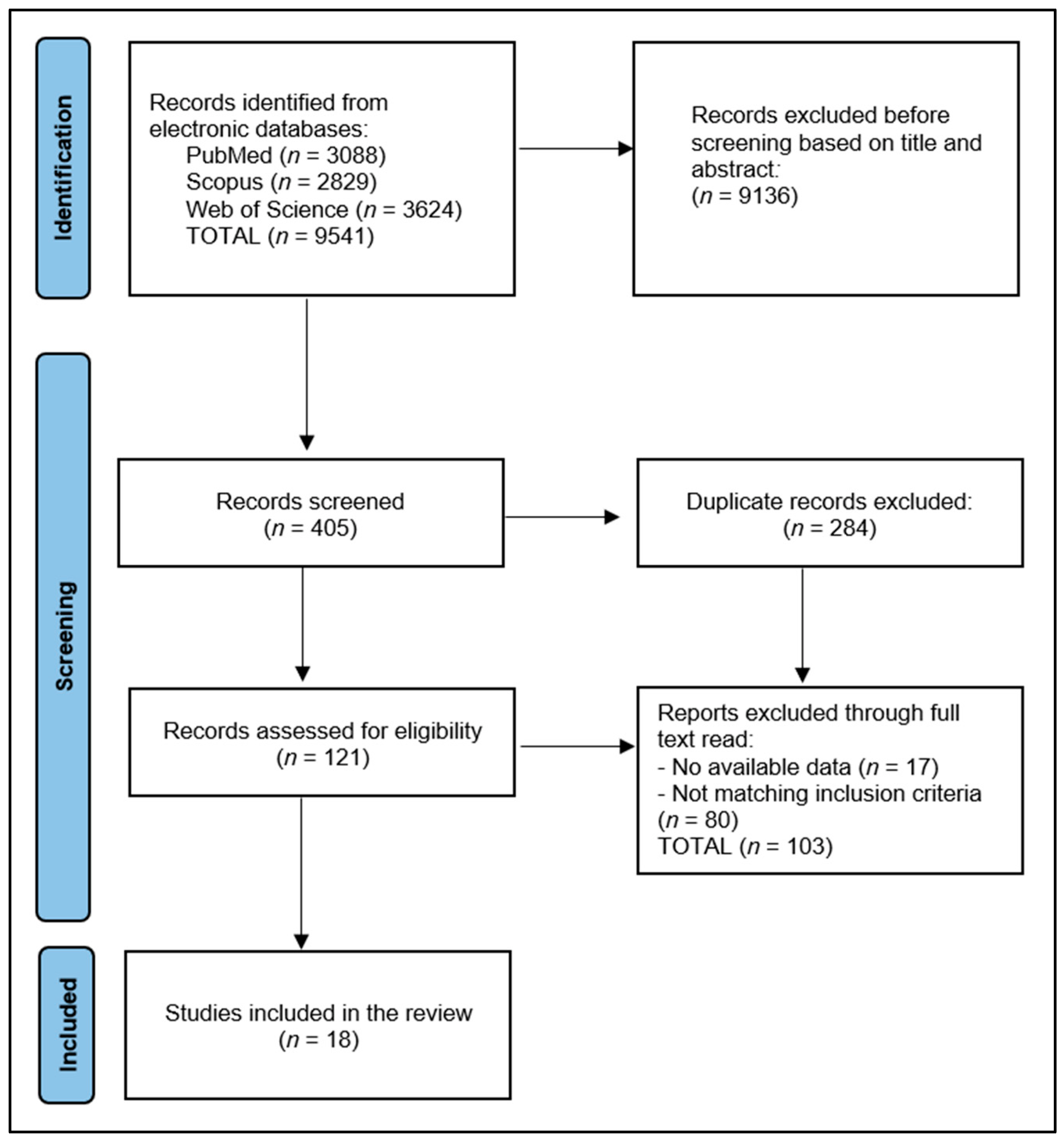
3. Results
4. Discussion
4.1. Literature Findings
4.2. Study Limitations
5. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Marincu, I.; Bratosin, F.; Vidican, I.; Bostanaru, A.-C.; Frent, S.; Cerbu, B.; Turaiche, M.; Tirnea, L.; Timircan, M. Predictive Value of Comorbid Conditions for COVID-19 Mortality. J. Clin. Med. 2021, 10, 2652. [Google Scholar] [CrossRef] [PubMed]
- Citu, C.; Gorun, F.; Motoc, A.; Ratiu, A.; Gorun, O.M.; Burlea, B.; Neagoe, O.; Citu, I.M.; Rosca, O.; Bratosin, F.; et al. Evaluation and Comparison of the Predictive Value of 4C Mortality Score, NEWS, and CURB-65 in Poor Outcomes in COVID-19 Patients: A Retrospective Study from a Single Center in Romania. Diagnostics 2022, 12, 703. [Google Scholar] [CrossRef] [PubMed]
- Hrubaru, I.; Motoc, A.; Bratosin, F.; Rosca, O.; Folescu, R.; Moise, M.L.; Neagoe, O.; Citu, I.M.; Feciche, B.; Gorun, F.; et al. Exploring Clinical and Biological Features of Premature Births among Pregnant Women with SARS-CoV-2 Infection during the Pregnancy Period. J. Pers. Med. 2022, 12, 1871. [Google Scholar] [CrossRef] [PubMed]
- Toma, A.-O.; Prodan, M.; Reddyreddy, A.R.; Seclaman, E.; Crainiceanu, Z.; Bloanca, V.; Bratosin, F.; Dumitru, C.; Pilut, C.N.; Alambaram, S.; et al. The Epidemiology of Malignant Melanoma during the First Two Years of the COVID-19 Pandemic: A Systematic Review. Int. J. Environ. Res. Public Health 2023, 20, 305. [Google Scholar] [CrossRef] [PubMed]
- Mahajan, R.K.; Paul, G.; Mahajan, R.; Gautam, P.L.; Paul, B. Systemic manifestations of COVID-19. J. Anaesthesiol. Clin. Pharmacol. 2020, 36, 435–442. [Google Scholar] [CrossRef]
- Grigoras, M.L.; Citu, I.M.; Citu, C.; Chiriac, V.D.; Gorun, F.; Levai, M.C.; Manolescu, D.; Rosca, O.; Bratosin, F.; Gurumurthy, S.; et al. Evaluation of FIB-4, NFS, APRI and Liver Function Tests as Predictors for SARS-CoV-2 Infection in the Elderly Population: A Matched Case-Control Analysis. J. Clin. Med. 2022, 11, 5149. [Google Scholar] [CrossRef]
- Fericean, R.M.; Citu, C.; Manolescu, D.; Rosca, O.; Bratosin, F.; Tudorache, E.; Oancea, C. Characterization and Outcomes of SARS-CoV-2 Infection in Overweight and Obese Patients: A Dynamic Comparison of COVID-19 Pandemic Waves. J. Clin. Med. 2022, 11, 2916. [Google Scholar] [CrossRef]
- Beyerstedt, S.; Casaro, E.B.; Rangel, É.B. COVID-19: Angiotensin-converting enzyme 2 (ACE2) expression and tissue susceptibility to SARS-CoV-2 infection. Eur. J. Clin. Microbiol. Infect. Dis. 2021, 40, 905–919. [Google Scholar] [CrossRef]
- Zheng, M. ACE2 and COVID-19 Susceptibility and Severity. Aging Dis. 2022, 13, 360–372. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Aleem, A.; Shah, H. Gastrointestinal And Hepatic Manifestations Of Coronavirus (COVID-19) [Updated 2022 May 4]. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, January 2022. Available online: https://www.ncbi.nlm.nih.gov/books/NBK570562/ (accessed on 10 January 2023).
- Otifi, H.M.; Adiga, B.K. Endothelial Dysfunction in COVID-19 Infection. Am. J. Med. Sci. 2022, 363, 281–287. [Google Scholar] [CrossRef]
- Barac, S.; Onofrei, R.R.; Lazureanu, C.; Barna, R.; Tutelca, A.; Rata, A.L. An In Vivo Observational Histological Study of Peripheral Arterial Damage in Patients with Acute Limb Ischemia in SARS-CoV-2 Infection. Diagnostics 2022, 12, 488. [Google Scholar] [CrossRef]
- Barac, S.; Onofrei, R.R.; Neagoe, P.V.; Popescu, A.I.; Pantea, S.; Rață, A.L. An Observational Study on Patients with Acute Limb Ischemia and SARS-CoV-2 Infection: Early and Late Results in Limb Salvage Rate. J. Clin. Med. 2021, 10, 5083. [Google Scholar] [CrossRef]
- Lamers, M.M.; Haagmans, B.L. SARS-CoV-2 pathogenesis. Nat. Rev. Microbiol. 2022, 20, 270–284. [Google Scholar] [CrossRef]
- V’kovski, P.; Kratzel, A.; Steiner, S.; Stalder, H.; Thiel, V. Coronavirus biology and replication: Implications for SARS-CoV-2. Nat. Rev. Microbiol. 2021, 19, 155–170. [Google Scholar] [CrossRef]
- Parekh, U.; Kanchan, T.; Krishan, K. Inexplicable COVID-19! Would pathological autopsy be the panacea? Acta Bio Med. Atenei Parm. 2021, 92, e2021175. [Google Scholar] [CrossRef]
- Evert, K.; Dienemann, T.; Brochhausen, C.; Lunz, D.; Lubnow, M.; Ritzka, M.; Keil, F.; Trummer, M.; Scheiter, A.; Salzberger, B.; et al. Autopsy findings after long-term treatment of COVID-19 patients with microbiological correlation. Virchows Arch. 2021, 479, 97–108. [Google Scholar] [CrossRef]
- Barata, P.I.; Crisan, A.F.; Maritescu, A.; Negrean, R.A.; Rosca, O.; Bratosin, F.; Citu, C.; Oancea, C. Evaluating Virtual and Inpatient Pulmonary Rehabilitation Programs for Patients with COPD. J. Pers. Med. 2022, 12, 1764. [Google Scholar] [CrossRef]
- Popescu, A.; Craina, M.; Pantea, S.; Pirvu, C.; Radu, D.; Marincu, I.; Bratosin, F.; Bogdan, I.; Hosin, S.; Citu, C.; et al. COVID-19 Pandemic Impact on Surgical Treatment Methods for Early-Stage Cervical Cancer: A Population-Based Study in Romania. Healthcare 2022, 10, 639. [Google Scholar] [CrossRef]
- Papava, I.; Dehelean, L.; Romosan, R.S.; Bondrescu, M.; Dimeny, C.Z.; Domuta, E.M.; Bratosin, F.; Bogdan, I.; Grigoras, M.L.; Tigmeanu, C.V.; et al. The Impact of Hyper-Acute Inflammatory Response on Stress Adaptation and Psychological Symptoms of COVID-19 Patients. Int. J. Environ. Res. Public Health 2022, 19, 6501. [Google Scholar] [CrossRef]
- Marincu, I.; Citu, C.; Bratosin, F.; Bogdan, I.; Timircan, M.; Gurban, C.V.; Bota, A.V.; Braescu, L.; Grigoras, M.L. Clinical Characteristics and Outcomes of COVID-19 Hospitalized Patients: A Comparison between Complete mRNA Vaccination Profile and Natural Immunity. J. Pers. Med. 2022, 12, 259. [Google Scholar] [CrossRef]
- Djaharuddin, I.; Munawwarah, S.; Nurulita, A.; Ilyas, M.; Tabri, N.A.; Lihawa, N. Comorbidities and mortality in COVID-19 patients. Gac. Sanit. 2021, 35 (Suppl. 2), S530–S532. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Citu, C.; Burlea, B.; Gorun, F.; Motoc, A.; Gorun, O.M.; Malita, D.; Ratiu, A.; Margan, R.; Grigoras, M.L.; Bratosin, F.; et al. Predictive Value of Blood Coagulation Parameters in Poor Outcomes in COVID-19 Patients: A Retrospective Observational Study in Romania. J. Clin. Med. 2022, 11, 2831. [Google Scholar] [CrossRef] [PubMed]
- Buja, L.M.; Wolf, D.A.; Zhao, B.; Akkanti, B.; McDonald, M.; Lelenwa, L.; Reilly, N.; Ottaviani, G.; Elghetany, M.T.; Trujillo, D.O.; et al. Emerging spectrum of cardiopulmonary pathology of the coronavirus disease 2019 (COVID-19): Report of three autopsies from Houston, Texas and review of autopsy findings from other United States cities. Cardiovasc. Pathol. 2020, 48, 107233. [Google Scholar] [CrossRef] [PubMed]
- Casagrande, M.; Fitzek, A.; Püschel, K.; Aleshcheva, G.; Schultheiss, H.-P.; Berneking, L.; Spitzer, M.S.; Schultheiss, M. Detection of SARS-CoV-2 in human retinal biopsies of deceased COVID-19 patients. Ocul. Immunol. Inflamm. 2020, 28, 721–725. [Google Scholar] [CrossRef] [PubMed]
- Loibner, M.; Langner, C.; Regitnig, P.; Gorkiewicz, G.; Zatloukal, K. Biosafety Requirements for Autopsies of Patients with COVID-19: Example of a BSL-3 Autopsy Facility Designed for Highly Pathogenic Agents. Pathobiology 2021, 88, 37–45. [Google Scholar] [CrossRef]
- Fericean, R.M.; Oancea, C.; Reddyreddy, A.R.; Rosca, O.; Bratosin, F.; Bloanca, V.; Citu, C.; Alambaram, S.; Vasamsetti, N.G.; Dumitru, C. Outcomes of Elderly Patients Hospitalized with the SARS-CoV-2 Omicron B.1.1.529 Variant: A Systematic Review. Int. J. Environ. Res. Public Health 2023, 20, 2150. [Google Scholar] [CrossRef]
- Dadras, O.; SeyedAlinaghi, S.; Karimi, A.; Shamsabadi, A.; Qaderi, K.; Ramezani, M.; Mirghaderi, S.P.; Mahdiabadi, S.; Vahedi, F.; Saeidi, S.; et al. COVID-19 mortality and its predictors in the elderly: A systematic review. Health Sci. Rep. 2022, 5, e657, Erratum in 2022, 5, e723. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Schiavo, J.H. PROSPERO: An International Register of Systematic Review Protocols. Med. Ref. Serv. Q. 2019, 38, 171–180. [Google Scholar] [CrossRef]
- Foster, E.D.; Deardorff, A. Open Science Framework (OSF). J. Med. Libr. Assoc. 2017, 105, 203–206. [Google Scholar] [CrossRef] [PubMed Central]
- Tran, L.; Tam, D.N.H.; Elshafay, A.; Dang, T.; Hirayama, K.; Huy, N.T. Quality assessment tools used in systematic reviews of in vitro studies: A systematic review. BMC Med. Res. Methodol. 2021, 21, 101. [Google Scholar] [CrossRef]
- Lax, S.F.; Skok, K.; Zechner, P.; Kessler, H.H.; Kaufmann, N.; Koelblinger, C.; Vander, K.; Bargfrieder, U.; Trauner, M. Pulmonary arterial thrombosis in COVID-19 with fatal outcome: Results from a prospective, single-center, clinicopathologic case series. Ann. Intern. Med. 2020, 173, 350–361. [Google Scholar] [CrossRef]
- Menter, T.; Haslbauer, J.D.; Nienhold, R.; Savic, S.; Hopfer, H.; Deigendesch, N.; Frank, S.; Turek, D.; Willi, N.; Pargger, H.; et al. Postmortem examination of COVID19 patients reveals diffuse alveolar damage with severe capillary congestion and variegated findings in lungs and other organs suggesting vascular dysfunction. Histopathology 2020, 77, 198–209. [Google Scholar] [CrossRef]
- Schaller, T.; Hirschbuhl, K.; Burkhardt, K.; Braun, G.; Trepel, M.; Märkl, B.; Claus, R. Postmortem examination of patients with COVID-19. JAMA 2020, 323, 2518–2520. [Google Scholar] [CrossRef]
- Martines, R.B.; Ritter, J.M.; Matkovic, E.; Gary, J.; Bollweg, B.C.; Bullock, H.; Goldsmith, C.S.; Silva-Flannery, L.; Seixas, J.N.; Reagan-Steiner, S.; et al. Pathology and pathogenesis of SARS-CoV-2 associated with fatal coronavirus disease, United States. Emerg. Infect. Dis. 2020, 26, 2005–2015. [Google Scholar] [CrossRef]
- Flikweert, A.W.; Grootenboers, M.J.; Yick, D.C.; du Mée, A.W.; van der Meer, N.J.; Rettig, T.C.; Kant, M.K. Late histopathologic characteristics of critically ill COVID-19 patients: Different phenotypes without evidence of invasive aspergillosis, a case series. J. Crit. Care 2020, 59, 149–155. [Google Scholar] [CrossRef]
- Wu, J.H.; Li, X.; Huang, B.; Su, H.; Li, Y.; Luo, D.J.; Chen, S.; Ma, L.; Wang, S.H.; Nie, X.; et al. Pathological changes of fatal coronavirus disease 2019 (COVID-19) in the lungs: Report of 10 cases by postmortem needle autopsy. Chi. J. Pathol. 2020, 49, 568–575. [Google Scholar]
- Grosse, C.; Grosse, A.; Salzer, H.J.; Dünser, M.W.; Motz, R.; Langer, R. Analysis of cardiopulmonary findings in COVID-19 fatalities: High incidence of pulmonary artery thrombi and acute suppurative bronchopneumonia. Cardiovasc. Pathol. 2020, 49, 107263. [Google Scholar] [CrossRef]
- Deinhardt-Emmer, S.; Wittschieber, D.; Sanft, J.; Kleemann, S.; Elschner, S.; Haupt, K.F.; Vau, V.; Häring, C.; Rödel, J.; Henke, A.; et al. Early postmortem mapping of SARS-CoV-2 RNA in patients with COVID-19 and the correlation with tissue damage. Elife 2021, 10, e60361. [Google Scholar] [CrossRef]
- Roden, A.C.; Bois, M.C.; Johnson, T.F.; Aubry, M.C.; Alexander, M.P.; Hagen, C.E.; Lin, P.T.; Quinton, R.A.; Maleszewski, J.J.; Boland, J.M. The spectrum of histopathologic findings in lungs of patients with fatal coronavirus disease 2019 (COVID-19) infection. Arch. Pathol. Lab. Med. 2021, 145, 11–21. [Google Scholar] [CrossRef]
- Barisione, E.; Grillo, F.; Ball, L.; Bianchi, R.; Grosso, M.; Morbini, P.; Pelosi, P.; Patroniti, N.A.; De Lucia, A.; Orengo, G.; et al. Fibrotic progression and radiologic correlation in matched lung samples from COVID-19 postmortems. Virchows Arch. 2021, 478, 471–485. [Google Scholar] [CrossRef] [PubMed]
- De Michele, S.; Sun, Y.; Yilmaz, M.M.; Katsyv, I.; Salvatore, M.; Dzierba, A.L.; Marboe, C.C.; Brodie, D.; Patel, N.M.; Garcia, C.K.; et al. Forty postmortem examinations in COVID-19 patients. Am. J. Clin. Pathol. 2020, 154, 748–760. [Google Scholar] [CrossRef] [PubMed]
- Youd, E.; Moore, L. COVID-19 autopsy in people who died in community settings: The first series. J. Clin. Pathol. 2020, 73, 840–844. [Google Scholar] [CrossRef] [PubMed]
- Skok, K.; Stelzl, E.; Trauner, M.; Kessler, H.H.; Lax, S.F. Postmortem viral dynamics and tropism in COVID-19 patients in correlation with organ damage. Virchows Arch. 2021, 478, 343–353. [Google Scholar] [CrossRef] [PubMed]
- Remmelink, M.; De Mendonca, R.; D’Haene, N.; De Clercq, S.; Verocq, C.; Lebrun, L.; Lavis, P.; Racu, M.-L.; Trépant, A.-L.; Maris, C.; et al. Unspecific postmortem findings despite multiorgan viral spread in COVID-19 patients. Crit. Care 2020, 24, 495. [Google Scholar] [CrossRef]
- Borczuk, A.C.; Salvatore, S.P.; Seshan, S.V.; Patel, S.S.; Bussel, J.B.; Mostyka, M.; Elsoukkary, S.; He, B.; Del Vecchio, C.; Fortarezza, F.; et al. COVID-19 pulmonary pathology: A multi-institutional autopsy cohort from Italy and New York City. Mod. Pathol. 2020, 33, 2156–2168. [Google Scholar] [CrossRef]
- D’Onofrio, V.; Keulen, L.; Vandendriessche, A.; Dubois, J.; Cartuyvels, R.; Vanden Abeele, M.E.; Fraussen, J.; Vandormael, P.; Somers, V.; Achten, R.; et al. Studying the clinical, radiological, histological, microbiological, and immunological evolution during the different COVID-19 disease stages using minimal invasive autopsy. Sci. Rep. 2022, 12, 1360. [Google Scholar] [CrossRef]
- Mikhaleva, L.M.; Cherniaev, A.L.; Samsonova, M.V.; Zayratyants, O.V.; Kakturskiy, L.V.; Vasyukova, O.A.; Birukov, A.E.; Kontorshchikov, A.S.; Sorokina, A.V.; Sinelnikov, M.Y. Pathological Features in 100 Deceased Patients With COVID-19 in Correlation With Clinical and Laboratory Data. Pathol. Oncol. Res. 2021, 27, 1609900. [Google Scholar] [CrossRef]
- Schwab, N.; Nienhold, R.; Henkel, M.; Baschong, A.; Graber, A.; Frank, A.; Mensah, N.; Koike, J.; Hernach, C.; Sachs, M.; et al. COVID-19 Autopsies Reveal Underreporting of SARS-CoV-2 Infection and Scarcity of Co-infections. Front. Med. 2022, 9, 868954. [Google Scholar] [CrossRef]
- Edler, C.; Schröder, A.S.; Aepfelbacher, M.; Fitzek, A.; Heinemann, A.; Heinrich, F.; Sperhake, J.P. Dying with SARS-CoV-2 infection—An autopsy study of the first consecutive 80 cases in Hamburg, Germany. Int. J. Legal Med. 2020, 134, 1275–1284. [Google Scholar] [CrossRef]
- Falasca, L.; Nardacci, R.; Colombo, D.; Lalle, E.; Di Caro, A.; Nicastri, E.; Del Nonno, F. Postmortem findings in Italian patients with COVID-19: A descriptive full autopsy study of cases with and without comorbidities. J. Infect. Dis. 2020, 222, 1807–1815. [Google Scholar] [CrossRef]
- Keresztesi, A.A.; Perde, F.; Ghita-Nanu, A.; Radu, C.C.; Negrea, M.; Keresztesi, G. Postmortem diagnosis and autopsy findings in SARS-CoV-2 infection: Forensic case series. Diagnostics 2020, 10, 1070. [Google Scholar] [CrossRef]
- Maiese, A.; Passaro, G.; Matteis, A.; Raffaele, R.; Paolo, M.D. Thromboinflammatory response in SARS-CoV-2 sepsis. Med.-Leg. J. 2020, 88, 78–80. [Google Scholar] [CrossRef]
- Zanza, C.; Racca, F.; Longhitano, Y.; Volonnino, G.; Russa, R.L. Risk management and treatment of coagulation disorders related to COVID-19 infection. Int. J. Environ. Res. Public Health 2021, 18, 1268. [Google Scholar] [CrossRef]
- Ackermann, M.; Verleden, S.E.; Kuehnel, M.; Haverich, A.; Welte, T.; Laenger, F.; Vanstapel, A.; Werlein, C.; Stark, H.; Tzankov, A.; et al. Pulmonary vascular endothelialitis, thrombosis, and angiogenesis in COVID-19. N. Engl. J. Med. 2020, 383, 120–128. [Google Scholar] [CrossRef]
- Tian, S.; Xiong, Y.; Liu, H.; Niu, L.; Guo, J.; Liao, M.; Xiao, S.-Y. Pathological study of the 2019 novel coronavirus disease (COVID-19) through postmortem core biopsies. Mod. Pathol. 2020, 33, 1007–1014. [Google Scholar] [CrossRef]
- Sauter, J.L.; Baine, M.K.; Butnor, K.J.; Buonocore, D.J.; Chang, J.C.; Jungbluth, A.A.; Szabolcs, M.J.; Morjaria, S.; Mount, S.L.; Rekhtman, N.; et al. Insights into pathogenesis of fatal COVID-19 pneumonia from histopathology with immunohistochemical and viral RNA studies. Histopathology 2020, 77, 915–925. [Google Scholar] [CrossRef]
- Nie, X.; Qian, L.; Sun, R.; Huang, B.; Dong, X.; Xiao, Q.; Zhang, Q.; Lu, T.; Yue, L.; Chen, S.; et al. Multi-organ proteomic landscape of COVID-19 autopsies. Cell 2021, 184, 775–791.e14. [Google Scholar] [CrossRef]
- Barton, L.M.; Duval, E.J.; Stroberg, E.; Ghosh, S.; Mukhopadhyay, S. COVID-19 autopsies, Oklahoma, USA. Am. J. Clin. Pathol. 2020, 153, 725–733. [Google Scholar] [CrossRef]
- Bruce-Brand, C.; Allwood, B.W.; Koegelenberg, C.F.; Lalla, U.; Louw, E.; Diacon, A.H.; Schubert, P.T. Postmortem lung biopsies from four patients with COVID-19 at a tertiary hospital in Cape Town, South Africa. S. Afr. Med. J. 2020, 110, 1195–1200. [Google Scholar] [CrossRef]
- Duarte-Neto, A.N.; Monteiro, R.A.; da Silva, L.F.; Malheiros, D.M.A.C.; De Oliveira, E.P.; Theodoro-Filho, J.; Pinho, J.R.R.; Gomes-Gouvêa, M.S.; Salles, A.P.M.; De Oliveira, I.R.S.; et al. Pulmonary and systemic involvement in COVID-19 patients assessed with ultrasound-guided minimally invasive autopsy. Histopathology 2020, 77, 186–197. [Google Scholar] [CrossRef] [PubMed]
- Carsana, L.; Sonzogni, A.; Nasr, A.; Rossi, R.S.; Pellegrinelli, A.; Zerbi, P.; Rech, R.; Colombo, R.; Antinori, S.; Corbellino, M.; et al. Pulmonary postmortem findings in a series of COVID-19 cases from northern Italy: A two-centre descriptive study. Lancet Infect. Dis. 2020, 20, 1135–1140. [Google Scholar] [CrossRef] [PubMed]
- Fox, S.E.; Akmatbekov, A.; Harbert, J.L.; Li, G.; Brown, J.Q.; Heide, R.S.V. Pulmonary and cardiac pathology in African American patients with COVID-19: An autopsy series from New Orleans. Lancet Respir. Med. 2020, 8, 681–686. [Google Scholar] [CrossRef] [PubMed]
- Chmielik, E.; Jazowiecka-Rakus, J.; Dyduch, G.; Nasierowska-Guttmejer, A.; Michalowski, L.; Sochanik, A.; Ulatowska-Bialas, M. COVID-19 autopsies: A case series from Poland. Pathobiology 2021, 88, 78–87. [Google Scholar] [CrossRef] [PubMed]
- Wichmann, D.; Sperhake, J.-P.; Lutgehetmann, M.; Steurer, S.; Edler, C.; Heinemann, A.; Heinrich, F.; Mushumba, H.; Kniep, I.; Schröder, A.S.; et al. Autopsy findings and venous thromboembolism in patients with COVID-19: A prospective cohort study. Ann. Intern. Med. 2020, 173, 268–277. [Google Scholar] [CrossRef] [PubMed]
- Beigmohammadi, M.T.; Jahanbin, B.; Safaei, M.; Amoozadeh, L.; Khoshavi, M.; Mehrtash, V.; Jafarzadeh, B.; Abdollahi, A. Pathological findings of postmortem biopsies from lung, heart, and liver of 7 deceased COVID-19 patients. Int. J. Surg. Pathol. 2021, 29, 135–145. [Google Scholar] [CrossRef]
- Bryce, C.; Grimes, Z.; Pujadas, E.; Ahuja, S.; Beasley, M.B.; Albrecht, R.; Hernandez, T.; Stock, A.; Zhao, Z.; Al Rasheed, M.; et al. Pathophysiology of SARS-CoV-2: Targeting of endothelial cells renders a complex disease with thrombotic microangiopathy and aberrant immune response: The Mount Sinai COVID-19 autopsy experience. medRxiv 2020. [Google Scholar] [CrossRef]
- Elsoukkary, S.S.; Mostyka, M.; Dillard, A.; Berman, D.R.; Ma, L.X.; Chadburn, A.; Salvatore, S.P. Autopsy findings in 32 patients with COVID-19: A single institution experience. Pathobiology 2021, 88, 55–67. [Google Scholar] [CrossRef]
- Satturwar, S.; Fowkes, M.; Farver, C.; Wilson, A.M.; Eccher, A.; Girolami, I.; Pujadas, E.; Bryce, C.; Salem, F.; El Jamal, S.M.; et al. Postmortem findings associated with SARS-CoV-2: Systematic review and meta-analysis. Am. J. Surg. Pathol. 2021, 45, 587. [Google Scholar] [CrossRef]
- Konopka, K.E.; Nguyen, T.; Jentzen, J.M.; Rayes, O.; Schmidt, C.J.; Wilson, A.M.; Farver, C.F.; Myers, J.L. Diffuse alveolar damage (DAD) from coronavirus disease 2019 infection is morphologically indistinguishable from other causes of DAD. Histopathology 2020, 77, 570–578. [Google Scholar] [CrossRef]
- Duarte-Neto, A.N.; Caldinia, E.G.; Gomes-Gouv, M.S.; Kanamura, C.T.; de Almeida Monteiro, R.A.; Ferranti, J.F.; Cordeiro Ventura, A.M.; Regalio, F.A.; Fiorenzano, D.M.; Gibelli, M.A.B.C.; et al. An autopsy study of the spectrum of severe COVID-19 in children: From SARS to different phenotypes of MIS-C. EClinicalMedicine 2021, 35, 100850. [Google Scholar] [CrossRef]
- Dolhnikoff, M.; Ferranti, J.F.; Monteiro, R.A.; Duarte-neto, A.N.; Gomes-Gouvea, M.S. SARS-CoV-2 in cardiac tissue of a child with COVID-19-related multisystem inflammatory syndrome. Lancet Child Adolesc. Health 2020, 20, 3025–3027. [Google Scholar] [CrossRef]
- Yao, X.H.; Li, T.Y.; He, Z.C.; Ping, Y.F.; Liu, H.W.; Yu, S.C.; Mou, H.M.; Wang, L.H.; Zhang, H.R.; Fu, W.J.; et al. A pathological report of three COVID-19 cases by minimal invasive autopsies. Chin. J. Pathol. 2020, 49, 411–417. [Google Scholar] [CrossRef]
- Suba, Z. Prevention and therapy of COVID-19 via exogenous estrogen treatment for both male and female patients: An opinion paper. J. Pharm. Pharm. Sci. 2020, 23, 75–85. [Google Scholar] [CrossRef]
- Miyara, M.; Tubach, F.; Pourcher, V.; Morelot-Panzini, C.; Pernet, J.; Harrois, J.; Lebbah, S.; Morawiec, E.; Gorochov, G.; Caumes, E.; et al. Low rate of daily active tobacco smoking in patients with symptomatic COVID-19. Qeios 2020. [Google Scholar] [CrossRef]
- Takahashi, T.; Ellingson, M.K.; Wong, P.; Israelow, B.; Lucas, C.; Klein, J.; Silva, J.; Mao, T.; Oh, J.E.; Tokuyama, M.; et al. Sex differences in immune responses to SARS-CoV-2 that underlie disease outcomes. Nature 2020, 588, 315–320. [Google Scholar] [CrossRef]
| Study & Author | Country | Study Year | Study Design | Study Quality |
|---|---|---|---|---|
| 1 [33] Lax et al. | Austria | 2020 | Prospective cohort | Excellent |
| 2 [34] Menter et al. | Switzerland | 2020 | Retrospective cohort | Good |
| 3 [35] Schaller et al. | Germany | 2020 | Prospective cohort | Fair |
| 4 [36] Martines et al. | USA | 2020 | Retrospective cohort | Excellent |
| 5 [37] Flikweert et al. | Netherlands | 2020 | Prospective cohort | Good |
| 6 [38] Wu et al. | China | 2020 | Prospective cohort | Fair |
| 7 [39] Grosse et al. | Austria | 2020 | Prospective cohort | Excellent |
| 8 [40] Deinhardt–Emmer et al. | Germany | 2021 | Prospective cohort | Good |
| 9 [41] Roden et al. | USA | 2021 | Retrospective cohort | Good |
| 10 [42] Barisione et al. | Italy | 2021 | Prospective cohort | Excellent |
| 11 [43] De Michele et al. | USA | 2020 | Prospective cohort | Excellent |
| 12 [44] Youd and Moore | UK | 2020 | Retrospective cohort | Good |
| 13 [45] Skok et al. | Austria | 2021 | Prospective cohort | Excellent |
| 14 [46] Remmelink et al. | Belgium | 2020 | Retrospective cohort | Good |
| 15 [47] Borczuk et al. | USA | 2020 | Prospective cohort | Excellent |
| 16 [48] D’Onofrio et al. | Belgium | 2022 | Prospective cohort | Excellent |
| 17 [49] Mikhaleva et al. | Russia | 2021 | Prospective cohort | Excellent |
| 18 [50] Schwab et al. | Switzerland | 2022 | Prospective cohort | Excellent |
| Study Number | Autopsies | Age, Years (Mean/Median) | Sex (Men, %) | Lung Disease, Smoking |
|---|---|---|---|---|
| 1 [33] | 11 | 80.5 | 72.7% | COPD—18.2% |
| 2 [34] | 21 | 76.0 | 80.9% | COPD—18.2%; Smoking—14.3% |
| 3 [35] | 12 | 79.0 | 58.3% | COPD—16.7%; Lung cancer—8.3% |
| 4 [36] | 8 | 73.5 | 50.0% | COPD—25.0% |
| 5 [37] | 7 | 73.0 | 71.4% | NR |
| 6 [38] | 10 | 70.0 | 70.0% | COPD—20.0%; Smoking—50.0% |
| 7 [39] | 14 | 82.0 | 64.3% | COPD—42.9% |
| 8 [40] | 11 | 72.2 | 63.6% | COPD—18.2% |
| 9 [41] | 8 | 79.0 | 87.5% | COPD—12.5% |
| 10 [42] | 8 | 76.0 | 75.0% | None |
| 11 [43] | 40 | 71.5 | 70.0% | COPD—25.0% |
| 12 [44] | 9 | 72.1 | 44.4% | COPD—44.4% |
| 13 [45] | 28 | 82.9 | 60.7% | NR |
| 14 [46] | 17 | 71.0 | 70.6% | COPD—11.8% |
| 15 [47] | 68 | 73.0 | 69.0% | COPD—8.8%; Smoking—30.0% |
| 16 [48] | 44 | 82.0 | 56.8% | COPD—3.1% |
| 17 [49] | 100 | 70.5 | 47.0% | COPD—1% |
| 18 [50] | 62 | 76.5 | 65.0% | COPD—33.5% |
| Study Number | Lung Weight (Grams) | Pathologic Findings (%) | Other Findings |
|---|---|---|---|
| 1 [33] | RL—998 g; LL—795 g | Massive bilateral congestion—81.8%; Moderate emphysematous changes—100%; Fibrous adhesions—63.6%; Focal or extensive pulmonary infarctions—72.7% | DAD, hyaline membranes, proliferation of pneumocytes and fibroblasts |
| 2 [34] | NR | Severe mucous tracheitis/tracheobronchitis—30.0%; Heavy and firm lung parenchyma with severe congestion—100%; Proliferative DAD—38.1%; Bronchopneumonia—47.6% | Extensive suppurative bronchopneumonic infiltrates, unevenly bluish–red color, alveolar hemorrhage in conjunction with pulmonary embolism |
| 3 [35] | NR | DAD—83.3%; Emphysema—16.7%; Minor neutrophil infiltration—41.7% | Hyaline membrane formation, intra-alveolar edema, and thickened alveolar septa |
| 4 [36] | NR | DAD—87.5%; Squamous metaplasia and atypical pneumocytes—37.5%; Hemosiderin-laden macrophages and hemorrhage—50.0%; Interstitial pneumonitis—50.0% | Desquamation of pneumocytes, hyaline membranes, alveolar edema, and fibrin deposits, type II pneumocyte hyperplasia, alveolar infiltrates, increased alveolar macrophages |
| 5 [37] | NR | Organized pneumonia—57.1%; DAD—14.7%; Intra-alveolar fibro myxoid/fibroblastic bodies—57.1%; | Micro-thrombi, hyperplasia with atypia, multinucleated giant cell, intranuclear inclusion bodies |
| 6 [38] | NR | Fibrinous and suppurative alveolar exudation—90.0%; | Hyaline membrane and fibroblastic proliferation of alveolar septum, reactive hyperplasia and desquamation of the type II pneumocytes, acute bronchiolitis with mucous membrane exfoliation |
| 7 [39] | RR—390 g to 1340 g; LL—350 g to 1100 g | DAD—100%; Type II pneumocyte hyperplasia—92.9%; Squamous metaplasia—92.9%; Multinucleated cells—85.7%; Secondary acute bronchopneumonia—78.6% | Pulmonary edema, significant bilateral vascular congestion, and dark red to grayish-red color of the lung parenchyma in the lower lobes and basal regions of the higher lobes |
| 8 [40] | RR—1380 g; LL—1107 g | DAD—75.0%; Purulent bronchitis and bronchopneumonia—34.5%; Hyperemia and edema—75.0%; Thrombemboli—50.0% | Severe intra-alveolar and interstitial hemorrhages, severe loss of structured pneumocytes, hyaline membranes, fibrinous edema, and interstitial proliferation |
| 9 [41] | RR+LL (average)—1220 g | Consolidation—62.5%; DAD—75.0%; Aspiration pneumonia—25.0%; Thrombemboli—62.5%; Perivascular chronic inflammation—50.0% | Hyaline membranes and interstitial fibroblast proliferation, interstitial fibroblast proliferation, organizing pneumonia |
| 10 [42] | NR | Vascular injury and platelet microthrombi—50.0%; Early DAD—25.0%; Mid proliferative DAD—37.5%; Late DAD—37.5% | Type II pneumocyte hyperplasia, atypical pneumocytes, occasional multi-nucleation, and intracytoplasmic eosinophilic Mallory-like inclusions in type 2 pneumocytes |
| 11 [43] | NR | Acute lung injury—72.5%; DAD—83.3%; Intravascular fibrin or platelet-rich aggregates—90.0%; Vascular congestion and hemangiomatosis-like changes—50.0% | Macroscopic and microscopic pulmonary thrombi |
| 12 [44] | RR—932 g; LL—642 g | Lung consolidation—77.8%; Pulmonary edema—77.8%; Bronchopneumonia—33.3% | No macroscopic thromboemboli or areas of infarction |
| 13 [45] | NR | Pulmonary artery thrombosis—100%; Bronchopneumonia—89.5%; Fibrosis—73.7% | Edema, proliferation, and hyaline membranes in all samples |
| 14 [46] | NR | DAD—88.2%; Lung microthrombosis—64.7%; Bronchopneumonia—47.1%; Lung infarct—23.5% | Interstitial and alveolar fibroblastic proliferation, atypical pneumocytes, hyaline membranes, and type II pneumocyte hyperplasia |
| 15 [47] | RR+LL (average)—>1300 g | Large vessel thrombi—42.0%; Focal and diffuse microthrombi—88.0%; DAD—16.2%; Type II pneumocytes hyperplasia—87.0%; Organizing pneumonia—34.0% | Hyaline membranes, atypical alveolar cells, basophilic intracytoplasmic inclusions |
| 16 [48] | NR | DAD—84.1%; Fibrosis—59.1% | Fibrin deposition, hyaline membranes, atypic pneumocytes |
| 17 [49] | NR | Pulmonary edema—61.0%; Hyaline membranes—67.0%; DAD—75.0%; Intra-alveolar hemorrhage—75.0% | Bronchial epithelium desquamation, alveolar macrophages, arterial thrombi, interstitial inflammation |
| 18 [50] | NR | DAD—20.9%; Thrombosis—38.7%; Secondary pneumonia—27.4% | NR |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Septimiu-Radu, S.; Gadela, T.; Gabriela, D.; Oancea, C.; Rosca, O.; Lazureanu, V.E.; Fericean, R.M.; Bratosin, F.; Dumitrescu, A.; Stoicescu, E.R.; et al. A Systematic Review of Lung Autopsy Findings in Elderly Patients after SARS-CoV-2 Infection. J. Clin. Med. 2023, 12, 2070. https://doi.org/10.3390/jcm12052070
Septimiu-Radu S, Gadela T, Gabriela D, Oancea C, Rosca O, Lazureanu VE, Fericean RM, Bratosin F, Dumitrescu A, Stoicescu ER, et al. A Systematic Review of Lung Autopsy Findings in Elderly Patients after SARS-CoV-2 Infection. Journal of Clinical Medicine. 2023; 12(5):2070. https://doi.org/10.3390/jcm12052070
Chicago/Turabian StyleSeptimiu-Radu, Susa, Tejaswi Gadela, Doros Gabriela, Cristian Oancea, Ovidiu Rosca, Voichita Elena Lazureanu, Roxana Manuela Fericean, Felix Bratosin, Andreea Dumitrescu, Emil Robert Stoicescu, and et al. 2023. "A Systematic Review of Lung Autopsy Findings in Elderly Patients after SARS-CoV-2 Infection" Journal of Clinical Medicine 12, no. 5: 2070. https://doi.org/10.3390/jcm12052070
APA StyleSeptimiu-Radu, S., Gadela, T., Gabriela, D., Oancea, C., Rosca, O., Lazureanu, V. E., Fericean, R. M., Bratosin, F., Dumitrescu, A., Stoicescu, E. R., Bagiu, I., Murariu, M., & Mavrea, A. (2023). A Systematic Review of Lung Autopsy Findings in Elderly Patients after SARS-CoV-2 Infection. Journal of Clinical Medicine, 12(5), 2070. https://doi.org/10.3390/jcm12052070








