Chronic Low Back Pain: A Narrative Review of Recent International Guidelines for Diagnosis and Conservative Treatment
Abstract
1. Introduction
2. Materials and Methods
3. Results
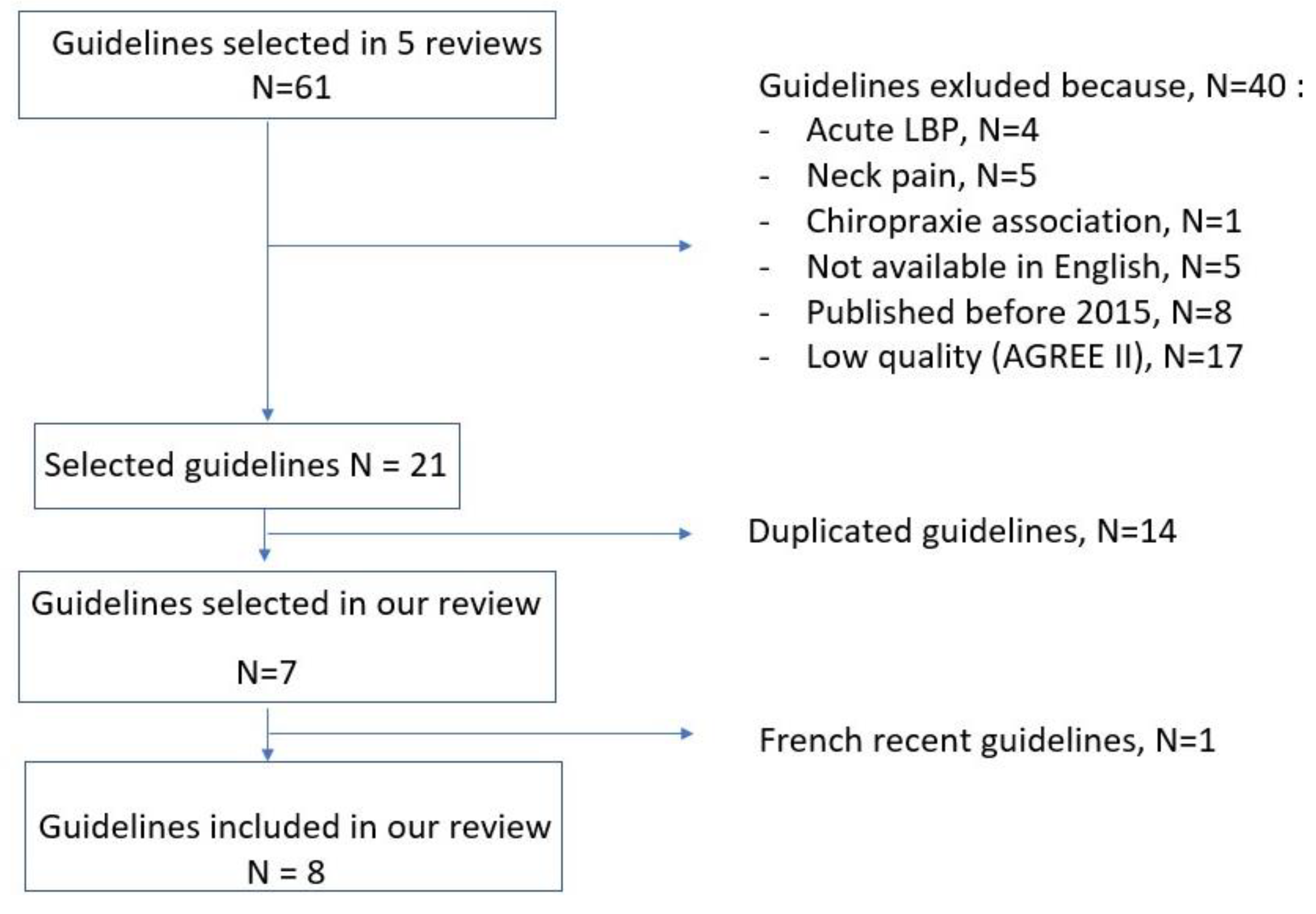
3.1. Literature Search
3.2. Diagnosis (Table 1)
| Author | Year | Country | Red Flags | Psychosocial Risk Factor | History and Physical Examination | Imaging |
|---|---|---|---|---|---|---|
| van Wambeke [18] | 2017 | Belgium | Assess for signs of serious underlying conditions including cancer, infection, trauma, inflammatory or severe neurological impairments (e.g., cauda equina syndrome) Search for differential diagnoses particularly for new or changed symptoms | Consider using screening tools for risk stratification (e.g., STarT Back or Örebro) for new episodes from 48 h after the pain onset. Risk stratification is aimed at informing shared decision making about stratified management | There is insufficient evidence to recommend for or against specific clinical tests, because no test considered in isolation has adequate sensitivity and specificity for determining the cause of pain The objective of history taking and physical examination is to assess for signs of serious underlying condition | Imaging should not be routinely offered in the absence of red flags Consider prescribing imaging if expected results may lead to change management Explain to patients with low back pain that imaging may not be necessary |
| TOP [13] | 2015 | Canada | Assess for signs of serious underlying conditions requiring specific evaluation and treatment Search for surgical emergency (e.g., cauda equina syndrome) | Assess for psychosocial risk factors (i.e., yellow flags including include fear, financial problems, anger, depression, job dissatisfaction, family problems or stress) Conduct a review of these factors if there is no improvement There is insufficient evidence to recommend for or against using screening tools for risk stratification | There is insufficient evidence to recommend for or against using the Clinically Organized Relevant Exam (CORE) | Lumbar spine X-rays are poor indicators of serious underlying conditions. In the absence of red flags, spinal and lumbar spine X-rays are not recommended Specific and appropriate diagnostic imaging should be selected on the basis of the condition being sought Lumbar spine X-rays may be considered prior to other diagnostic imaging to assess stability and stenosis (e.g., MRI): views should be limited to standing antero-posterior and lateral views. MRI scanning has limited value in the absence of red flags, radiculopathy or neurogenic claudication CT scans may be considered when vertebral fractures are suspected, or MRI contraindicated |
| Chenot [14] | 2017 | Germany | Assess for signs requiring specific imaging or laboratory tests and/or referral to a specialist | Assess for psychosocial and workplace risk factors from the beginning Consider using screening tools for psychosocial (i.e., yellow flags) and workplace risk factors from 4 weeks after the pain onset if pain persists despite adequate treatment (i.e., provided in accordance with guidelines) | The objective of history taking and physical examination is to assess for signs of a dangerous course of the disease or serious underlying condition When such signs are absent, no further diagnostic steps should be undertaken, because they will exceptionally result in a specific diagnosis, and may promote chronic pain | Current evidence does not support routine imaging Indication for diagnostic imaging should be reassessed from 4 to 6 weeks after the pain onset if pain or activity limitations persist despite adequate treatment (i.e., provided in accordance with guidelines) Indication for diagnostic imaging may be reassessed earlier, from 2 to 4 weeks after the pain onset, if a currently employed patient has been unable to work for a long period of time, or if a diagnostic evaluation is required before multimodal treatment Imaging that lacks any potential therapeutic relevance should be avoided |
| HAS [2] | 2019 | France | Assess for signs of underlying conditions requiring specific and/or urgent care in case of recent lumbar pain or worsening of symptoms or new symptoms (i.e., acute flare-up of low back pain or change in symptoms) | Assess early for psychosocial risk factors (i.e., yellow flags). Fears and beliefs, psychological and social contexts must be identified early Consider using screening tools for risk stratification (e.g., STarT Back or Örebro) to assess the risk for chronic pain. Other specific questionnaires assessing the level of fears and avoidances (e.g., FABQ) or symptoms of anxiety and depression (e.g., HADS) can also be used Assess for risk factors of prolonged inability to work and/or to return to work (i.e., blue flags and black flags) in the event of repeated or prolonged (>4 weeks) sick leave. Consider requesting the expertise of an occupational physician in this case | No data | It is recommended to explain to the patient why imaging is not necessary in the first place, and if there is absence of systematic correlation between the symptoms and the radiological signs In the absence of a red flag, spinal imaging (i.e., MRI or a CT scan if MRI is contra-indicated) should be considered if pain persists beyond 3 months, or if an invasive procedure (epidural infiltration or spinal surgery) is planned In the absence of a red flag, there is no indication to perform isolated X-rays, except to asses for instability or spinal deformity There is no indication to repeat imaging in the absence of changes in symptoms |
| NICE [12] | 2016 | UK | Assess for alternative conditions, particularly for new or changed symptoms Search for specific causes of low back pain including cancer, infection, trauma or inflammatory disease | Consider using screening tools for risk stratification (e.g., STarT Back), at first point of contact with a healthcare professional, for each new episode of low back pain, in order to inform shared decision making about stratified management | No data | Imaging should not be routinely offered Explain to patients that they may not need imaging Consider imaging in specialist settings of care, only if the result is likely to change management |
| Qaseem [17] | 2017 | USA | No data | No data | No data | No data |
| NASS [16] | 2020 | North America | No data | Assess for psychosocial and workplace risk factors for chronic pain Consider previous episodes of low back pain as a prognostic factor for chronic pain Consider pain severity and functional impairment to stratify the risk for chronic pain Consider psychosocial factors as prognostic factors for return to work following an episode of acute low back pain | Consider a nonstructural cause of low back pain in patients with diffuse low back pain and tenderness Using fear avoidance behavior to determine the likelihood of a structural cause of low back pain There is insufficient evidence to recommend for or against using diffuse low back tenderness to predict the presence of disc degeneration | There is insufficient evidence to recommend for or against an association between low back pain and spondylosis There is insufficient evidence to recommend for or against imaging in the absence of a red flag to recommend for or against imaging findings correlating with low back pain There is insufficient evidence to determine whether imaging findings contribute to decision making to guide treatment |
| ACOEM [23] | 2020 | USA | Assess for red flags through medical history and physical examination | Assess for psychosocial risk factors at follow-up visits | Physical examination includes straight leg raising test and neurological examination. | Assess for nerve root compression by MRI or CT-scan in patients with symptoms that are not improving over 4 to 6 weeks with signs of nerve root dysfunction |
| VA/DOD [4] | 2017 | USA | Assess for neurologic deficits through medical history and physical examination (e.g., radiculopathy, neurogenic claudication) Assess for signs of serious underlying conditions including malignancy, fracture, infection | Perform mental health screening to inform selection of treatment | History taking and physical examination are critical to identify treatable causes of low back pain | Diagnostic imaging may be considered in patients with serious or progressive neurologic deficits, or when a red flag is present There is insufficient evidence to recommend for or against imaging in patients with pain for longer than 1 month who have not improved or responded to initial treatments |
3.3. Treatments (Table 2)
3.3.1. Non-Pharmacological Treatments
| Author | Year | Country | Pharmacological Treatment | Non Pharmacological Treatment | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| General Treatment | Topical Treatment | Spinal Injection | Physical Activity | Physiotherapy | Information/Education | Psychotherapy | Multidisciplinary Treatment | Other Treatments | |||
| van Wambeke [18] | 2017 | Belgium | Recommended, if a medication is required: - first line: oral NSAIDs - second line: weak opioids ± acetaminophen Not recommended: - acetaminophen as a single medication nor opioids in routine - selective serotonin-norepinephrine reuptake inhibitors - tricyclic antidepressants or non-selective serotonin-norepinephrine reuptake inhibitors in routine - anticonvulsants - antibiotics, muscle relaxants | No clear recommandation: topical NSAIDs | Not recommended: non epidural spinal injections No clear recommandation: facet joint infiltration | Recommended: exercise programme (specific exercises or a combination of approaches) | Recommended: manipulation, mobilization or soft tissue techniques: only as part of a multimodal treatment with a supervised exercise program | Recommended: - provide advice and information to help self-management - promote and facilitate return to work or normal activities of daily living as soon as possible | Recommended: psychological intervention using a cognitive behavioral approach: - only as part of a multimodal treatment with a supervised exercise program - optional and depending on patients risk stratification | Recommended: multidisciplinary rehabilitation program which combines physical and psychological component (cognitive behavioral approach, takes into account the person’s specific needs and capabilities): - when people have psychological obstacles to recovery, - when previous evidence-based management has not been effective No clear recommandation: back school | Not recommended: - belts and corsets - foot orthotics, rocker sole shoes - manual traction - ultrasounds - percutaneous electrical nerve stimulation - transcutaneous electrical nerve stimulation - interferential therapy No clear recommandation: acupuncture |
| TOP [13] | 2015 | Canada | Recommended: - acetaminophen - NSAIDs - muscle relaxants - tricyclic antidepressants - herbal medicines Not recommended: - selective Serotonin reuptake inhibitors - antibiotics (based on MRI Modic Changes) No clear recommendation: - opioids and tapentadol - marijuana (dried cannabis) - Duloxetine | Recommended: capsaicin frutescens No clear recommendation: topical NSAIDs, Buprenorphine transdermal system | Not recommended: prolotherapy as a sole treatment No clear recommendation: - prolotherapy as an adjunct - epidural steroid injections - therapeutic sacroiliac joint injections insufficient evidence - trigger point injections | Recommended: exercise and therapeutic exercise: - initiate gentle exercise and gradually increase the exercise level within pain tolerance - may include unsupervised walking and group exercise programs - when exercise exacerbates pain, programme should be assessed by a qualified physical therapist - if exercise exacerbates pain, patients should be assessed by a physician - therapeutic aquatic exercise -Viniyoga and Iyengar types of yoga | Recommended: massage therapy (as an adjunct to an active rehabilitation program) | Recommended: provide brief education to optimize function - review of clinical examination results - provision of low back pain information and advice to stay active - reduce fear and catastrophizing | Recommended: - when group chronic pain cognitive behavioral therapy programs are not available, consider referral for individual cognitive behavioral therapy - respondent behavioral therapies (progressive relaxation or EMG biofeedback) | Recommended: - structured community-based self-management group program: - for patients interested in learning pain coping skills- most community-based programs also include exercise and activity programming - if not available: individual self-management counselling (trained professional) - multidisciplinary treatment program: after no improvement with primary care management | Recommended: - acupuncture: short-term therapy or as an adjunct to a broader active rehabilitation program Not recommended: - motorized traction - transcutaneous electrical nerve stimulation (as a sole treatment) No clear recommendation: - manual therapy (spinal manipulative treatment or spinal mobilization) - therapeutic ultrasound - gravity tables (inverted traction, self-traction, gravitational traction) - ow-level laser therapy - mindfulness-based meditation - shock-wave treatment - spa therapy - back belts, corsets, - non-motorized traction - craniosacral massage/therapy - intramuscular stimulation - interferential current therapy - touch therapies |
| Chenot [14] | 2017 | Germany | Recommended: - NSAID - Metamizole Not recommended: - acetaminophen - Flupirtine - intravenously, intramuscularly or subcutaneously administered analgesic drugs, local anesthetics, glucocorticoids, or mixed infusions No clear recommendation: - COX-2-inhibitors: can be used if NSAIDs are contraindicated or poorly tolerated - opioids:
| Recommended: - instruction to continue usual physical activities - rehabilitative sports and functional training - progressive muscle relaxation | No clear recommendation: massage | Recommended: - explain the condition and the treatment to the patient - encourage the pursuit of a healthful lifestyle, including regular physical exercise - patients should be advised against bed rest - initiation and coordination of psychotherapeutic care, if necessary - possibly social counseling | Recommended: initiation and coordination of psychotherapeutic care, if necessary | Recommended: - exercise therapy combined with educative measures based on behavioral-therapeutic principles should be used in the primary treatment of chronic non-specific low back pain - multimodal programs if less intensive evidence-based treatments have yielded an insufficient benefit:
| No clear recommendation: - self-administered heat therapy - manual therapies (manipulation and mobilization) - ergotherapy - back school - acupuncture => could be used to treat chronic low back pain in combination with activating therapeutic measures Not recommended: - interference-current therapy - kinesiotaping - short-wave diathermy - laser therapy - magnetic field therapy - medical aids - percutaneous electrical nerve stimulation (PENS) - traction devices - cryotherapy - transcutaneous electrical nerve stimulation (TENS), - therapeutic ultrasound | ||
| HAS [2] | 2019 | France | First line: acetaminophen, non-steroidal anti-inflammatory drugs (low dose, short duration); Second line: opioids (risk of misuse). Antidepressants and anticonvulsants are not indicated in acute LBP, possible use in case of chronic pain. No opinion for nefopam, cortico-steroids. Not recommended: muscle relaxants. No indication for: vitamin D, antibiotic, anti-TNF alpha. | No indication for lidocaine patch | Generally no indication for LBP infiltration without root pain | Physical exercise is the main treatment: self-management in first line: return to daily activities (and professional activities if possible), adapted physical activities and sports (progressive and fractional) | Suggested/Recommended: physiotherapy (active participation of patient); patient education; mobilizations, manual therapy (only as part of a multimodal combination of treatments with supervised exercises and on second-line treatment) | Suggested/Recommended: deliver reassuring information | Suggested/Recommended: second-line treatment: cognitive behavioral therapy (only as part of a multimodal combination of treatments with supervised exercises) | Suggested/Recommended: third-line treatment for patients with persistant pain and psychosocial risk factors or in case of failure of first- and second-line treatments | Not recommended: ultra sound therapy; lumbar tractions; plantar orthosis No clear recommendation: acupuncture, acupressure, dry needling; sophrology; relaxation; mindfulness; hypnosis; lumbar brace; lumbar belt |
| NICE [12] | 2016 | UK | Recommended: - oral NSAIDs: - weak opioids (±acetaminophen): only if NSAID is contraindicated, not tolerated or has been ineffective. Not recommended: - acetaminophen alone - opioids - selective serotonin reuptake inhibitors, serotonin–norepinephrine reuptake inhibitors or tricyclic antidepressants - gabapentinoids or anticonvulsants | Not recommended: spinal injections in LBP Recommended: - radiofrequency denervation, to consider in chronic LBP:
| Recommended: group exercise program: - biomechanical, aerobic, mind–body or a combination of approaches - take people’s specific needs, preferences and capabilities into account when choosing the type of exercise | Recommended: - advice and information, tailored to their needs and capabilities, - help them self-manage low back pain - information on the nature of low back pain - encouragement to continue with normal activities | Recommended: - psychological therapies using a cognitive behavioral approach - as part of a treatment package including exercise | Recommended: - combined physical and psychological program
| Recommended: - consider manual therapy (spinal manipulation, mobilization or soft tissue techniques) as part of a treatment package including exercise Not recommended: - belts or corsets - foot orthotics - rocker sole shoes - traction - acupuncture - ultrasound - percutaneous electrical nerve simulation (PENS) - transcutaneous electrical nerve simulation (TENS) - interferential therapy | ||
| Qaseem [17] | 2017 | USA | Recommended (in patients who have had an inadequate response to nonpharmacologic therapy): - first line: NSAIDs - second line: tramadol or Duloxetine - If failure: opioids if the potential benefits outweigh the risks | Recommended: exercise, Tai Chi, yoga | Recommended: - motor control exercise - progressive relaxation | Recommended: multidisciplinary rehabilitation | Recommended: - acupuncture - mindfulness-based stress reduction - electromyography biofeedback - low-level laser therapy, operant therapy - spinal manipulation (low-quality evidence) | ||||
| NASS [16] | 2020 | North America | Suggested/Recommended: opioid pain medications (short duration) Not recommended: oral or IV steroids; antidepressants No clear recommendation: anticonvulsants; vitamin D; selective NSAIDs | Suggested/Recommended: topical capsaicin No clear recommendation: lidocaine patch | No clear recommendation: caudal epidural steroid injections; interlaminar epidural steroid injections; zygapophyseal joint injection; intradiscal steroids; intradiscal platelet rich plasma | Suggested/Recommended: yoga; aerobic exercise | Suggested/Recommended: McKenzie method No recommended: traction; ultrasound; addition of massage to an exercise program; lumbar stabilization No clear recommendation: transcutaneous electrical nerve stimulation (TENS); dry needling | Suggested/Recommended: back school No clear recommendation: patient education | Suggested/Recommended: cognitive behavioral therapy (in combination with physical therapy) treatments targeting fear avoidance (combined with physical therapy) | ||
| ACOEM [23] | 2020 | USA | Recommended: - NSAIDs - acetaminophen - antidepressants - skeletal muscle relaxants: for acute exacerbations of chronic LBP Not recommended: opioids, antibiotics, antidepressants, anticonvulsants, bisphosphonates, calcitonin and oral and intravenous colchicine NMDA receptor/antagonists skeletal muscle relaxants glucocorticosteroids TNF-a vitamin supplementation No Clear recommendation: Thiocolchicoside | Recommended: capsaicin Not recommended: - lidocaine patches - Spiroflor - DMSO, N-acetylcysteine, EMLA, and wheatgrass cream No Clear recommendation: topical NSAIDs or other creams | Recommended: - exercise prescription - self-administered or enacted through formal therapy appointments - aerobic exercises (progressive walking program) - directional exercises which centralize or abolish the pain - slump stretching exercises 3 to 5 times a day - strengthening exercises - specific strengthening exercises - yoga and tai chi for select, motivated patients Not recommended: - stretching exercises in the absence of significant range of motion deficits - abdominal strengthening exercises as a sole or central goal of a strengthening program No Clear recommendation: Pilates | Recommended: - massages for select use as an adjunct to more efficacious treatments (aerobic and strengthening exercise program) - self-applications of low-tech heat therapies and cryotherapies - aquatic therapy for select chronic LBP patients (extreme obesity, significant degenerative joint disease, etc.) Not recommended: - mechanical devices for administering massage - reflexology - high-tech devices of heat and/or cryotherapy - diathermy - lumbar extension machines No Clear recommendation: myofascial release | Recommended: - maintaining maximal levels of activity, including work activities, - work modifications should be tailored taking into consideration 3 main factors: (1) job physical requirements; (2) severity of the problem; and (3) the patient’s understanding of his or her condition - fear avoidance belief training for patients with elevated fear avoidance beliefs Not recommended: bed rest | Recommended: - lordotic sitting posture - sleep posture comfortable - manipulation or mobilization (component of an active exercise program) - acupuncture - transcutaneous electrical nerve simulation TENS Not recommended: - specific beds or other commercial sleep products - kinesiotaping - shoe lifts or insoles except for individuals with significant leg length discrepancy of more than 2 cm - lumbar supports - magnets - traction - low-level laser therapy - microcurrent electrical stimulation No Clear recommendation: - specific mattresses, bedding, and water bed - medical foods (Ther- amine, an amino acid formulation) - herbal - iontophoresis - inversion therapy - infrared therapy - ultrasounds | |||
| VA/DOD [4] | 2017 | USA | Recommended: - NAIDs - duloxetine - non-benzodiazepine muscle relaxant for acute exacerbations of chronic low back pain Not recommended: - non-benzodiazepine muscle relaxant - benzodiazepines - oral or intramuscular injection corticosteroids - long-term opioid therapy - chronic use of oral acetaminophen No clear recommendation: time-limited opioid therapy, for acute exacerbations of chronic low back pain time-limited (less than 7 days) acetaminophen therapy anticonvulsants nutritional, herbal, and homeopathic supplements | No clear recommendation: topical preparations | Not recommended: - spinal epidural steroid injections - intra-articular facet joint steroid injections | Recommended: - clinician-directed exercises - exercise program, which may include Pilates, yoga, and tai chi | Recommended: - provide evidence-based information with regard to their expected course - advise patients to remain active, - provide information about self-care options - add structured education component as part of multicomponent self-management intervention | Recommended: - cognitive behaviral therapy | Recommended: - Multidisciplinary or interdisciplinary rehabilitation programme which should include at least one physical component and at least one other component of the biopsychosocial model (psychological, social, occupational) - for selected patients not satisfactorily responding to more limited approaches | Recommended: - spinal mobilization/manipulation as part of a multimodal programme - acupuncture - mindfulness-based stress reduction. No clear recommendation: - lumbar supports - ultrasound - transcutaneous electrical nerve stimulation (TENS) - lumbar traction - electrical muscle stimulation - medial branch blocks | |
3.3.2. Pharmacological Treatments
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Airaksinen, O.; Brox, J.I.; Cedraschi, C.; Hildebrandt, J.; Klaber-Moffett, J.; Kovacs, F.; Mannion, A.F.; Reis, S.; Staal, J.B.; Ursin, H.; et al. Chapter 4. European guidelines for the management of chronic nonspecific low back pain. Eur. Spine J. 2006, 15 (Suppl. S2), S192–S300. [Google Scholar] [CrossRef] [PubMed]
- Bailly, F.; Trouvin, A.P.; Bercier, S.; Dadoun, S.; Deneuville, J.P.; Faguer, R.; Fassier, J.B.; Koleck, M.L.; Lassalle, L.; Le Vraux, T.; et al. Clinical guidelines and care pathway for management of low back pain with or without radicular pain. Jt. Bone Spine 2021, 88, 105227. [Google Scholar] [CrossRef] [PubMed]
- Maher, C.; Underwood, M.; Buchbinder, R. Non-specific low back pain. Lancet 2017, 389, 736–747. [Google Scholar] [CrossRef] [PubMed]
- Pangarkar, S.S.; Kang, D.G.; Sandbrink, F.; Bevevino, A.; Tillisch, K.; Konitzer, L.; Sall, J. Va/dod clinical practice guideline: Diagnosis and treatment of low back pain. J. Gen. Intern. Med. 2019, 34, 2620–2629. [Google Scholar] [CrossRef]
- Nguyen, C.; De Sèze, M.; Rannou, F. The challenges of precision medicine in chronic low back pain: Lessons learned from active discopathy. Ann. Phys. Rehabil. Med. 2021, 64, 101504. [Google Scholar] [CrossRef]
- Nguyen, C.; Lefèvre-Colau, M.M.; Kennedy, D.J.; Schneider, B.J.; Rannou, F. Low back pain. Lancet 2018, 392, 2547. [Google Scholar] [CrossRef]
- Corp, N.; Mansell, G.; Stynes, S.; Wynne-Jones, G.; Morsø, L.; Hill, J.C.; van der Windt, D.A. Evidence-based treatment recommendations for neck and low back pain across europe: A systematic review of guidelines. Eur. J. Pain. 2021, 25, 275–295. [Google Scholar] [CrossRef]
- Krenn, C.; Horvath, K.; Jeitler, K.; Zipp, C.; Siebenhofer-Kroitzsch, A.; Semlitsch, T. Management of non-specific low back pain in primary care—A systematic overview of recommendations from international evidence-based guidelines. Prim. Health Care Res. Dev. 2020, 21, e64. [Google Scholar] [CrossRef]
- Longtin, C.; Décary, S.; Cook, C.E.; Tousignant-Laflamme, Y. What does it take to facilitate the integration of clinical practice guidelines for the management of low back pain into practice? Part 1: A synthesis of recommendation. Pain Pract. 2021, 21, 943–954. [Google Scholar] [CrossRef]
- Meroni, R.; Piscitelli, D.; Ravasio, C.; Vanti, C.; Bertozzi, L.; De Vito, G.; Perin, C.; Guccione, A.A.; Cerri, C.G.; Pillastrini, P. Evidence for managing chronic low back pain in primary care: A review of recommendations from high-quality clinical practice guidelines. Disabil. Rehabil. 2021, 43, 1029–1043. [Google Scholar] [CrossRef]
- Oliveira, C.B.; Maher, C.G.; Pinto, R.Z.; Traeger, A.C.; Lin, C.C.; Chenot, J.F.; van Tulder, M.; Koes, B.W. Clinical practice guidelines for the management of non-specific low back pain in primary care: An updated overview. Eur. Spine J. 2018, 27, 2791–2803. [Google Scholar] [CrossRef] [PubMed]
- Bernstein, I.A.; Malik, Q.; Carville, S.; Ward, S. Low back pain and sciatica: Summary of nice guidance. BMJ 2017, 356, i6748. [Google Scholar] [CrossRef] [PubMed]
- College of Family Physicians of Canada. Evidence-informed primary care management of low back pain: Towards Optimized Pratice. In Clinical Practice Guideline; College of Family Physicians of Canada: Mississauga, ON, Canada, 2015. [Google Scholar]
- Chenot, J.F.; Greitemann, B.; Kladny, B.; Petzke, F.; Pfingsten, M.; Schorr, S.G. Non-specific low back pain. Dtsch. Arztebl. Int. 2017, 114, 883–890. [Google Scholar] [CrossRef] [PubMed]
- Hegmann, K.T.; Travis, R.; Andersson, G.B.J.; Belcourt, R.M.; Carragee, E.J.; Donelson, R.; Eskay-Auerbach, M.; Galper, J.; Goertz, M.; Haldeman, S.; et al. Non-invasive and minimally invasive management of low back disorders. J. Occup. Environ. Med. 2020, 62, e111–e138. [Google Scholar] [CrossRef]
- Kreiner, D.S.; Matz, P.; Bono, C.M.; Cho, C.H.; Easa, J.E.; Ghiselli, G.; Ghogawala, Z.; Reitman, C.A.; Resnick, D.K.; Watters, W.C., 3rd; et al. Guideline summary review: An evidence-based clinical guideline for the diagnosis and treatment of low back pain. Spine J. 2020, 20, 998–1024. [Google Scholar] [CrossRef]
- Qaseem, A.; McLean, R.M.; O’Gurek, D.; Batur, P.; Lin, K.; Kansagara, D.L. Nonpharmacologic and pharmacologic management of acute pain from non-low back, musculoskeletal injuries in adults: A clinical guideline from the american college of physicians and american academy of family physicians. Ann. Intern. Med. 2020, 173, 739–748. [Google Scholar] [CrossRef]
- Van Wambeke, P.; Desomer, A.; Jonckheer, P.; Depreitere, B. The belgian national guideline on low back pain and radicular pain: Key roles for rehabilitation, assessment of rehabilitation potential and the prm specialist. Eur. J. Phys. Rehabil. Med. 2020, 56, 220–227. [Google Scholar] [CrossRef]
- Hill, J.C.; Dunn, K.M.; Lewis, M.; Mullis, R.; Main, C.J.; Foster, N.E.; Hay, E.M. A primary care back pain screening tool: Identifying patient subgroups for initial treatment. Arthritis Rheum 2008, 59, 632–641. [Google Scholar] [CrossRef]
- Linton, S.J.; Halldén, K. Can we screen for problematic back pain? A screening questionnaire for predicting outcome in acute and subacute back pain. Clin. J. Pain 1998, 14, 209–215. [Google Scholar] [CrossRef]
- Waddell, G.; Newton, M.; Henderson, I.; Somerville, D.; Main, C.J. A fear-avoidance beliefs questionnaire (fabq) and the role of fear-avoidance beliefs in chronic low back pain and disability. Pain 1993, 52, 157–168. [Google Scholar] [CrossRef]
- Zigmond, A.S.; Snaith, R.P. The hospital anxiety and depression scale. Acta Psychiatr. Scand. 1983, 67, 361–370. [Google Scholar] [CrossRef] [PubMed]
- Hegmann, K.T.; Travis, R.; Belcourt, R.M.; Donelson, R.; Eskay-Auerbach, M.; Galper, J.; Haldeman, S.; Hooper, P.D.; Lessenger, J.E.; Mayer, T.; et al. Diagnostic tests for low back disorders. J. Occup. Environ. Med. 2019, 61, e155–e168. [Google Scholar] [CrossRef]
- Poiraudeau, S.; Rannou, F.; Baron, G.; Henanff, L.A.; Coudeyre, E.; Rozenberg, S.; Huas, D.; Martineau, C.; Jolivet-Landreau, I.; Garcia-Mace, J.; et al. Fear-avoidance beliefs about back pain in patients with subacute low back pain. Pain 2006, 124, 305–311. [Google Scholar] [CrossRef] [PubMed]
- Coudeyre, E.; Rannou, F.; Tubach, F.; Baron, G.; Coriat, F.; Brin, S.; Revel, M.; Poiraudeau, S. General practitioners’ fear-avoidance beliefs influence their management of patients with low back pain. Pain 2006, 124, 330–337. [Google Scholar] [CrossRef] [PubMed]
- Barrey, C.Y.; Le Huec, J.C.; French Society for Spine, S. Chronic low back pain: Relevance of a new classification based on the injury pattern. Orthop. Traumatol. Surg. Res. 2019, 105, 339–346. [Google Scholar] [CrossRef]
- Maas, E.T.; Juch, J.N.; Ostelo, R.W.; Groeneweg, J.G.; Kallewaard, J.W.; Koes, B.W.; Verhagen, A.P.; Huygen, F.J.; van Tulder, M.W. Systematic review of patient history and physical examination to diagnose chronic low back pain originating from the facet joints. Eur. J. Pain 2017, 21, 403–414. [Google Scholar] [CrossRef]
- Ma, K.; Zhuang, Z.G.; Wang, L.; Liu, X.G.; Lu, L.J.; Yang, X.Q.; Lu, Y.; Fu, Z.J.; Song, T.; Huang, D.; et al. The chinese association for the study of pain (casp): Consensus on the assessment and management of chronic nonspecific low back pain. Pain Res. Manag. 2019, 2019, 8957847. [Google Scholar] [CrossRef]
- Jensen, M.C.; Brant-Zawadzki, M.N.; Obuchowski, N.; Modic, M.T.; Malkasian, D.; Ross, J.S. Magnetic resonance imaging of the lumbar spine in people without back pain. N. Engl. J. Med. 1994, 331, 69–73. [Google Scholar] [CrossRef]
- Daste, C.; Laclau, S.; Boisson, M.; Segretin, F.; Feydy, A.; Lefevre-Colau, M.M.; Rannou, F.; Nguyen, C. Intervertebral disc therapies for non-specific chronic low back pain: A systematic review and meta-analysis. Ther. Adv. Musculoskelet. Dis. 2021, 13. [Google Scholar] [CrossRef]
- Nguyen, C.; Boutron, I.; Baron, G.; Sanchez, K.; Palazzo, C.; Benchimol, R.; Paris, G.; James-Belin, E.; Lefevre-Colau, M.M.; Beaudreuil, J.; et al. Intradiscal glucocorticoid injection for patients with chronic low back pain associated with active discopathy: A randomized trial. Ann. Intern. Med. 2017, 166, 547–556. [Google Scholar] [CrossRef]
- Tavares, I.; Thomas, E.; Cyteval, C.; Picot, M.C.; Manna, F.; Macioce, V.; Laffont, I.; Thouvenin, Y.; Viala, P.; Larbi, A.; et al. Intradiscal glucocorticoids injection in chronic low back pain with active discopathy: A randomized controlled study. Ann. Phys. Rehabil. Med. 2021, 64, 101396. [Google Scholar] [CrossRef] [PubMed]
- Boisson, M.; Lefevre-Colau, M.M.; Rannou, F.; Nguyen, C. Active discopathy: A clinical reality. RMD Open 2018, 4, e000660. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, C.; Poiraudeau, S.; Rannou, F. From modic 1 vertebral-endplate subchondral bone signal changes detected by mri to the concept of ‘active discopathy’. Ann. Rheum Dis. 2015, 74, 1488–1494. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nicol, V.; Verdaguer, C.; Daste, C.; Bisseriex, H.; Lapeyre, É.; Lefèvre-Colau, M.-M.; Rannou, F.; Rören, A.; Facione, J.; Nguyen, C. Chronic Low Back Pain: A Narrative Review of Recent International Guidelines for Diagnosis and Conservative Treatment. J. Clin. Med. 2023, 12, 1685. https://doi.org/10.3390/jcm12041685
Nicol V, Verdaguer C, Daste C, Bisseriex H, Lapeyre É, Lefèvre-Colau M-M, Rannou F, Rören A, Facione J, Nguyen C. Chronic Low Back Pain: A Narrative Review of Recent International Guidelines for Diagnosis and Conservative Treatment. Journal of Clinical Medicine. 2023; 12(4):1685. https://doi.org/10.3390/jcm12041685
Chicago/Turabian StyleNicol, Vanina, Claire Verdaguer, Camille Daste, Hélène Bisseriex, Éric Lapeyre, Marie-Martine Lefèvre-Colau, François Rannou, Alexandra Rören, Julia Facione, and Christelle Nguyen. 2023. "Chronic Low Back Pain: A Narrative Review of Recent International Guidelines for Diagnosis and Conservative Treatment" Journal of Clinical Medicine 12, no. 4: 1685. https://doi.org/10.3390/jcm12041685
APA StyleNicol, V., Verdaguer, C., Daste, C., Bisseriex, H., Lapeyre, É., Lefèvre-Colau, M.-M., Rannou, F., Rören, A., Facione, J., & Nguyen, C. (2023). Chronic Low Back Pain: A Narrative Review of Recent International Guidelines for Diagnosis and Conservative Treatment. Journal of Clinical Medicine, 12(4), 1685. https://doi.org/10.3390/jcm12041685






