Clinical Outcomes of XEN45®-Stent Implantation after Failed Trabeculectomy: A Retrospective Single-Center Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Statistical Analysis
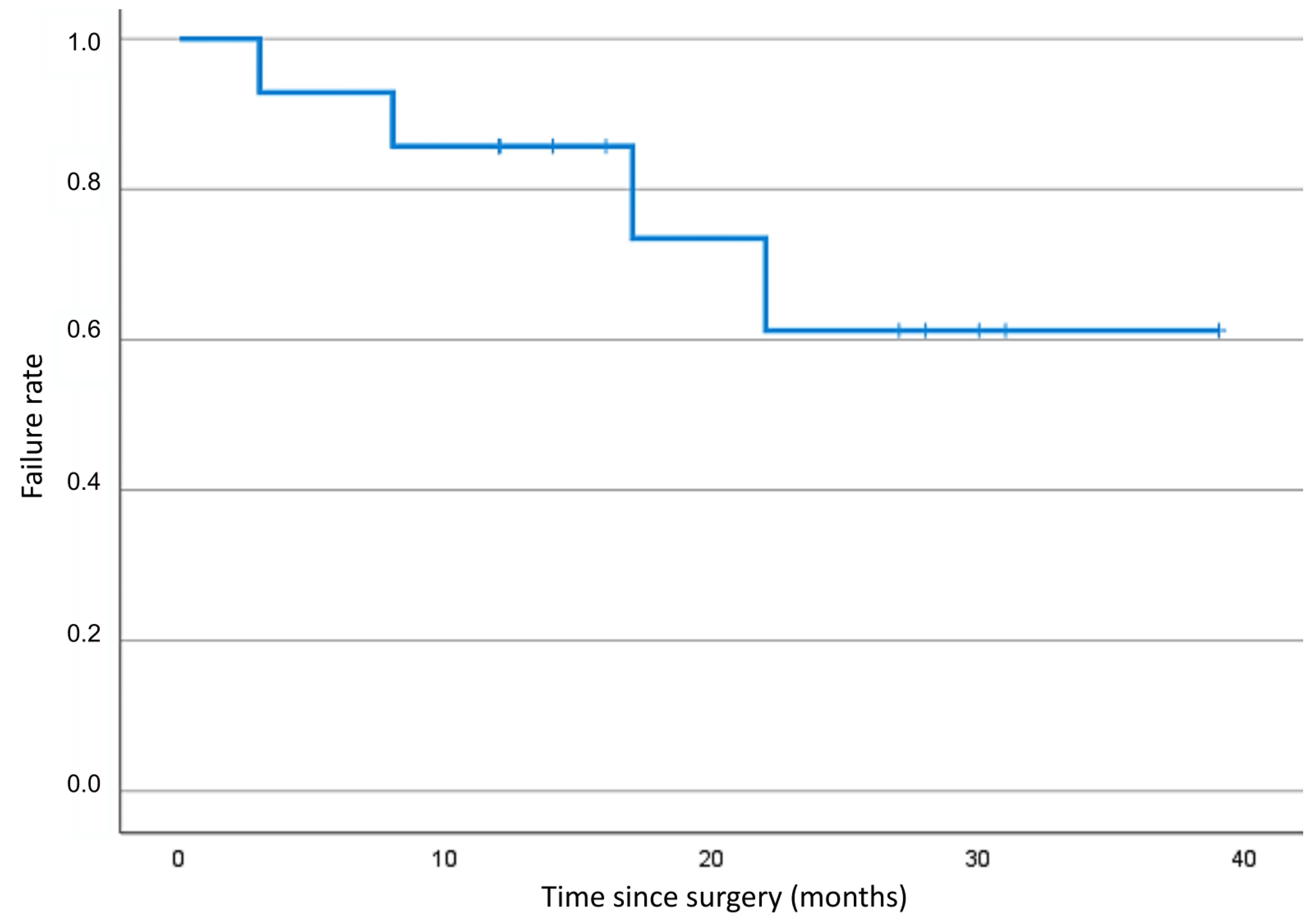
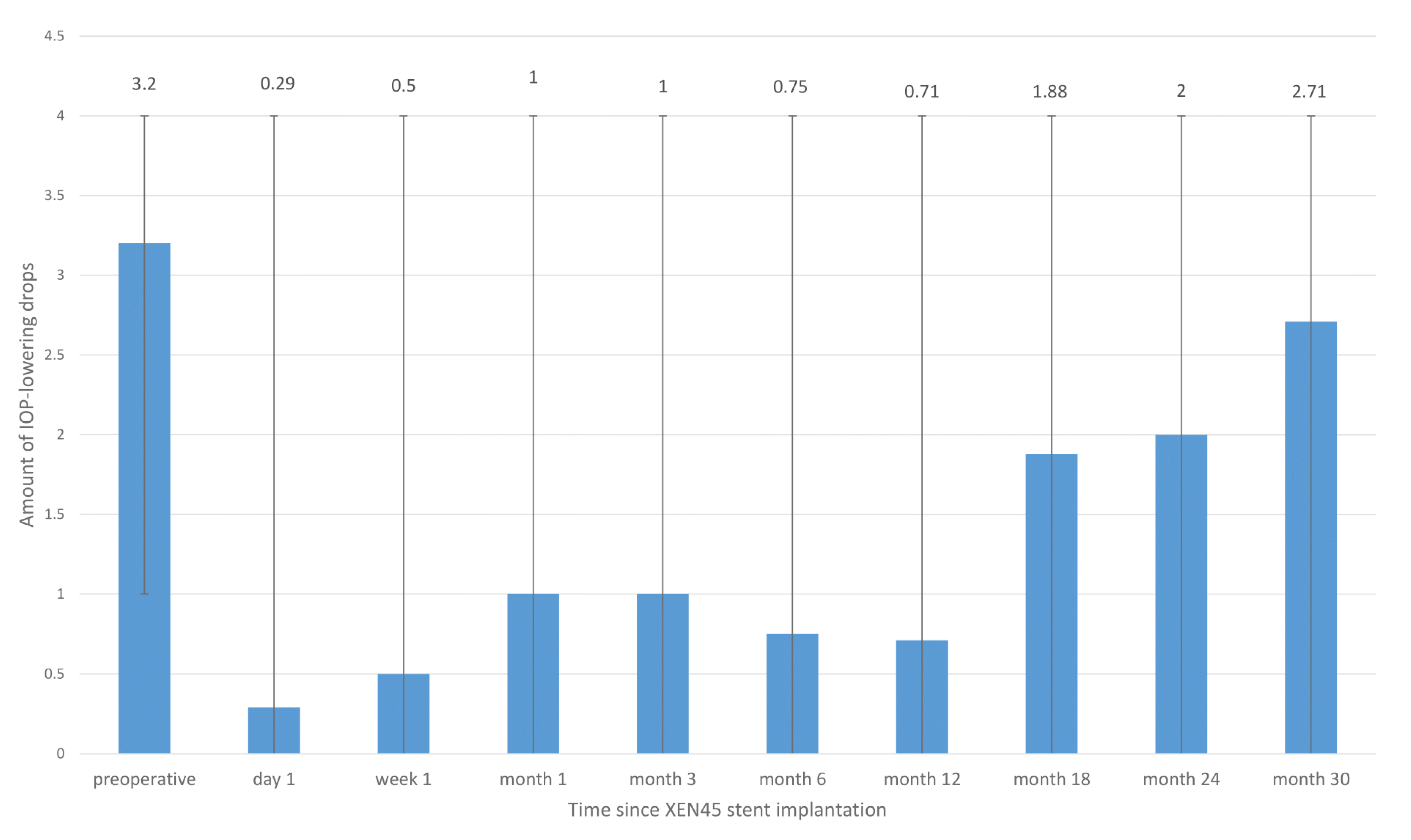
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Cairns, J.E. Trabeculectomy. Preliminary report of a new method. Am. J. Ophthalmol. 1968, 66, 673–679. [Google Scholar] [CrossRef] [PubMed]
- Kwong, A.; Law, S.K.; Kule, R.R.; Nouri-Mahdavi, K.; Coleman, A.L.; Caprioli, J.; Giaconi, J.A. Long-term outcomes of resident- versus attending-performed primary trabeculectomy with mitomycin C in a United States residency program. Am. J. Ophthalmol. 2014, 157, 1190–1201. [Google Scholar] [CrossRef] [PubMed]
- Klink, T.; Kann, G.; Ellinger, P.; Klink, J.; Grehn, F.; Guthoff, R. The prognostic value of the wuerzburg bleb classification score for the outcome of trabeculectomy. Ophthalmologica 2011, 225, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Landers, J.; Martin, K.; Sarkies, N.; Bourne, R.; Watson, P. A twenty-year follow-up study of trabeculectomy: Risk factors and outcomes. Ophthalmology 2012, 119, 694–702. [Google Scholar] [CrossRef] [PubMed]
- Vesti, E.; Raitta, C. A review of the outcome of trabeculectomy in open-angle glaucoma. Ophthalmic Surg Lasers 1997, 28, 128–132. [Google Scholar] [CrossRef] [PubMed]
- Romero, P.; Hirunpatravong, P.; Alizadeh, R.; Kim, E.A.; Nouri-Mahdavi, K.; Morales, E.; Law, S.K.; Caprioli, J. Trabeculectomy with Mitomycin-C: Outcomes and Risk Factors for Failure in Primary Angle-closure Glaucoma. J. Glaucoma 2018, 27, 101–107. [Google Scholar] [CrossRef]
- Mittal, D.; Bhoot, M.; Dubey, S. Trabeculectomy with Mitomycin-C: Outcomes and Risk Factors for Failure in Primary Angle-closure Glaucoma. J. Glaucoma 2018, 27, e186. [Google Scholar] [CrossRef]
- Saheb, H.; Ahmed, I.I.K. Micro-invasive glaucoma surgery: Current perspectives and future directions. Curr. Opin. Ophthalmol. 2012, 23, 96–104. [Google Scholar] [CrossRef]
- Nichani, P.; Popovic, M.M.; Schlenker, M.B.; Park, J.; Ahmed, I.I.K. Microinvasive glaucoma surgery: A review of 3476 eyes. Surv. Ophthalmol. 2021, 66, 714–742. [Google Scholar] [CrossRef]
- SooHoo, J.R.; Seibold, L.K.; Radcliffe, N.; Kahook, M.Y. Minimally invasive glaucoma surgery: Current implants and future innovations. Can. J. Ophthalmol. 2014, 49, 528–533. [Google Scholar] [CrossRef]
- Lewis, R.A. Ab interno approach to the subconjunctival space using a collagen glaucoma stent. J. Cataract. Refract. Surg. 2014, 40, 1301–1306. [Google Scholar] [CrossRef]
- Galal, A.; Bilgic, A.; Eltanamly, R.; Osman, A. XEN Glaucoma Implant with Mitomycin C 1-Year Follow-Up: Result and Complications. J. Ophthalmol. 2017, 2017, 5457246. [Google Scholar] [CrossRef]
- Lenzhofer, M.; Kersten-Gomez, I.; Sheybani, A.; Gulamhusein, H.; Strohmaier, C.; Hohensinn, M.; Dick, H.B.; Hitzl, W.; Eisenkopf, L.; Sedarous, F.; et al. Four-year results of a minimally invasive transscleral glaucoma gel stent implantation in a prospective multi-centre study. Clin. Exp. Ophthalmol. 2019, 47, 581–587. [Google Scholar] [CrossRef]
- Rauchegger, T.; Angermann, R.; Willeit, P.; Schmid, E.; Teuchner, B. Two-year outcomes of minimally invasive XEN Gel Stent implantation in primary open-angle and pseudoexfoliation glaucoma. Acta Ophthalmol. 2021, 99, 369–375. [Google Scholar] [CrossRef]
- Lee, N.; Ma, K.T.; Bae, H.W.; Hong, S.; Seong, G.J.; Hong, Y.J.; Kim, C.Y. Surgical results of trabeculectomy and Ahmed valve implantation following a previous failed trabeculectomy in primary congenital glaucoma patients. Korean J. Ophthalmol. 2015, 29, 109–114. [Google Scholar] [CrossRef]
- Hirunpatravong, P.; Reza, A.; Romero, P.; Kim, E.A.; Nouri-Mahdavi, K.; Law, S.K.; Morales, E.; Caprioli, J. Same-site Trabeculectomy Revision for Failed Trabeculectomy: Outcomes and Risk Factors for Failure. Am. J. Ophthalmol. 2016, 170, 110–118. [Google Scholar] [CrossRef]
- Dubey, S.; Rajurkar, K. Same-site Trabeculectomy Revision for Failed Trabeculectomy: Outcomes and Risk Factors for Failure. Am. J. Ophthalmol. 2017, 177, 239–240. [Google Scholar] [CrossRef]
- Khaw, P.T.; Chiang, M.; Shah, P.; Sii, F.; Lockwood, A.; Khalili, A. Enhanced Trabeculectomy: The Moorfields Safer Surgery System. Dev. Ophthalmol. 2017, 59, 15–35. [Google Scholar]
- Karimi, A.; Hopes, M.; Martin, K.R.; Lindfield, D. Efficacy and Safety of the Ab-interno Xen Gel Stent After Failed Trabeculectomy. J. Glaucoma 2018, 27, 864–868. [Google Scholar] [CrossRef]
- Bormann, C.; Schmidt, M.; Busch, C.; Rehak, M.; Scharenberg, C.T.; Unterlauft, J.D. Implantation of XEN After Failed Trabeculectomy: An Efficient Therapy? Klin. Monbl. Augenheilkd. 2021, 239, 86–93. [Google Scholar] [CrossRef]
- Düzgün, E.; Olgun, A.; Karapapak, M.; Alkan, A.A.; Ustaoğlu, M. Outcomes of XEN Gel Stent Implantation in the Inferonasal Quadrant after Failed Trabeculectomy. J. Curr. Glaucoma Pract. 2021, 15, 64–69. [Google Scholar] [CrossRef] [PubMed]
- Gedde, S.J.; Feuer, W.; Lim, K.; Barton, K.; Goyal, K.; Ahmed, I.; Brandt, J. Treatment Outcomes in the Primary Tube Versus Trabeculectomy Study after 3 Years of Follow-u. Ophthalmology 2020, 127, 333–345. [Google Scholar] [CrossRef] [PubMed]
- Fontana, H.; Nouri-Mahdavi, K.; Lumba, J.; Ralli, M.; Caprioli, J. Trabeculectomy with mitomycin C: Outcomes and risk factors for failure in phakic open-angle glaucoma. Ophthalmology 2006, 113, 930–936. [Google Scholar] [CrossRef] [PubMed]

| Patient Characteristics | n = 14 (%) |
|---|---|
| Gender | |
| Male/Female | 9 (64.3)/5 (35.7) |
| Age | |
| Mean, median (range, SD) | 71.14, 74.50 (51–84; 9.51) |
| Glaucoma type | |
| Primary open-angle glaucoma | 8 (57.1) |
| Pseudoexfoliation glaucoma | 5 (35.7) |
| Secondary neovascular glaucoma | 1 (7.1) |
| Follow-up time (months) | |
| Mean, median (range, SD) | 20.36, 20.50 (12–34; 8.43) |
| Time between TE and XEN (months) | |
| Mean, median (range, SD) | 110.54, 100.00 (36–270; 10.33) |
| Acetazolamide | |
| Yes/no | 1 (7.1)/13 (92.9) |
| Number of glaucoma medications | |
| 0 | 0 |
| 1 | 2 (14.3) |
| 2 | 3 (21.4) |
| 3 | 2 (14.3) |
| 4 | 7 (50.0) |
| 5 | 0 |
| BCVA, measured preoperatively (logMAR) | |
| Mean, median (range, SD) | 0.52, 0.35 (0–2.3; 0.62) |
| IOP preoperatively | |
| Mean, median (range, SD) | 17.93, 18.00 (11–25; 4.45) |
| Maximum IOP without drops in the past | |
| Mean, median (range, SD) | 22.27, 21.00 (16–35; 4.40) |
| Mean deviation (MD) (Humphrey 24-2) | |
| Mean, median (range, SD) | −13.75, −15,50 (−1.96–−25.25; 8.13) |
| Pattern standard deviation (PSD) | |
| Mean, median (range, SD) | 8.6, 9.53 (3.1–14.54; 3.79) |
| Cup-to-disc ratio (CDR) | |
| Mean, median (range, SD) | 0.78, 0.80 (0.50–0.99; 0.17) |
| Complications | |
|---|---|
| Yes/no | 5 (35.7)/9 (64.3) |
| Complications (3 patients with more than 1 complication) | |
| Hyphema | 1 (7.1) |
| XEN-Revision + Mitomycin C | 2 (14.2) |
| Needling | 1 (7.1) |
| Partial XEN removal | 1 (7.1) |
| Corneal oedema | 1 (7.1) |
| Numerical hypotony | 1 (7.1) |
| Failure yes/no | |
| Yes/no | 4 (28.6)/10 (71.4) |
| Failure reason | |
| Loss of visual acuity to levels below light perception | 0 |
| Hypotony < 6 mmHg over two consecutive visits | 0 |
| Persistent high pressure > 21 mmHg over two consecutive visits | 1 (25.0) |
| Additional glaucoma surgery needed | 3 (75.0) |
| Time interval since XEN operation (months) | |
| Mean (range) | 10.33 (1–22) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Weber, C.; Hundertmark, S.; Petrak, M.; Ludwig, E.; Brinkmann, C.K.; Holz, F.G.; Mercieca, K. Clinical Outcomes of XEN45®-Stent Implantation after Failed Trabeculectomy: A Retrospective Single-Center Study. J. Clin. Med. 2023, 12, 1296. https://doi.org/10.3390/jcm12041296
Weber C, Hundertmark S, Petrak M, Ludwig E, Brinkmann CK, Holz FG, Mercieca K. Clinical Outcomes of XEN45®-Stent Implantation after Failed Trabeculectomy: A Retrospective Single-Center Study. Journal of Clinical Medicine. 2023; 12(4):1296. https://doi.org/10.3390/jcm12041296
Chicago/Turabian StyleWeber, Constance, Sarah Hundertmark, Michael Petrak, Elisabeth Ludwig, Christian Karl Brinkmann, Frank G. Holz, and Karl Mercieca. 2023. "Clinical Outcomes of XEN45®-Stent Implantation after Failed Trabeculectomy: A Retrospective Single-Center Study" Journal of Clinical Medicine 12, no. 4: 1296. https://doi.org/10.3390/jcm12041296
APA StyleWeber, C., Hundertmark, S., Petrak, M., Ludwig, E., Brinkmann, C. K., Holz, F. G., & Mercieca, K. (2023). Clinical Outcomes of XEN45®-Stent Implantation after Failed Trabeculectomy: A Retrospective Single-Center Study. Journal of Clinical Medicine, 12(4), 1296. https://doi.org/10.3390/jcm12041296





