Artificial Intelligence Based Analysis of Corneal Confocal Microscopy Images for Diagnosing Peripheral Neuropathy: A Binary Classification Model
Abstract
1. Introduction
2. Materials and Methods
2.1. Dataset and Participants
2.2. Algorithm Architecture and Implementation
2.3. Performance Evaluation
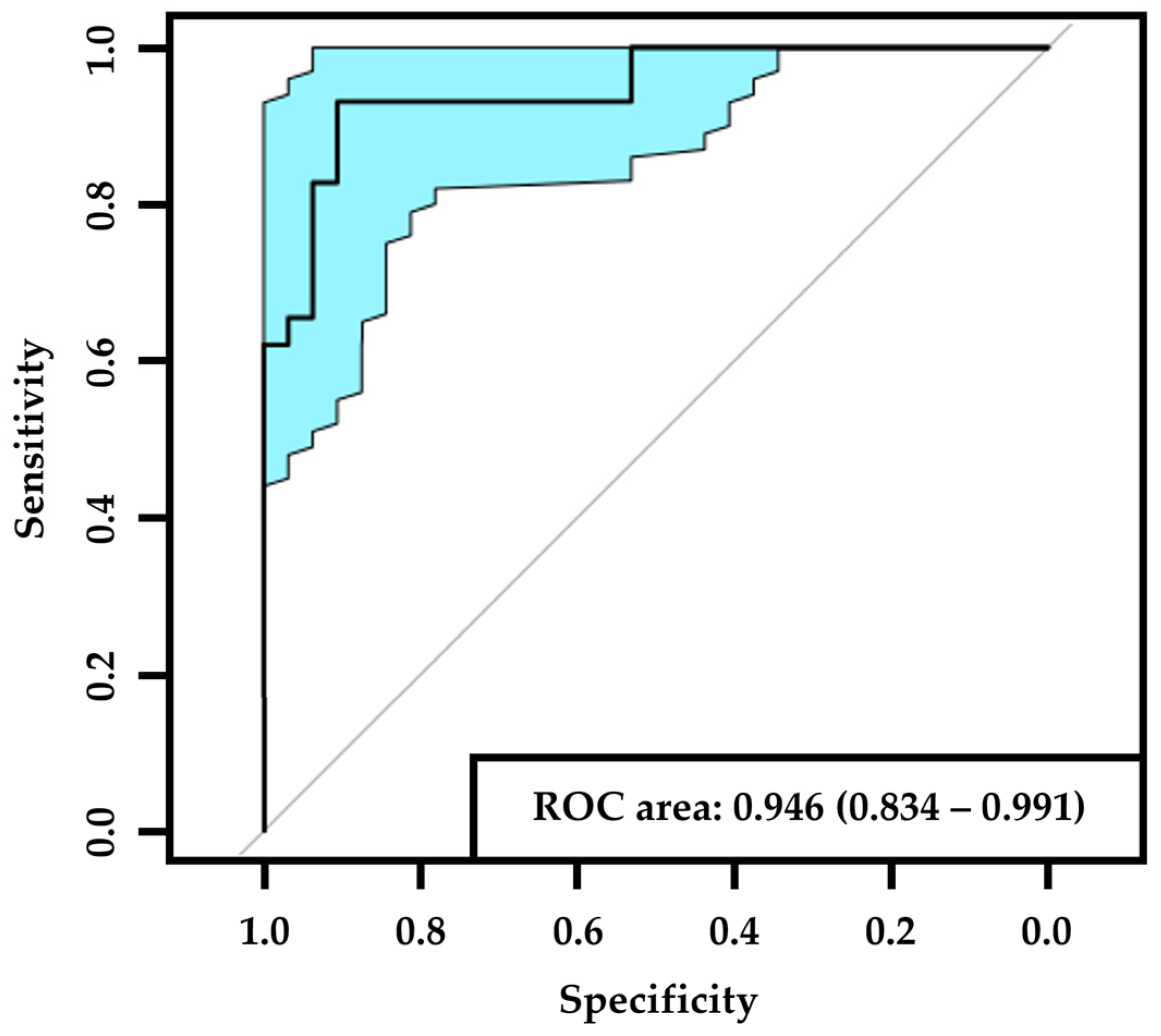
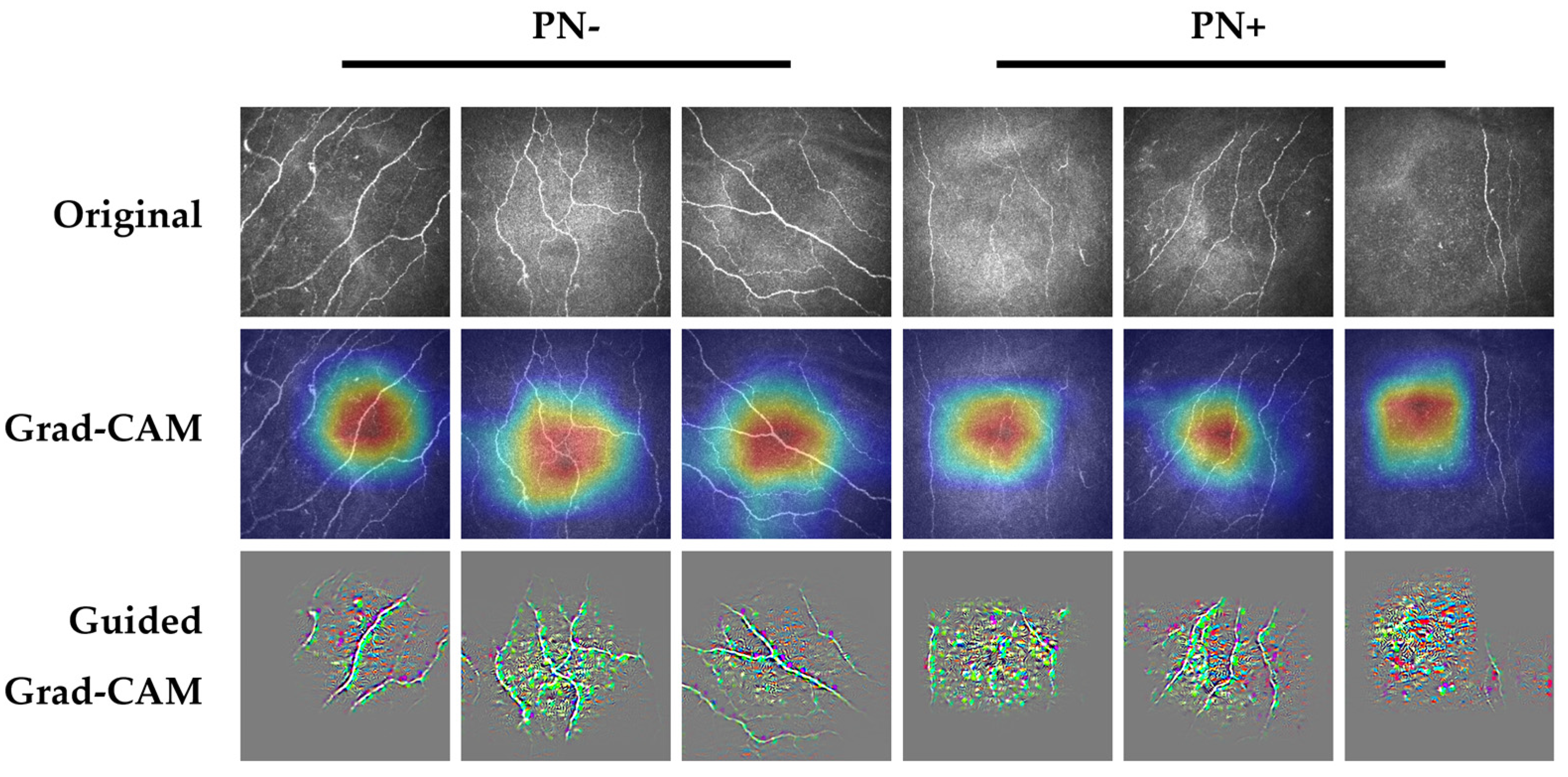
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Iqbal, Z.; Azmi, S.; Yadav, R.; Ferdousi, M.; Kumar, M.; Cuthbertson, D.J.; Lim, J.; Malik, R.A.; Alam, U. Diabetic Peripheral Neuropathy: Epidemiology, Diagnosis, and Pharmacotherapy. Clin. Ther. 2018, 40, 828–849. [Google Scholar] [CrossRef] [PubMed]
- Alam, U.; Jeziorska, M.; Petropoulos, I.N.; Asghar, O.; Fadavi, H.; Ponirakis, G.; Marshall, A.; Tavakoli, M.; Boulton, A.J.M.; Efron, N.; et al. Diagnostic utility of corneal confocal microscopy and intra-epidermal nerve fibre density in diabetic neuropathy. PLoS ONE 2017, 12, e0180175. [Google Scholar] [CrossRef]
- Roszkowska, A.M.; Licitra, C.; Tumminello, G.; Postorino, E.I.; Colonna, M.R.; Aragona, P. Corneal nerves in diabetes—The role of the in vivo corneal confocal microscopy of the subbasal nerve plexus in the assessment of peripheral small fiber neuropathy. Surv. Ophthalmol. 2020, 66, 493–513. [Google Scholar] [CrossRef]
- Preston, F.G.; Meng, Y.; Burgess, J.; Ferdousi, M.; Azmi, S.; Petropoulos, I.N.; Kaye, S.; Malik, R.A.; Zheng, Y.; Alam, U. Artificial intelligence utilising corneal confocal microscopy for the diagnosis of peripheral neuropathy in diabetes mellitus and prediabetes. Diabetologia 2021, 65, 457–466. [Google Scholar] [CrossRef]
- Salahouddin, T.; Petropoulos, I.N.; Ferdousi, M.; Ponirakis, G.; Asghar, O.; Alam, U.; Kamran, S.; Mahfoud, Z.R.; Efron, N.; Malik, R.A.; et al. Artificial Intelligence–Based Classification of Diabetic Peripheral Neuropathy From Corneal Confocal Microscopy Images. Diabetes Care 2021, 44, e151–e153. [Google Scholar] [CrossRef]
- Scarpa, F.; Colonna, A.; Ruggeri, A. Multiple-Image Deep Learning Analysis for Neuropathy Detection in Corneal Nerve Images. Cornea 2019, 39, 342–347. [Google Scholar] [CrossRef]
- Williams, B.M.; Borroni, D.; Liu, R.; Zhao, Y.; Zhang, J.; Lim, J.; Ma, B.; Romano, V.; Qi, H.; Ferdousi, M.; et al. An artificial intelligence-based deep learning algorithm for the diagnosis of diabetic neuropathy using corneal confocal microscopy: A development and validation study. Diabetologia 2019, 63, 419–430. [Google Scholar] [CrossRef]
- Tesfaye, S.; Boulton, A.J.M.; Dyck, P.J.; Freeman, R.; Horowitz, M.; Kempler, P.; Lauria, G.; Malik, R.A.; Spallone, V.; Vinik, A.; et al. Diabetic Neuropathies: Update on Definitions, Diagnostic Criteria, Estimation of Severity, and Treatments. Diabetes Care 2010, 33, 2285–2293. [Google Scholar] [CrossRef]
- He, K.; Zhang, X.; Ren, S.; Sun, J. Deep residual learning for image recognition. In Proceedings of the IEEE Conference on Computer Vision and Pattern Recognition, Las Vegas, NV, USA, 27–30 June 2016; pp. 770–778. [Google Scholar]
- Deng, J.; Dong, W.; Socher, R.; Li, L.-J.; Li, K.; Fei-Fei, L. Imagenet: A large-scale hierarchical image database. In Proceedings of the 2009 IEEE Conference on Computer Vision and Pattern Recognition, Miami, FL, USA, 20–25 June 2009; pp. 248–255. [Google Scholar]
- Selvaraju, R.R.; Cogswell, M.; Das, A.; Vedantam, R.; Parikh, D.; Batra, D. Grad-cam: Visual explanations from deep networks via gradient-based localization. In Proceedings of the IEEE International Conference on Computer Vision, Venice, Italy, 22–29 October 2017; pp. 618–626. [Google Scholar]
- Ghassemi, M.; Oakden-Rayner, L.; Beam, A.L. The false hope of current approaches to explainable artificial intelligence in health care. Lancet Digit. Health 2021, 3, e745–e750. [Google Scholar] [CrossRef]
- National Institute for Health and Care Excellence (NICE). NICE Guideline [NG19] Diabetic Foot Problems: Prevention and Management; National Institute for Health and Care Excellence (NICE): London, UK, 2023. [Google Scholar]
- Vinik, A.I.; Strotmeyer, E.S.; Nakave, A.A.; Patel, C.V. Diabetic Neuropathy in Older Adults. Clin. Geriatr. Med. 2008, 24, 407–435. [Google Scholar] [CrossRef]
- Abbott, C.A.; Malik, R.A.; van Ross, E.R.; Kulkarni, J.; Boulton, A.J. Prevalence and Characteristics of Painful Diabetic Neuropathy in a Large Community-Based Diabetic Population in the U.K. Diabetes Care 2011, 34, 2220–2224. [Google Scholar] [CrossRef] [PubMed]
- Callaghan, B.C.; A Little, A.; Feldman, E.; Hughes, R.A.C. Enhanced glucose control for preventing and treating diabetic neuropathy. Cochrane Database Syst. Rev. 2012, 6, CD007543. [Google Scholar] [CrossRef]
- Kirthi, V.; Perumbalath, A.; Brown, E.; Nevitt, S.; Petropoulos, I.N.; Burgess, J.; Roylance, R.; Cuthbertson, D.J.; Jackson, T.L.; A Malik, R.; et al. Prevalence of peripheral neuropathy in pre-diabetes: A systematic review. BMJ Open Diabetes Res. Care 2021, 9, e002040. [Google Scholar] [CrossRef]
- Pop-Busui, R.; Boulton, A.J.; Feldman, E.L.; Bril, V.; Freeman, R.; Malik, R.A.; Sosenko, J.M.; Ziegler, D. Diabetic Neuropathy: A Position Statement by the American Diabetes Association. Diabetes Care 2016, 40, 136–154. [Google Scholar] [CrossRef]
- Burgess, J.; Frank, B.; Marshall, A.; Khalil, R.; Ponirakis, G.; Petropoulos, I.; Cuthbertson, D.; Malik, R.; Alam, U. Early Detection of Diabetic Peripheral Neuropathy: A Focus on Small Nerve Fibres. Diagnostics 2021, 11, 165. [Google Scholar] [CrossRef]
- Heydon, P.; Egan, C.; Bolter, L.; Chambers, R.; Anderson, J.; Aldington, S.; Stratton, I.M.; Scanlon, P.H.; Webster, L.; Mann, S.; et al. Prospective evaluation of an artificial intelligence-enabled algorithm for automated diabetic retinopathy screening of 30 000 patients. Br. J. Ophthalmol. 2020, 105, 723–728. [Google Scholar] [CrossRef]
- Brown, J.J.; Pribesh, S.L.; Baskette, K.G.; Vinik, A.I.; Colberg, S.R. A Comparison of Screening Tools for the Early Detection of Peripheral Neuropathy in Adults with and without Type 2 Diabetes. J. Diabetes Res. 2017, 2017, 1467213. [Google Scholar] [CrossRef]
- Alam, U.; Asghar, O.; Petropoulos, I.N.; Jeziorska, M.; Fadavi, H.; Ponirakis, G.; Marshall, A.; Tavakoli, M.; Boulton, A.J.; Efron, N.; et al. Small Fiber Neuropathy in Patients With Latent Autoimmune Diabetes in Adults. Diabetes Care 2015, 38, e102–e103. [Google Scholar] [CrossRef]
- Asghar, O.; Petropoulos, I.N.; Alam, U.; Jones, W.; Jeziorska, M.; Marshall, A.; Ponirakis, G.; Fadavi, H.; Boulton, A.J.; Tavakoli, M.; et al. Corneal Confocal Microscopy Detects Neuropathy in Subjects With Impaired Glucose Tolerance. Diabetes Care 2014, 37, 2643–2646. [Google Scholar] [CrossRef]
- Azmi, S.; Jeziorska, M.; Ferdousi, M.; Petropoulos, I.N.; Ponirakis, G.; Marshall, A.; Alam, U.; Asghar, O.; Atkinson, A.; Jones, W.; et al. Early nerve fibre regeneration in individuals with type 1 diabetes after simultaneous pancreas and kidney transplantation. Diabetologia 2019, 62, 1478–1487. [Google Scholar] [CrossRef]
- Chen, X.; Graham, J.; Dabbah, M.A.; Petropoulos, I.N.; Ponirakis, G.; Asghar, O.; Alam, U.; Marshall, A.; Fadavi, H.; Ferdousi, M.; et al. Small Nerve Fiber Quantification in the Diagnosis of Diabetic Sensorimotor Polyneuropathy: Comparing Corneal Confocal Microscopy With Intraepidermal Nerve Fiber Density. Diabetes Care 2015, 38, 1138–1144. [Google Scholar] [CrossRef] [PubMed]
- Petropoulos, I.N.; Alam, U.; Fadavi, H.; Marshall, A.; Asghar, O.; Dabbah, M.A.; Chen, X.; Graham, J.; Ponirakis, G.; Boulton, A.J.M.; et al. Rapid Automated Diagnosis of Diabetic Peripheral Neuropathy With In Vivo Corneal Confocal Microscopy. Investig. Opthalmol. Vis. Sci. 2014, 55, 2071–2078. [Google Scholar] [CrossRef]
- Petropoulos, I.N.; Manzoor, T.; Morgan, P.; Fadavi, H.; Asghar, O.; Alam, U.; Ponirakis, G.; Dabbah, M.A.; Chen, X.; Graham, J.; et al. Repeatability of In Vivo Corneal Confocal Microscopy to Quantify Corneal Nerve Morphology. Cornea 2013, 32, e83–e89. [Google Scholar] [CrossRef]
- Tavakoli, M.; Mitu-Pretorian, M.; Petropoulos, I.N.; Fadavi, H.; Asghar, O.; Alam, U.; Ponirakis, G.; Jeziorska, M.; Marshall, A.; Efron, N.; et al. Corneal Confocal Microscopy Detects Early Nerve Regeneration in Diabetic Neuropathy After Simultaneous Pancreas and Kidney Transplantation. Diabetes 2012, 62, 254–260. [Google Scholar] [CrossRef]
- Wang, F.; Zhang, J.; Yu, J.; Liu, S.; Zhang, R.; Ma, X.; Yang, Y.; Wang, P. Diagnostic Accuracy of Monofilament Tests for Detecting Diabetic Peripheral Neuropathy: A Systematic Review and Meta-Analysis. J. Diabetes Res. 2017, 2017, 8787261. [Google Scholar] [CrossRef]
- Kanagasingam, Y.; Xiao, D.; Vignarajan, J.; Preetham, A.; Tay-Kearney, M.-L.; Mehrotra, A. Evaluation of Artificial Intelligence–Based Grading of Diabetic Retinopathy in Primary Care. JAMA Netw. Open 2018, 1, e182665. [Google Scholar] [CrossRef]
| Predicted Class | |||
|---|---|---|---|
| PN− | PN+ | ||
| True Class | PN− | 27 | 2 |
| PN+ | 3 | 29 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meng, Y.; Preston, F.G.; Ferdousi, M.; Azmi, S.; Petropoulos, I.N.; Kaye, S.; Malik, R.A.; Alam, U.; Zheng, Y. Artificial Intelligence Based Analysis of Corneal Confocal Microscopy Images for Diagnosing Peripheral Neuropathy: A Binary Classification Model. J. Clin. Med. 2023, 12, 1284. https://doi.org/10.3390/jcm12041284
Meng Y, Preston FG, Ferdousi M, Azmi S, Petropoulos IN, Kaye S, Malik RA, Alam U, Zheng Y. Artificial Intelligence Based Analysis of Corneal Confocal Microscopy Images for Diagnosing Peripheral Neuropathy: A Binary Classification Model. Journal of Clinical Medicine. 2023; 12(4):1284. https://doi.org/10.3390/jcm12041284
Chicago/Turabian StyleMeng, Yanda, Frank George Preston, Maryam Ferdousi, Shazli Azmi, Ioannis Nikolaos Petropoulos, Stephen Kaye, Rayaz Ahmed Malik, Uazman Alam, and Yalin Zheng. 2023. "Artificial Intelligence Based Analysis of Corneal Confocal Microscopy Images for Diagnosing Peripheral Neuropathy: A Binary Classification Model" Journal of Clinical Medicine 12, no. 4: 1284. https://doi.org/10.3390/jcm12041284
APA StyleMeng, Y., Preston, F. G., Ferdousi, M., Azmi, S., Petropoulos, I. N., Kaye, S., Malik, R. A., Alam, U., & Zheng, Y. (2023). Artificial Intelligence Based Analysis of Corneal Confocal Microscopy Images for Diagnosing Peripheral Neuropathy: A Binary Classification Model. Journal of Clinical Medicine, 12(4), 1284. https://doi.org/10.3390/jcm12041284







