Integrative and Mechanistic Approach to the Hair Growth Cycle and Hair Loss
Abstract
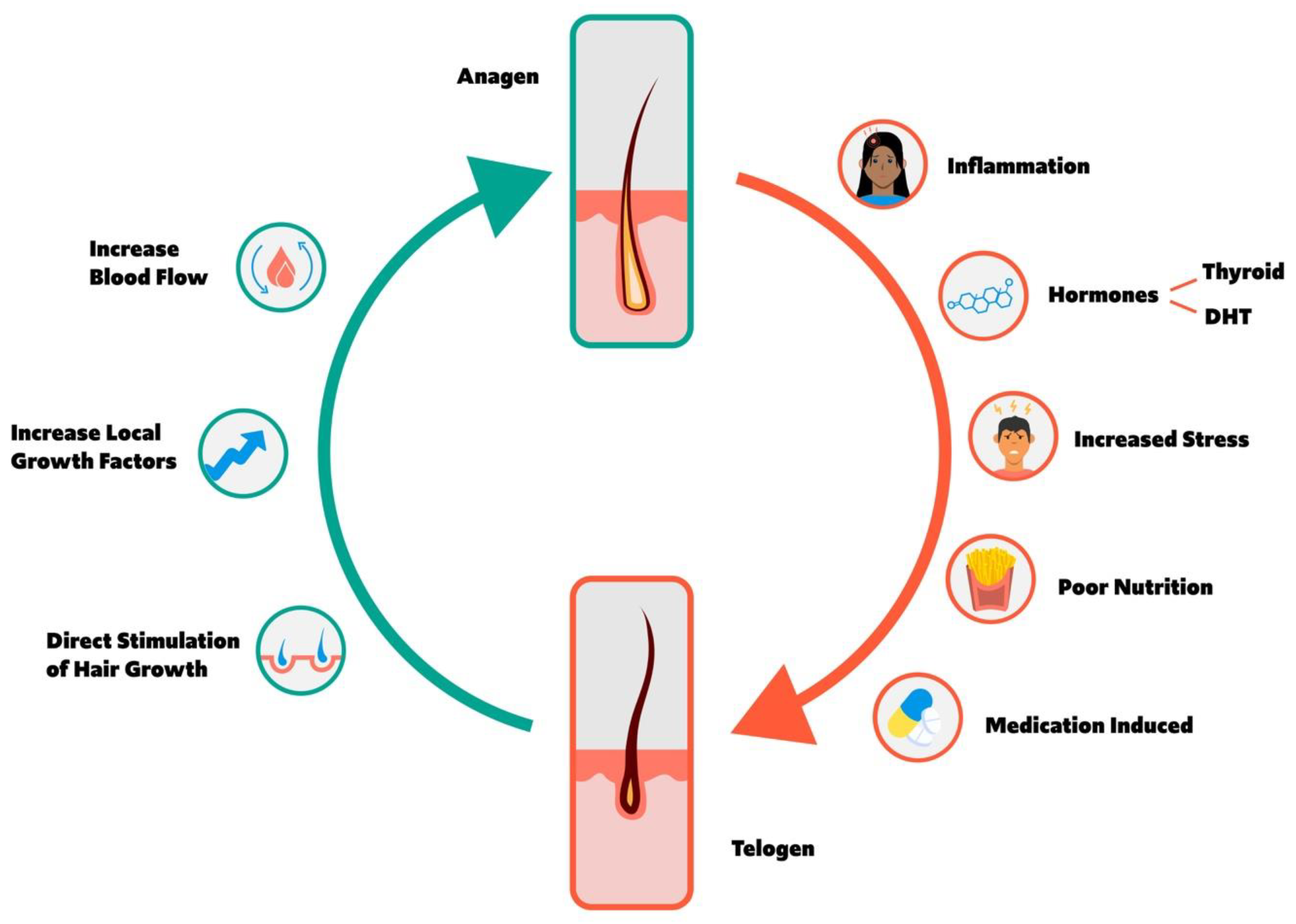
1. Overview of Hair Cycle
1.1. Anagen and Telogen Balance
2. Factors Increasing Anagen to Telogen Transition and Hair Loss
2.1. Inflammation
2.2. Hormones
2.2.1. Thyroid Hormone
2.2.2. Dihydrotestosterone
2.2.3. Estrogen to Testosterone Ratio
2.3. Stress
2.4. Nutritional Deficiencies
2.4.1. Amino Acids and Protein
2.4.2. Fatty Acids
2.4.3. Vitamins
Vitamin A
B Vitamins
Vitamin D
Vitamin E
2.4.4. Minerals
2.5. Poor Sleep
Circadian Rhythm and Clock Genes
2.6. Cell Division Inhibiting Medication
2.7. History-Taking Tips
- Duration of hair shedding;
- Episodic or continuous patterns;
- Estimated percent hair loss;
- Potential triggers and temporal relationships;
- Recent surgery, fever, illness, childbirth, psychological stress;
- History of chronic disease, malignancy, infection, autoimmune disease, liver or renal disease;
- Menstrual history;
- Hair care products and procedures;
- Dietary history including vitamins and supplements;
- Family history of AnA, AA, autoimmune disease, or thyroid disorder;
- Medication history including botanicals;
- History of radiation therapy or heavy metal exposure.
3. Factors Increasing Telogen to Anagen Transition and Hair Growth
3.1. Increased Blood Flow
3.1.1. Scalp Massage
3.1.2. Minoxidil
3.1.3. HIF-1α
3.2. Direct Stimulation of the Hair Follicle
3.2.1. Herbs and Phytochemicals
3.2.2. Supplements
3.2.3. Light-Based Approaches
3.2.4. Prostaglandins
3.3. Growth Factors and Platelet Rich Plasma
4. Diagnostic Lab Testing
4.1. Ferritin
4.1.1. Premenopausal vs. Postmenopausal Women
4.1.2. Men vs. Women
4.2. ANA
4.3. RPR
4.4. Thyroid Hormones
4.5. Functional Testing
4.5.1. Diurnal Cortisol Slope Testing
Correlations to Hair Cortisol
4.5.2. Mitochondrial Function Testing
Thyroid Impact on Mitochondrial Function
Organic Acid Testing of Krebs Cycle and Electron Transport Chain
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
Disclosures
Abbreviations
| AA: | Alopecia areata |
| AnA: | Androgenetic alopecia |
| CI: | Confidence interval |
| CRY: | Circadian regulator gene |
| DHEAS: | Dehydroepiandrosterone sulfate |
| DHT: | Dihydrotestosterone |
| FGF: | Fibroblast growth factor |
| FPAnA: | Female-pattern androgenetic alopecia |
| FSH: | Follicle-stimulating hormone |
| Ig: | Immunoglobulin |
| LH: | Luteinizing hormone |
| LO: | Lavender oil |
| MPAnA: | Male-pattern androgenetic alopecia |
| OR: | Odds ratio |
| PRP: | Platelet rich plasma |
| PSQI: | Pittsburgh Sleep Quality Index |
| SHBG: | Sex hormone binding globulin |
| SMD: | Standardized mean difference |
| TE: | Telogen Effluvium |
| TSH: | Thyroid-stimulating hormone |
| VDR: | Vitamin D receptor |
| ROS: | Reactive oxygen species |
| ETC: | Electron transport chain |
| OAT: | Organic Acid Testing |
| HA: | Hyaluronic acid |
| HIF: | Hypoxia inducible factor |
| HSF: | HIF strengthening factor |
| HPA: | Hypothalamic pituitary adrenal axis |
| SA: | Syphilitic alopecia |
| PER: | Period gene |
| RPR: | Rapid plasma reagin |
| ANA: | Antinuclear antibody |
| MPHL: | Male-pattern hair loss |
| FPHL: | Female-pattern hair loss |
| IGF: | Insulin-like growth factor |
References
- Paus, R.; Cotsarelis, G. The Biology of Hair Follicles. N. Engl. J. Med. 1999, 341, 491–497. [Google Scholar] [CrossRef] [PubMed]
- Harrison, S.; Bergfeld, W. Diffuse hair loss: Its triggers and management. Clevel. Clin. J. Med. 2009, 76, 361–367. [Google Scholar] [CrossRef] [PubMed]
- Millar, S.E. Molecular Mechanisms Regulating Hair Follicle Development. J. Investig. Dermatol. 2002, 118, 216–225. [Google Scholar] [CrossRef] [PubMed]
- Burg, D.; Yamamoto, M.; Namekata, M.; Haklani, J.; Koike, K.; Halasz, M. Promotion of anagen, increased hair density and reduction of hair fall in a clinical setting following identification of FGF5-inhibiting compounds via a novel 2-stage process. Clin. Cosmet. Investig. Dermatol. 2017, 10, 71–85. [Google Scholar] [CrossRef]
- Alonso, L.; Fuchs, E. The hair cycle. J. Cell Sci. 2006, 119, 391–393. [Google Scholar] [CrossRef]
- Sinclair, R. Male pattern androgenetic alopecia. BMJ 1998, 317, 865–869. [Google Scholar] [CrossRef] [PubMed]
- Asghar, F.; Shamim, N.; Farooque, U.; Sheikh, H.; Aqeel, R. Telogen effluvium: A review of the literature. Cureus 2020, 12, e8320. [Google Scholar] [CrossRef]
- Vincent, M.; Yogiraj, K. A descriptive study of alopecia patterns and their relation to thyroid dysfunction. Int. J. Trichol. 2013, 5, 57–60. [Google Scholar] [CrossRef]
- Spano, F.; Donovan, J.C. Alopecia areata. Can. Fam. Phys. 2015, 61, 751–755. [Google Scholar]
- Saceda-Corralo, D.; Pindado-Ortega, C.; Moreno-Arrones, O.M.; Ortega-Quijano, D.; Fernández-Nieto, D.; Jiménez-Cauhe, J.; Vañó-Galván, S. Association of Inflammation With Progression of Hair Loss in Women With Frontal Fibrosing Alopecia. JAMA Dermatol. 2020, 156, 700–702. [Google Scholar] [CrossRef]
- Lattanand, A.; Johnson, W.C. Male Pattern Alopecia A Histopathologic and Histochemical Study. J. Cutan. Pathol. 1975, 2, 58–70. [Google Scholar] [CrossRef] [PubMed]
- Whiting, D.A. Diagnostic and predictive value of horizontal sections of scalp biopsy specimens in male pattern androgenetic alopecia. J. Am. Acad. Dermatol. 1993, 28, 755–763. [Google Scholar] [CrossRef] [PubMed]
- Whiting, D.A. Chronic telogen effluvium: Increased scalp hair shedding in middle-aged women. J. Am. Acad. Dermatol. 1996, 35, 899–906. [Google Scholar] [CrossRef]
- Magro, C.M.; Rossi, A.; Poe, J.; Manhas-Bhutani, S.; Sadick, N. The role of inflammation and immunity in the pathogenesis of androgenetic alopecia. J. Drugs Dermatol. JDD 2011, 10, 1404–1411. [Google Scholar]
- Piérard, G.; Piérard-Franchimont, C.; Nikkels-Tassoudji, N.; Nikkels, A.; Léger, D.S. Improvement in the inflammatory aspect of androgenetic alopecia. A pilot study with an antimicrobial lotion. J. Dermatol. Treat. 1996, 7, 153–157. [Google Scholar] [CrossRef]
- Arck, P.C.; Handjiski, B.; Peters, E.M.J.; Peter, A.S.; Hagen, E.; Fischer, A.; Klapp, B.F.; Paus, R. Stress Inhibits Hair Growth in Mice by Induction of Premature Catagen Development and Deleterious Perifollicular Inflammatory Events via Neuropeptide Substance P-Dependent Pathways. Am. J. Pathol. 2003, 162, 803–814. [Google Scholar] [CrossRef] [PubMed]
- O’Connor, T.M.; O’Connell, J.; O’Brien, D.I.; Goode, T.; Bredin, C.P.; Shanahan, F. The role of substance P in inflammatory disease. J. Cell. Physiol. 2004, 201, 167–180. [Google Scholar] [CrossRef] [PubMed]
- Siebenhaar, F.; Sharov, A.A.; Peters, E.M.J.; Sharova, T.Y.; Syska, W.; Mardaryev, A.N.; Freyschmidt-Paul, P.; Sundberg, J.P.; Maurer, M.; Botchkarev, V.A. Substance P as an Immunomodulatory Neuropeptide in a Mouse Model for Autoimmune Hair Loss (Alopecia Areata). J. Investig. Dermatol. 2007, 127, 1489–1497. [Google Scholar] [CrossRef]
- Zhao, Y.; Zhang, B.; Caulloo, S.; Chen, X.; Li, Y.; Zhang, X. Diffuse alopecia areata is associated with intense inflammatory infiltration and CD8+ T cells in hair loss regions and an increase in serum IgE level. Indian J. Dermatol. Venereol. Leprol. 2012, 78, 709–714. [Google Scholar] [CrossRef]
- Zhang, B.; Zhao, Y.; Cai, Z.; Caulloo, S.; McElwee, K.; Li, Y.; Chen, X.; Yu, M.; Yang, J.; Chen, W.; et al. Early stage alopecia areata is associated with inflammation in the upper dermis and damage to the hair follicle infundibulum. Australas. J. Dermatol. 2013, 54, 184–191. [Google Scholar] [CrossRef]
- Contreras-Jurado, C.; Lorz, C.; García-Serrano, L.; Paramio, J.M.; Aranda, A. Thyroid hormone signaling controls hair follicle stem cell function. Mol. Biol. Cell 2015, 26, 1263–1272. [Google Scholar] [CrossRef] [PubMed]
- Kinter, K.J.; Anekar, A.A. Biochemistry, Dihydrotestosterone; StatPearls Publishing: Treasure Island, FL, USA, 2022. Available online: http://www.ncbi.nlm.nih.gov/books/NBK557634/ (accessed on 27 December 2022).
- Thom, E. Stress and the hair growth cycle: Cortisol-induced hair growth disruption. J. Drugs Dermatol. 2016, 15, 1001–1004. [Google Scholar] [PubMed]
- Bassino, E.; Gasparri, F.; Munaron, L. Protective role of nutritional plants containing flavonoids in hair follicle disruption: A review. Int. J. Mol. Sci. 2020, 21, 523. [Google Scholar] [CrossRef] [PubMed]
- Fu, D.; Huang, J.; Li, K.; Chen, Y.; He, Y.; Sun, Y.; Guo, Y.; Du, L.; Qu, Q.; Miao, Y.; et al. Dihydrotestosterone-induced hair regrowth inhibition by activating androgen receptor in C57BL6 mice simulates androgenetic alopecia. Biomed. Pharmacother. 2021, 137, 111247. [Google Scholar] [CrossRef] [PubMed]
- Kaufman, K.D.; Olsen, E.A.; Whiting, D.; Savin, R.; DeVillez, R.; Bergfeld, W.; Price, V.H.; Van Neste, D.; Roberts, J.L.; Hordinsky, M.; et al. Finasteride in the treatment of men with androgenetic alopecia. Finasteride Male Pattern Hair Loss Study Group. J. Am. Acad. Dermatol. 1998, 39, 578–589. [Google Scholar] [CrossRef]
- Urysiak-Czubatka, I.; Kmieć, M.L.; Broniarczyk-Dyła, G. Assessment of the usefulness of dihydrotestosterone in the diagnostics of patients with androgenetic alopecia. Adv. Dermatol. Allergol. 2014, 31, 207–215. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Xu, J.; Jing, J.; Wu, X.; Lv, Z. Serum levels of androgen-associated hormones are correlated with curative effect in androgenic alopecia in young men. Med. Sci. Monit. Int. Med. J. Exp. Clin. Res. 2018, 24, 7770–7777. [Google Scholar] [CrossRef]
- Grymowicz, M.; Rudnicka, E.; Podfigurna, A.; Napierala, P.; Smolarczyk, R.; Smolarczyk, K.; Meczekalski, B. Hormonal effects on hair follicles. Int. J. Mol. Sci. 2020, 21, 5342. [Google Scholar] [CrossRef]
- Levy, L.L.; Emer, J.J. Female pattern alopecia: Current perspectives. Int. J. Womens Health 2013, 5, 541–556. [Google Scholar] [CrossRef]
- Riedel-Baima, B.; Riedel, A. Female pattern hair loss may be triggered by low oestrogen to androgen ratio. Endocr. Regul. 2008, 42, 13–16. [Google Scholar]
- Adenuga, P.; Summers, P.; Bergfeld, W. Hair regrowth in a male patient with extensive androgenetic alopecia on estrogen therapy. J. Am. Acad. Dermatol. 2012, 67, e121–e123. [Google Scholar] [CrossRef] [PubMed]
- Worret, I.; Arp, W.; Zahradnik, H.P.; Andreas, J.O.; Binder, N. Acne resolution rates: Results of a single-blind, randomized, controlled, parallel phase III trial with EE/CMA (Belara) and EE/LNG (Microgynon). Dermatology 2001, 203, 38–44. [Google Scholar] [CrossRef] [PubMed]
- Kozicka, K.; Kozicka, K.; Łukasik, A.; Pastuszczak, M.; Jaworek, A.; Spałkowska, M.; Kłosowicz, A.; Dyduch, G.; Wojas-Pelc, A. Is hormone testing worthwhile in patients with female pattern hair loss? Pol. Merkur. Lek. Organ Pol. Tow. Lek. 2020, 48, 323–326. [Google Scholar]
- York, J.; Nicholson, T.; Minors, P.; Duncan, D.F. Stressful life events and loss of hair among adult women, a case-control study. Psychol. Rep. 1998, 82, 1044–1046. [Google Scholar] [CrossRef] [PubMed]
- Hughes, E.C.; Saleh, D. Telogen Effluvium; StatPearls Publishing: Treasure Island, FL, USA, 2022. Available online: http://www.ncbi.nlm.nih.gov/books/NBK430848/ (accessed on 28 December 2022).
- Schmidt, J.B. Hormonal basis of male and female androgenic alopecia: Clinical relevance. Skin Pharmacol. 1994, 7, 61–66. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, J.B.; Lindmaier, A.; Spona, J. Hormonal parameters in androgenetic hair loss in the male. Dermatologica 1991, 182, 214–217. [Google Scholar] [CrossRef]
- Agren, U.M.; Tammi, M.; Tammi, R. Hydrocortisone regulation of hyaluronan metabolism in human skin organ culture. J. Cell. Physiol. 1995, 164, 240–248. [Google Scholar] [CrossRef]
- Piérard-Franchimont, C.; De Doncker, P.; Cauwenbergh, G.; Piérard, G.E. Ketoconazole shampoo: Effect of long-term use in androgenic alopecia. Dermatology 1998, 196, 474–477. [Google Scholar] [CrossRef]
- Inui, S.; Itami, S. Reversal of androgenetic alopecia by topical ketoconzole: Relevance of anti-androgenic activity. J. Dermatol. Sci. 2007, 45, 66–68. [Google Scholar] [CrossRef]
- El-Garf, A.; Mohie, M.; Salah, E. Trichogenic effect of topical ketoconazole versus minoxidil 2% in female pattern hair loss: A clinical and trichoscopic evaluation. Biomed. Dermatol. 2019, 3, 8. [Google Scholar] [CrossRef]
- Almohanna, H.M.; Ahmed, A.A.; Tsatalis, J.P.; Tosti, A. The Role of Vitamins and Minerals in Hair Loss: A Review. Dermatol. Ther. 2018, 9, 51–70. [Google Scholar] [CrossRef] [PubMed]
- Guo, E.L.; Katta, R. Diet and hair loss: Effects of nutrient deficiency and supplement use. Dermatol. Pract. Concept. 2017, 7, 1. [Google Scholar] [CrossRef]
- Gowda, D.; Premalatha, V.; Imtiyaz, D.B. Prevalence of nutritional deficiencies in hair loss among Indian participants: Results of a cross-sectional study. Int. J. Trichol. 2017, 9, 101–104. [Google Scholar] [CrossRef] [PubMed]
- Lengg, N.; Heidecker, B.; Seifert, B.; Trüeb, R. Dietary supplement increases anagen hair rate in women with telogen effluvium: Results of a double-blind, placebo-controlled trial. Therapy 2007, 4, 59–65. [Google Scholar] [CrossRef]
- Ablon, G. A 3-month, randomized, double-blind, placebo-controlled study evaluating the ability of an extra-strength marine protein supplement to promote hair growth and decrease shedding in women with self-perceived thinning hair. Dermatol. Res. Pract. 2015, 2015, 841570. [Google Scholar] [CrossRef]
- Rizer, R.L.; Stephens, T.J.; Herndon, J.H.; Sperber, B.R.; Murphy, J.; Ablon, G.R. A marine protein-based dietary supplement for subclinical hair thinning/loss: Results of a multisite, double-blind, placebo-controlled clinical trial. Int. J. Trichol. 2015, 7, 156–166. [Google Scholar] [CrossRef]
- Munkhbayar, S.; Munkhbayar, S.; Jang, S.; Cho, A.-R.; Choi, S.-J.; Shin, C.Y.; Eun, H.C.; Kim, K.H.; Kwon, O. Role of Arachidonic Acid in Promoting Hair Growth. Ann. Dermatol. 2016, 28, 55–64. [Google Scholar] [CrossRef]
- Hamel, A.F.; Menard, M.T.; Novak, M.A. Fatty acid supplements improve hair coat condition in rhesus macaques. J. Med. Primatol. 2017, 46, 248–251. [Google Scholar] [CrossRef]
- Skolnik, P.; Eaglstein, W.H.; Ziboh, V.A. Human essential fatty acid deficiency: Treatment by topical application of linoleic acid. Arch. Dermatol. 1977, 113, 939–941. [Google Scholar] [CrossRef]
- Le Floc’h, C.; Cheniti, A.; Connétable, S.; Piccardi, N.; Vincenzi, C.; Tosti, A.A. Effect of a nutritional supplement on hair loss in women. J. Cosmet. Dermatol. 2015, 14, 76–82. [Google Scholar] [CrossRef]
- Suo, L.; Sundberg, J.P.; Everts, H.B. Dietary vitamin A regulates wingless-related MMTV integration site signaling to alter the hair cycle. Exp. Biol. Med. 2014, 240, 618–623. [Google Scholar] [CrossRef] [PubMed]
- VanBuren, C.A.; Everts, H.B. Vitamin A in Skin and Hair: An Update. Nutrients 2022, 14, 2952. [Google Scholar] [CrossRef] [PubMed]
- Shmunes, E. Hypervitaminosis A in a patient with alopecia receiving renal dialysis. Arch. Dermatol. 1979, 115, 882–883. [Google Scholar] [CrossRef] [PubMed]
- Kmieć, M.L.; Pajor, A.; Broniarczyk-Dyła, G. Evaluation of biophysical skin parameters and assessment of hair growth in patients with acne treated with isotretinoin. Postepy Dermatol. Alergol. 2013, 6, 343–349. [Google Scholar] [CrossRef]
- Ertugrul, D.T.; Karadag, A.S.; Takcı, Z.; Bilgili, S.G.; Ozkol, H.U.; Tutal, E.; Akin, K.O. Serum holotranscobalamine, vitamin B12, folic acid and homocysteine levels in alopecia areata patients. Cutan. Ocul. Toxicol. 2012, 32, 1–3. [Google Scholar] [CrossRef]
- Durusoy, C.; Ozenli, Y.; Adiguzel, A.; Budakoglu, I.Y.; Tugal, O.; Arikan, S.; Uslu, A.; Gulec, A.T. The role of psychological factors and serum zinc, folate and vitamin B12levels in the aetiology of trichodynia: A case-control study. Clin. Exp. Dermatol. 2009, 34, 789–792. [Google Scholar] [CrossRef]
- Trüeb, R.M. Serum biotin levels in women complaining of hair loss. Int. J. Trichol. 2016, 8, 73–77. [Google Scholar] [CrossRef]
- Ardabilygazir, A.; Afshariyamchlou, S.; Mir, D.; Sachmechi, I. Effect of high-dose biotin on thyroid function tests: Case report and literature review. Cureus 2018, 10, e2845. [Google Scholar] [CrossRef]
- Li, D.; Rooney, M.R.; Burmeister, L.A.; Basta, N.E.; Lutsey, P.L. Trends in daily use of biotin supplements among US adults, 1999–2016. JAMA 2020, 324, 605–607. [Google Scholar] [CrossRef]
- Patel, D.P.; Swink, S.M.; Castelo-Soccio, L. A review of the use of biotin for hair loss. Skin Appendage Disord. 2017, 3, 166–169. [Google Scholar] [CrossRef]
- Nair, R.; Maseeh, A. Vitamin D: The “sunshine” vitamin. J. Pharmacol. Pharmacother. 2012, 3, 118–126. [Google Scholar] [CrossRef] [PubMed]
- Gerkowicz, A.; Chyl-Surdacka, K.; Krasowska, D.; Chodorowska, G. The role of vitamin D in non-scarring alopecia. Int. J. Mol. Sci. 2017, 18, 2653. [Google Scholar] [CrossRef] [PubMed]
- Amor, K.T.; Rashid, R.M.; Mirmirani, P. Does D matter? The role of vitamin D in hair disorders and hair follicle cycling. Dermatol. Online J. 2010, 16, 3. [Google Scholar] [CrossRef]
- Tahlawy, S.; Alkhayat, M.; Ali, H.; Samhoud, E. Serum vitamin D and serum ferritin levels in male pattern hair loss: Is there a role? Fayoum Univ. Med. J. 2021, 8, 1–8. [Google Scholar] [CrossRef]
- Rasheed, H.; Mahgoub, D.; Hegazy, R.; El-Komy, M.; Hay, R.A.; Hamid, M.; Hamdy, E. Serum ferritin and vitamin D in female hair loss: Do they play a role? Skin Pharmacol. Physiol. 2013, 26, 101–107. [Google Scholar] [CrossRef]
- Karadağ, A.S.; Ertuğrul, D.T.; Tutal, E.; Akin, K.O. The role of anemia and vitamin D levels in acute and chronic telogen effluvium. Turk. J. Med. Sci. 2011, 41, 827–833. [Google Scholar] [CrossRef]
- Zubair, Z.; Kantamaneni, K.; Jalla, K.; Renzu, M.; Jena, R.; Jain, R.; Muralidharan, S.; Yanamala, V.L.; Alfonso, M. Prevalence of low serum vitamin D levels in patients presenting with androgenetic alopecia: A review. Cureus 2021, 13, e20431. [Google Scholar] [CrossRef] [PubMed]
- Hassan, G.F.R.; Sadoma, M.E.T.; Elbatsh, M.M.; Ibrahim, Z.A. Treatment with oral vitamin D alone, topical minoxidil, or combination of both in patients with female pattern hair loss: A comparative clinical and dermoscopic study. J. Cosmet. Dermatol. 2022, 21, 3917–3924. [Google Scholar] [CrossRef]
- Ring, C.; Heitmiller, K.; Correia, E.; Gabriel, Z.; Saedi, N. Nutraceuticals for androgenetic alopecia. J. Clin. Aesthetic Dermatol. 2022, 15, 26–29. [Google Scholar]
- Beoy, L.A.; Woei, W.J.; Hay, Y.K. Effects of tocotrienol supplementation on hair growth in human volunteers. Trop. Life Sci. Res. 2010, 21, 91–99. [Google Scholar]
- Kil, M.S.; Kim, C.W.; Kim, S.S. Analysis of serum zinc and copper concentrations in hair loss. Ann. Dermatol. 2013, 25, 405–409. [Google Scholar] [CrossRef] [PubMed]
- Karashima, T.; Tsuruta, D.; Hamada, T.; Ono, F.; Ishii, N.; Abe, T.; Ohyama, B.; Nakama, T.; Dainichi, T.; Hashimoto, T. Oral zinc therapy for zinc deficiency-related telogen effluvium. Dermatol. Ther. 2012, 25, 210–213. [Google Scholar] [CrossRef] [PubMed]
- Park, H.; Kim, C.W.; Kim, S.S.; Park, C.W. The Therapeutic Effect and the Changed Serum Zinc Level after Zinc Supplementation in Alopecia Areata Patients Who Had a Low Serum Zinc Level. Ann. Dermatol. 2009, 21, 142–146. [Google Scholar] [CrossRef] [PubMed]
- Bates, J.M.; Spate, V.L.; Morris, J.S.; Germain, D.L.S.; Galton, V.A. Effects of selenium deficiency on tissue selenium content, deiodinase activity, and thyroid hormone economy in the rat during development. Endocrinology 2000, 141, 2490–2500. [Google Scholar] [CrossRef]
- Sengupta, A.; Lichti, U.F.; Carlson, B.A.; Ryscavage, A.O.; Gladyshev, V.N.; Yuspa, S.H.; Hatfield, D.L. Selenoproteins are essential for proper keratinocyte function and skin development. PLoS ONE 2010, 5, e12249. [Google Scholar] [CrossRef]
- Vinton, N.E.; Dahlstrom, K.A.; Strobel, C.T.; Ament, M.E. Macrocytosis and pseudoalbinism: Manifestations of selenium deficiency. J. Pediatr. 1987, 111, 711–771. [Google Scholar] [CrossRef]
- Masumoto, K.; Nagata, K.; Higashi, M.; Nakatsuji, T.; Uesugi, T.; Takahashi, Y.; Nishimoto, Y.; Kitajima, J.; Hikino, S.; Hara, T.; et al. Clinical features of selenium deficiency in infants receiving long-term nutritional support. Nutrition 2007, 23, 782–787. [Google Scholar] [CrossRef]
- Liamsombut, S.; Pomsoong, C.; Kositkuljorn, C.; Leerunyakul, K.; Tantrakul, V.; Suchonwanit, P. Sleep quality in men with androgenetic alopecia. Sleep Breath. 2022. Epub: Apr 25. [Google Scholar] [CrossRef]
- Yi, Y.; Qiu, J.; Jia, J.; Djakaya, G.D.N.; Li, X.; Fu, J.; Chen, Y.; Chen, Q.; Miao, Y.; Hu, Z. Severity of androgenetic alopecia associated with poor sleeping habits and carnivorous eating and junk food consumption—A web-based investigation of male pattern hair loss in China. Dermatol. Ther. 2020, 33, e13273. [Google Scholar] [CrossRef]
- Shakoei, S.; Torabimirzaee, A.; Saffarian, Z.; Abedini, R. Sleep disturbance in alopecia areata: A cross-sectional study. Health Sci. Rep. 2022, 5, e576. [Google Scholar] [CrossRef]
- Seo, H.-M.; Kim, T.L.; Kim, J.S. The risk of alopecia areata and other related autoimmune diseases in patients with sleep disorders: A Korean population-based retrospective cohort study. Sleep 2018, 41, zsy111. [Google Scholar] [CrossRef] [PubMed]
- Reddy, S.; Reddy, V.; Sharma, S. Physiology, Circadian Rhythm; StatPearls Publishing: Treasure Island, FL, USA, 2022. Available online: http://www.ncbi.nlm.nih.gov/books/NBK519507/ (accessed on 30 December 2022).
- Geyfman, M.; Andersen, B. Clock genes, hair growth and aging. Aging 2010, 2, 122–128. [Google Scholar] [CrossRef]
- Rossi, A.; Fortuna, M.C.; Caro, G.; Pranteda, G.; Garelli, V.; Pompili, U.; Carlesimo, M. Chemotherapy-induced alopecia management: Clinical experience and practical advice. J. Cosmet. Dermatol. 2017, 16, 537–541. [Google Scholar] [CrossRef]
- West, H. Chemotherapy-induced hair loss (Alopecia). JAMA Oncol. 2017, 3, 1147. [Google Scholar] [CrossRef] [PubMed]
- Trüeb, R.M. Chemotherapy-induced alopecia. Curr. Opin. Support. Palliat. Care 2010, 4, 281–284. [Google Scholar] [CrossRef] [PubMed]
- Young, A.; Arif, A. The use of scalp cooling for chemotherapy-induced hair loss. Br. J. Nurs. 2016, 25, S22–S27. [Google Scholar] [CrossRef]
- Murphrey, M.B.; Agarwal, S.; Zito, P.M. Anatomy, Hair; StatPearls Publishing: Treasure Island, FL, USA,, 2022. Available online: http://www.ncbi.nlm.nih.gov/books/NBK513312/ (accessed on 30 December 2022).
- Koyama, T.; Kobayashi, K.; Hama, T.; Murakami, K.; Ogawa, R. Standardized scalp massage results in increased hair thickness by inducing stretching forces to dermal papilla cells in the subcutaneous tissue. Eplasty 2016, 16, e8. [Google Scholar]
- English, R.S.; Barazesh, J.M. Self-Assessments of Standardized Scalp Massages for Androgenic Alopecia: Survey Results. Dermatol. Ther. 2019, 9, 167–178. [Google Scholar] [CrossRef]
- Feaster, B.; Onamusi, T.; Cooley, J.E.; McMichael, A.J. Oral minoxidil use in androgenetic alopecia and telogen effluvium. Arch. Dermatol. Res. 2022. Epub: Mar 4. [Google Scholar] [CrossRef]
- Sharma, A.; Michelle, L.; Juhasz, M.; Ramos, P.; Mesinkovska, N.A. Low-dose oral minoxidil as treatment for non-scarring alopecia: A systematic review. Int. J. Dermatol. 2020, 59, 1013–1019. [Google Scholar] [CrossRef]
- Fenton, D.A.; Wilkinson, J.D. Topical minoxidil in the treatment of alopecia areata. BMJ 1983, 287, 1015–1017. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.K.; Talukder, M.; Venkataraman, M.; Bamimore, M.A. Minoxidil: A comprehensive review. J. Dermatol. Treat. 2021, 33, 1896–1906. [Google Scholar] [CrossRef] [PubMed]
- Mori, O.; Uno, H. The effect of topical minoxidil on hair follicular cycles of rats. J. Dermatol. 1990, 17, 276–281. [Google Scholar] [CrossRef] [PubMed]
- Messenger, A.G.; Rundegren, J. Minoxidil: Mechanisms of action on hair growth. Br. J. Dermatol. 2004, 150, 186–194. [Google Scholar] [CrossRef]
- Gupta, A.K.; Charrette, A. Topical minoxidil: Systematic review and meta-analysis of its efficacy in androgenetic alopecia. SkinMed 2015, 13, 185–189. [Google Scholar]
- Randolph, M.; Tosti, A. Oral minoxidil treatment for hair loss: A review of efficacy and safety. J. Am. Acad. Dermatol. 2021, 84, 737–746. [Google Scholar] [CrossRef]
- Moussa, A.; Kazmi, A.; Bokhari, L.; Sinclair, D. Bicalutamide improves minoxidil-induced hypertrichosis in female pattern hair loss: A retrospective review of 35 patients. J. Am. Acad. Dermatol. 2022, 87, 488–490. [Google Scholar] [CrossRef]
- Kim, M.J.; Seong, K.-Y.; Kim, D.S.; Jeong, J.S.; Kim, S.Y.; Lee, S.; Yang, S.Y.; An, B.-S. Minoxidil-loaded hyaluronic acid dissolving microneedles to alleviate hair loss in an alopecia animal model. Acta Biomater. 2022, 143, 189–202. [Google Scholar] [CrossRef]
- Fresta, M.; Mancuso, A.; Cristiano, M.C.; Urbanek, K.; Cilurzo, F.; Cosco, D.; Iannone, M.; Paolino, D. Targeting of the pilosebaceous follicle by liquid crystal nanocarriers: In vitro and in vivo effects of the entrapped minoxidil. Pharmaceutics 2020, 12, 1127. [Google Scholar] [CrossRef]
- Thor, D.; Pagani, A.; Bukowiecki, J.; Houschyar, K.; Kølle, S.F.; Wyles, S.P.; Duscher, D. Novel hair restoration technology counteracts androgenic hair loss and promotes hair growth in a blinded clinical trial. J. Clin. Med. 2023, 12, 470. [Google Scholar] [CrossRef]
- Škulj, A.Z.; Poljšak, N.; Glavač, N.K.; Kreft, S. Herbal preparations for the treatment of hair loss. Arch. Dermatol. Res. 2020, 312, 395–406. [Google Scholar] [CrossRef] [PubMed]
- Hosking, A.M.; Juhasz, M.; Mesinkovska, N.A. Complementary and alternative treatments for alopecia: A comprehensive review. Skin Appendage Disord. 2019, 5, 72–89. [Google Scholar] [CrossRef] [PubMed]
- Hyun, J.; Im, J.; Kim, S.-W.; Kim, H.Y.; Seo, I.; Bhang, S.H. Morus alba root extract induces the anagen phase in the human hair follicle dermal papilla cells. Pharmaceutics 2021, 13, 1155. [Google Scholar] [CrossRef] [PubMed]
- Roy, R.K.; Thakur, M.; Dixit, V.K. Hair growth promoting activity of Eclipta alba in male albino rats. Arch. Dermatol. Res. 2008, 300, 357–364. [Google Scholar] [CrossRef]
- Kim, J.; Kim, S.R.; Choi, Y.-H.; Shin, J.Y.; Kim, C.D.; Kang, N.-G.; Park, B.C.; Lee, S. Quercitrin stimulates hair growth with enhanced expression of growth factors via activation of MAPK/CREB signaling pathway. Molecules 2020, 25, 4004. [Google Scholar] [CrossRef]
- Wikramanayake, T.C.; Villasante, A.C.; Mauro, L.M.; Perez, C.I.; Schachner, L.A.; Jimenez, J.J. Prevention and treatment of alopecia areata with quercetin in the C3H/HeJ mouse model. Cell Stress Chaperones 2012, 17, 267–274. [Google Scholar] [CrossRef]
- Panahi, Y.; Taghizadeh, M.; Marzony, E.T.; Sahebkar, A. Rosemary oil vs minoxidil 2% for the treatment of androgenetic alopecia: A randomized comparative trial. SkinMed 2015, 13, 15–21. [Google Scholar]
- Lee, B.H.; Lee, J.S.; Kim, Y.C. Hair growth-promoting effects of lavender oil in C57BL/6 mice. Toxicol. Res. 2016, 32, 103–108. [Google Scholar] [CrossRef]
- Takahashi, T.; Kamiya, T.; Tokoo, Y. Proanthocyanidins from grape seeds promote proliferation of mouse hair follicle cells in vitro and convert hair cycle in vivo. Acta Derm.-Venereol. 1998, 78, 428–432. [Google Scholar] [CrossRef]
- Takahashi, T.; Kamimura, A.; Yokoo, Y.; Watanabe, Y. Procyanidin B-2 and the hair-growing activity of proanthocyanidins. Cosmet. Toilet. 2001, 116, 335–351. [Google Scholar]
- Ablon, G.; Kogan, S. A six-month, randomized, double-blind, placebo-controlled study evaluating the safety and efficacy of a nutraceutical supplement for promoting hair growth in women with self-perceived thinning hair. J. Drugs Dermatol. 2018, 17, 558–565. Available online: https://jddonline.com/articles/a-six-month-randomized-double-blind-placebo-controlled-study-evaluating-the-safety-and-efficacy-of-a-S1545961618P0558X/ (accessed on 31 December 2022). [PubMed]
- Cho, Y.H.; Lee, S.Y.; Jeong, D.W.; Choi, E.J.; Kim, Y.J.; Lee, J.G.; Yi, Y.H.; Cha, H.S. Effect of pumpkin seed oil on hair growth in men with androgenetic alopecia: A randomized, double-blind, placebo-controlled trial. Evid.-Based Complement. Altern. Med. ECAM 2014, 2014, 549721. [Google Scholar] [CrossRef] [PubMed]
- Kang, X.-C.; Chen, T.; Zhou, J.-L.; Shen, P.-Y.; Dai, S.-H.; Gao, C.-Q.; Zhang, J.-Y.; Xiong, X.-Y.; Liu, D.-B. Phytosterols in hull-less pumpkin seed oil, rich in ∆7-phytosterols, ameliorate benign prostatic hyperplasia by lowing 5α-reductase and regulating balance between cell proliferation and apoptosis in rats. Food Nutr. Res. 2021, 65, 7537. [Google Scholar] [CrossRef] [PubMed]
- Avci, P.; Gupta, A.; Sadasivam, M.; Vecchio, D.; Pam, Z.; Pam, N.; Hamblin, M.R. Low-level laser (light) therapy (LLLT) in skin: Stimulating, healing, restoring. Semin. Cutan. Med. Surg. 2013, 32, 41–52. [Google Scholar]
- Avci, P.; Gupta, G.K.; Clark, J.; Wikonkal, N.; Hamblin, M.R. Low-level laser (Light) therapy (LLLT) for treatment of hair loss. Lasers Surg. Med. 2014, 46, 144–151. [Google Scholar] [CrossRef]
- Esmat, S.M.; Hegazy, R.A.; Gawdat, H.I.; Hay, R.M.A.; El Naggar, R.S.A.R.; Moneib, H. Low level light-minoxidil 5% combination versus either therapeutic modality alone in management of female patterned hair loss: A randomized controlled study. Lasers Surg. Med. 2017, 49, 835–843. [Google Scholar] [CrossRef] [PubMed]
- Liu, K.-H.; Liu, D.; Chen, Y.-T.; Chin, S.-Y. Comparative effectiveness of low-level laser therapy for adult androgenic alopecia: A system review and meta-analysis of randomized controlled trials. Lasers Med. Sci. 2019, 34, 1063–1069. [Google Scholar] [CrossRef]
- Johnstone, M.A.; Albert, D.M. Prostaglandin-induced hair growth. Surv. Ophthalmol. 2002, 47, S185–S202. [Google Scholar] [CrossRef]
- Blume-Peytavi, U.; Lönnfors, S.; Hillmann, K.; Bartels, N.G. A randomized double-blind placebo-controlled pilot study to assess the efficacy of a 24-week topical treatment by latanoprost 0.1% on hair growth and pigmentation in healthy volunteers with androgenetic alopecia. J. Am. Acad. Dermatol. 2012, 66, 794–800. [Google Scholar] [CrossRef]
- Khidhir, K.G.; Woodward, D.F.; Farjo, N.P.; Farjo, B.K.; Tang, E.S.; Wang, J.W.; Picksley, S.M.; Randall, V. The prostamide-related glaucoma therapy, bimatoprost, offers a novel approach for treating scalp alopecias. FASEB J. 2013, 27, 557–567. [Google Scholar] [CrossRef]
- Garza, L.A.; Liu, Y.; Yang, Z.; Alagesan, B.; Lawson, J.A.; Norberg, S.M.; Loy, D.E.; Zhao, T.; Blatt, H.B.; Stanton, D.C.; et al. Prostaglandin D2 inhibits hair growth and is elevated in bald scalp of men with androgenetic alopecia. Sci. Transl. Med. 2012, 4, 126ra34. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.; Xiang, L.-J.; Shi, H.-X.; Zhang, J.; Jiang, L.; Cai, P.; Lin, Z.-L.; Lin, B.-B.; Huang, Y.; Zhang, H.-L.; et al. Fibroblast growth factors stimulate hair growth through β-catenin and Shh expression in C57BL/6 mice. BioMed Res. Int. 2015, 2015, 730139. [Google Scholar] [CrossRef] [PubMed]
- Pavlovic, V.; Ciric, M.; Jovanovic, V.; Stojanovic, P. Platelet Rich Plasma: A short overview of certain bioactive components. Open Med. 2016, 11, 242–247. [Google Scholar] [CrossRef] [PubMed]
- Stevens, J.; Khetarpal, S. Platelet-rich plasma for androgenetic alopecia: A review of the literature and proposed treatment protocol. Int. J. Womens Dermatol. 2018, 5, 46–51. [Google Scholar] [CrossRef]
- Giordano, S.; Romeo, M.; Lankinen, P. Platelet-rich plasma for androgenetic alopecia: Does it work? Evidence from meta analysis. J. Cosmet. Dermatol. 2017, 16, 374–381. [Google Scholar] [CrossRef]
- Mao, G.; Zhang, G.; Fan, W. Platelet-rich plasma for treating androgenic alopecia: A systematic review. Aesthetic Plast. Surg. 2019, 43, 1326–1336. [Google Scholar] [CrossRef]
- Dervishi, G.; Liu, H.; Peternel, S.; Labeit, A.; Peinemann, F. Autologous platelet-rich plasma therapy for pattern hair loss: A systematic review. J. Cosmet. Dermatol. 2020, 19, 827–835. [Google Scholar] [CrossRef]
- Gupta, A.K.; Renaud, H.J.; Bamimore, M. Platelet-rich plasma for androgenetic alopecia: Efficacy differences between men and women. Dermatol. Ther. 2020, 33, e14143. [Google Scholar] [CrossRef]
- Kuffler, D.P. Variables affecting the potential efficacy of PRP in providing chronic pain relief. J. Pain Res. 2019, 12, 109–116. [Google Scholar] [CrossRef]
- Knovich, M.A.; Storey, J.A.; Coffman, L.G.; Torti, S.V. Ferritin for the clinician. Blood Rev. 2009, 23, 95–104. [Google Scholar] [CrossRef]
- Trost, L.B.; Bergfeld, W.F.; Calogeras, E. The diagnosis and treatment of iron deficiency and its potential relationship to hair loss. J. Am. Acad. Dermatol. 2006, 54, 824–844. [Google Scholar] [CrossRef] [PubMed]
- Amornpinyo, W.; Thuangtong, R.; Wongdama, S.; Triwongwaranat, D. Clinical characteristics of female patterned hair loss in patients attending hair clinic in Thailand. Siriraj Med. J. 2022, 74, 1. [Google Scholar] [CrossRef]
- Park, S.Y.; Na, S.Y.; Kim, J.H.; Cho, S.; Lee, J.H. Iron plays a certain role in patterned hair loss. J. Korean Med. Sci. 2013, 28, 934–938. [Google Scholar] [CrossRef] [PubMed]
- Coad, J.; Pedley, K. Iron deficiency and iron deficiency anemia in women. Scand. J. Clin. Lab. Investig. Suppl. 2014, 244, 82–89, discussion 89. [Google Scholar] [CrossRef]
- Bossuyt, X.; De Langhe, E.; Borghi, M.O.; Meroni, P.L. Understanding and interpreting antinuclear antibody tests in systemic rheumatic diseases. Nat. Rev. Rheumatol. 2020, 16, 715–726. [Google Scholar] [CrossRef]
- Choi, W.J.; Kim, J.E.; Kang, H. Frequency of antinuclear antibody positivity in patients with pattern hair loss. Ann. Dermatol. 2015, 27, 210–212. [Google Scholar] [CrossRef][Green Version]
- Santhosh, P.; Ajithkumar, K. Anti-nuclear antibodies: A practical approach to testing and interpretation. J. Skin Sex. Transm. Dis. 2021, 3, 175–179. [Google Scholar] [CrossRef]
- Li, Q.-Z.; Karp, D.R.; Quan, J.; Branch, V.; Zhou, J.; Lian, Y.; Chong, B.F.; Wakeland, E.K.; Olsen, N.J. Risk factors for ANA positivity in healthy persons. Arthritis Res. Ther. 2011, 13, R38. [Google Scholar] [CrossRef]
- Lum, B.; Sergent, S.R. Rapid Plasma Reagin; StatPearls Publishing: Treasure Island, FL, USA, 2022. Available online: http://www.ncbi.nlm.nih.gov/books/NBK557732/ (accessed on 30 December 2022).
- Tognetti, L.; Cinotti, E.; Perrot, J.-L.; Campoli, M.; Rubegni, P. Syphilitic alopecia: Uncommon trichoscopic findings. Dermatol. Pract. Concept. 2017, 7, 55–59. [Google Scholar] [CrossRef]
- Doche, I.; Hordinsky, M.K.; Valente, N.Y.S.; Romiti, R.; Tosti, A. Syphilitic alopecia: Case reports and trichoscopic findings. Skin Appendage Disord. 2017, 3, 222–224. [Google Scholar] [CrossRef]
- Van Beek, N.; Bodo, E.; Kromminga, A.; Ga, E.; Meyer, K.; Zmijewski, M.A.; Slominski, A.; Wenzel, B.E.; Paus, R. Thyroid hormones directly alter human hair follicle functions: Anagen prolongation and stimulation of both hair matrix keratinocyte proliferation and hair pigmentation. J. Clin. Endocrinol. Metab. 2008, 93, 4381–4388. [Google Scholar] [CrossRef] [PubMed]
- Thau, L.; Gandhi, J.; Sharma, S. Physiology, Cortisol; StatPearls Publishing: Treasure Island, FL, USA, 2022. Available online: http://www.ncbi.nlm.nih.gov/books/NBK538239/ (accessed on 30 December 2022).
- Hannibal, K.E.; Bishop, M.D. Chronic stress, cortisol dysfunction, and pain: A psychoneuroendocrine rationale for stress management in pain rehabilitation. Phys. Ther. 2014, 94, 1816–1825. [Google Scholar] [CrossRef] [PubMed]
- Adam, E.K.; Kumari, M. Assessing salivary cortisol in large-scale, epidemiological research. Psychoneuroendocrinology 2009, 34, 1423–1436. [Google Scholar] [CrossRef] [PubMed]
- Hodes, A.; Meyer, J.; Lodish, M.B.; Stratakis, C.A.; Zilbermint, M. Mini-review of hair cortisol concentration for evaluation of cushing syndrome. Expert Rev. Endocrinol. Metab. 2018, 13, 225–231. [Google Scholar] [CrossRef]
- Wright, K.D.; Hickman, R.; Laudenslager, M.L. Hair cortisol analysis: A promising biomarker of hpa activation in older adults. Gerontologist 2015, 55, S140–S145. [Google Scholar] [CrossRef]
- Russell, E.; Koren, G.; Rieder, M.; Van Uum, S. Hair cortisol as a biological marker of chronic stress: Current status, future directions and unanswered questions. Psychoneuroendocrinology 2012, 37, 589–601. [Google Scholar] [CrossRef]
- Novak, M.A.; Hamel, A.F.; Coleman, K.; Lutz, C.K.; Worlein, J.; Menard, M.; Ryan, A.; Rosenberg, K.; Meyer, J.S. Hair loss and hypothalamic–pituitary–adrenocortical axis activity in captive rhesus macaques (Macaca mulatta). J. Am. Assoc. Lab. Anim. Sci. JAALAS 2014, 53, 261–266. [Google Scholar]
- Vidali, S.; Knuever, J.; Lerchner, J.; Giesen, M.; Bíró, T.; Klinger, M.; Kofler, B.; Funk, W.; Poeggeler, B.; Paus, R. Hypothalamic-pituitary-thyroid axis hormones stimulate mitochondrial function and biogenesis in human hair follicles. J. Investig. Dermatol. 2014, 134, 33–42. [Google Scholar] [CrossRef]
- Short, K.R.; Nygren, J.; Barazzoni, R.; Levine, J.; Nair, K.S. T3 increases mitochondrial ATP production in oxidative muscle despite increased expression of UCP2 and -3. Am. J. Physiol.-Endocrinol. Metab. 2001, 280, E761–E769. [Google Scholar] [CrossRef]
- Singh, B.; Schoeb, T.R.; Bajpai, P.; Slominski, A.; Singh, K.K. Reversing wrinkled skin and hair loss in mice by restoring mitochondrial function. Cell Death Dis. 2018, 9, 735. [Google Scholar] [CrossRef]
- Tang, Y.; Luo, B.; Deng, Z.; Wang, B.; Liu, F.; Li, J.; Shi, W.; Xie, H.; Hu, X.; Li, J. Mitochondrial aerobic respiration is activated during hair follicle stem cell differentiation, and its dysfunction retards hair regeneration. PeerJ 2016, 4, e1821. [Google Scholar] [CrossRef] [PubMed]
- Shin, J.-M.; Ko, J.-W.; Choi, C.-W.; Lee, Y.; Seo, Y.-J.; Lee, J.-H.; Kim, C.-D. Deficiency of Crif1 in hair follicle stem cells retards hair growth cycle in adult mice. PLoS ONE 2020, 15, e0232206. [Google Scholar] [CrossRef] [PubMed]
- Thiel, A. Assessing Mitochrondrial Function. Available online: https://www.integrativepro.com/articles/assessing-mitochrondrial-function (accessed on 30 December 2022).
| Intervention | Support of Hair Cycle | Topical Dose or Dose Range | Evidence (Humans, Animals) |
|---|---|---|---|
| Finasteride | Inhibit 5-alpha-reductase | 1 mg daily | Humans (Men)–FDA Approved |
| Pumpkin seed oil | Inhibit 5-alpha-reductase | 400 mg oral | Humans |
| Herbal based supplement (Nutrafol) | Anti-inflammatory, reduce Stress, and DHT inhibiting properties | Humans | |
| Minoxidil (Topical) | Increase local blood flow | Women—3% or 5% daily Men—5% twice daily | Humans—FDA Approved |
| Morbus alba | Activation of anagen phase | Humans | |
| Low level light therapies | Increased telogen to anagen phase transition | Humans | |
| Latanoprost | Activation of anagen phase | 0.1% latanoprost solution topical | Humans |
| Bimatoprost | Stimulate anagen phase | 0.03% bimatoprost solution topical | Humans |
| Marine protein-based supplement (Viviscal) | Prolongs anagen phase | 450 mg oral | Humans |
| Platelet rich plasma injections | Induces anagen phase | Humans | |
| Bhringaraj (Eclipta alba) | Activation of anagen phase | 5% petroleum ether extract topical | Mice |
| Quercetin | Supports mitochondrial function and anagen phase | Mice |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Natarelli, N.; Gahoonia, N.; Sivamani, R.K. Integrative and Mechanistic Approach to the Hair Growth Cycle and Hair Loss. J. Clin. Med. 2023, 12, 893. https://doi.org/10.3390/jcm12030893
Natarelli N, Gahoonia N, Sivamani RK. Integrative and Mechanistic Approach to the Hair Growth Cycle and Hair Loss. Journal of Clinical Medicine. 2023; 12(3):893. https://doi.org/10.3390/jcm12030893
Chicago/Turabian StyleNatarelli, Nicole, Nimrit Gahoonia, and Raja K. Sivamani. 2023. "Integrative and Mechanistic Approach to the Hair Growth Cycle and Hair Loss" Journal of Clinical Medicine 12, no. 3: 893. https://doi.org/10.3390/jcm12030893
APA StyleNatarelli, N., Gahoonia, N., & Sivamani, R. K. (2023). Integrative and Mechanistic Approach to the Hair Growth Cycle and Hair Loss. Journal of Clinical Medicine, 12(3), 893. https://doi.org/10.3390/jcm12030893







