Pleural Mesothelioma: Advances in Blood and Pleural Biomarkers
Abstract
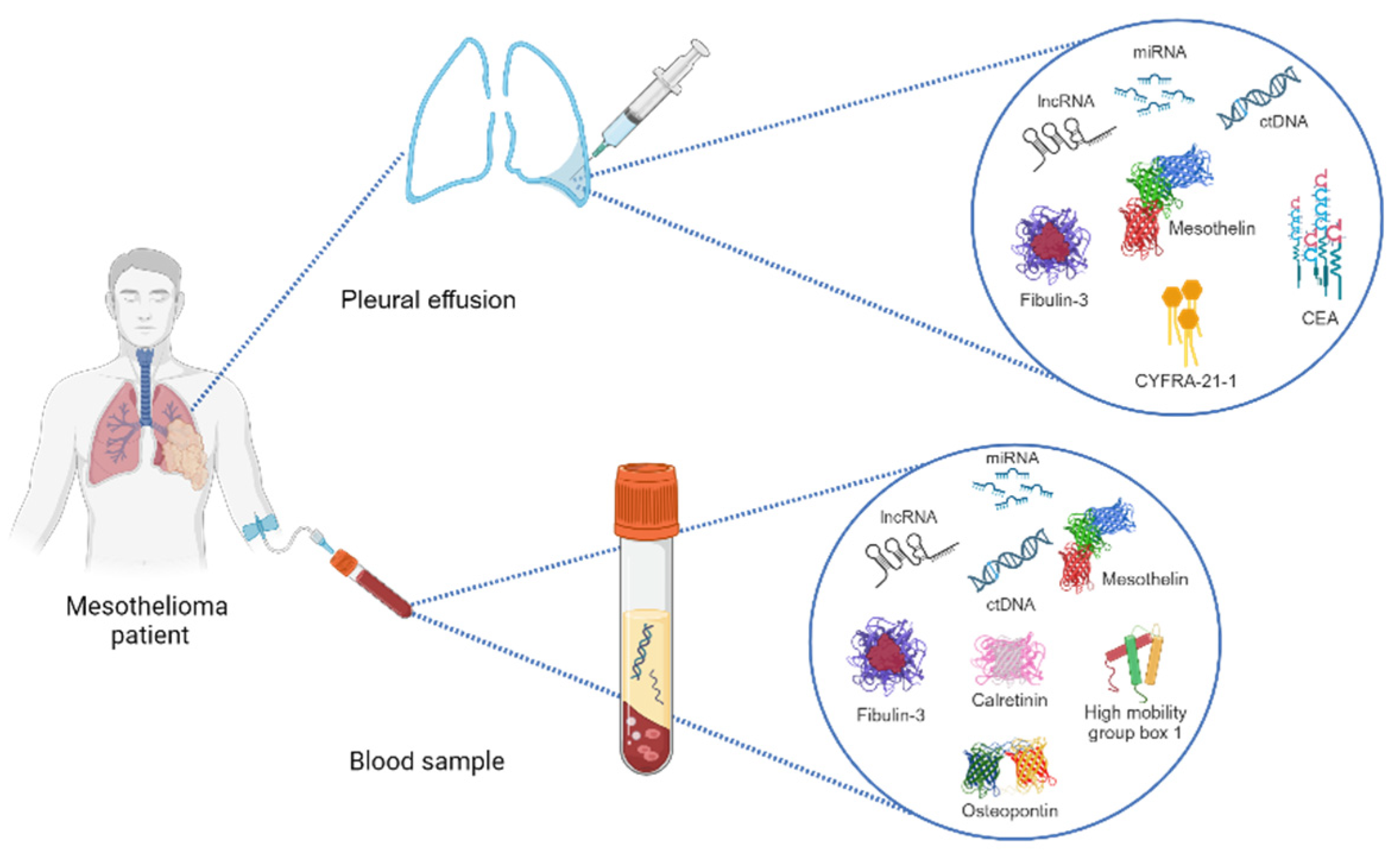
1. Introduction
2. Methods
3. Pleural Fluid Biomarkers of PM
3.1. Mesothelin
3.2. Fibulin-3
3.3. Hyaluronic Acid
3.4. Cell-Free microRNAs
3.5. CYFRA-21-1 and CEA
3.6. Combined Markers Panels
3.7. Cytology
4. Blood Biomarkers of PM
4.1. Mesothelin and SMRP
4.2. Osteopontin (OPN)
4.3. Fibulin-3
4.4. Calretinin
4.5. MicroRNAs (miRNAs) and Long Non-Coding RNAs (lncRNAs)
4.6. Circulating Tumor DNA (ctDNA) and Epigenetic Biomarkers
4.7. High-Mobility Group Box 1 (HMGB1)
4.8. Other Blood Biomarkers
4.9. Combination of Serum Biomarkers
5. Conclusions and Future Perspectives
Funding
Data Availability Statement
Conflicts of Interest
References
- Asciak, R.; George, V.; Rahman, N.M. Update on biology and management of mesothelioma. Eur. Respir. Rev. 2021, 30, 200226. [Google Scholar] [CrossRef] [PubMed]
- Sauter, J.L.; Dacic, S.; Galateau-Salle, F.; Attanoos, R.L.; Butnor, K.J.; Churg, A.; Husain, A.N.; Kadota, K.; Khoor, A.; Nicholson, A.G.; et al. The 2021 WHO Classification of Tumors of the Pleura: Advances Since the 2015 Classification. J. Thorac. Oncol. 2022, 17, 608–622. [Google Scholar] [CrossRef] [PubMed]
- Mlika, M.; Lamzirbi, O.; Limam, M.; Mejri, N.; Saad, S.B.; Chaouch, N.; Miled, K.B.; Marghli, A.; Mezni, F. Clinical and pathological profile of the pleural malignant mesothelioma: A retrospective study about 30 cases. Rev. Pneumol. Clin. 2018, 74, 427–435. [Google Scholar] [CrossRef] [PubMed]
- Congedo, M.T.; West, E.C.; Evangelista, J.; Mattingly, A.A.; Calabrese, G.; Sassorossi, C.; Nocera, A.; Chiappetta, M.; Flamini, S.; Abenavoli, L.; et al. The genetic susceptibility in the development of malignant pleural mesothelioma: Somatic and germline variants, clinicopathological features and implication in practical medical/surgical care: A narrative review. J. Thorac. Dis, 2023; in press. [Google Scholar]
- Moolgavkar, S.H.; Chang, E.T.; Mezei, G.; Mowat, F.S. Epidemiology of Mesothelioma. In Asbestos and Mesothelioma; Testa, J.R., Ed.; Springer International Publishing: Philadelphia, PA, USA, 2017; pp. 43–72. [Google Scholar]
- Price, B. Projection of future numbers of mesothelioma cases in the US and the increasing prevalence of background cases: An update based on SEER data for 1975 through 2018. Crit. Rev. Toxicol. 2022, 52, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Wen, W.; Xu, D.; Piao, Y.; Li, X. Prognostic value of maximum standard uptake value, metabolic tumour volume, and total lesion glycolysis of 18F-FDG PET/CT in patients with malignant pleural mesothelioma: A systematic review and meta-analysis. Cancer Cell Int. 2022, 22, 60. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, R.; Gu, Y.; LiZhu, Y.; Liu, X.; Zhang, S. Clinical, Laboratory, Histological, Radiological, and Metabolic Features and Prognosis of Malignant Pleural Mesothelioma. Medicina 2022, 58, 1874. [Google Scholar] [CrossRef]
- Royal College of Physicians (RCP). National Mesothelioma Audit Report 2020 (Audit Period 2016–18); RCP: London, UK, 2020; Available online: www.rcplondon.ac.uk/projects/outputs/national-mesothelioma-audit-report-2020-audit-period-2016-18 (accessed on 2 May 2023).
- Popat, S.; Baas, P.; Faivre-Finn, C.; Girard, N.; Nicholson, A.G.; Nowak, A.K.; Opitz, I.; Scherpereel, A.; Reck, M.; ESMO Guidelines Committee. Malignant pleural mesothelioma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2022, 33, 129–142. [Google Scholar] [CrossRef]
- Shah, R.; Klotz, L.V.; Glade, J. Current Management and Future Perspective in Pleural Mesothelioma. Cancers 2022, 14, 1044. [Google Scholar] [CrossRef] [PubMed]
- Vogelzang, N.J.; Rusthoven, J.J.; Symanowski, J.; Denham, C.; Kaukel, E.; Ruffie, P.; Gatzemeier, U.; Boyer, M.; Emri, S.; Manegold, C.; et al. Phase III study of pemetrexed in combination with cisplatin versus cisplatin alone in patients with malignant pleural mesothelioma. J. Clin. Oncol. 2003, 21, 2636–2644. [Google Scholar] [CrossRef]
- Zalcman, G.; Mazieres, J.; Margery, J.; Greillier, L.; Audigier-Valette, C.; Moro-Sibilot, D.; Molinier, O.; Corre, R.; Monnet, I.; Gounant, V.; et al. Bevacizumab for newly diagnosed pleural mesothelioma in the Mesothelioma Avastin Cisplatin Pemetrexed Study (MAPS): A randomised, controlled, open-label, phase 3 trial. Lancet 2016, 387, 1405–1414. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, E.C.; Vellanki, P.J.; Larkins, E.; Chatterjee, S.; Mishra-Kalyani, P.S.; Bi, Y.; Qosa, H.; Liu, J.; Zhao, H.; Biable, M.; et al. FDA Approval Summary: Nivolumab in Combination with Ipilimumab for the Treatment of Unresectable Malignant Pleural Mesothelioma. Clin. Cancer Res. 2022, 28, 446–451. [Google Scholar] [CrossRef] [PubMed]
- Baas, P.; Scherpereel, A.; Nowak, A.K.; Fujimoto, N.; Peters, S.; Tsao, A.S.; Mansfield, A.S.; Popat, S.; Jahan, T.; Antonia, S.; et al. First-line nivolumab plus 15 ipilimumab in unresectable malignant pleural mesothelioma (CheckMate 743): A multicentre, 16 randomised, open-label, phase 3 trial. Lancet 2021, 397, 375–386.17. [Google Scholar] [CrossRef] [PubMed]
- Woolhouse, I.; Bishop, L.; Darlison, L.; De Fonseka, D.; Edey, A.; Edwards, J.; Faivre-Finn, C.; Fennell, D.A.; Holmes, S.; Kerr, K.M.; et al. British Thoracic Society Guideline for the investigation and management of malignant pleural mesothelioma. Thorax 2018, 73 (Suppl. S1), i1–i30. [Google Scholar] [CrossRef]
- Scherpereel, A.; Opitz, I.; Berghmans, T.; Psallidas, I.; Glatzer, M.; Rigau, D.; Astoul, P.; Bölükbas, S.; Boyd, J.; Coolen, J.; et al. ERS/ESTS/EACTS/ESTRO guidelines for the management of malignant pleural mesothelioma. Eur. Respir. J. 2020, 55, 1900953. [Google Scholar] [CrossRef]
- Pesch, B.; Brüning, T.; Johnen, G.; Casjens, S.; Bonberg, N.; Taeger, D.; Müller, A.; Weber, D.G.; Behrens, T. Biomarker research with prospective study designs for the early detection of cancer. Biochim. Biophys. Acta. 2014, 1844, 874–883. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.T.; Dong, X.L.; Zhang, Y.; Fang, P.; Shi, H.Y.; Ming, Z.J. Diagnostic value and safety of medical thoracoscopy for pleural effusion of different causes. World J. Clin. Cases. 2022, 10, 3088–3100. [Google Scholar] [CrossRef]
- Dixon, G.; de Fonseka, D.; Maskell, N. Pleural controversies: Image guided biopsy vs. thoracoscopy for undiagnosed pleural effusions? J. Thorac. Dis. 2015, 7, 1041–1051. [Google Scholar] [PubMed]
- Zhang, Y.; Li, N.; Li, R.; Gu, Y.; Liu, X.; Zhang, S. Predicting survival for patients with mesothelioma: Development of the PLACE prognostic model. BMC Cancer 2023, 23, 698. [Google Scholar] [CrossRef] [PubMed]
- Yeap, B.Y.; De Rienzo, A.; Gill, R.R.; Oster, M.E.; Dao, M.N.; Dao, N.T.; Levy, R.D.; Vermilya, K.; Gustafson, C.E.; Ovsak, G.; et al. Mesothelioma Risk Score: A New Prognostic Pretreatment, Clinical-Molecular Algorithm for Malignant Pleural Mesothelioma. J. Thorac. Oncol. 2021, 16, 1925–1935. [Google Scholar] [CrossRef]
- Ahmad, A.; Imran, M.; Ahsan, H. Biomarkers as Biomedical Bioindicators: Approaches and Techniques for the Detection, Analysis, and Validation of Novel Biomarkers of Diseases. Pharmaceutics 2023, 15, 1630. [Google Scholar] [CrossRef]
- Zhang, M.; Yan, L.; Lippi, G.; Hu, Z.D. Pleural biomarkers in diagnostics of malignant pleural effusion: A narrative review. Transl. Lung Cancer Res. 2021, 10, 1557–1570. [Google Scholar] [CrossRef] [PubMed]
- Schillebeeckx, E.; van Meerbeeck, J.P.; Lamote, K. Clinical utility of diagnostic biomarkers in malignant pleural mesothelioma: A systematic review and meta-analysis. Eur. Respir. Rev. 2021, 30, 210057. [Google Scholar] [CrossRef] [PubMed]
- Gao, R.; Wang, F.; Wang, Z.; Wu, Y.; Xu, L.; Qin, Y.; Shi, H.; Tong, Z. Diagnostic value of soluble mesothelin-related peptides in pleural effusion for malignant pleural mesothelioma: An updated meta-analysis. Medicine 2019, 98, e14979. [Google Scholar] [CrossRef] [PubMed]
- Cui, A.; Jin, X.G.; Zhai, K.; Tong, Z.H.; Shi, H.Z. Diagnostic values of soluble mesothelin-related peptides for malignant pleural mesothelioma: Updated meta-analysis. BMJ Open 2014, 4, e004145. [Google Scholar] [CrossRef]
- Pass, H.I.; Levin, S.M.; Harbut, M.R.; Melamed, J.; Chiriboga, L.; Donington, J.; Huflejt, M.; Carbone, M.; Chia, D.; Goodglick, L.; et al. Fibulin-3 as a blood and effusion biomarker for pleural mesothelioma. N. Engl. J. Med. 2012, 367, 1417–1427, Erratum in N. Engl. J. Med. 2012, 367, 1768. [Google Scholar] [CrossRef]
- Fujimoto, N.; Gemba, K.; Asano, M.; Fuchimoto, Y.; Wada, S.; Ono, K.; Ozaki, S.; Kishimoto, T. Hyaluronic acid in the pleural fluid of patients with malignant pleural mesothelioma. Respir. Investig. 2013, 51, 92–97. [Google Scholar] [CrossRef]
- Birnie, K.A.; Prêle, C.M.; Musk, A.W.B.; de Klerk, N.; Lee, Y.C.G.; Fitzgerald, D.; Allcock, R.J.N.; Thompson, P.J.; Creaney, J.; Badrian, B.; et al. MicroRNA signatures in malignant pleural mesothelioma effusions. Dis. Markers 2019, 2019, 8628612. [Google Scholar] [CrossRef]
- Filiberti, R.; Parodi, S.; Libener, R.; Ivaldi, G.P.; Canessa, P.A.; Ugolini, D.; Bobbio, B.; Marroni, P. Diagnostic value of mesothelin in pleural fluids: Comparison with CYFRA 21-1 and CEA. Med. Oncol. 2013, 30, 543. [Google Scholar] [CrossRef]
- Husain, A.N.; Colby, T.V.; Ordóñez, N.G.; Allen, T.C.; Attanoos, R.L.; Beasley, M.B.; Butnor, K.J.; Chirieac, L.R.; Churg, A.M.; Dacic, S.; et al. Guidelines for Pathologic Diagnosis of Malignant Mesothelioma 2017 Update of the Consensus Statement from the International Mesothelioma Interest Group. Arch. Pathol. Lab. Med. 2018, 142, 89–108. [Google Scholar] [CrossRef]
- Casjens, S.; Weber, D.G.; Johnen, G.; Raiko, I.; Taeger, D.; Meinig, C.; Moebus, S.; Jöckel, K.-H.; Brüning, T.; Pesch, B. Assessment of potential predictors of calretinin and mesothelin to improve the diagnostic performance to detect malignant mesothelioma: Results from a population-based cohort study. BMJ Open 2017, 7, e017104. [Google Scholar] [CrossRef]
- Creaney, J.; Sneddon, S.; Dick, I.M.; Dare, H.; Boudville, N.; Musk, A.W.; Skates, S.J.; Robinson, B.W. Comparison of the diagnostic accuracy of the MSLN gene products, mesothelin and megakaryocyte potentiating factor, as biomarkers for mesothelioma in pleural effusions and serum. Dis. Markers 2013, 35, 119–127. [Google Scholar] [CrossRef]
- Yu, Y.; Ryan, B.M.; Thomas, A.; Morrow, B.; Zhang, J.; Kang, Z.; Zingone, A.; Onda, M.; Hassan, R.; Pastan, I.; et al. Elevated Serum Megakaryocyte Potentiating Factor as a Predictor of Poor Survival in Patients with Mesothelioma and Primary Lung Cancer. J. Appl. Lab. Med. 2018, 3, 166–177. [Google Scholar] [CrossRef]
- Napolitano, A.; Antoine, D.J.; Pellegrini, L.; Baumann, F.; Pagano, I.; Pastorino, S.; Goparaju, C.M.; Prokrym, K.; Canino, C.; Pass, H.I.; et al. HMGB1 and Its Hyperacetylated Isoform are Sensitive and Specific Serum Biomarkers to Detect Asbestos Exposure and to Identify Mesothelioma Patients. Clin. Cancer Res. 2016, 22, 3087–3096. [Google Scholar] [CrossRef] [PubMed]
- Creaney, J.; Yeoman, D.; Demelker, Y.; Segal, A.; Musk, A.; Skates, S.J.; Robinson, B.W. Comparison of osteopontin, megakaryocyte potentiating factor, and mesothelin proteins as markers in the serum of patients with malignant mesothelioma. J. Thorac. Oncol. 2008, 3, 851–857. [Google Scholar] [CrossRef]
- Arnold, D.T.; De Fonseka, D.; Hamilton, F.W.; Rahman, N.M.; Maskell, N.A. Prognostication and monitoring of mesothelioma using biomarkers: A systematic review. Br. J. Cancer 2017, 116, 731–741. [Google Scholar] [CrossRef]
- Pass, H.I.; Alimi, M.; Carbone, M.; Yang, H.; Goparaju, C.M. Mesothelioma Biomarkers: A Review Highlighting Contributions from the Early Detection Research Network. Cancer Epidemiol. Biomark. Prev. 2020, 29, 2524–2540. [Google Scholar] [CrossRef]
- Hollevoet, K.; Nackaerts, K.; Gosselin, R.; De Wever, W.; Bosquée, L.; De Vuyst, P.; Germonpré, P.; Kellen, E.; Legrand, C.; Kishi, Y.; et al. Soluble mesothelin, megakaryocyte potentiating factor, and osteopontin as markers of patient response and outcome in mesothelioma. J. Thorac. Oncol. 2011, 6, 1930–1937. [Google Scholar] [CrossRef]
- Weber, D.G.; Gawrych, K.; Casjens, S.; Brik, A.; Lehnert, M.; Taeger, D.; Pesch, B.; Kollmeier, J.; Bauer, T.T.; Johnen, G.; et al. Circulating miR-132-3p as a Candidate Diagnostic Biomarker for Malignant Mesothelioma. Dis. Markers. 2017, 2017, 9280170. [Google Scholar] [CrossRef] [PubMed]
- Munson, P.B.; Hall, E.M.; Farina, N.H.; Pass, H.I.; Shukla, A. Exosomal miR-16-5p as a target for malignant mesothelioma. Sci. Rep. 2019, 9, 11688. [Google Scholar] [CrossRef]
- Cavalleri, T.; Angelici, L.; Favero, C.; Dioni, L.; Mensi, C.; Bareggi, C.; Palleschi, A.; Rimessi, A.; Consonni, D.; Bordini, L.; et al. Plasmatic extracellular vesicle microRNAs in malignant pleural mesothelioma and asbestos-exposed subjects suggest a 2-miRNA signature as potential biomarker of disease. PLoS ONE 2017, 12, e0176680. [Google Scholar] [CrossRef]
- Tomasetti, M.; Monaco, F.; Strogovets, O.; Volpini, L.; Valentino, M.; Amati, M.; Neuzil, J.; Santarelli, L. ATG5 as biomarker for early detection of malignant mesothelioma. BMC Res. Notes 2023, 16, 61. [Google Scholar] [CrossRef] [PubMed]
- Weber, D.G.; Casjens, S.; Brik, A.; Raiko, I.; Lehnert, M.; Taeger, D.; Gleichenhagen, J.; Kollmeier, J.; Bauer, T.T.; Brüning, T. Circulating long non-coding RNA GAS5 (growth arrest-specific transcript 5) as a complement marker for the detection of malignant mesothelioma using liquid biopsies. Biomark. Res. 2020, 8, 15. [Google Scholar] [CrossRef]
- Matboli, M.; Shafei, A.E.; Ali, M.A.; Gaber, A.I.; Galal, A.; Tarek, O.; Marei, M.; Khairy, E.; El-Khazragy, N.; Anber, N.; et al. Clinical significance of serum DRAM1 mRNA, ARSA mRNA, hsa-miR-2053 and lncRNA-RP1-86D1.3 axis expression in malignant pleural mesothelioma. J. Cell Biochem. 2019, 120, 3203–3211. [Google Scholar] [CrossRef] [PubMed]
- Ho, M.; Bera, T.K.; Willingham, M.C.; Onda, M.; Hassan, R.; FitzGerald, D.; Pastan, I. Mesothelin expression in human lung cancer. Clin. Cancer Res. 2007, 13, 1571–1575. [Google Scholar] [CrossRef] [PubMed]
- Lv, J.; Li, P. Mesothelin as a biomarker for targeted therapy. Biomark. Res. 2019, 7, 18. [Google Scholar] [CrossRef]
- Maeda, M.; Hino, O. Blood tests for asbestos-related mesothelioma. Oncology 2006, 71, 26–31. [Google Scholar] [CrossRef] [PubMed]
- Chang, K.; Pai, L.H.; Pass, H.; Pogrebniak, H.W.; Tsao, M.S.; Pastan, I.; Willingham, M.C. Monoclonal antibody K1 reacts with epithelial mesothelioma but not with lung adenocarcinoma. Am. J. Surg. Pathol. 1992, 16, 259–268. [Google Scholar] [CrossRef]
- Ordóñez, N.G. Value of mesothelin immunostaining in the diagnosis of mesothelioma. Mod. Pathol. 2003, 16, 192–197. [Google Scholar] [CrossRef] [PubMed]
- Ordóñez, N.G. What are the current best immunohistochemical markers for the diagnosis of epithelioid mesothelioma? A review and update. Hum. Pathol. 2007, 38, 1–16. [Google Scholar] [CrossRef]
- Kaya, H.; Demir, M.; Taylan, M.; Sezgi, C.; Tanrikulu, A.C.; Yilmaz, S.; Bayram, M.; Kaplan, I.; Senyigit, A. Fibulin-3 as a diagnostic biomarker in patients with malignant mesothelioma. Asian Pac. J. Cancer Prev. 2015, 16, 1403–1407. [Google Scholar] [CrossRef]
- Livingstone, I.; Uversky, V.N.; Furniss, D.; Wiberg, A. The Pathophysiological Significance of Fibulin-3. Biomolecules 2020, 10, 1294. [Google Scholar] [CrossRef]
- Battolla, E.; Canessa, P.A.; Ferro, P.; Franceschini, M.C.; Fontana, V.; Dessanti, P.; Pinelli, V.; Morabito, A.; Fedeli, F.; Pistillo, M.P.; et al. Comparison of the Diagnostic Performance of Fibulin-3 and Mesothelin in Patients with Pleural Effusions from Malignant Mesothelioma. Anticancer Res. 2017, 37, 1387–1391. [Google Scholar] [PubMed]
- Ledda, C.; Senia, P.; Rapisarda, V. Biomarkers for Early Diagnosis and Prognosis of Malignant Pleural Mesothelioma: The Quest Goes on. Cancers 2018, 10, 203. [Google Scholar] [CrossRef]
- Creaney, J.; Dick, I.M.; Meniawy, T.M.; Leong, S.L.; Leon, J.S.; Demelker, Y.; Segal, A.; Musk, A.W.; Lee, Y.C.G.; Skates, S.J.; et al. Comparison of fibulin-3 and mesothelin as markers in malignant mesothelioma. Thorax 2014, 69, 895–902. [Google Scholar] [CrossRef]
- Joy, R.A.; Vikkath, N.; Ariyannur, P.S. Metabolism and mechanisms of action of hyaluronan in human biology. Drug Metab. Pers. Ther. 2018, 33, 15–32. [Google Scholar] [CrossRef]
- Asplund, T.; Versnel, M.A.; Laurent, T.C.; Heldin, P. Human mesothelioma cells produce factors that stimulate the production of hyaluronan by mesothelial cells and fibroblasts. Cancer Res. 1993, 53, 388–392. [Google Scholar]
- Creaney, J.; Dick, I.M.; Segal, A.; Musk, A.W.; Robinson, B.W. Pleural effusion hyaluronic acid as a prognostic marker in pleural malignant mesothelioma. Lung Cancer 2013, 82, 491–498. [Google Scholar] [CrossRef] [PubMed]
- Smolarz, B.; Durczyński, A.; Romanowicz, H.; Szyłło, K.; Hogendorf, P. miRNAs in Cancer (Review of Literature). Int. J. Mol. Sci. 2022, 23, 2805. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Xie, W.; Xue, P.; Wei, Z.; Liang, X.; Chen, N. Diagnostic accuracy and prognostic applications of CYFRA 21-1 in head and neck cancer: A systematic review and meta-analysis. PLoS ONE 2019, 14, e0216561. [Google Scholar] [CrossRef] [PubMed]
- Hegmans, J.; Veltman, J.D.; Fung, E.T.; Verch, T.; Glover, C.; Zhang, F.; Allard, W.J.; T’Jampens, D.; Hoogsteden, H.C.; Lambrecht, B.N.; et al. Protein profiling of pleural effusions to identify malignant pleural mesothelioma using SELDI-TOF MS. Technol. Cancer Res. Treat. 2009, 8, 323–332. [Google Scholar] [CrossRef] [PubMed]
- Fuhrman, C.; Duche, J.C.; Chouaid, C.; Alsamad, I.A.; Atassi, K.; Monnet, I.; Tillement, J.; Housset, B. Use of tumor markers for differential diagnosis of mesothelioma and secondary pleural malignancies. Clin. Biochem. 2000, 33, 405–410. [Google Scholar] [CrossRef]
- Wang, X.F.; Wu, Y.H.; Wang, M.S.; Wang, Y.S. CEA, AFP, CA125, CA153 and CA199 in malignant pleural effusions predict the cause. Asian Pac. J. Cancer Prev. 2014, 15, 363–368. [Google Scholar] [CrossRef] [PubMed]
- Mundt, F.; Nilsonne, G.; Arslan, S.; Csürös, K.; Hillerdal, G.; Yildirim, H.; Metintas, M.; Dobra, K.; Hjerpe, A. Hyaluronan and N-ERC/mesothelin as key biomarkers in a specific two-step model to predict pleural malignant mesothelioma. PLoS ONE 2013, 8, e72030. [Google Scholar] [CrossRef]
- Palstrøm, N.B.; Overgaard, M.; Licht, P.; Beck, H.C. Identification of Highly Sensitive Pleural Effusion Protein Biomarkers for Malignant Pleural Mesothelioma by Affinity-Based Quantitative Proteomics. Cancers 2023, 15, 641. [Google Scholar] [CrossRef] [PubMed]
- Henderson, D.W.; Reid, G.; Kao, S.C.; van Zandwijk, N.; Klebe, S. Challenges and controversies in the diagnosis of mesothelioma: Part 1. Cytology-only diagnosis, biopsies, immunohistochemistry, discrimination between mesothelioma and reactive mesothelial hyperplasia, and biomarkers. J. Clin. Pathol. 2013, 66, 847–853. [Google Scholar] [CrossRef] [PubMed]
- Erber, R.; Warth, A.; Muley, T.; Hartmann, A.; Herpel, E.; Agaimy, A. BAP1 Loss is a Useful Adjunct to Distinguish Malignant Mesothelioma Including the Adenomatoid-like Variant From Benign Adenomatoid Tumors. Appl. Immunohistochem. Mol. Morphol. 2020, 28, 67–73. [Google Scholar] [CrossRef]
- Biancosino, C.; van der Linde, L.I.S.; Sauter, G.; Stellmacher, F.; Krüger, M.; Welker, L. Cytological Diagnostic Procedures in Malignant Mesothelioma. Adv. Exp. Med. Biol. 2022, 1374, 41–49. [Google Scholar]
- Straccia, P.; Magnini, D.; Trisolini, R.; Lococo, F.; Chiappetta, M.; Cancellieri, A. The value of cytology in distinguishing malignant mesothelioma: An institutional experience of 210 cases reclassified according to the International System for Reporting Serous Fluid Cytopathology (ISRSFC). Cytopathology 2022, 33, 77–83. [Google Scholar] [CrossRef]
- Wang, J.J.; Yan, L. Serum diagnostic markers for malignant pleural mesothelioma: A narrative review. Transl. Cancer Res. 2022, 11, 4434–4440. [Google Scholar] [CrossRef]
- Yeo, D.; Castelletti, L.; van Zandwijk, N.; Rasko, J.E.J. Hitting the Bull’s-Eye: Mesothelin’s Role as a Biomarker and Therapeutic Target for Malignant Pleural Mesothelioma. Cancers 2021, 13, 3932. [Google Scholar] [CrossRef]
- Hollevoet, K.; Reitsma, J.B.; Creaney, J.; Grigoriu, B.D.; Robinson, B.W.; Scherpereel, A.; Cristaudo, A.; Pass, H.I.; Nackaerts, K.; Portal, J.A.R.; et al. Serum mesothelin for diagnosing malignant pleural mesothelioma: An individual patient data meta-analysis. J. Clin. Oncol. 2012, 30, 1541–1549. [Google Scholar] [CrossRef]
- Hollevoet, K.; Nackaerts, K.; Thimpont, J.; Germonpré, P.; Bosquée, L.; De Vuyst, P.; Legrand, C.; Kellen, E.; Kishi, Y.; Delanghe, J.R.; et al. Diagnostic performance of soluble mesothelin and megakaryocyte potentiating factor in mesothelioma. Am. J. Respir. Crit. Care Med. 2010, 181, 620–625. [Google Scholar] [CrossRef]
- Tian, L.; Zeng, R.; Wang, X.; Shen, C.; Lai, Y.; Wang, M.; Che, G. Prognostic significance of soluble mesothelin in malignant pleural mesothelioma: A meta-analysis. Oncotarget 2017, 8, 46425–46435. [Google Scholar] [CrossRef] [PubMed]
- Goricar, K.; Kovac, V.; Dodic-Fikfak, M.; Dolzan, V.; Franko, A. Evaluation of soluble mesothelin-related peptides and MSLN genetic variability in asbestos-related diseases. Radiol. Oncol. 2020, 54, 86–95. [Google Scholar] [CrossRef]
- Fontana, V.; Pistillo, M.P.; Vigani, A.; Canessa, P.A.; Berisso, G.; Giannoni, U.; Ferro, P.; Franceschini, M.C.; Carosio, R.; Tonarelli, M.; et al. Determination of Mesothelin Levels in Pleural Effusion Does Not Help Predict Survival of Patients With Malignant Pleural Mesothelioma. Anticancer Res. 2019, 39, 5219–5223. [Google Scholar] [CrossRef]
- Forest, F.; Patoir, A.; Dal Col, P.; Sulaiman, A.; Camy, F.; Laville, D.; Bayle-Bleuez, S.; Fournel, P.; Habougit, C. Nuclear grading, BAP1, mesothelin and PD-L1 expression in malignant pleural mesothelioma: Prognostic implications. Pathology 2018, 50, 635–641. [Google Scholar] [CrossRef]
- Hollevoet, K.; Nackaerts, K.; Thas, O.; Thimpont, J.; Germonpré, P.; De Vuyst, P.; Bosquée, L.; Legrand, C.; Kellen, E.; Kishi, Y.; et al. The effect of clinical covariates on the diagnostic and prognostic value of soluble mesothelin and megakaryocyte potentiating factor. Chest 2012, 141, 477–484. [Google Scholar] [CrossRef]
- Katz, S.I.; Roshkovan, L.; Berger, I.; Friedberg, J.S.; Alley, E.W.; Simone, C.B., 2nd; Haas, A.R.; Cengel, K.A.; Sterman, D.H.; Albelda, S.M. Serum soluble mesothelin-related protein (SMRP) and fibulin-3 levels correlate with baseline malignant pleural mesothelioma (PM) tumor volumes but are not useful as biomarkers of response in an immunotherapy trial. Lung Cancer 2021, 154, 5–12. [Google Scholar] [CrossRef]
- Grigoriu, B.D.; Chahine, B.; Vachani, A.; Gey, T.; Conti, M.; Sterman, D.H.; Marchandise, G.; Porte, H.; Albelda, S.M.; Scherpereel, A. Kinetics of soluble mesothelin in patients with malignant pleural mesothelioma during treatment. Am. J. Respir. Crit. Care Med. 2009, 179, 950–954. [Google Scholar] [CrossRef] [PubMed]
- Creaney, J.; Francis, R.J.; Dick, I.M.; Musk, A.W.; Robinson, B.W.S.; Byrne, M.J.; Nowak, A.K. Serum soluble mesothelin concentrations in malignant pleural mesothelioma: Relationship to tumor volume, clinical stage and changes in tumor burden. Clin. Cancer Res. 2011, 17, 1181–1189. [Google Scholar] [CrossRef] [PubMed]
- Wheatley-Price, P.; Yang, B.; Patsios, D.; Patel, D.; Ma, C.; Xu, W.; Leighl, N.; Feld, R.; Cho, B.J.; O’Sullivan, B.; et al. Soluble mesothelin-related Peptide and osteopontin as markers of response in malignant mesothelioma. J. Clin. Oncol. 2010, 28, 3316–3322. [Google Scholar] [CrossRef] [PubMed]
- De Fonseka, D.; Arnold, D.T.; Stadon, L.; Morley, A.; Keenan, E.; Darby, M.; Armstrong, L.; Virgo, P.; Maskell, N.A. A prospective study to investigate the role of serial serum mesothelin in monitoring mesothelioma. BMC Cancer 2018, 18, 199. [Google Scholar] [CrossRef]
- Grosso, F.; Mannucci, M.; Ugo, F.; Ferro, P.; Cassinari, M.; Vigani, A.; De Angelis, A.M.; Delfanti, S.; Lia, M.; Guaschino, R.; et al. Pilot Study to Evaluate Serum Soluble Mesothelin-Related Peptide (SMRP) as Marker for Clinical Monitoring of Pleural Mesothelioma (PM): Correlation with Modified RECIST Score. Diagnostics 2021, 11, 2015. [Google Scholar] [CrossRef] [PubMed]
- Burt, B.M.; Lee, H.-S.; De Rosen, V.L.; Hamaji, M.; Groth, S.S.; Wheeler, T.M.; Sugarbaker, D.J. Soluble Mesothelin-Related Peptides to Monitor Recurrence after Resection of Pleural Mesothelioma. Ann. Thorac. Surg. 2017, 104, 1679–1687. [Google Scholar] [CrossRef] [PubMed]
- Roshini, A.; Goparaju, C.; Kundu, S.; Nandhu, M.S.; Longo, S.L.; Longo, J.A.; Chou, J.; Middleton, F.A.; Pass, H.I.; Viapiano, M.S. The extracellular matrix protein fibulin-3/EFEMP1 promotes pleural mesothelioma growth by activation of PI3K/Akt signaling. Front. Oncol. 2022, 12, 1014749. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Jiang, Z.; Yan, J.; Ying, S. HMGB1 as a Potential Biomarker and Therapeutic Target for Malignant Mesothelioma. Dis. Markers 2019, 2019, 4183157. [Google Scholar] [CrossRef] [PubMed]
- Tomasetti, M.; Staffolani, S.; Nocchi, L.; Neuzil, J.; Strafella, E.; Manzella, N.; Mariotti, L.; Bracci, M.; Valentino, M.; Amati, M.; et al. Clinical significance of circulating miR-126 quantification in malignant mesothelioma patients. Clin. Biochem. 2012, 45, 575–581. [Google Scholar] [CrossRef]
- Zhang, L.; Zhou, Q.; Han, Y.Q.; Li, P.; Ouyang, P.H.; Wang, M.Y.; Hu, Z.D. Diagnostic accuracy of circulating miR-126 for malignant pleural mesothelioma: A systematic review and meta-analysis. Transl. Cancer Res. 2021, 10, 1856–1862. [Google Scholar] [CrossRef]
- Reid, G.; Johnson, T.G.; van Zandwijk, N. Manipulating microRNAs for the Treatment of Malignant Pleural Mesothelioma: Past, Present and Future. Front. Oncol. 2020, 10, 105. [Google Scholar] [CrossRef]
- Birnie, K.A.; Prêle, C.M.; Thompson, P.J.; Badrian, B.; Mutsaers, S.E. Targeting microRNA to improve diagnostic and therapeutic approaches for malignant mesothelioma. Oncotarget 2017, 8, 78193–78207. [Google Scholar] [CrossRef] [PubMed]
- Creaney, J.; Yeoman, D.; Musk, A.W.; de Klerk, N.; Skates, S.J.; Robinson, B.W. Plasma versus serum levels of osteopontin and mesothelin in patients with malignant mesothelioma—Which is best? Lung Cancer. 2011, 74, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Grigoriu, B.D.; Scherpereel, A.; Devos, P.; Chahine, B.; Letourneux, M.; Lebailly, P.; Grégoire, M.; Porte, H.; Copin, M.-C.; Lassalle, P. Utility of osteopontin and serum mesothelin in malignant pleural mesothelioma diagnosis and prognosis assessment. Clin. Cancer Res. 2007, 13, 2928–2935. [Google Scholar] [CrossRef] [PubMed]
- Cristaudo, A.; Foddis, R.; Bonotti, A.; Simonini, S.; Vivaldi, A.; Guglielmi, G.; Ambrosino, N.; Canessa, P.A.; Chella, A.; Lucchi, M.; et al. Comparison between plasma and serum osteopontin levels: Usefulness in diagnosis of epithelial malignant pleural mesothelioma. Int. J. Biol. Markers 2010, 25, 164–170. [Google Scholar] [CrossRef]
- Hattori, T.; Iwasaki-Hozumi, H.; Bai, G.; Chagan-Yasutan, H.; Shete, A.; Telan, E.F.; Takahashi, A.; Ashino, Y.; Matsuba, T. Both Full-Length and Protease-Cleaved Products of Osteopontin Are Elevated in Infectious Diseases. Biomedicines 2021, 9, 1006. [Google Scholar] [CrossRef] [PubMed]
- Kirschner, M.B.; Pulford, E.; Hoda, M.A.; Rozsas, A.; Griggs, K.; Cheng, Y.Y.; Edelman, J.J.B.; Kao, S.C.; Hyland, R.; Dong, Y.; et al. Fibulin-3 levels in malignant pleural mesothelioma are associated with prognosis but not diagnosis. Br. J. Cancer 2015, 113, 963–969. [Google Scholar] [CrossRef] [PubMed]
- Tsim, S.; Alexander, L.; Kelly, C.; Shaw, A.; Hinsley, S.; Clark, S.; Evison, M.; Holme, J.; Cameron, E.J.; Sharma, D.; et al. Serum Proteomics and Plasma Fibulin-3 in Differentiation of Mesothelioma from Asbestos-Exposed Controls and Patients with Other Pleural Diseases. J. Thorac. Oncol. 2021, 16, 1705–1717. [Google Scholar] [CrossRef] [PubMed]
- Pei, D.; Li, Y.; Liu, X.; Yan, S.; Guo, X.; Xu, X.; Guo, X. Diagnostic and prognostic utilities of humoral fibulin-3 in malignant pleural mesothelioma: Evidence from a meta-analysis. Oncotarget 2017, 8, 13030–13038. [Google Scholar] [CrossRef]
- Raiko, I.; Sander, I.; Weber, D.G.; Raulf-Heimsoth, M.; Gillissen, A.; Kollmeier, J.; Scherpereel, A.; Brüning, T.; Johnen, G. Development of an enzyme-linked immunosorbent assay for the detection of human calretinin in plasma and serum of mesothelioma patients. BMC Cancer 2010, 10, 242. [Google Scholar] [CrossRef]
- Blum, W.; Pecze, L.; Felley-Bosco, E.; Schwaller, B. Overexpression or absence of calretinin in mouse primary mesothelial cells inversely affects proliferation and cell migration. Respir. Res. 2015, 16, 153. [Google Scholar] [CrossRef] [PubMed]
- Johnen, G.; Gawrych, K.; Raiko, I.; Casjens, S.; Pesch, B.; Weber, D.G.; Taeger, D.; Lehnert, M.; Kollmeier, J.; Bauer, T.; et al. Calretinin as a blood-based biomarker for mesothelioma. BMC Cancer 2017, 17, 386. [Google Scholar] [CrossRef]
- Johnen, G.; Burek, K.; Raiko, I.; Wichert, K.; Pesch, B.; Weber, D.G.; Lehnert, M.; Casjens, S.; Hagemeyer, O.; Taeger, D.; et al. Prediagnostic detection of mesothelioma by circulating calretinin and mesothelin—A case-control comparison nested into a prospective cohort of asbestos-exposed workers. Sci. Rep. 2018, 8, 14321. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.Q.; Xu, S.C.; Zheng, W.Q.; Hu, Z.D. Diagnostic value of microRNAs for malignant pleural mesothelioma: A mini-review. Thorac. Cancer 2021, 12, 8–12. [Google Scholar] [CrossRef]
- Santarelli, L.; Strafella, E.; Staffolani, S.; Amati, M.; Emanuelli, M.; Sartini, D.; Pozzi, V.; Carbonari, D.; Bracci, M.; Pignotti, E.; et al. Association of MiR-126 with soluble mesothelin-related peptides, a marker for malignant mesothelioma. PLoS ONE 2011, 6, e18232. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Pruett, N.; Pahwa, R.; Mahajan, A.P.; Schrump, D.S.; Hoang, C.D. MicroRNA-206 suppresses mesothelioma progression via the Ras signaling axis. Mol. Ther. Nucleic Acids 2021, 24, 669–681. [Google Scholar] [CrossRef] [PubMed]
- Raei, N.; Safaralizadeh, R.; Latifi-Navid, S. Clinical application of circulating tumor DNA in metastatic cancers. Expert Rev. Mol. Diagn. 2023, 1–12. [Google Scholar] [CrossRef]
- Hylebos, M.; de Beeck, K.O.; Pauwels, P.; Zwaenepoel, K.; van Meerbeeck, J.P.; Van Camp, G. Tumor-specific genetic variants can be detected in circulating cell-free DNA of malignant pleural mesothelioma patients. Lung Cancer 2018, 124, 19–22. [Google Scholar] [CrossRef] [PubMed]
- Christensen, B.C.; Godleski, J.J.; Marsit, C.J.; Houseman, E.A.; Lopez-Fagundo, C.Y.; Longacker, J.L.; Bueno, R.; Sugarbaker, D.J.; Nelson, H.H.; Kelsey, K.T. Asbestos exposure predicts cell cycle control gene promoter methylation in pleural mesothelioma. Carcinogenesis 2008, 29, 1555–1559. [Google Scholar] [CrossRef] [PubMed]
- Nocchi, L.; Tomasetti, M.; Amati, M.; Neuzil, J.; Santarelli, L.; Saccucci, F. Thrombomodulin is silenced in malignant mesothelioma by a poly(ADP-ribose) polymerase-1-mediated epigenetic mechanism. J. Biol. Chem. 2011, 286, 19478–19488. [Google Scholar] [CrossRef]
- Santarelli, L.; Staffolani, S.; Strafella, E.; Nocchi, L.; Manzella, N.; Grossi, P.; Bracci, M.; Pignotti, E.; Alleva, R.; Borghi, B.; et al. Combined circulating epigenetic markers to improve mesothelin performance in the diagnosis of malignant mesothelioma. Lung Cancer 2015, 90, 457–464. [Google Scholar] [CrossRef]
- Guarrera, S.; Viberti, C.; Cugliari, G.; Allione, A.; Casalone, E.; Betti, M.; Ferrante, D.; Aspesi, A.; Casadio, C.; Grosso, F.; et al. Peripheral Blood DNA Methylation as Potential Biomarker of Malignant Pleural Mesothelioma in Asbestos-Exposed Subjects. J. Thorac. Oncol. 2019, 14, 527–539. [Google Scholar] [CrossRef]
- Napolitano, A.; Antoine, D.J.; Pellegrini, L.; Baumann, F.; Pagano, I.; Pastorino, S.; Goparaju, C.M.; Prokrym, K.; Canino, C.; Pass, H.I.; et al. Expression of Concern: HMGB1 and Its Hyperacetylated Isoform are Sensitive and Specific Serum Biomarkers to Detect Asbestos Exposure and to Identify Mesothelioma Patients. Clin. Cancer Res. 2020, 26, 1529. [Google Scholar] [CrossRef]
- Ying, S.; Jiang, Z.; He, X.; Yu, M.; Chen, R.; Chen, J.; Ru, G.; Chen, Y.; Chen, W.; Zhu, L.; et al. Serum HMGB1 as a Potential Biomarker for Patients with Asbestos-Related Diseases. Dis. Markers 2017, 2017, 5756102. [Google Scholar] [CrossRef] [PubMed]
- Cunningham, G.M.; Roman, M.G.; Flores, L.C.; Hubbard, G.B.; Salmon, A.B.; Zhang, Y.; Gelfond, J.; Ikeno, Y. The paradoxical role of thioredoxin on oxidative stress and aging. Arch. Biochem. Biophys. 2015, 576, 32–38. [Google Scholar] [CrossRef] [PubMed]
- Demir, M.; Kaya, H.; Taylan, M.; Ekinci, A.; Yılmaz, S.; Teke, F.; Sezgi, C.; Tanrikulu, A.C.; Meteroglu, F.; Senyigit, A. Evaluation of New Biomarkers in the Prediction of Malignant Mesothelioma in Subjects with Environmental Asbestos Exposure. Lung 2016, 194, 409–417. [Google Scholar] [CrossRef]
- Hirayama, N.; Tabata, C.; Tabata, R.; Maeda, R.; Yasumitsu, A.; Yamada, S.; Kuribayashi, K.; Fukuoka, K.; Nakano, T. Pleural effusion VEGF levels as a prognostic factor of malignant pleural mesothelioma. Respir. Med. 2011, 105, 137–142. [Google Scholar] [CrossRef] [PubMed]
- Yasumitsu, A.; Tabata, C.; Tabata, R.; Hirayama, N.; Murakami, A.; Yamada, S.; Terada, T.; Iida, S.; Tamura, K.; Fukuoka, K.; et al. Clinical significance of serum vascular endothelial growth factor in malignant pleural mesothelioma. J. Thorac. Oncol. 2010, 5, 479–483. [Google Scholar] [CrossRef] [PubMed]
- Cristaudo, A.; Bonotti, A.; Guglielmi, G.; Fallahi, P.; Foddis, R. Serum mesothelin and other biomarkers: What have we learned in the last decade? J. Thorac. Dis. 2018, 10 (Suppl. S2), S353–S359. [Google Scholar] [CrossRef]
- Rai, A.J.; Flores, R.M. Association of malignant mesothelioma and asbestos related conditions with ovarian cancer: Shared biomarkers and a possible etiological link? Clin. Chem. Lab. Med. 2011, 49, 5–7. [Google Scholar] [CrossRef]
- Muley, T.; Dienemann, H.; Herth, F.J.; Thomas, M.; Meister, M.; Schneider, J. Combination of mesothelin and CEA significantly improves the differentiation between malignant pleural mesothelioma, benign asbestos disease, and lung cancer. J. Thorac. Oncol. 2013, 8, 947–951. [Google Scholar] [CrossRef]
- Corradi, M.; Goldoni, M.; Alinovi, R.; Tiseo, M.; Ampollini, L.; Bonini, S.; Carbognani, P.; Casalini, A.; Mutti, A. YKL-40 and mesothelin in the blood of patients with malignant mesothelioma, lung cancer and asbestosis. Anticancer Res. 2013, 33, 5517–5524. [Google Scholar]
- Bonotti, A.; Foddis, R.; Landi, S.; Melaiu, O.; De Santi, C.; Giusti, L.; Donadio, E.; Ciregia, F.; Mazzoni, M.R.; Lucacchini, A.; et al. A Novel Panel of Serum Biomarkers for PM Diagnosis. Dis. Markers 2017, 2017, 3510984. [Google Scholar] [CrossRef] [PubMed]
- Lu, Z.; Zhang, W.; Huang, K.; Zhu, M.; Gu, X.; Wei, D.; Shi, M.; Chen, Y.; Wang, H. Systematic Review, Meta-Analysis and Bioinformatic Analysis of Biomarkers for Prognosis of Malignant Pleural Mesothelioma. Diagnostics 2022, 12, 2210. [Google Scholar] [CrossRef] [PubMed]
- Lagniau, S.; Lamote, K.; van Meerbeeck, J.P.; Vermaelen, K.Y. Biomarkers for early diagnosis of malignant mesothelioma: Do we need another moonshot? Oncotarget 2017, 8, 53751–53762. [Google Scholar] [CrossRef] [PubMed][Green Version]
| Method | Advantages | Disadvantages |
|---|---|---|
| Antigen-antibody reaction (eg ELISA) | Fast to perform Inexpensive reagents and equipment Easy to perform with simple procedure Effective for soluble substances such as proteins, hormones, peptides, and antibodies | Risk of cross-reactivity High possibility of false positive/negative Wells to be read quickly due to short-term enzyme/substrate reaction Inadequate for microRNAs |
| Proteomics | Comprehensive analysis of proteins Identifies protein modifications Can uncover novel biomarkers Provides insights into functional changes | Technical expertise needed Expensive equipment Data analysis complexity Limited to known proteins |
| Genomics | Provides genetic information Identifies gene mutations Potential for early detection Can be used for risk assessment | Limited to DNA mutations May not capture all changes Complex data analysis Incomplete understanding |
| Transcriptomics | Captures gene expression levels Reveals potential therapeutic targets Helps to identify novel biomarkers Can distinguish subtypes | RNA instability Data analysis challenges Limited to RNA-based markers May not reflect true protein levels |
| Metabolomics | Provides insight into metabolic changes Identifies unique metabolic profiles Potential for early detection Can offer insights into treatment response | Metabolite variation Data interpretation Limited coverage of metabolites Technical challenges |
| Immunohistochemistry (IHC) | Validates biomarkers in tissue Useful for clinical applications Identifies protein expression Well-established technique | Limited to known targets Semi-quantitative information Inter-observer variability Dependent on tissue availability and quality |
| MicroRNA Profiling | Captures microRNA expression Useful for diagnostic and prognostic purposes Potential for early detection Minimally invasive samples are enough | Specific to microRNAs Data analysis complexity May require validation studies Limited to known microRNAs |
| Next-Generation Sequencing (NGS) | Comprehensive analysis of various data types Can integrate genomics, transcriptomics, and more Provides holistic view of molecular changes Identifies novel biomarkers | Data volume and complexity Requires cost and technical expertise Presents bioinformatics challenges Demands large sample sizes |
| Matrix | Biomarker | Cut-Off | AUCROC | Sensitivity | Specificity |
|---|---|---|---|---|---|
| Pleural | Mesothelin | 1.96–2.5 nM | 0.83 [25,26,27] | 44–100% | 46–100% |
| Fibulin-3 | 346.01–378.33 ng/mL | 0.68–0.97 [25,28] | 78.4–83.8% | 92.4–97.6% | |
| Hyaluronic acid | 100,000 ng/mL | 0.78–0.83 [25,29] | 44.0% | 96.5% | |
| Cell-free microRNAs | - | 0.92 (miR-143 + miR-210 + miR-200c) [30] | 92.3% | 96.1% | |
| CYFRA-21-1 vs. benign PE | 71.5 ng/mL | 0.76 [31] | 76% | 85% | |
| CYFRA-21-1 vs. other cancers | 100 ng/mL | 0.61 [31] | 81% | 47% | |
| CEA vs. benign PE | 5.5 ng/mL | 0.32 [31] | 30% | 98% | |
| CEA vs. other cancers | 3.8 ng/mL | 0.20 [31] | 98% | 56% | |
| Blood | Mesothelin vs. healthy | - | 0.87 [27] | 66% | 97% |
| Mesothelin vs. other cancers | - | 0.73 [27] | 60% | 81% | |
| Mesothelin vs. benign AE | - | 0.84 [27] | 58% | 89% | |
| Calretinin | 0.42 ng/mL | 0.86 [32] | 71% | 95% | |
| Calretinin + Mesothelin | 0.60–0.85 ng/mL; 2.32–2.91 nM | 0.83 [33] | 46–66% | 98–99% | |
| MPF | 12.38 ng/ml | 0.78 [25,34,35] | 68% | 95% | |
| Osteopontin vs. healthy | 48.3 ng/mL | 0.89 [24,36,37] | 65% | 81% | |
| Fibulin-3 | 52 ng/mL | 0.91 [25] | 62% | 82% | |
| miR-197-3p | - | 0.76 [38,39,40] | - | - | |
| miR-625-3p | 0.80 [41] | 73.3% | 78.6% | ||
| miR-20a | 0.98 [38,39,40] | - | - | ||
| miR-126It | 0.74 [42] | 71% | 69% | ||
| miR-132-3p + miR-126 | - | 0.76 [41] | 77% | 86% | |
| miR-103a-3p + miR-30e-3p | - | 0.94 [43] | 95.5% | 80% | |
| ATG5 | 23 ng/mL | 0.81 [44] | 43% | 98% | |
| GAS5 | - | 0.86 [45] | 64–73% | 97% | |
| ARSA mRNA.1 | - | 0.94 [46] | 90% | 93.7% | |
| DRAM mRNA | - | 0.82 [46] | 78.3% | 87.5% | |
| hsa-miR-2053 | - | 0.91 [46] | 85% | 97.5% | |
| LncRNA-RP1-86D1.3 | - | 0.88 [46] | 83.3% | 95% | |
| HMGB1 vs. healthy | 52.2 ng/mL | 0.88 [25] | 100% | 88.3% | |
| HMGB1 vs. asbestosis | 52.3 ng/mL | 0.56 [25] | 100% | 29.3% |
| Targeted Biomarker | Tested Treatment Options | Study Type |
|---|---|---|
| Mesothelin [73] | Chimeric monoclonal antibodies (Amatuximab) | Human, phase II |
| Antibody–drug conjugates Anetumab ravtansine (BAY94-9343) BMS-986148 BAY2287411 | Human, phase I, II phase II phase I | |
| Immunotoxins SS1P LMB-100 | Human, phase I phase I, II | |
| Cancer vaccines CRS-207 | Human, phase I, II | |
| Mesothelin-targeted cellular therapy CAR-T | Human phase I, II | |
| Fibulin-3 [88] | Anti-Fibulin-3 monoclonal antibody | Preclinical (animal) |
| HMBG1 [89] | Polypeptides Recombinant HMG Box-A Anti-HMGB1 neutralizing monoclonal antibody Chemical pharmaceuticals Ethyl pyruvate Aspirin and its metabolite, salicylic acid Plant extract Flaxseed lignans | Preclinical (animal) |
| microRNA [90,91,92,93] | TargomiRs (miR-16 mimic-drug delivery vehicle) miR-206 miR-215-5p miR-486-5p miR-16, mi-16-5p miR-126 miR-193a-3p | Human, phase 1 Preclinical (cell lines, animal) Preclinical (cell lines, animal) Preclinical (cell lines) Preclinical (animal) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sorino, C.; Mondoni, M.; Marchetti, G.; Agati, S.; Inchingolo, R.; Mei, F.; Flamini, S.; Lococo, F.; Feller-Kopman, D. Pleural Mesothelioma: Advances in Blood and Pleural Biomarkers. J. Clin. Med. 2023, 12, 7006. https://doi.org/10.3390/jcm12227006
Sorino C, Mondoni M, Marchetti G, Agati S, Inchingolo R, Mei F, Flamini S, Lococo F, Feller-Kopman D. Pleural Mesothelioma: Advances in Blood and Pleural Biomarkers. Journal of Clinical Medicine. 2023; 12(22):7006. https://doi.org/10.3390/jcm12227006
Chicago/Turabian StyleSorino, Claudio, Michele Mondoni, Giampietro Marchetti, Sergio Agati, Riccardo Inchingolo, Federico Mei, Sara Flamini, Filippo Lococo, and David Feller-Kopman. 2023. "Pleural Mesothelioma: Advances in Blood and Pleural Biomarkers" Journal of Clinical Medicine 12, no. 22: 7006. https://doi.org/10.3390/jcm12227006
APA StyleSorino, C., Mondoni, M., Marchetti, G., Agati, S., Inchingolo, R., Mei, F., Flamini, S., Lococo, F., & Feller-Kopman, D. (2023). Pleural Mesothelioma: Advances in Blood and Pleural Biomarkers. Journal of Clinical Medicine, 12(22), 7006. https://doi.org/10.3390/jcm12227006








