Cerebrolysin in Patients with Subarachnoid Hemorrhage: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Search Strategy
2.2. Data Abstraction
2.3. Statistical Analysis
2.4. Risk of Bias
2.5. The Quality of Studies
3. Results
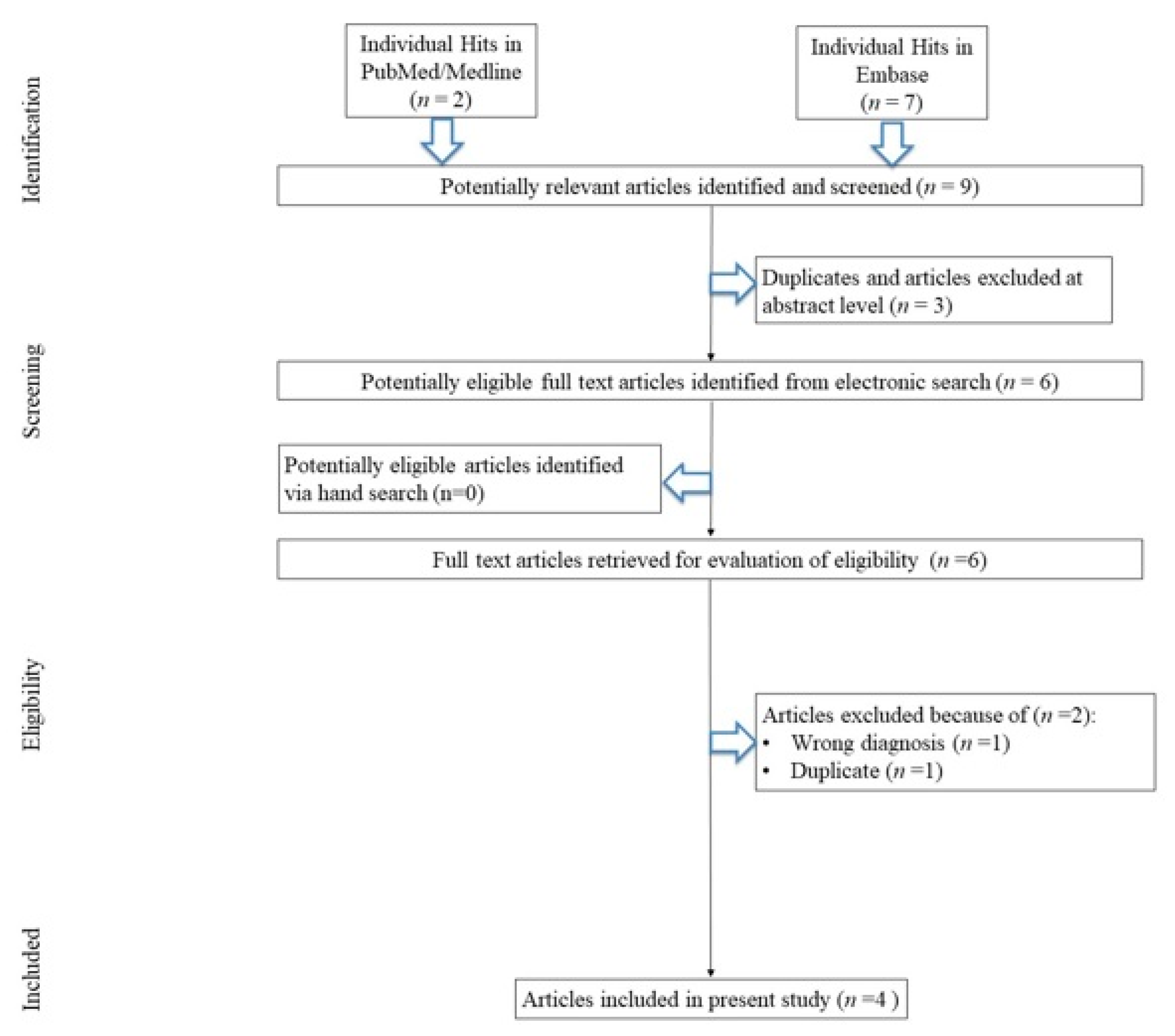
3.1. Search Results
3.2. Study, Patients, and Treatment Characteristics
3.3. Outcomes
3.4. Effect of Cerebrolysin on LOS
3.5. Effect of Cerebrolysin on Mortality
3.6. Limitations of the Study
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Correction Statement
References
- Macdonald, R.L.; Schweizer, T.A. Spontaneous subarachnoid haemorrhage. Lancet 2017, 389, 655–666. [Google Scholar] [CrossRef] [PubMed]
- Bosche, B.; Graf, R.; Ernestus, R.I.; Dohmen, C.; Reithmeier, T.; Brinker, G.; Strong, A.J.; Dreier, J.P.; Woitzik, J. Members of the Cooperative Study of Brain Injury Depolarizations (COSBID). Recurrent spreading depolarizations after subarachnoid hemorrhage decreases oxygen availability in human cerebral cortex. Ann. Neurol. 2010, 67, 607–617. [Google Scholar] [CrossRef] [PubMed]
- Nishimura, K.; Ogasawara, K.; Kitazono, T.; Iihara, K.; J-ASPECT Study Collaborators. Impact of Physician Volume and Specialty on In-Hospital Mortality of Ischemic and Hemorrhagic Stroke. Circ. J. 2021, 85, 1876–1884. [Google Scholar] [CrossRef] [PubMed]
- Korja, M.; Silventoinen, K.; McCarron, P.; Zdravkovic, S.; Skytthe, A.; Haapanen, A.; Faire, U.; Pedersen, N.L.; Christensen, K.; Koskenvuo, M. Genetic Epidemiology of Spontaneous Subarachnoid Hemorrhage. Stroke 2010, 41, 2458–2462. [Google Scholar] [CrossRef]
- Woo, P.Y.M.; Ho, J.W.K.; Ko, N.M.W.; Li, R.; Jian, L.; Chu, A.C.; Kwan, M.C.; Chan, Y.; Wong, A.K.; Wong, H.T.; et al. Randomized, placebo-controlled, double-blind, pilot trial to investigate safety and efficacy of Cerebrolysin in patients with aneurysmal subarachnoid hemorrhage. BMC Neurol. 2020, 20, 401. [Google Scholar] [CrossRef]
- Ingall, T.; Asplund, K.; Mähönen, M.; Bonita, R. A multinational comparison of subarachnoid hemorrhage epidemiology in the WHO MONICA stroke study. Stroke 2000, 31, 1054–1061. [Google Scholar] [CrossRef]
- Bonita, R.; Thomson, S. Subarachnoid hemorrhage: Epidemiology, diagnosis, management, and outcome. Stroke 1985, 16, 591–594. [Google Scholar] [CrossRef]
- Sarti, C.; Tuomilehto, J.; Salomaa, V.; Sivenius, J.; Kaarsalo, E.; Narva, E.V.; Salmi, K.; Torppa, J. Epidemiology of subarachnoid hemorrhage in Finland from 1983 to 1985. Stroke 1991, 22, 848–853. [Google Scholar] [CrossRef]
- Isselbacher, E.M.; Preventza, O.; Black, J.H.; Augoustides, J.G.; Beck, A.W.; Bolen, M.A.; Braverman, A.C.; Bray, B.E.; Brown-Zimmerman, M.M.; Chen, E.P. 2022 ACC/AHA guideline for the diagnosis and management of aortic disease: A report of the American Heart Association/American College of Cardiology Joint Committee on Clinical Practice Guidelines. J. Thorac. Cardiovasc. Surg. 2023, 3, e334–e482. [Google Scholar] [CrossRef]
- Song, J.P.; Ni, W.; Gu, Y.-X.; Zhu, W.; Chen, L.; Xu, B.; Leng, B.; Tian, Y.-L.; Mao, Y. Epidemiological Features of Nontraumatic Spontaneous Subarachnoid Hemorrhage in China: A Nationwide Hospital-based Multicenter Study. Chin. Med. J. 2017, 130, 776–781. [Google Scholar] [CrossRef]
- Feigin, V.L.; Rinkel, G.J.E.; Lawes, C.M.M.; Algra, A.; Bennett, D.A.; Gijn, J.; Anderson, G.S. Risk factors for subarachnoid hemorrhage: An updated systematic review of epidemiological studies. Stroke 2005, 36, 2773–2780. [Google Scholar] [CrossRef] [PubMed]
- Audibert, G.; Bousquet, S.; Charpentier, C.; Devaux, Y.; Mertes, P.M. Subarachnoid haemorrhage: Epidemiology, genomic, clinical presentation. Ann. Fr. Anesth. Reanim. 2007, 26, 943–947. [Google Scholar] [CrossRef] [PubMed]
- Dabus, G.; Nogueira, R.G. Current options for the management of aneurysmal subarachnoid hemorrhage-induced cerebral vasospasm: A comprehensive review of the literature. Interv. Neurol. 2013, 2, 30–51. [Google Scholar] [CrossRef]
- Muresanu, D.F.; Florian, S.; Hömberg, V.; Matula, C.; von Steinbüchel, N.; Vos, P.E.; von Wild, K.; Birle, C.; Muresanu, I.; Slavoaca, D.; et al. Efficacy and safety of cerebrolysin in neurorecovery after moderate-severe traumatic brain injury: Results from the CAPTAIN II trial. Neurol. Sci. 2020, 41, 1171–1181. [Google Scholar] [CrossRef] [PubMed]
- Tso, M.K.; Turgeon, P.; Bosche, B.; Lee, C.K.; Nie, T.; D’Abbondanza, J.; Ai, J.; Marsden, P.A.; Macdonald, R.L. Gene expression profiling of brain endothelial cells after experimental subarachnoid haemorrhage. Sci. Rep. 2021, 9, 7818. [Google Scholar] [CrossRef]
- Bosche, B.; Molcanyi, M.; Noll, T.; Rej, S.; Zatschler, B.; Doeppner, T.R.; Hescheler, J.; Müller, D.J.; Macdonald, R.L.; Härtel, F.V. A differential impact of lithium on endothelium-dependent but not on endothelium-independent vessel relaxation. Prog. Neuropsychopharmacol. Biol. Psychiatry 2016, 67, 98–106. [Google Scholar] [CrossRef] [PubMed]
- Haupt, M.; Zheng, X.; Kuang, Y.; Lieschke, S.; Janssen, L.; Bosche, B.; Jin, F.; Hein, K.; Kilic, E.; Venkataramani, V.; et al. Lithium modulates miR-1906 levels of mesenchymal stem cell-derived extracellular vesicles contributing to poststroke neuroprotection by toll-like receptor 4 regulation. Stem Cells Transl. Med. 2021, 10, 357–373. [Google Scholar] [CrossRef]
- Bentz, K.; Molcanyi, M.; Schneider, A.; Riess, P.; Maegele, M.; Bosche, B.; Hampl, J.A.; Hescheler, J.; Patz, S.; Schäfer, U. Extract derived from rat brains in the acute phase following traumatic brain injury impairs survival of undifferentiated stem cells and induces rapid differentiation of surviving cells. Cell Physiol. Biochem. 2010, 26, 821–830. [Google Scholar] [CrossRef] [PubMed]
- Neifert, S.N.; Chapman, E.K.; Martini, M.L.; Shuman, W.H.; Schupper, A.J.; Oermann, E.K.; Mocco, J.; Macdonald, R.L. Aneurysmal Subarachnoid Hemorrhage: The Last Decade. Transl. Stroke Res. 2021, 12, 428–446. [Google Scholar] [CrossRef] [PubMed]
- Rosenthal, E.S.; Biswal, S.; Zafar, S.F.; O’Connor, K.L.; Bechek, S.; Shenoy, A.V.; Boyle, E.J.; Shafi, M.M.; Gilmore, E.J.; Foreman, B.P.; et al. Continuous electroencephalography predicts delayed cerebral ischemia after subarachnoid hemorrhage: A prospective study of diagnostic accuracy. Ann. Neurol. 2018, 5, 958–969. [Google Scholar] [CrossRef]
- Adamczyk, P.; He, S.; Amar, A.P.; Mack, W.J. Medical Management of Cerebral Vasospasm following Aneurysmal Subarachnoid Hemorrhage: A Review of Current and Emerging Therapeutic Interventions. Neurol. Res. Int. 2013, 2013, 462491. [Google Scholar] [CrossRef] [PubMed]
- Jarosz, K.; Kojder, K.; Andrzejewska, A.; Solek-Pastuszka, J.; Jurczak, A. Cerebrolysin in Patients with TBI: Systematic Review and Meta-Analysis. Brain Sci. 2023, 13, 507. [Google Scholar] [CrossRef] [PubMed]
- Rejdak, K.; Sienkiewicz-Jarosz, H.; Bienkowski, P.; Alvarez, A. Modulation of neurotrophic factors in the treatment of dementia, stroke and TBI: Effects of Cerebrolysin. Med. Res. Rev. 2023, 43, 1668–1700. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Chopp, M.; Meier, D.H.; Winter, S.; Wang, L.; Szalad, A.; Lu, M.; Wei, M.; Cui, Y.; Zhang, Z.G. Sonic hedgehog signaling pathway mediates cerebrolysin-improved neurological function after stroke. Stroke 2013, 44, 1965–1972. [Google Scholar] [CrossRef]
- Heiss, W.D.; Brainin, M.; Bornstein, N.M.; Tuomilehto, J.; Hong, Z. Cerebrolysin Acute Stroke Treatment in Asia (CASTA) Investigators. Cerebrolysin in patients with acute ischemic stroke in Asia: Results of a double-blind, placebo-controlled randomized trial. Stroke 2012, 43, 630–636. [Google Scholar] [CrossRef]
- Strilciuc, S.; Vécsei, L.; Boering, D.; Pražnikar, A.; Kaut, O.; Riederer, P.; Battistin, L. Safety of Cerebrolysin for Neurorecovery after Acute Ischemic Stroke: A Systematic Review and Meta-Analysis of Twelve Randomized-Controlled Trials. Pharmaceuticals 2021, 14, 1297. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- DerSimonian, R.; Laird, N. Meta-analysis in clinical trials. Control Clin. Trials 1986, 7, 177–188. [Google Scholar] [CrossRef]
- Egger, M.; Smith, G.D.; Schneider, M.; Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997, 315, 629–634. [Google Scholar] [CrossRef]
- Park, Y.K.; Yi, H.J.; Choi, K.S.; Lee, Y.J.; Kim, D.W.; Kwon, S.M. Cerebrolysin for the Treatment of Aneurysmal Subarachnoid Hemorrhage in Adults: A Retrospective Chart Review. Adv. Ther. 2018, 35, 2224–2235. [Google Scholar] [CrossRef]
- Kojder, K.; Jarosz, K.; Andrzejewska, A. Evaluation of the effect of early administrated neuroprotective drug on treatment results in patients after traumatic brain injury (SAH)—PILOT trial. ESICM LIVES 2020. Intensive Care Med. Exp. 2020, 14, 73. [Google Scholar]
- Poljakovic, Z. Cerebrolysin in patients with subarachnoidal haemorrhage and high initial hunt-hess grade ESOC Abstract Book. Eur. Stroke J. 2022, 7, 3–545. [Google Scholar]
- Bornstein, N.M.; Guekht, A.; Vester, J.; Heiss, W.D.; Gusev, E.; Homberg, V.; Rahlfs, V.W.; Bajenaru, O.; Popescu, B.O.; Muresanu, D. Safety and efficacy of Cerebrolysin in early post-stroke recovery: A meta-analysis of nine randomized clinical trials. Neurol. Sci. 2018, 39, 629–640. [Google Scholar] [CrossRef] [PubMed]
- Sharm, H.S.; Zimmermann-Meinzingen, S.; Johanson, C.E. Cerebrolysin reduces blood-cerebrospinal fluid barrier permeability change, brain pathology, and functional deficits following traumatic brain injury in the rat. Ann. N. Y. Acad. Sci. 2010, 1199, 125–137. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Zheng, Z.V.; Lu, G.; Chan, W.Y.; Zhang, Y.; Wong, G.K.C. Microglia activation, classification and microglia-mediated neuroinflammatory modulators in subarachnoid hemorrhage. Neural. Regen. Res. 2022, 17, 1404–1411. [Google Scholar] [PubMed]
- Hasegawa, Y.; Suzuki, H.; Altay, O.; Chen, H.; Zhang, J.H. Treatment with sodium orthovanadate reduces blood-brain barrier disruption via phosphatase and tensin homolog deleted on chromosome 10 (PTEN) phosphorylation in experimental subarachnoid hemorrhage. J. Neurosci. Res. 2012, 90, 691–697. [Google Scholar] [CrossRef]
- Peng, J.; Pang, J.; Huang, L.; Enkhjargal, B.; Zhang, T.; Mo, J.; Wu, P.; Xu, W.; Zuo, Y.; Peng, J.; et al. LRP1 activation attenuates white matter injury by modulating microglial polarization through Shc1/PI3K/Akt pathway after subarachnoid hemorrhage in rats. Redox. Biol. 2019, 21, 101121. [Google Scholar] [CrossRef]
- Zhou, C.H.; Wang, C.X.; Xie, G.B.; Wu, L.Y.; Wei, Y.X.; Wang, Q.; Zhang, H.S.; Hang, C.H.; Zhou, M.L.; Shi, J.X. Fisetin alleviates early brain injury following experimental subarachnoid hemorrhage in rats possibly by suppressing TLR 4/NF-κB signaling pathway. Brain Res. 2015, 1629, 250–259. [Google Scholar] [CrossRef]
- Gao, Y.; Zhuang, Z.; Lu, Y.; Tao, T.; Zhou, Y.; Liu, G.; Wang, H.; Zhang, D.; Wu, L.; Dai, H.; et al. Curcumin mitigates neuro-inflammation by modulating microglia polarization through inhibiting TLR4 axis signaling pathway following experimental subarachnoid hemorrhage. Front. Neurosci. 2019, 13, 1223. [Google Scholar] [CrossRef]
- Liu, F.Y.; Cai, J.; Wang, C.; Ruan, W.; Guan, G.P.; Pan, H.Z.; Li, J.R.; Qian, C.; Chen, J.S.; Wang, L.; et al. Fluoxetine attenuates neuroinflammation in early brain injury after subarachnoid hemorrhage: A possible role for the regulation of TLR4/MyD88/NF-κB signaling pathway. J. Neuroinflamm. 2018, 15, 347. [Google Scholar] [CrossRef]
- Galea, J.; Ogungbenro, K.; Hulme, S.; Patel, H.; Scarth, S.; Hoadley, M.; Illingworth, K.; McMahon, C.J.; Tzerakis, N.; King, A.T.; et al. Reduction of inflammation after administration of interleukin-1 receptor antagonist following aneurysmal subarachnoid hemorrhage: Results of the Subcutaneous Interleukin-1Ra in SAH (SCIL-SAH) study. J. Neurosurg. 2018, 128, 515–523. [Google Scholar] [CrossRef] [PubMed]
- Formichi, P.; Radi, E.; Battisti, C.; Di Maio, G.; Muresanu, D.; Federico, A. Cerebrolysin administration reduces oxidative stress-induced apoptosis in lymphocytes from healthy individuals. J. Cell Mol. Med. 2012, 16, 2840–2843. [Google Scholar] [CrossRef] [PubMed]
- Hartbauer, M.; Hutter-Paier, B.; Skofitsch, G.; Windisch, M. Antiapoptotic effects of the peptidergic drug cerebrolysin on primary cultures of embryonic chick cortical neurons. J. Neural. Transm. 2001, 108, 459–473. [Google Scholar] [CrossRef]
- Lu, W.; Zhu, Z.; Shi, D.; Li, X.; Luo, J.; Liao, X. Cerebrolysin alleviates early brain injury after traumatic brain injury by inhibiting neuroinflammation and apoptosis via TLR signaling pathway. Acta Cir. Bras. 2022, 5, e370605. [Google Scholar] [CrossRef] [PubMed]
- Sarode, L.P.; Ghatage, T.; Mardhekar, V.; Verma, B.; Prakash, A.; Ugale, R.R. Cerebrolysin reduces excitotoxicity by modulation of cell-death proteins in delayed hours of ischemic reperfusion injury. Metab. Brain Dis. 2023, 38, 2401–2416. [Google Scholar] [CrossRef]
- Avci, S.; Gunaydin, S.; Ari, N.S.; Karaca Sulukoglu, E.; Polat, O.E.; Gecili, I.; Yeni, Y.; Yilmaz, A.; Genc, S.; Hacimuftuoglu, A.; et al. Cerebrolysin Alleviating Effect on Glutamate-Mediated Neuroinflammation Via Glutamate Transporters and Oxidative Stress. J. Mol. Neurosci. 2022, 72, 2292–2302. [Google Scholar] [CrossRef] [PubMed]
| Surgical Approach | HH 1,2 | HH 3,4,5 | F 1,2 | F 3,4 | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Reference | Craniectomy/Clipping (n) | Endovascular (n) | C− | C+ | C− | C+ | C− | C+ | C− | C+ |
| Park YK et al. 1 [30] | 328 | 134 | 151 | 65 | 177 | 69 | 36 | 17 | 292 | 117 |
| Park YK et al. 2 [30] | 148 | 68 | 151 | 65 | - | - | 29 | 11 | 122 | 54 |
| Park YK et al. 3 [30] | 180 | 66 | - | - | 177 | 69 | 7 | 6 | 170 | 63 |
| Woo PYM et al. [5] | 15 | 35 | ND | ND | ND | ND | 11 | 6 | 13 | 19 |
| Kojder K et al. [31] | 3 | 5 | 1 | 0 | 4 | 3 | 0 | 0 | 5 | 5 |
| Poljakovic et al. [32] | ND | ND | 0 | 10 | 0 | 0 | ND | ND | ND | ND |
| Study Characteristics | Intervention | Comparator | Sample Characteristics | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Reference | Country | Sponsorship | Blinding (Y/N) | Trial Duration (Days) | N Total Analyzed | Cerebrolysin Mean Dose/Day (mL) | PBO or Other Intervention | Age (Mean) | % Male |
| Park YK et al. 1 [30] | Republic of Korea | No | No | 10–20 | 462 | 30 | - | 55 | 34 |
| Park YK et al. 2 [30] | Republic of Korea | No | No | 9–17 | 216 | 30 | - | 54 | 34 |
| Park YK et al. 3 [30] | Republic of Korea | No | No | 12–21 | 246 | 30 | - | 56 | 34 |
| Woo PYM et al. [5] | Hong Kong | No | Yes | 10–21 | 50 | 30 | - | 53 | 28 |
| Kojder K et al. [31] | Poland | No | No | 6–21 | 8 | 50 | - | 61 | 40 |
| Poljakovic et al. [32] | Croatia | No | No | ND | 10 | ND | - | ND | ND |
| Variable | Length of Stay | |||||||
|---|---|---|---|---|---|---|---|---|
| Cerebrolysin | Control | |||||||
| Reference | Median | Range | SD | n | Median | Range | SD | n |
| Park et al., 2018 [30] | 18 | [15.0; 26.0] | ND | 134 | 22.0 | [17.0; 34.0] | ND | 328 |
| Park et al., 2018 [30] | 33.0 | [16.0; 70.0] | ND | 65 | 30.0 | [15.0; 67.0] | ND | 151 |
| Park et al., 2018 [30] | 23.0 | [16.0; 15.5] | ND | 69 | 27.0 | [17.0; 60.0] | ND | 177 |
| Woo et al., 2020 [5] | 33 | ND | 15 | 25 | 39 | ND | 32 | 25 |
| Kojder et al., 2020 [31] | 9 | 3–22 | ND | 5 | 13 | 3–28 | ND | 5 |
| Poljakovic, et al., 2022 [32] | ND | ND | ND | 10 | ND | ND | ND | ND |
| Reference | GCS C− [Mean] | GCS C+ [Mean] | Mortality C− | Mortality C+ |
|---|---|---|---|---|
| Park YK et al. 1 [30] | 13.0 | 14.0 | 57 (17.4%) | 12 (9%) |
| Park YK et al. 2 [30] | 15.0 | 15.0 | 12 (7.9%) | 6 (9.2%) |
| Park YK et al. 3 [30] | 11.0 | 8.0 | 45 (25.4%) | 6 (8.7%) |
| Woo PYM et al. [5] | ND | ND | 4 (16%) | 0 |
| Kojder K et al. [31] | 8.2 | 6 | 2 (40%) | 1 (20%) |
| Poljakovic et al. [32] | ND | ND | - | 3 (30%) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kojder, K.; Jarosz, K.; Bosiacki, M.; Andrzejewska, A.; Zacha, S.; Solek-Pastuszka, J.; Jurczak, A. Cerebrolysin in Patients with Subarachnoid Hemorrhage: A Systematic Review and Meta-Analysis. J. Clin. Med. 2023, 12, 6638. https://doi.org/10.3390/jcm12206638
Kojder K, Jarosz K, Bosiacki M, Andrzejewska A, Zacha S, Solek-Pastuszka J, Jurczak A. Cerebrolysin in Patients with Subarachnoid Hemorrhage: A Systematic Review and Meta-Analysis. Journal of Clinical Medicine. 2023; 12(20):6638. https://doi.org/10.3390/jcm12206638
Chicago/Turabian StyleKojder, Klaudyna, Konrad Jarosz, Mateusz Bosiacki, Agata Andrzejewska, Sławomir Zacha, Joanna Solek-Pastuszka, and Anna Jurczak. 2023. "Cerebrolysin in Patients with Subarachnoid Hemorrhage: A Systematic Review and Meta-Analysis" Journal of Clinical Medicine 12, no. 20: 6638. https://doi.org/10.3390/jcm12206638
APA StyleKojder, K., Jarosz, K., Bosiacki, M., Andrzejewska, A., Zacha, S., Solek-Pastuszka, J., & Jurczak, A. (2023). Cerebrolysin in Patients with Subarachnoid Hemorrhage: A Systematic Review and Meta-Analysis. Journal of Clinical Medicine, 12(20), 6638. https://doi.org/10.3390/jcm12206638







