Validation of a Brief Form of the Self-Administered Multidimensional Prognostic Index: The SELFY-BRIEF-MPI Project
Abstract
:1. Introduction
2. Materials and Methods
2.1. Participants
2.2. The Multidimensional Prognostic Index (Standard Version)
- Activities of Daily Living (ADL [21]): six items to assess the patient’s independence in the management of basic physical needs;
- Instrumental Activities of Daily Living (IADL [22]): eight items to measure one’s capacity for self-care and home maintenance;
- Short Portable Mental Status Questionnaire (SPMSQ [23]): ten items for the evaluation of cognitive status;
- Exton-Smith Scale (ESS [24]): five items to estimate the likelihood of developing pressure sores;
- Mini-Nutritional Assessment Short Form (MNA-SF [25]): six questions that investigate a person’s nutritional status, the presence of neuropsychological disorders, and exposure to acute stressors;
- Cumulative Illness Rating Scale (CIRS [26]): fourteen items to identify the existence and severity of concomitant conditions;
- Number of drugs consumed at home;
- Cohabitation status (alone, with family, or institutionalized): to indirectly acknowledge psychosocial information.
2.3. The SELFY-BRIEF-MPI
- Three dichotomous answers for the ADL test to evaluate independence in the most relevant areas of feeding, dressing, and continence.
- Three questions about the capacity to make calls, consume medicines, and conduct independent shopping were used to evaluate IADL.
- The Cognitive Change Index (CCI [28]), which uses three yes-or-no questions to assess cognitive status, was used to measure how people perceived their cognitive abilities, particularly their attention, language, and memory skills.
- The mobility domain measures an individual’s capacity to move in and out of a chair or bed, walk, and climb stairs. Each aptitude is classified as either yes or no.
- Two questions about changes in food consumption and weight along with an objective measure (BMI) are used to evaluate the nutritional status.
- The presence of comorbid conditions is assessed in this version through a single question about the number of diseases that require chronic therapies.
- The assessment of the number of medications was unchanged from the standard version.
- Cohabitation status is still tripartite: living alone, with family, or institutionalized, unchanged from the standard version.
2.4. Administration Procedure
2.5. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hoogendijk, E.O.; Afilalo, J.; Ensrud, K.E.; Kowal, P.; Onder, G.; Fried, L.P. Frailty: Implications for clinical practice and public health. Lancet 2019, 394, 1365–1375. [Google Scholar] [CrossRef] [PubMed]
- Clegg, A.; Young, J.; Iliffe, S.; Rikkert, M.O.; Rockwood, K. Frailty in elderly people. Lancet 2013, 381, 752–762. [Google Scholar] [CrossRef] [PubMed]
- Collard, R.M.; Boter, H.; Schoevers, R.A.; Oude Voshaar, R.C. Prevalence of Frailty in Community-Dwelling Older Persons: A Systematic Review. J. Am. Geriatr. Soc. 2012, 60, 1487–1492. [Google Scholar] [CrossRef] [PubMed]
- Gómez Jiménez, E.; Avendaño Céspedes, A.; Cortés Zamora, E.B.; García Molina, R.; Abizanda, P. [Frailty prevalence in hospitalized older adults. A systematic review]. Rev. Esp. Salud Publica 2021, 95, e202110158. [Google Scholar] [PubMed]
- Soysal, P.; Veronese, N.; Thompson, T.; Kahl, K.G.; Fernandes, B.S.; Prina, A.M.; Solmi, M.; Schofield, P.; Koyanagi, A.; Tseng, P.-T.; et al. Relationship between depression and frailty in older adults: A systematic review and meta-analysis. Ageing Res. Rev. 2017, 36, 78–87. [Google Scholar] [CrossRef] [PubMed]
- Kojima, G.; Iliffe, S.; Jivraj, S.; Walters, K. Association between frailty and quality of life among community-dwelling older people: A systematic review and meta-analysis. J. Epidemiol. Community Health 2016, 70, 716–721. [Google Scholar] [CrossRef] [PubMed]
- Parker, S.G.; McCue, P.; Phelps, K.; McCleod, A.; Arora, S.; Nockels, K.; Kennedy, S.; Roberts, H.; Conroy, S. What is Comprehensive Geriatric Assessment (CGA)? An umbrella review. Age Ageing 2018, 47, 149–155. [Google Scholar] [CrossRef] [PubMed]
- Stuck, A.E.; Siu, A.L.; Wieland, G.D.; Rubenstein, L.Z.; Adams, J. Comprehensive geriatric assessment: A meta-analysis of controlled trials. The Lancet 1993, 342, 1032–1036. [Google Scholar] [CrossRef]
- Ellis, G.; Langhorne, P. Comprehensive geriatric assessment for older hospital patients. Br. Med. Bull. 2005, 71, 45–59. [Google Scholar] [CrossRef]
- Rubenstein, L.Z.; Josephson, K.R.; Wieland, G.D.; English, P.A.; Sayre, J.A.; Kane, R.L. Effectiveness of a Geriatric Evaluation Unit: A Randomized Clinical Trial. N. Engl. J. Med. 1984, 311, 1664–1670. [Google Scholar] [CrossRef]
- Rubin, C.D.; Sizemore, M.T.; Loftis, P.A.; Adams-Huet, B.; Anderson, R.J. The Effect of Geriatric Evaluation and Management on Medicare Reimbursement in a Large Public Hospital: A Randomized Clinical Trial. J. Am. Geriatr. Soc. 1992, 40, 989–995. [Google Scholar] [CrossRef] [PubMed]
- Pilotto, A.; Ferrucci, L.; Franceschi, M.; D’Ambrosio, L.P.; Scarcelli, C.; Cascavilla, L.; Paris, F.; Placentino, G.; Seripa, D.; Dallapiccola, B.; et al. Development and Validation of a Multidimensional Prognostic Index for One-Year Mortality from Comprehensive Geriatric Assessment in Hospitalized Older Patients. Rejuvenation Res. 2008, 11, 151–161. [Google Scholar] [CrossRef] [PubMed]
- Pilotto, A.; Custodero, C.; Maggi, S.; Polidori, M.C.; Veronese, N.; Ferrucci, L. A multidimensional approach to frailty in older people. Ageing Res. Rev. 2020, 60, 101047. [Google Scholar] [CrossRef] [PubMed]
- Cella, A.; Veronese, N.; Custodero, C.; Castagna, A.; Cammalleri, L.A.; Capitano, W.M.; Solimando, L.; Carruba, L.; Sabbà, C.; Ruotolo, G.; et al. Validation of Abbreviated Form of the Multidimensional Prognostic Index (MPI): The BRIEF-MPI Project. Clin. Interv. Aging 2022, 17, 789–796. [Google Scholar] [CrossRef] [PubMed]
- Pilotto, A.; Veronese, N.; Quispe Guerrero, K.L.; Zora, S.; Boone, A.L.D.; Puntoni, M.; Giorgeschi, A.; Cella, A.; Rey Hidalgo, I.; Pers, Y.-M.; et al. Development and Validation of a Self-Administered Multidimensional Prognostic Index to Predict Negative Health Outcomes in Community-Dwelling Persons. Rejuvenation Res. 2019, 22, 299–305. [Google Scholar] [CrossRef] [PubMed]
- Barreto, P.D.S.; Greig, C.; Ferrandez, A.-M. Detecting and categorizing frailty status in older adults using a self-report screening instrument. Arch. Gerontol. Geriatr. 2012, 54, e249–e254. [Google Scholar] [CrossRef] [PubMed]
- Nunes, D.P.; Duarte, Y.A.D.O.; Santos, J.L.F.; Lebrão, M.L. Screening for frailty in older adults using a self-reported instrument. Rev. Saúde Pública 2015, 49, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Singh-Manoux, A. What does self rated health measure? Results from the British Whitehall II and French Gazel cohort studies. J. Epidemiol. Community Health 2006, 60, 364–372. [Google Scholar] [CrossRef]
- Quesnel Vallee, A. Self-rated health: Caught in the crossfire of the quest for “true” health? Int. J. Epidemiol. 2007, 36, 1161–1164. [Google Scholar] [CrossRef]
- Wilkinson, R.; Marmot, M.; World Health Organization R. O. for Europe. Social Determinants of Health: The Solid Facts, 2nd ed.; World Health Organization Regional Office for Europe: Copenhagen, Denmark, 2003; Available online: https://apps.who.int/iris/handle/10665/326568 (accessed on 4 August 2023).
- Katz, S.; Downs, T.D.; Cash, H.R.; Grotz, R.C. Progress in Development of the Index of ADL. Gerontologist 1970, 10, 20–30. [Google Scholar] [CrossRef]
- Lawton, M.P.; Brody, E.M. Assessment of Older People: Self-Maintaining and Instrumental Activities of Daily Living. Gerontologist 1969, 9, 179–186. [Google Scholar] [CrossRef] [PubMed]
- Pfeiffer, E. A Short Portable Mental Status Questionnaire for the Assessment of Organic Brain Deficit in Elderly Patients. J. Am. Geriatr. Soc. 1975, 23, 433–441. [Google Scholar] [CrossRef] [PubMed]
- Bliss, M.R.; McLaren, R.; Exton-Smith, A.N. Mattresses for preventing pressure sores in geriatric patients. Mon. Bull. Minist. Health Public Health Lab. Serv. 1966, 25, 238–268. [Google Scholar] [PubMed]
- MNA-International Group; Kaiser, M.J.; Bauer, J.M.; Ramsch, C.; Uter, W.; Guigoz, Y.; Cederholm, T.; Thomas, D.R.; Anthony, P.; Charlton, K.E.; et al. Validation of the Mini Nutritional Assessment short-form (MNA®-SF): A practical tool for identification of nutritional status. J. Nutr. Health Aging 2009, 13, 782–788. [Google Scholar] [CrossRef] [PubMed]
- Linn, B.S.; Linn, M.W.; Gurel, L. Cumulative illness rating scale. J. Am. Geriatr. Soc. 1968, 16, 622–626. [Google Scholar] [CrossRef] [PubMed]
- Bonnekoh, S.I.; Meyer, A.M.; Pickert, L.; Schulz, R.-J.; Becker, I.; Polidori, M.C. The multidimensional prognostic index in hospitalized older adults: Practicability with regard to time needs. Aging Clin. Exp. Res. 2023, 35, 711–716. [Google Scholar] [CrossRef] [PubMed]
- Rattanabannakit, C.; Risacher, S.L.; Gao, S.; Lane, K.A.; Brown, S.A.; McDonald, B.C.; Unverzagt, F.W.; Apostolova, L.G.; Saykin, A.J.; Farlow, M.R. The Cognitive Change Index as a Measure of Self and Informant Perception of Cognitive Decline: Relation to Neuropsychological Tests. J. Alzheimers Dis. 2016, 51, 1145–1155. [Google Scholar] [CrossRef]
- Jessen, F. Prediction of Dementia by Subjective Memory Impairment Effects of Severity and Temporal Association With Cognitive Impairment Dementia and Subjective Memory Impairment. Arch. Gen. Psychiatry 2010, 67, 414. [Google Scholar] [CrossRef]
- Bland, J.M.; Altman, D.G. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet 1986, 1, 307–310. [Google Scholar] [CrossRef]
- Bland, J.M.; Altman, D.G. Measuring agreement in method comparison studies. Stat. Methods Med. Res. 1999, 8, 135–160. [Google Scholar] [CrossRef]
- Veronese, N.; Custodero, C.; Cella, A.; Demurtas, J.; Zora, S.; Maggi, S.; Barbagallo, M.; Sabbà, C.; Ferrucci, L.; Pilotto, A. Prevalence of multidimensional frailty and pre-frailty in older people in different settings: A systematic review and meta-analysis. Ageing Res. Rev. 2021, 72, 101498. [Google Scholar] [CrossRef]
- Youden, W.J. Index for rating diagnostic tests. Cancer 1950, 3, 32–35. [Google Scholar] [CrossRef]
- Yourman, L.C.; Lee, S.J.; Schonberg, M.A.; Widera, E.W.; Smith, A.K. Prognostic Indices for Older Adults: A Systematic Review. JAMA 2012, 307, 182. [Google Scholar] [CrossRef]
- Wleklik, M.; Uchmanowicz, I.; Jankowska, E.A.; Vitale, C.; Lisiak, M.; Drozd, M.; Pobrotyn, P.; Tkaczyszyn, M.; Lee, C. Multidimensional Approach to Frailty. Front. Psychol. 2020, 11, 564. [Google Scholar] [CrossRef]

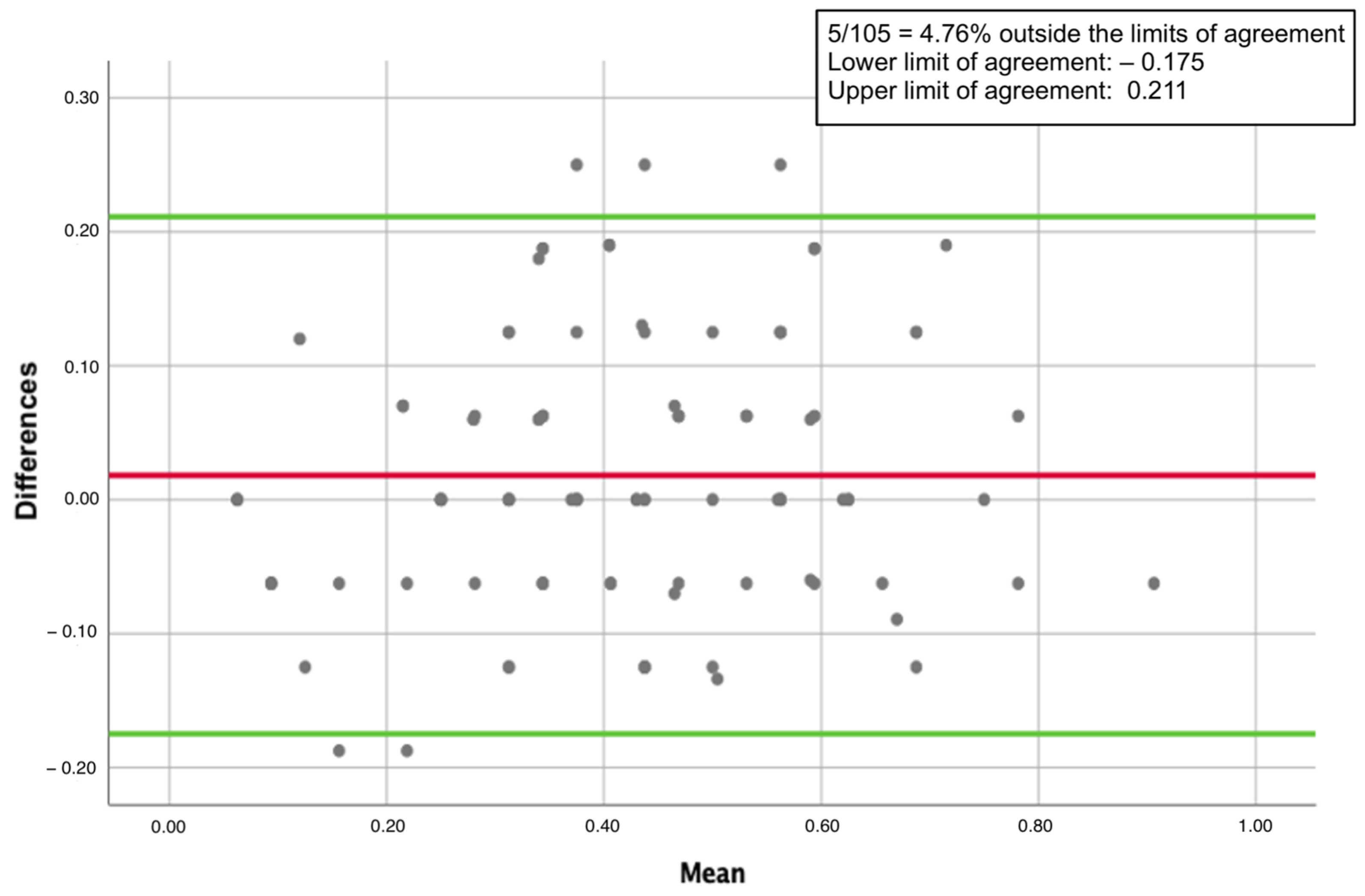

| Parameter | Mean | Standard Deviation | Range |
|---|---|---|---|
| Age | 78.75 | 7.03 | 65–99 |
| Standard-MPI | 0.42 | 0.19 | 0.06–0.88 |
| ADL | 3.70 | 1.87 | 0–6 |
| IADL | 3.91 | 2.57 | 0–8 |
| SPMSQ | 2.17 | 2.27 | 0–10 |
| ESS | 12.36 | 6.36 | 0–20 |
| MNA-SF | 7.38 | 4.63 | 0–14 |
| CIRS | 4.49 | 2.62 | 0–13 |
| N. OF DRUGS | 6.46 | 3.34 | 1–14 |
| Cohabitation status: Living alone 27 (25.7%) | |||
Cause of hospitalization/visiting outpatients’ clinics:
| |||
| MPI’s Domains | Standard-MPI | SELFY-BRIEF-MPI | p-Value | |
|---|---|---|---|---|
| MPI index (n = 105) | 0.42 (0.19) | 0.41 (0.18) | 0.104 | |
| ADL (n = 105) | Low (n, %) | 62 (59%) | 58 (55.2%) | 1.000 |
| Medium (n, %) | 19 (18.1%) | 27 (25.7%) | ||
| High (n, %) | 24 (22.9%) | 20 (19%) | ||
| IADL (n = 105) | Low (n, %) | 49 (46.7%) | 50 (47.6%) | 0.007 |
| Medium (n, %) | 16 (15.2%) | 29 (27.6%) | ||
| High (n, %) | 40 (38.1%) | 26 (24.8%) | ||
| MNA-SF (n = 105) | Low (n, %) | 29 (27.6%) | 66 (62.9%) | <0.001 |
| Medium (n, %) | 48 (45.7%) | 34 (32.4%) | ||
| High (n, %) | 28 (26.7%) | 5 (4.8%) | ||
| CIRS-CI (Standard-MPI n = 104; SELFY-BRIEF-MPI n = 105) | Low (n, %) | 5 (4.8%) | 0 (0%) | 0.279 |
| Medium (n, %) | 16 (15.4%) | 32 (30.5%) | ||
| High (n, %) | 83 (79.8%) | 73 (69.5%) | ||
| ESS-MOB (n = 105) | Low (n, %) | 62 (59%) | 79 (75.2%) | 0.029 |
| Medium (n, %) | 34 (32.4%) | 16 (15.2%) | ||
| High (n, %) | 9 (8.6%) | 10 (9.5%) | ||
| SPMSQ (n = 105) CCI (n = 104) | Low (n, %) | 74 (70.5%) | 54 (51.9%) | <0.001 |
| Medium (n, %) | 23 (21.9%) | 24 (23.1%) | ||
| High (n, %) | 8 (7.6%) | 26 (25%) | ||
| Cohabitation status (n = 105) | Low (n, %) | 76 (72.4%) | 76 (72.4%) | 1.000 |
| Medium (n, %) | 2 (1.9%) | 2 (1.9%) | ||
| High (n, %) | 27 (25.7%) | 27 (25.7%) | ||
| Number of drugs (n = 104) | Low (n, %) | 23 (22.1%) | 23 (22.1%) | 1.000 |
| Medium (n, %) | 27 (26%) | 27 (26%) | ||
| High (n, %) | 54 (51.9%) | 54 (51.9%) | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Morganti, W.; Veronese, N.; Barbagelata, M.; Castagna, A.; Custodero, C.; Solimando, L.; Burgio, M.I.; Montana Lampo, S.E.; Seminerio, E.; Puleo, G.; et al. Validation of a Brief Form of the Self-Administered Multidimensional Prognostic Index: The SELFY-BRIEF-MPI Project. J. Clin. Med. 2023, 12, 6026. https://doi.org/10.3390/jcm12186026
Morganti W, Veronese N, Barbagelata M, Castagna A, Custodero C, Solimando L, Burgio MI, Montana Lampo SE, Seminerio E, Puleo G, et al. Validation of a Brief Form of the Self-Administered Multidimensional Prognostic Index: The SELFY-BRIEF-MPI Project. Journal of Clinical Medicine. 2023; 12(18):6026. https://doi.org/10.3390/jcm12186026
Chicago/Turabian StyleMorganti, Wanda, Nicola Veronese, Marina Barbagelata, Alberto Castagna, Carlo Custodero, Luisa Solimando, Marianna Ilarj Burgio, Sofia Elena Montana Lampo, Emanuele Seminerio, Gianluca Puleo, and et al. 2023. "Validation of a Brief Form of the Self-Administered Multidimensional Prognostic Index: The SELFY-BRIEF-MPI Project" Journal of Clinical Medicine 12, no. 18: 6026. https://doi.org/10.3390/jcm12186026
APA StyleMorganti, W., Veronese, N., Barbagelata, M., Castagna, A., Custodero, C., Solimando, L., Burgio, M. I., Montana Lampo, S. E., Seminerio, E., Puleo, G., Senesi, B., Cammalleri, L., Ruotolo, G., Sabbà, C., Barbagallo, M., & Pilotto, A. (2023). Validation of a Brief Form of the Self-Administered Multidimensional Prognostic Index: The SELFY-BRIEF-MPI Project. Journal of Clinical Medicine, 12(18), 6026. https://doi.org/10.3390/jcm12186026







