1. Introduction
Diffuse midline glioma (DMG) is a type of malignant tumor derived from glial cells, primarily occurring in children. It is characterized by a diffuse infiltrative growth of tissue affecting the brain stem, which often renders surgical resection unfeasible [
1]. Brain stem tumors are relatively prevalent among children, accounting for approximately one in five cases of nervous system tumors in this age group. Of these, approximately four out of five are DMGs. The clinical symptoms in this type of tumor depend to a large extent on the brain areas affected producing sensory or motor syndromes such as facial paralysis, balance disorders, visual disturbances, dysarthria, dysphagia, and dysphagia, among others [
2,
3,
4].
From a histological perspective, it is a highly heterogeneous disease with a wide range of histological variations. These tumors exhibit significant diffusion and frequently invade neighboring brain structures beyond the pons. Numerous studies have reported that leptomeningeal dissemination and subventricular spread are common findings in up to one-third of DIPG cases. In fact, tumor cells have been identified as far rostrally as the frontal lobe, highlighting the extensive nature of the disease [
5]. Like other brain stem tumors, the degree of malignancy is determined by the differentiation level of the tumor cells, with low-grade tumors exhibiting higher differentiation and high-grade tumors characterized by poorly differentiated glial cells and significant anaplasia [
5,
6].
Previously and still commonly referred to as diffuse intrinsic pontine glioma (DIPG), this tumor was formerly recognized as a grade IV brain stem glioma and has recently been renamed according to the World Health Organization (WHO) CNS5 nomenclature as “diffuse midline glioma” [
6].
Indeed, it is essential to emphasize the significant distinction in diagnosing DMG and DIPG. The publication of WHO CNS5 in 2021 has resulted in a noteworthy change. WHO grade IV tumors located in the thalami, brainstem, and spinal cord of both pediatric and young adult populations are now reclassified as DMG, particularly if they bear the H3-K27 mutation. Recent advancements in tissue acquisition and molecular profiling have revealed that DMG and GBM (glioblastoma multiforme) are distinct disease entities with separate tissue characteristics and genetic profiles. Additional profiling has shed light on the origin of the disease and the influence of various mutations, such as the highly recurring K27M mutation in histone H3, on its tumorigenesis. Furthermore, early evidence suggests that DMG has a unique immune microenvironment, characterized by low levels of immune cell infiltration, inflammation, and immunosuppression, which may have implications for the disease development and outcomes [
7,
8].
The reclassification of tumors into the DMG category not only impacts the patient demographic but also broadens the disease’s classification to encompass not only pediatric cases but also those affecting young adults [
9].
In the past decade, the primary method for diagnosing DMG has been magnetic resonance imaging (MRI) [
1,
2,
3,
4,
10]. The tumor grows within the brain stem, responsible for vital functions like breathing, heart rate, and blood pressure. Therefore, initially, the diagnosis relied solely on MRI since the distinct imaging characteristics of DMG provided definitive diagnosis. However, in recent years, biopsies have become increasingly important for histological confirmation and, more significantly, for molecular biology studies.
The combination of a high mortality rate and a lack of effective therapeutic options renders DMG one of the most challenging tumors in the field of pediatric neuro-oncology. By the early 2010s, the estimated survival rate for DMG treatment stood at a mere 10% two years after diagnosis [
5,
11].
In terms of effectiveness, the available therapies used for this type of tumor at the beginning of the previous decade were not effective enough in preventing tumor relapse and achieving long-term survival [
2,
3,
4]. Achieving satisfactory treatment outcomes for DMG poses significant challenges, largely due to its location and high likelihood of developing refractory disease. In addition, its infiltrative and diffuse patterns further complicate surgical intervention, making resection impossible and diminishing the efficacy of radiotherapy [
3,
4,
5,
6]. Due to these challenges, stereotactic radiotherapy has become a common approach for treating DMG. However, its effectiveness is often limited due to the infiltrative nature of the tumor and the challenge of targeting all malignant cells [
3].
Despite being classified as a rare tumor, DMG accounts for approximately 10–15% of all brain tumor deaths in children, representing one of the most important causes of tumor-related deaths in this age group [
1,
11,
12], so is particularly relevant today due to the high mortality rate and the lack of effective therapeutic options. DMG has emerged as a compelling subject of scientific investigation, particularly in the realm of clinical studies and trials aimed at assessing novel therapeutic approaches such as immunotherapy, T-cell therapy, and targeted therapies. However, substantial further research is still required to improve the prognosis of patients with DMG [
13,
14].
The prognosis for DMG has remained discouraging for over five decades and biological research has been hindered by the scarcity of pre-treatment tissue samples, as biopsies were typically reserved for atypical cases [
2,
3,
4,
15]. However, recent advances in surgical techniques and the possibility of performing molecular analysis have improved the safety and potential utility of biopsies. Brainstem biopsies have now been incorporated into several prospective clinical trials [
16]. These and other recent efforts have yielded new insights into the molecular pathogenesis of DMG and have opened up new avenues for research.
Ongoing clinical studies are currently investigating promising therapeutic approaches (
Supplementary Table S1), such as T-cell therapy, immunotherapy, and targeted therapies [
11,
16]. Consequently, this comprehensive review of the existing literature on DMG aims to gather reliable information that can contribute to improving the prognosis of DMG patients in the long term. Despite the existence of other reviews that specifically address DIPG/DMG [
17,
18,
19,
20] (
Supplementary Table S3), this review of DMG is necessary to provide updated insights, as this field has been rapidly evolving in recent years and the recent advances can potentially improve patients’ survival.
A comprehensive review study is proposed to address key aspects related to this tumor such as its definition, epidemiological aspects, diagnostic methods, classifications, and treatment options. By examining these fundamental aspects, the review aims to contribute to the existing body of knowledge and provide valuable insights into DMG. Such research will be valuable to clinicians, researchers, and other stakeholders in the field, ultimately driving progress in DMG management and patient outcomes.
2. Methods and Search Strategy
In order to achieve the proposed objective, a systematic literature review of relevant studies published between 1 January 2012 and 31 July 2023 was conducted following the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) statement. This bibliographical search was conducted using the PubMed, Cochrane Library, and SciELO (through the LILACS) databases.
The search terms used were: “Diffuse intrinsic pontine glioma”, “DIPG”, “Diffuse midline glioma”, “DMG”, “epidemiology”, “diagnosis”, “molecular biology”, “SPECT”, “biopsy”, “stereotactic biopsy”, “classification”, “radiotherapy”, “radiation therapy”, “proton therapy”, “chemotherapy”, “histone”, “H3”, “H3K27”, “M3.H3K27”, “New therapies”, “peptide vaccines”, and “CNS WHO”.
To structure the search in the different engines, we used the Boolean operator “AND” to connect two different terms and “OR” was employed to represent an alternative between one term and another. Inclusion criteria included addressing any of the following topics: diagnosis, treatment of DMG, epidemiology, molecular classification, or WHO tumor classification. Studies were included in English or Spanish, available for full reading online. Three authors independently screened the titles and abstracts of articles identified in the search and selected those that met the inclusion criteria. Any disagreement between authors was resolved through consensus.
The final inclusion of articles was determined following an assessment of their evidence quality using Sackett’s levels of evidence. This review has not been registered.
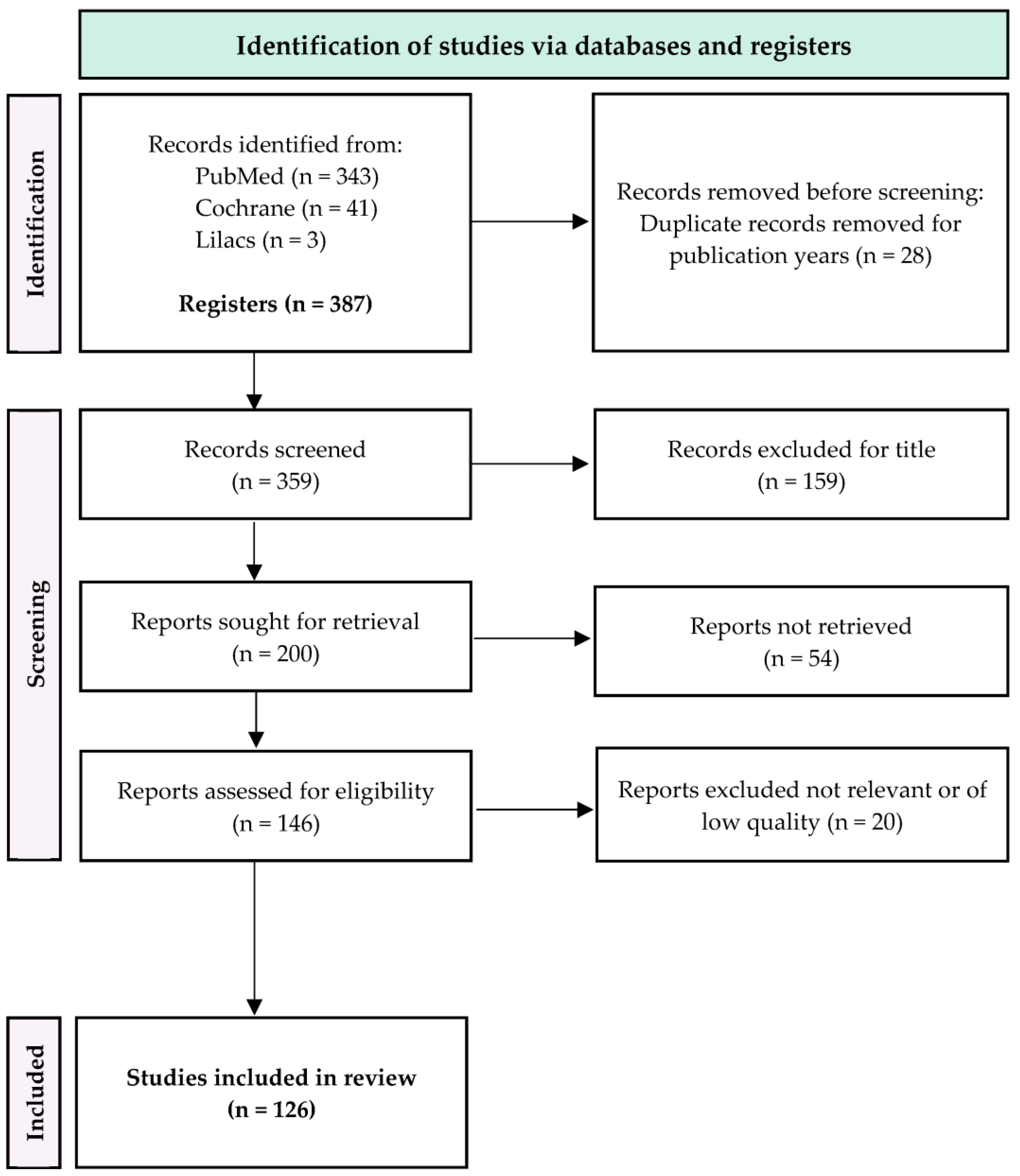
3. Results
Following the identification and screening process depicted in
Figure 1, a total of 126 articles were included in this review.
3.1. DMG Definition
DMG is a rare and aggressive brain tumor that primarily affects children and young adults. It predominantly arises in the brainstem and is characterized by a remarkably low median overall survival time, with over 50% of affected individuals surviving less than 12 months after diagnosis [
13,
17,
18,
19,
21,
22,
23,
24]. DMGs represent a heterogeneous group of tumors that exhibit distinct biological features compared to other high-grade brain tumors [
25]. With the addition of other midline structures to DIPG, the diagnosis of DMG is broader. Due to their unpredictable location and growth pattern, complete surgical resection of DMG is typically unattainable, posing significant challenges in pediatric neuro-oncology [
13,
24].
Damodharan et al. (2022) show that, although DMG is one of the tumors with the bleakest prognosis at present, in recent years the molecular study of DMG has advanced, leading to new avenues of research [
24]. This tumor has been related to oncogenesis that is produced from proto-oncogenes and the suppression of specific cell differentiation [
17,
18]. Advanced diagnostic strategies not only hold a crucial role in confirming the diagnosis of DMG but also possess significant potential in guiding personalized treatment approaches, ultimately improving patient outcomes.
3.2. Epidemiology
According to Argersinger et al. (2021) and other authors [
22,
23,
24,
25], DMG is recognized as one of the most malignant brain tumors in childhood, comprising over two-thirds of all brainstem tumor cases. It has been reported that DMG may contribute to up to 15% of all childhood brain tumor-related deaths [
23]. The survival rate for DMG is particularly low, with only one in ten cases surviving beyond two years after diagnosis [
22,
23,
24,
25,
26].
Approximately one in five childhood brain tumors is identified as a form of DMG and nearly four in five of all brainstem tumors fall into this category [
17,
23]. In the United States alone, 150 to 400 new cases are reported annually [
23,
24,
26].
3.3. Diagnostic Methods
3.3.1. Magnetic Resonance Imaging (MRI)
The conventional MRI used for diagnosing DMG typically reveals hypointense signals on T1-weighted images and hyperintense signals on T2-weighted images, without contrast enhancement (
Figure 2). This characteristic distinguishes DMG from other brain stem tumors. The tumor frequently localizes in the brain stem pons, occupying more than two-thirds of the total volume of the pons and sometimes extending to surrounding areas [
23,
26]. Enhancement techniques for imaging vary based on the tumor type and have not been standardized for cases with specific mutations [
23].
Some of the most common macroscopic findings on imaging techniques for DMG include necrosis in one in five cases, growth from the cephalocaudal direction towards the midbrain, cerebral peduncles, cerebellum or medulla oblongata, and poorly defined margins [
10,
26].
These tumors are usually exophytic, solid, and outgrowing, compressing the basilar artery, with rare involvement of the meninges [
6,
26]. Spectroscopy may show significantly higher choline/N-acetylaspartate and creatine/choline ratios compared to other tumors. Contrast MRI is used after radiotherapy, as radiation necrosis favors contrast uptake [
19,
26].
3.3.2. Biopsy and Laboratory Diagnostic Techniques
Obtaining an accurate histological sample for DMG diagnosis poses a significant challenge, necessitating the use of a reliable and safe method such as stereotactic biopsy. This approach enables precise collection of tissue samples from the tumor, facilitating histological examination and molecular analysis (
Figure 3), leading to improved diagnostic accuracy [
22,
23]. Additionally, the analysis of the tumor’s surrounding serum for the presence of tumor cells or specific biomarkers has emerged as a promising diagnostic technique [
22,
23].
Recent evidence suggests that biopsy is not only safe and diagnostically accurate, but also has the potential to influence treatment decisions for individual patients [
27,
28]. Advancements in neuroimaging techniques, like diffusion tensor imaging and frameless stereotactic technologies, have led to a re-evaluation of performing biopsies on brainstem tumors [
29,
30,
31,
32].
The safety of brainstem biopsy further supports the feasibility of obtaining biopsy-derived DIPG/DMG models for research purposes. Various approaches, including supratentorial transfrontal, infratentorial, and transcerebellar–transpeduncular, have been applied with high diagnostic accuracy and low morbidity in brainstem lesions [
29,
30,
31,
32]. The selection of a specific trajectory according to the lesion’s location is crucial when approaching brainstem lesions.
The majority of models used for studying DIPG and diffuse midline glioma, particularly those with the H3 K27M mutation (DMG), are either derived from autopsies or genetically engineered [
31,
32,
33,
34]. However, these models have limitations for translational studies. To address these limitations, utilizing biopsy tissue for developing laboratory models offers a unique opportunity to create systems that mimic tumors in their untreated state, thereby replicating the initial disease condition.
The advancements in analyzing DMG tumor tissue have provided researchers with a deeper understanding of the disease biology, which may lead to the development of more effective treatment strategies. Stereotactic biopsy, despite yielding limited tumor tissue due to the size of the needle, has demonstrated its ability to provide sufficient material for histopathologic diagnosis and immunohistochemical staining [
30,
31,
32]. As such, performing a biopsy in any lesion suspected of DIPG is highly recommendable, as the new molecular classification and therapeutics under investigation may offer an opportunity for the patient to be included in ongoing clinical trials.
Figure 3.
Representative histology of pediatric DMG. Histologic geographic variability of DMG. ×4 (
A,
C) and ×20 (
B,
D). Hematoxylin and eosin stains from different sections of a single tumor showing low-grade (
A,
B) and high-grade (
C,
D) areas. (
E) Immunohistochemical staining of the same sample using antibody directed to the H3K27M epitope. The sample is strongly positive. (
F) Immunohistochemical staining of mutant p53, a common co-occurring mutation in DMG. (
A–
D) Reprinted with permission and adapted from Warren et al. (2012) [
15] and (
E,
F) reprinted with permission and adapted from Srikanthan et al. (2021) [
35].
Figure 3.
Representative histology of pediatric DMG. Histologic geographic variability of DMG. ×4 (
A,
C) and ×20 (
B,
D). Hematoxylin and eosin stains from different sections of a single tumor showing low-grade (
A,
B) and high-grade (
C,
D) areas. (
E) Immunohistochemical staining of the same sample using antibody directed to the H3K27M epitope. The sample is strongly positive. (
F) Immunohistochemical staining of mutant p53, a common co-occurring mutation in DMG. (
A–
D) Reprinted with permission and adapted from Warren et al. (2012) [
15] and (
E,
F) reprinted with permission and adapted from Srikanthan et al. (2021) [
35].
3.3.3. Molecular Studies
The advent of molecular studies has ushered in a new era in the diagnosis and management of DMG, as affirmed by multiple experts in the field. Molecular testing has emerged as a pivotal paraclinical tool, augmenting the diagnostic and prognostic capabilities of biopsy. At the forefront of its importance is the detection of H3K27M mutations in tumor DNA, which holds immense clinical significance and serves as a robust biomarker for DMG [
18,
19] (
Figure 3E).
The determination of the H3K27M mutation through immunohistochemistry (IHC) is essential for enhancing the histological classification and prognosis of pediatric brain tumors. Initially, H3K27M mutations were exclusively identified in diffuse midline gliomas (DMGs), establishing a unique molecular hallmark for this specific condition. Subsequently, these mutations were also detected in other types of brain tumors. However, it seems that the clinical prognostic value associated with these mutations is particularly notable within the domain of diffuse midline gliomas [
36,
37].
The assessment of H3K27M mutation status extends beyond immunohistochemistry (IHC) and encompasses alternative techniques such as Sanger sequencing, next-generation sequencing (NGS), droplet–digital polymerase chain reaction, and pyrosequencing. Notably, diffuse midline gliomas (DMG) have been found to potentially harbor subclonal H3K27M mutations with mosaic patterns. Instances have arisen where tumor cells exhibited positivity in the cytoplasm or immuno-positivity in lymphocytes, only to be subsequently verified as H3 wild-type through Sanger sequencing. Consequently, there are situations wherein additional sequencing methods are imperative to accurately ascertain the H3K27M mutation status [
38].
Genome-wide DNA methylation profiling has emerged as a crucial tool for diagnosing central nervous system (CNS) tumors. This innovative technique holds significant value for both pediatric and adult patients. By classifying brain tumors according to their unique DNA methylation patterns, this approach has revolutionized how we diagnose cases where traditional histology alone may not provide clear answers. This advancement has notably transformed the diagnostic strategy for CNS tumors with uncertain characteristics [
37,
39,
40,
41].
The 2021 WHO Classification of Tumors of the Central Nervous System incorporates various tumor types and subtypes that require, in part, the use of whole genome methylation profiling for accurate diagnosis. The contemporary methodology for array DNA methylation profiling involves employing a reference library of tumor DNA methylation data, coupled with a machine learning-based tumor classifier. This pioneering approach was introduced and popularized by the German Cancer Research Network (DKFZ) and University Hospital Heidelberg. Remarkably, this research collective has generously made their CNS tumor classifier accessible for research purposes through an online platform [
42,
43].
Furthermore, the application of high-density methylation arrays has emerged as a robust technique for effectively categorizing primary brain neoplasms into distinct molecular subgroups. The development of the DKFZ/Heidelberg CNS tumor methylation classifier significantly contributes to our comprehension of methylation patterns and their interplay with clinical variables. This advancement greatly enhances the precision of molecular pathological classification in the realm of brain tumors [
42,
44].
A study conducted by Ceccarelli et al. (2016) [
45] on diffuse gliomas revealed that these epigenetic subgroups offer prognostic insights independent of factors like age and tumor grade. This underscores the significant value of epigenetic information in predicting disease outcomes and tailoring treatment strategies; they used the Illumina Infinium Human Methylation array platform. The first version of this platform 450 (450 K) array not only enables methylation profiling but also facilitates concurrent copy number analysis. Additionally, a more recent validation has been carried out with the 850 K array. This application has led to the identification of six distinct subgroups, each enriched for specific DNA mutations. Notably, these subgroups exhibit discernible clinical characteristics encompassing factors such as age, anatomical location, and treatment outcomes [
45].
3.4. Previous and Current WHO Classification of DMG
Until 2016, all DMGs were classified solely based on histological and morphological MRI characteristics of the tumor [
25,
46,
47]. At the time, tumor grading could be summarized as: low grade or grade I and grade II tumors were those with low proliferative potential. Whereas grade I tumors could be possibly cured via surgical resection, grade II were usually infiltrative and tended to recur and progress to higher grades. Grade III tumors were those considered malignant, determined by histological evidence of mitosis or anaplasia, with frequent recurrence and rarely curable. High grade or grade IV tumors were those malignant and highly proliferative with active mitosis, and prone to necrosis, which is associated with a rapid evolution and therefore fatal prognosis causing death within months [
6,
48,
49].
In most cases, DMGs were considered high grade due to their histological appearance (such as glioblastoma multiforme grade IV or anaplastic astrocytoma grade III), but this classification was incorrect, as tumors such as well-differentiated astrocytoma (grade II) were considered to be lower grade lesions.
Recognizing the limitations of this approach, a paradigm shift occurred in 2016 when molecular classifications emerged from earlier discoveries, prompting a re-evaluation of DMG severity based on both histological and molecular–genetic characteristics [
5,
6,
17,
22,
50,
51]. The 2016 CNS WHO classification considers phenotypic and genotypic parameters. This additional layer of refinement results in narrowly defined tumor types, creating larger groups of entities that do not fit in any category, designated NOS (not otherwise specified). The combination of both histology and genetics creates the possibility of discordant results. Therefore, the 2016 classification still considers not possible to understand nosological and clinical significance of certain genetic changes without histology. The genotype is integrated with the phenotype but still maintaining histology as the main criteria for grade determinations, as well as for the understanding of NOS tumors [
49].
The periodicity of major WHO classification updates depends on many factors. However, the acceleration in our understanding of the molecular features of CNS tumors and their clinical impact has created a need for more frequent updates in the past years. Intended to fill the gap between official WHO classifications, the cIMPACT-NOW (Consortium to Inform Molecular and Practical Approaches to CNS Tumor Taxonomy—Not Official WHO) was created, in order to provide possible guidelines for clinicians and waypoints for the next WHO classification update [
48]. Since the publication of the 2016 CNS WHO update, seven cIMPACT-NOW updates have reviewed newly acquired criteria on CNS tumors classification, including the definition and diagnostic pathways of NOS tumors, and the relevance of the mutations in histone 3 (K23M mutation), B-Raf (V600E mutation), and isocitrate dehydrogenase (IDH), among others [
52].
These updates are reflected in the revised classification scheme by the WHO published in 2021, which now integrates the histological and immunohistochemical features of DMG mutations [
24]. Notably, recent molecular biology studies have led to the identification of histone 3 or “H3” mutants as a distinct subgroup within DMG. Among the various mutations studied, H3K27M has gained prominence and has been incorporated into the WHO classification of central nervous system tumors as a sign of severity [
6,
21,
22,
47,
51].
There are three types of genetic modifications in histone 3 mutated DMG, which are summarized in the
Table 1 with their respective frequencies.
H3K27M mutations in H3.3/H3.1 have been extensively documented by multiple authors, with a prevalence of ~80% in cases of DMGs, making it the most frequently observed mutation [
22,
23,
50]. H3K27M represents a recurrent somatic gain-of-function mutation characterized by the substitution of lysine 27 to methionine (p.Lys27Met: K27M) within the histone 3 (H3) protein. Consequently, the classification of these tumors is commonly referred to as H3 classification [
5,
6,
51,
54]. Additionally, ACVR1 mutations have been identified in approximately 25% of DIPGs, contributing to the genetic landscape of these tumors [
22,
23,
50].
In addition to H3K27M mutations, the new classification includes several other high-grade tumors. These include diffuse hemispheric glioma with H3G34 mutation, pediatric-type diffuse high-grade glioma with H3-wildtype and IDH-wildtype, and infantile-type hemispheric glioma [
46]. Tumors lacking the H3K27M mutation are referred to as “wild-type” DMGs and they generally have a more favorable prognosis compared to those with the mutation, as the H3 mutation is associated with higher mortality [
22,
50,
54]. In addition to a different biological behaviour caused by this mutation, the worse prognosis could be partially attributed by the midline localization of H3K27 tumors, which induces severe symptoms and prevents effective treatments as resection.
The TP53 mutation is frequently observed in wild-type DMGs, as highlighted by Buczkowicz and Hawkins in 2015 [
5]. Another important mutation is ACVR1, which is considered a major factor in tumorigenesis and is present in nearly one-third of DMGs with the H3K27M mutation [
22]. Tumors with the ACVR1 mutation may exhibit encoding of serine/threonine kinase (ALK2), while mutations in EZH2, an enzyme involved in histone methylation, have been associated with a very poor prognosis.
3.5. Treatments
3.5.1. Radiotherapy (RT)
Radiotherapy has been the mainstay of treatment for improving the life expectancy of patients with DMG [
55,
56,
57]. The standard guideline recommends delivering doses of 54 to 60 Gy over a period of 6 weeks upon DMG diagnosis. This approach, in practice for over 20 years, has been observed to halt tumor progression for approximately 3 months in nearly four out of five cases [
13,
21,
24,
25,
47,
58,
59,
60,
61]. However, DMGs are highly aggressive tumors and, even with early initiation of RT, overall survival is minimally affected, as noted by Pai Panandiker et al. (2014) [
61].
Hyperfractionated RT has shown potential in improving clinical management by caregivers and has a significant positive impact on the quality of life for patients with DMG [
8,
15]. In contrast, a study by Park et al. (2020) compared hyperfractionated therapy with standard RT and found similar survival outcomes for patients with DMG [
62].
A phase III non-inferiority randomized trial comparing hypofractionated (39 Gy in 13 fractions and 45 Gy in 15 fractions) and conventional fractionated (54 Gy in 30 fractions) radiotherapy was performed [
63]. They conducted a randomized trial involving 253 patients, divided into three arms receiving different radiation regimens. The findings revealed median overall survival (OS) rates of 9.6, 8.2, and 8.7 months in the groups receiving 39 Gy in 13 fractions, 45 Gy in 15 fractions, and conventional radiation, respectively. The study met their non-inferiority assumption (with a non-inferiority margin of 15%) at 18 months OS. After these results, hypofractionation radiotherapy can be considered for patients with DMG, with a dose of 39 Gy in 13 fractions, at the clinician’s discretion, taking into account the patient’s condition and tumor location.
3.5.2. Proton Therapy
Proton therapy is a form of particle radiotherapy that offers precise dose localization and reduced late toxicity by minimizing radiation exposure to normal brain tissues surrounding the tumor. This treatment modality allows for safe irradiation of tumors that cannot be effectively treated with photon radiotherapy, as critical organs can be spared due to its high degree of dose conformity [
64]. In cases of reirradiation, proton therapy can mitigate the risk of radiation necrosis in the surrounding brain tissue [
64,
65].
Currently, proton therapy is typically administered using a similar schedule to photon radiotherapy. Early studies with limited patient follow-up have demonstrated comparable therapeutic efficacy between proton therapy and photon radiotherapy. Mizumoto et al. (2016, 2017) conducted studies in Japan on pediatric malignancies treated with proton therapy and reported a low incidence of late toxicity [
66,
67].
However, there is a lack of studies with sufficient sample sizes and long-term follow-up to compare the long-term effects of proton therapy and photon radiotherapy. Therefore, in-depth studies involving large cohorts of children and extended follow-up periods are needed to fully assess the potential benefits of proton therapy in reducing long-term side effects.
3.5.3. Chemotherapy (CTX)
DMG is distinguished by the presence of diverse tumor tissue, often accompanied by an intact BBB, which might account for its resistance to chemotherapy [
9]. CTX is commonly administered in conjunction with RT to leverage their synergistic effect in treating DMG. To date, there are still no guidelines for both chemo- and radiotherapy, resulting in a wide variety of treatment schedules, particularly when facing disease progression.
Some evidence supports the combination of treatment using radiosensitizers like gemcitabine and capecitabine, originated from the results of two phase II trials [
68,
69], although their impact on overall mortality is minimal or negligible.
Kilburn et al. (2018) conducted a phase II trial with capecitabine, an alkylating agent, given alongside radiotherapy, followed by adjuvant capecitabine in 44 patients with newly diagnosed DIPG [
68]. The findings showed earlier progression, with a 1-year progression-free survival (PFS) of 7.21% (SE = 3.47%) in the capecitabine-treated group compared to 15.59% (SE = 3.05%) in the historical control group (
p = 0.007). While there was no significant survival benefit (overall survival (OS) and PFS were comparable to historical controls), the treatment regimen was well-tolerated. Similarly, Veldhuijzen van Zanten et al. (2017) conducted a phase I/II trial to evaluate the efficacy, safety, and tolerability of gemcitabine, a nucleoside metabolic inhibitor, during radiotherapy in nine children with newly diagnosed DIPG [
69]. All patients experienced a reduction in tumor-related symptoms and quality of life (QoL) tended to improve during treatment. The PFS and median OS were 4.8 months (95% CI 4.0–5.7) and 8.7 months (95% CI 7.0–10.4), respectively. However, there were no significant survival benefits observed, with a median OS of 12.4 months and 8.7 months in intermediate- and high-risk patients, respectively.
Bailey et al. (2013) evaluated the efficacy of combining temozolomide (TMZ) with standard radiotherapy in children with DMG [
59]. They administered concomitant temozolomide (75 mg/m
2) during radiotherapy, followed by adjuvant temozolomide (75–100 mg/m
2) for up to 12 cycles in a 21-day schedule. Their study did not demonstrate a survival improvement [
59], which is consistent with findings reported by other authors [
5,
58,
70]. However, Jackaki et al. (2016) tested the combination of oral TMZ with lomustine and observed significantly improved outcomes compared to TMZ alone [
70].
Re-irradiation in combination with CTX or immunotherapy has been investigated [
22,
25]. These studies have shown a prolongation in overall survival compared to non-irradiated patients, although the observed changes, while statistically significant, have been modest. The data suggest that combining re-irradiation with other therapies may be more beneficial than implementing re-irradiation alone [
22,
25,
65]. Other agents with no effect on overall mortality are described in the literature, such as veliparib, tested by Baxter et al. (2020) in a phase I/II study [
60].
In addition to the aforementioned agents, other drugs frequently used in the treatment of DMG patients aim to alleviate symptoms or slow disease progression. These include etoposide, bevacizumab, irinotecan, nimotuzumab, and valproic acid. However, in most cases, these drugs have proven ineffective in significantly altering the fatal prognosis within a short period of time [
13,
58].
3.5.4. Novel Approaches for Local Administration of Therapies
Recent advancements in technology and surgical techniques have enabled the exploration of novel approaches for local administration of therapies in the treatment of DMG. One such technique is convection-enhanced delivery (CED). Gwak and Park, 2017 and other authors have extensively discussed the potential of CED in DMG treatment [
22,
50,
58,
71], while preclinical trials using murine models have demonstrated the efficacy of CED in prolonging drug retention of multiple molecules at the infusion site and maintaining a sustained therapeutic effect against DMG [
71]. However, it is important to note that there are currently no established guidelines regarding the therapeutic safety, potential systemic risks, or toxicity of this route of administration. Consequently, ongoing research and active debates in the field continue to explore these aspects [
22,
58,
71].
Another recent therapeutic strategy that has gained attention is the utilization of magnetic resonance-guided focused ultrasound (MRgFUS) [
72,
73]. This innovative approach enables the targeted delivery of substances to the tumor site through the application of ultrasound waves in combination with microbubbles. By temporarily increasing the permeability of the blood vessels and the BBB, MRgFUS allows for localized delivery of therapeutic agents, making it a promising therapeutic option for the future [
74,
75].
MRgFUS holds significant potential in providing precise and non-invasive treatment for diffuse midline gliomas (DMGs). The ability to precisely target the tumor area and control the release of therapeutic substances offers advantages in terms of reducing systemic toxicity and enhancing treatment efficacy [
74,
76,
77].
While MRgFUS shows promise, further research and clinical investigations are needed to optimize this approach, and evaluate its safety and effectiveness in DMG treatment. As this technique continues to evolve, it has the potential to revolutionize the field of neuro-oncology by providing a targeted and minimally invasive treatment option for patients with DMG.
3.5.5. Targeted Therapies Based on the Molecular Classification
Drugs targeting deregulated histone acetylation and methylation (H3 mutations), such as GSKJ4, an H3K27M demethylase inhibitor that increases H3K27me3 in H3K27me3-expressing cells, have been tested in humans. These therapies aim to induce tumor cell death, arrest the cell cycle, modulate chromatin expression, and limit differentiation. Preclinical studies have shown improved outcomes when these therapies are combined with panobinostat, a histone deacetylase inhibitor [
21,
22,
58,
78]. A summary of preclinical studies can be found in
Supplementary Table S2.
Large international collaborative efforts have facilitated the molecular analysis of a significant number of these rare tumors, revealing that H3K27M gliomas also encompass biologically and genetically diverse tumors. These comprehensive studies have led to the discovery of additional oncogenic driver alterations in H3K27M gliomas [
79,
80,
81]. Interestingly, these events include mutations in well-described oncogenic pathways, such as cell-/DNA-damage repair mechanisms (TP53, PPM1D, ATM, and ATRX), and receptor tyrosine kinase signaling pathways (ACVR1, FGFR1, PIK3CA, PIK3R1, and BRAF). Double mutant gliomas with H3K27M and BRAFV600E alterations have been reported in numerous studies. This suggests a biological overlap between histologically defined low- and high-grade gliomas, and may be associated with a better prognosis than expected when compared to BRAF wildtype and H3K27-mutant DMGs [
82,
83,
84,
85,
86,
87].
On 16 March 2023, the FDA approved dabrafenib (Tafinlar, Novartis) in combination with trametinib (Mekinist, Novartis) for pediatric patients aged 1 year and older with low-grade glioma (LGG) harboring a BRAF V600E mutation and requiring systemic therapy (NCT02684058). The FDA also approved new oral formulations of both drugs suitable for patients who cannot swallow pills [
88]. This marks the first FDA approval of a systemic therapy for the first-line treatment of pediatric patients with LGG carrying a BRAF V600E mutation. Patients with BRAF mutations may benefit from this therapy even if their tumors are not low-grade gliomas.
In tumors with ACVR1 mutations, ALK2 inhibitors have been identified as potential therapeutic agents, even when this mutation occurs in wild-type DMG. While in vitro studies have shown promise, there is a lack of in vivo studies in this context [
22]. Similarly, EZH2 inhibitors and metabolic inhibitors such as the polyamide synthesis inhibitor difluoromethylornithine (DFMO) have shown efficacy in mouse models, but clinical studies are yet to be conducted [
22,
89,
90]. Wei et al. (2018) conducted a comprehensive bioinformatics analysis on tumor composition leading to propose potential avenues for future therapies, highlighting the calcium signaling pathways and the neuroactive ligand-receptor interactions [
91].
Additionally, the use of a dopamine receptor D2 (DRD2) antagonist and a caseinolytic protease P agonist (ClpP), known as ONC201, has been proposed as a potentially safe treatment option [
92]. This specific DRD2/3 antagonist has the ability to penetrate the blood–brain barrier and demonstrates p53-independent effectiveness in preclinical models of high-grade glioma [
93]. The mode of action of ONC201 involves triggering the integrated stress response while simultaneously deactivating Akt/ERK and other prosurvival signaling pathways [
94]. Furthermore, ONC201 is capable of targeting and depleting cancer stem cells present in gliomas and other advanced cancers [
95]. The first pediatric clinical trial of ONC201 confirmed that the adult recommended phase II dose, administered weekly through oral capsules and scaled by body weight, was well tolerated in pediatric patients. Results from this study indicate a safety and pharmacokinetic profile of ONC201 similar to that observed in adults. There was no dose-limiting toxicity, with most adverse effects being grade 1 or 2. Five patients (22.7%), all of whom started ONC201 after radiation and before recurrence, were alive at 2 years from the time of diagnosis [
96]. New strategies are being studied with this drug as monotherapy and in combination with other agents such as PI3K inhibitors. While its use has been primarily after radiation or in cases of recurrence, the new strategies aim to incorporate ONC201 in earlier stages of the disease [
97,
98,
99,
100]. ONC201, panobinostat, and paxalisib are enzyme inhibitors that exhibit the potential to halt the growth of tumor cells by obstructing crucial enzymes required for cell proliferation. Currently, a phase II trial (NCI-2021-08386) is underway to evaluate various combinations of these drugs for the treatment of DMGs (NCI-2021-08386). Participants in the study will be randomly assigned to one of three study arms upon entry. Subsequently, they will be categorized into one to three phases and one of three cohorts based on their disease stage and prior treatment history. The target enrollment for the trial is 324 patients, with the estimated completion date set for 2027.
Another therapeutic strategy that has emerged in the field of neuro-oncology is the use of antibody–drug conjugates (ADCs) [
101]. ADCs are a class of immunoconjugates that chemically link protein-specific antibodies with cytotoxic agents to selectively target cells expressing the antigen of interest with high specificity. However, the role of ADCs in patients with gliomas has been reconsidered due to various factors, such as toxicity associated with targeting the EGFR family with certain cytotoxic payloads and the challenge of penetrating the blood–brain barrier to reach tumor cells [
101,
102].
Several ADCs have been evaluated in clinical trials for gliomas. For example, depatuxizumab mafodotin (ABT-414), an ADC targeting EGFR, has shown potential in the treatment of recurrent gliomas [
103]. However, a phase 3 trial did not demonstrate a benefit in overall survival when treating de novo glioblastomas with EGFR amplification [
104].
Furthermore, nanobodies can be engineered to deliver therapeutic agents directly to glioma cells, enhancing the specificity and effectiveness of treatment while minimizing off-target effects. This targeted approach holds great promise for improving the outcomes of glioma patients and overcoming the limitations of conventional therapies.
The development of nanobodies targeting molecular targets in gliomas in addition to BBB shuttle peptides [
105,
106] represents an exciting advancement in neuro-oncology research and opens new possibilities for personalized medicine in the treatment of gliomas.
3.5.6. Tumor Treating Fields
TTF utilizes low-intensity electric fields to inhibit cell division, showing efficacy in neoplastic conditions such as glioblastoma, pancreatic cancer, ovarian cancer, non-small cell lung carcinoma, and mesothelioma [
107]. TTFs have been approved by the FDA as adjuvant therapy for newly diagnosed and recurrent glioblastoma [
108,
109]. Ongoing clinical trials are further exploring its potential in various malignancies, while interdisciplinary healthcare teams play a crucial role in the management of patients receiving TTF therapy.
An inherent limitation in the context of generating transcranial fields is the restricted depth of targeted tissue and the insulating impact of the skull [
110,
111]. An study by Ibn Essayed et al. (2023) investigates the potential use of TTFs for DMG focusing on the anatomical constraints associated with endoscopic endonasal implantation of an electrode array in the clivus region [
112]. The findings reveal that, in the pediatric population studied, the dimensions allow for the placement of a 2.5 × 1 cm electrode array in 94% of patients (16/17), highlighting the feasibility of this approach. These results suggest that implantable transclival devices for TTF could serve as a viable adjunctive therapy for DMG, emphasizing the need for further investigation and development in this area.
3.5.7. Immunotherapies
Vaccines
The development and utilization of autologous dendritic cell response-based vaccines offer a potential avenue for personalized immunotherapy in targeting specific tumor mutations and enhancing the immune response against cancer cells. Further research is warranted to explore the efficacy and long-term effects of this approach in clinical settings. An approach using peptide vaccines for this matter has been proposed and shown to be safe and to generate a specific immune response in the treatment of H3K27M mutated tumors [
22]. An example of these approaches are peptide vaccines derived from neoantigens that occur within tumors. Mueller et al. (2020) demonstrated in a phase I/II clinical trial (NCT02960230) the efficacy of an H3.3K27M-targeted peptide vaccine, including newly diagnosed patients (3–21 years old) with HLA-A*02.01
+ and H3-3K27M
+ genotype [
113]. The vaccine was well tolerated by patients and increased the overall survival of patients that showed specific anti-H3.3K27M
+ CD8+ response. Another phase I clinical trial (NCT04749641) is currently in an enacting status using an H3.3K27M neoantigen vaccine.
Presently, there are other clinical trials that are being conducted for the study of other peptide vaccines for patients with DMG tumors, such as PEP-CMV (NCT05096481), composed of the synthetic long peptide Component A from human pp65, or rHSC-DIPGVax (NCT04943848), a mixture of peptides of neo-epitopes from the neo-antigen heat shock protein.
Virus-based vaccines are also under study, including DNX-2401 (Delta-24-RGD), an oncolytic adenovirus that has been genetically modified to induce in the patient a response similar to the natural immunity of a tumor, particularly in cases of H3K27M-mutated tumors [
22,
114]. Gállego Pérez-Larraya et al. (2022) conducted a single-centre dose escalation study in patients with newly diagnosed DMG. The study included 12 patients and two treatment regimens were employed: DNX-2401 viral particles: 1 × 10
10 (for the first 4 patients) or 5 × 10
10 (for the next 8 patients), with 11 receiving subsequent radiotherapy. The viral infusion was administered as a single dose through a catheter placed in the cerebellar peduncle. While the study demonstrated the feasibility of applying this therapy in humans, the authors emphasized the need for further research to establish safety profiles and assess potential adverse effects [
114].
Despite the promising potential of oncolytic virotherapy, there remain several significant barriers that need to be addressed to enhance its efficacy. These obstacles encompass various factors, including viral tropism, delivery platforms, viral distribution, dosing strategies, antiviral immunity, and the oncolysis capability.
Immunomodulators
The use of some antibodies, such as nimotuzumab, administered simultaneously with standard-dose radiotherapy may have effects similar and comparable to those of radiotherapy plus chemotherapy. Kebudi et al. (2019) conducted a trial where they administered nimotuzumab alone in 17 cases and nimotuzumab combined with vinorelbine until death in patients with progressive disease. The survival rates were similar to those reported with standard treatment reported by other authors who describe treatment only with RT + CTX. Although the study did not serve to demonstrate superior outcomes, it established a basis supporting nimotuzumab as a well-tolerated drug without significant adverse effects [
115]. Immunomodulatory antibodies such as MDV9300 (pidilizumab) have been used with promising results [
22,
116].
Immunotherapy with Tumor Infiltrating Lymphocytes (TILs)
Immunotherapy using tumor-infiltrating lymphocytes (TILs) has been considered as a potential treatment option for H3K27M-mutated tumors. TIL therapy involves extracting immune cells, particularly T cells, from the tumor tissue, expanding them in the laboratory, and then reinfusing them back into the patient with the aim of boosting the anti-tumor immune response [
117,
118].
However, the application of TIL therapy in H3K27M-mutated tumors has been limited due to challenges associated with obtaining a sufficient volume of tumor tissue through biopsy [
118,
119]. Collecting an adequate number of TILs is crucial for the success of this therapy, as it requires a significant quantity of tumor-infiltrating immune cells to be isolated and expanded. Given the diffuse nature of H3K27M-mutated tumors and their often limited accessibility, obtaining a suitable amount of tumor tissue for TIL extraction may be difficult [
6,
118].
CAR T-Cells and CAR-NK Anti GD2
Immunotherapy has gained ground in the area of nervous system cancer treatment research at least in the last 5 years. The molecular classification of these tumors has been very important in incorporating this knowledge in the development of chimeric antigen receptor (CAR) T-cell and NK therapies. Among them, CARs directed against the disialoganglioside GD2, which is an antigen that is almost uniquely associated with DMG tumor-related tissues, are showing some promise [
22,
50,
120].
In vitro studies have demonstrated a significant reduction in the number of cells expressing the H3K27M mutation using GD2-targeting CAR T-cells [
25,
120]. Mount et al. (2018) conducted a study using orthotopic xenograft models of H3-K27M+ tumors and demonstrated that systemic administration of GD2-targeted CAR T-cells resulted in the elimination of grafted tumors, with only a small number of GD2-positive cells remaining [
120]. Furthermore, Liu et al. (2022) recently described how the induction of immunogenic cell death in tumors is accompanied by the expression of molecular patterns associated with tissue damage in the tumor [
121].
The possible penetration of the therapy into the brain tissue or the neurotoxicity of the therapy is uncertain, which has made it difficult to carry out human studies. The potential of immunotherapy as an adjuvant to traditional chemo-radiation has been proposed [
22]. Majzner et al. (2022) conducted a well-designed phase I study testing GD2-CAR T-cell therapy for diffuse midline gliomas (DMGs) carrying the H3K27M mutation, which provided valuable insights into the therapeutic approach. Unlike other studies that focused on treatment-related inflammation, this study evidenced the development of signs and symptoms consistent with CAR T-cell-mediated inflammation (TIAN). The authors associated this inflammation with treatment toxicity, intracranial pressure alterations, and primary brain dysfunction [
122].
The study findings confirmed the potential safety of GD2-CAR T-cell therapy as a therapeutic strategy for DMG. However, it is important to note that the experience is still limited and based on a small number of patients. Considering this, approximately 75% of the patients showed positive clinical and radiographic changes after the specific therapy improving the functionality of the infiltrated brain areas, a finding that was amplified in second therapies with GD2-CAR T-cells. As of the date of this review, a clinical trial in patients with H3K27M+ DMG and spinal DMG using GD2-CAR T-cell therapy is ongoing to determine the optimal dose, route, and schedule, and to determine the efficacy of GD2-CAR T-cell therapy in patients with H3K27M + DMG and spinal DMG [
122].
4. Scope and Future Directions
In addition to all exposed above, the development of new tools to identify circulating biomarkers is on the rise. The quantification of tumor DNA in plasma has revolutionized treatment response monitoring, offering an accurate and non-invasive method for assessing therapeutic efficacy [
22], facilitating the detection of such mutations.
The identification of molecular targets in gliomas has also sparked the development of nanobodies, the single variable domain-heavy chain fragment (VHH) of the heavy-chain-only antibodies (HCAbs) derived from the
Camelidae species [
92,
123]. Nanobodies are designed to specifically bind to these molecular targets, such as overexpressed proteins or receptors in glioma cells. They offer several advantages over traditional antibodies, including their small size, high stability, and ability to penetrate the blood–brain barrier (BBB) [
123,
124].
Moreover, by targeting specific molecular markers in gliomas, nanobodies are emerging as a new tool with the potential to be used for various applications, including diagnostic imaging and targeted therapy. In diagnostic imaging, nanobodies can be labeled with imaging agents to selectively detect and visualize glioma cells using non-invasive imaging techniques including positron emission tomography (PET) or magnetic resonance imaging (MRI) [
92,
125,
126].
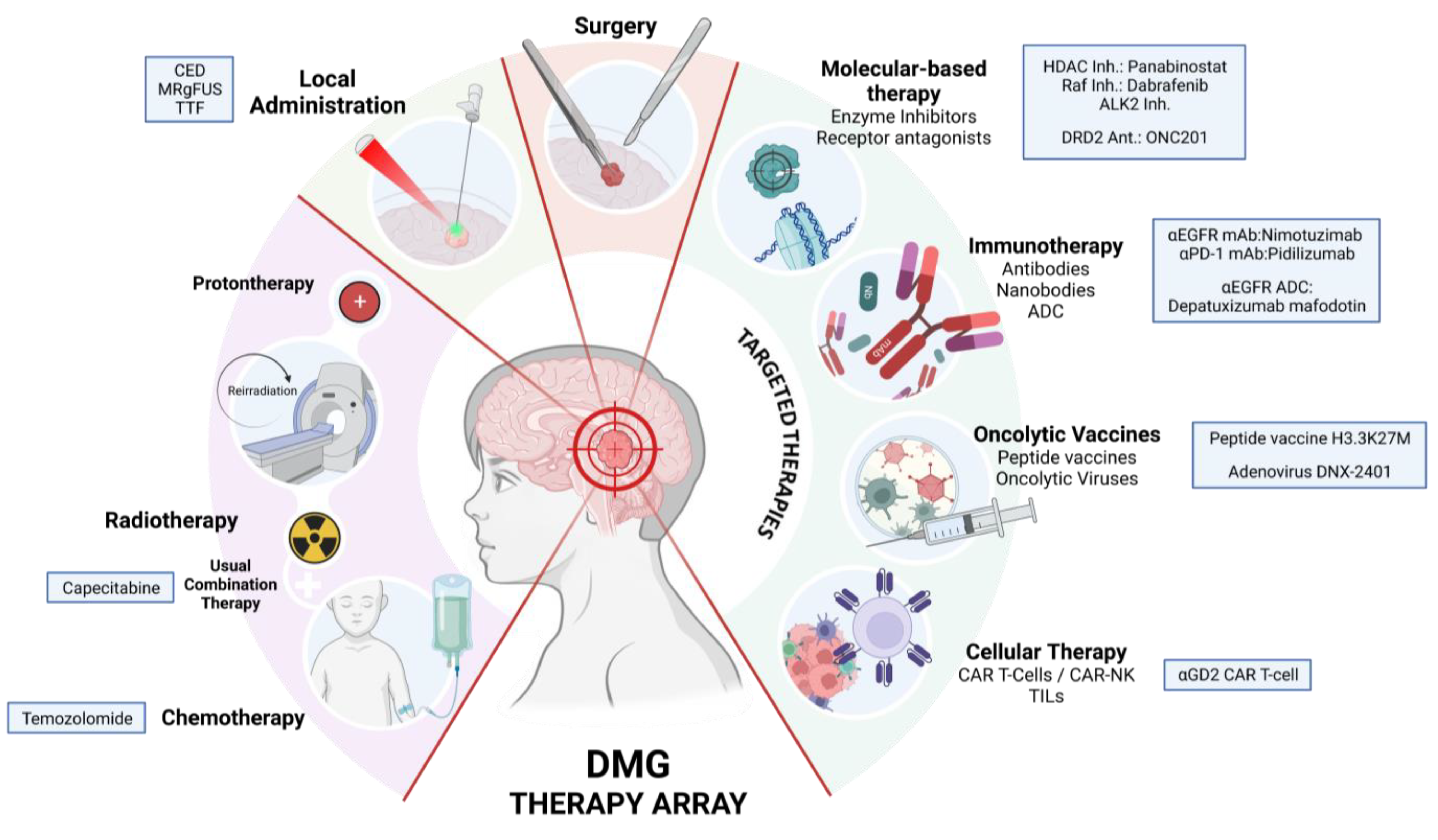
This review highlights the importance of molecular classification in guiding therapeutic strategies for DMGs (
Figure 4). Recent advances in understanding the genetic and molecular features of these tumors are leading to the development of targeted therapies, including immunotherapies and oncolytic vaccines, which show promising results in preclinical and early clinical studies.
However, there are limitations and challenges that need to be addressed. The infiltrative nature of DMGs, the limited accessibility to tumor tissue, and uncertainties regarding the penetration of therapies into the brain tissue pose obstacles to the implementation of effective treatments. Furthermore, the low frequency of DMGs and the limited number of patients available for clinical trials contribute to the slow progress in developing optimal therapeutic approaches.
Despite these limitations, ongoing clinical trials evaluating novel therapies based on the molecular characteristics of DMGs provide hope for improved outcomes. It is of importance to consider the geographic locations of clinical trials for potential treatment options. Some patients may be willing to travel for treatment and knowing the available trial locations can assist physicians in guiding their patients towards appropriate therapeutic avenues. While the treatment approaches for DMG in the USA, Europe, and Australia share similarities and prioritize a multidisciplinary approach, several key groups are driving significant advancements in DMG research and treatment. Notably, the United States hosts the largest network of centers specializing in DMG treatment, encompassing biopsy, molecular classification, and clinical trials. In addition, Spain and Switzerland in Europe, as well as Australia in the Oceania region, have centers that adhere to high-quality standards in DMG treatment. These regions are contributing substantially to the understanding and management of DMG. Collaborative efforts by prominent groups such as the Pediatric Brain Tumor Consortium (PBTC), the Children’s Oncology Group (COG) Phase I/Pilot Consortium, and the Pacific Pediatric Neuro-Oncology Consortium (PNOC) are actively involved in DMG research and treatment.
This review emphasizes the need for highly specific and tailored approaches to effectively target DMGs and improve patient outcomes. Molecular profiling and personalized medicine hold great potential for optimizing treatment strategies and improving prognosis for patients with DMGs.
5. Conclusions
Despite the recent advancements in the diagnosis and treatment of DMG, it continues to be a complex and challenging disease. However, the recent advances in the molecular classification of DMG have opened new avenues for research.
MRI remains the main diagnostic method. However, other techniques, such as biopsy and cerebrospinal fluid, plasma or serum analysis, are growing in importance due to the advent of molecular studies of new biomarkers that provide valuable prognostic insight.
Regarding therapy, the current standard of care, which combines radiation therapy and chemotherapy, provides limited improvement in the overall survival of these patients. Early studies suggest proton therapy as a safer modality, especially in cases that require re-irradiation, as well as novel local administration techniques of drugs and radiation or BBB permeability increase approaches, that prove promising in preclinical studies.
Moreover, novel molecular-based targeted therapies bring new opportunities noteworthy of consideration. These techniques offer a broad variety of possibilities, from targeted therapies against histone methylation abnormalities to immunotherapies that range between single antibodies or nanobodies to whole cells, like CAR T-cells or CAR-NK therapies, and even oncolytic vaccines.
All in all, although DMG is still a clinically challenging tumor, these new strategies offer hope for the future. Continued research and clinical trials are necessary to fully understand the safety, efficacy, and long-term effects of this approaches in order to advance its clinical application and to establish effective and safe treatment protocols.
6. Limitations of the Study
The limitations of the study include the restricted timeframe of 1 January 2012 to 30 June 2023, which may exclude relevant studies published before or after that period. Language bias is present as only English and Spanish articles available online were included missing other languages. The study relies on specific databases (PubMed, Cochrane Library, and SciELO through the LILACS), potentially overlooking relevant studies from other sources. The broad inclusion criteria may lead to varying levels of evidence and quality in the selected studies. Publication bias may be present due to the inclusion of online articles. The findings may not be generalizable to other brain tumors or populations. These limitations should be taken into account when interpreting the study’s conclusions.
Supplementary Materials
The following supporting information can be downloaded at:
https://www.mdpi.com/article/10.3390/jcm12165261/s1, Table S1: Highlights of results of clinical studies. Table S2: Highlights of the results of preclinical studies in animals and in culture. Table S3: Highlights of results from review studies.
Author Contributions
Conceptualization, G.M.L., V.M.M.P. and A.J.S.; investigation, G.M.L., V.M.M.P., B.C.S., I.C.-Y., S.V.S. and A.J.S.; writing—original draft preparation, G.M.L., V.M.M.P., B.C.S., I.C.-Y. and A.J.S.; writing—review and editing G.M.L., V.M.M.P., I.C.-Y. and A.J.S.; supervision, and funding acquisition, A.J.S. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the XIII Beca FERO en Investigación Oncológica Traslacional from Fundación FERO, Ayudas a la investigación del cáncer infantil from Asociación de Padres de Niños con Cáncer de Aragón (ASPANOA), Instituto de Salud Carlos III through the Fondo de Investigación en Salud Projects “PI18/01665” and “PI21/00441” and co-funded by European Union (ERDF, “A way to make Europe”), Proyectos líneas prioritarias y de carácter multidisciplinar de la RIS3 2021–2023 DGA (LMP248_21), (A.J.S.). Ayuda Clínico Junior en el Territorio from Asociación Española Contra el Cáncer (AECC) and Fundación Científica AECC (Reference: CLJUN223444MIGU for G.M-L). Ayudas para iberoamericanos y ecuatoguineanos en Estudios de Doctorado-Curso 2022/2023, convocada por la Universidad de Zaragoza en colaboración con Santander Universidades. (V.M.M-P.). Ayudas para la Formación de Profesorado Universitario (FPU) from Ministerio de Universidades (FPU19/06371) (I.C.-Y.).
Institutional Review Board Statement
All image scans were obtained from our Center dataset, performed for clinical purposes using routine techniques, properly anonymized, and an ethical review board approved written informed consent was obtained at the time of original clinical image collection.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
The authors thank the Molecular Oncology Lab members at IIS Aragón for critical reading of the manuscript. The contribution of A.J.S. to the review honors the memory of Paul E. Sanz whose absence keeps inspiring my work.
Conflicts of Interest
The authors declare no conflict of interest. A.J.S. is a shareholder of Oniria Therapeutics and founder of Great Air S.L. These two companies are not related to this research.
Abbreviations
| BBB | Blood–brain barrier |
| CAR | Chimeric antigen receptor |
| CTX | Chemotherapy |
| DFMO | Difluoromethylornithine |
| DIPG | Diffuse intrinsic pontine glioma |
| DMG | Diffuse midline glioma |
| FDA | Food and Drug Administration |
| GD2 | Disialoganglioside-2 |
| H3 | Histone 3 |
| NOS | Not otherwise specified |
| OS | Overall survival |
| PFS | Progression-free survival |
| QoL | Quality of life |
| RT | Radiotherapy |
| TILs | Tumor-infiltrating lymphocytes |
| WHO | World Health Organization |
References
- Farrukh, S.; Habib, S.; Rafaqat, A.; Sarfraz, Z.; Sarfraz, A.; Sarfraz, M.; Robles-Velasco, K.; Felix, M.; Cherrez-Ojeda, I. Emerging Therapeutic Strategies for Diffuse Intrinsic Pontine Glioma: A Systematic Review. Healthcare 2023, 11, 559. [Google Scholar] [CrossRef] [PubMed]
- Hennika, T.; Becher, O.J. Diffuse intrinsic pontine glioma: Time for cautious optimism. J. Child Neurol. 2016, 31, 1377–1385. [Google Scholar] [CrossRef] [PubMed]
- Robison, N.J.; Kieran, M.W. Diffuse intrinsic pontine glioma: A reassessment. J. Neurooncol. 2014, 119, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Hayat, M.A. (Ed.) Pediatric Cancer; Springer: Dordrecht, The Netherlands, 2012; Volume 2, ISBN 978-94-007-2956-8. [Google Scholar]
- Buczkowicz, P.; Hawkins, C. Pathology, molecular genetics, and epigenetics of diffuse intrinsic pontine glioma. Front. Oncol. 2015, 5, 147. [Google Scholar] [CrossRef]
- Louis, D.N.; Perry, A.; Wesseling, P.; Brat, D.J.; Cree, I.A.; Figarella-Branger, D.; Hawkins, C.; Ng, H.K.; Pfister, S.M.; Reifenberger, G.; et al. The 2021 WHO Classification of Tumors of the Central Nervous System: A summary. Neuro. Oncol. 2021, 23, 1231–1251. [Google Scholar] [CrossRef]
- Price, G.; Bouras, A.; Hambardzumyan, D.; Hadjipanayis, C.G. Current knowledge on the immune microenvironment and emerging immunotherapies in diffuse midline glioma. eBioMedicine 2021, 69, 103453. [Google Scholar] [CrossRef]
- Ross, J.L.; Velazquez Vega, J.; Plant, A.; MacDonald, T.J.; Becher, O.J.; Hambardzumyan, D. Tumour immune landscape of paediatric high-grade gliomas. Brain 2021, 144, 2594–2609. [Google Scholar] [CrossRef]
- Pachocki, C.J.; Hol, E.M. Current perspectives on diffuse midline glioma and a different role for the immune microenvironment compared to glioblastoma. J. Neuroinflamm. 2022, 19, 276. [Google Scholar] [CrossRef]
- Villarejo, F.J.; Ferrara, M.P.; Belinchón, J.M.; Madero, L.; Rivero, B.; Cordobés, F.; Pascual, A.; Pérez-Díaz, C.; de Prada, I. Tumores de tronco cerebral en la población pediátrica. Neurocirugia 2008, 19, 551–555. [Google Scholar] [CrossRef]
- Fontebasso, A.M.; Bechet, D.; Jabado, N. Molecular biomarkers in pediatric glial tumors: A needed wind of change. Curr. Opin. Oncol. 2013, 25, 665–673. [Google Scholar] [CrossRef]
- Jansen, M.H.A.; van Vuurden, D.G.; Vandertop, W.P.; Kaspers, G.J.L. Diffuse intrinsic pontine gliomas: A systematic update on clinical trials and biology. Cancer Treat. Rev. 2012, 38, 27–35. [Google Scholar] [CrossRef] [PubMed]
- Walker, D.A.; Liu, J.; Kieran, M.; Jabado, N.; Picton, S.; Packer, R.; St Rose, C.; Van Meeteren, A.S.; Carvalho, A.; Van Damme, A.; et al. A multi-disciplinary consensus statement concerning surgical approaches to low-grade, high-grade astrocytomas and diffuse intrinsic pontine gliomas in childhood (CPN Paris 2011) using the Delphi method. Neuro. Oncol. 2013, 15, 462–468. [Google Scholar] [CrossRef] [PubMed]
- Grimm, S.A.; Chamberlain, M.C. Brainstem glioma: A review. Curr. Neurol. Neurosci. Rep. 2013, 13, 346. [Google Scholar] [CrossRef]
- Warren, K.E. Diffuse intrinsic pontine glioma: Poised for progress. Front. Oncol. 2012, 2, 205. [Google Scholar] [CrossRef]
- Veldhuijzen van Zanten, S.E.; Jansen, M.H.; Sanchez Aliaga, E.; van Vuurden, D.G.; Vandertop, W.P.; Kaspers, G.J. A twenty-year review of diagnosing and treating children with diffuse intrinsic pontine glioma in The Netherlands. Expert Rev. Anticancer Ther. 2015, 15, 157–164. [Google Scholar] [CrossRef] [PubMed]
- Pollack, I.F.; Agnihotri, S.; Broniscer, A. Childhood brain tumors: Current management, biological insights, and future directions. J. Neurosurg. Pediatr. 2019, 23, 261–273. [Google Scholar] [CrossRef]
- El-Hashash, A.H.K. Histone H3K27M Mutation in Brain Tumors. In Advances in Experimental Medicine and Biology: Histone Mutations and Cancer; Fang, D., Han, J., Eds.; Springer Nature: Singapore, 2021; pp. 43–52. [Google Scholar]
- Aziz-Bose, R.; Monje, M. Diffuse intrinsic pontine glioma: Molecular landscape and emerging therapeutic targets. Curr. Opin. Oncol. 2019, 31, 522–530. [Google Scholar] [CrossRef]
- Hayden, E.; Holliday, H.; Lehmann, R.; Khan, A.; Tsoli, M.; Rayner, B.S.; Ziegler, D.S. Therapeutic Targets in Diffuse Midline Gliomas—An Emerging Landscape. Cancers 2021, 13, 6251. [Google Scholar] [CrossRef]
- Morales La Madrid, A.; Hashizume, R.; Kieran, M.W. Future Clinical Trials in DIPG: Bringing Epigenetics to the Clinic. Front. Oncol. 2015, 5, 148. [Google Scholar] [CrossRef]
- Hoffman, L.M.; Veldhuijzen van Zanten, S.E.M.; Colditz, N.; Baugh, J.; Chaney, B.; Hoffmann, M.; Lane, A.; Fuller, C.; Miles, L.; Hawkins, C.; et al. Clinical, Radiologic, Pathologic, and Molecular Characteristics of Long-Term Survivors of Diffuse Intrinsic Pontine Glioma (DIPG): A Collaborative Report From the International and European Society for Pediatric Oncology DIPG Registries. J. Clin. Oncol. 2018, 36, 1963–1972. [Google Scholar] [CrossRef]
- Argersinger, D.P.; Rivas, S.R.; Shah, A.H.; Jackson, S.; Heiss, J.D. New Developments in the Pathogenesis, Therapeutic Targeting, and Treatment of H3K27M-Mutant Diffuse Midline Glioma. Cancers 2021, 13, 5280. [Google Scholar] [CrossRef] [PubMed]
- Damodharan, S.; Lara-Velazquez, M.; Williamsen, B.C.; Helgager, J.; Dey, M. Diffuse Intrinsic Pontine Glioma: Molecular Landscape, Evolving Treatment Strategies and Emerging Clinical Trials. J. Pers. Med. 2022, 12, 840. [Google Scholar] [CrossRef] [PubMed]
- Vanan, M.I.; Eisenstat, D.D. DIPG in children—What can we learn from the past? Front. Oncol. 2015, 5, 237. [Google Scholar] [CrossRef] [PubMed]
- González, O.E.; Casas, C.; Bermúdez, Y.M. State of the art: Pediatric brain stem gliomas. Rev. Colomb. Cancerol. 2017, 21, 202–211. [Google Scholar] [CrossRef]
- Kline, C.; Jain, P.; Kilburn, L.; Bonner, E.R.; Gupta, N.; Crawford, J.R.; Banerjee, A.; Packer, R.J.; Villanueva-Meyer, J.; Luks, T.; et al. Upfront Biology-Guided Therapy in Diffuse Intrinsic Pontine Glioma: Therapeutic, Molecular, and Biomarker Outcomes from PNOC003. Clin. Cancer Res. 2022, 28, 3965–3978. [Google Scholar] [CrossRef]
- Jaradat, A.; Nowacki, A.; Fichtner, J.; Schlaeppi, J.-A.; Pollo, C. Stereotactic biopsies of brainstem lesions: Which approach? Acta Neurochir. 2021, 163, 1957–1964. [Google Scholar] [CrossRef]
- Hersh, D.S.; Kumar, R.; Moore, K.A.; Smith, L.G.F.; Tinkle, C.L.; Chiang, J.; Patay, Z.; Gajjar, A.; Choudhri, A.F.; Lee-Diaz, J.A.; et al. Safety and efficacy of brainstem biopsy in children and young adults. J. Neurosurg. Pediatr. 2020, 26, 552–562. [Google Scholar] [CrossRef]
- Pfaff, E.; El Damaty, A.; Balasubramanian, G.P.; Blattner-Johnson, M.; Worst, B.C.; Stark, S.; Witt, H.; Pajtler, K.W.; van Tilburg, C.M.; Witt, R.; et al. Brainstem biopsy in pediatric diffuse intrinsic pontine glioma in the era of precision medicine: The INFORM study experience. Eur. J. Cancer 2019, 114, 27–35. [Google Scholar] [CrossRef]
- Salah, M.; Shalaby, A. Computed tomography-guided stereotactic surgery in the management of brain lesions: A single-center experience. Surg. Neurol. Int. 2023, 14, 184. [Google Scholar] [CrossRef]
- Cage, T.A.; Samagh, S.P.; Mueller, S.; Nicolaides, T.; Haas-Kogan, D.; Prados, M.; Banerjee, A.; Auguste, K.I.; Gupta, N. Feasibility, safety, and indications for surgical biopsy of intrinsic brainstem tumors in children. Child Nerv. Syst. 2013, 29, 1313–1319. [Google Scholar] [CrossRef]
- Joud, A.; Stella, I.; Klein, O. Diffuse infiltrative pontine glioma biopsy in children with neuronavigation, frameless procedure: A single center experience of 10 cases. Neurochirurgie 2020, 66, 345–348. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, L.M.; DeWire, M.; Ryall, S.; Buczkowicz, P.; Leach, J.; Miles, L.; Ramani, A.; Brudno, M.; Kumar, S.S.; Drissi, R.; et al. Spatial genomic heterogeneity in diffuse intrinsic pontine and midline high-grade glioma: Implications for diagnostic biopsy and targeted therapeutics. Acta Neuropathol. Commun. 2016, 4, 1. [Google Scholar] [CrossRef]
- Srikanthan, D.; Taccone, M.S.; Van Ommeren, R.; Ishida, J.; Krumholtz, S.L.; Rutka, J.T. Diffuse intrinsic pontine glioma: Current insights and future directions. Chin. Neurosurg. J. 2021, 7, 6. [Google Scholar] [CrossRef] [PubMed]
- Louis, D.N.; Giannini, C.; Capper, D.; Paulus, W.; Figarella-Branger, D.; Lopes, M.B.; Batchelor, T.T.; Cairncross, J.G.; van den Bent, M.; Wick, W.; et al. cIMPACT-NOW update 2: Diagnostic clarifications for diffuse midline glioma, H3 K27M-mutant and diffuse astrocytoma/anaplastic astrocytoma, IDH-mutant. Acta Neuropathol. 2018, 135, 639–642. [Google Scholar] [CrossRef] [PubMed]
- An, S.; Camarillo, J.M.; Huang, T.Y.-T.; Li, D.; Morris, J.A.; Zoltek, M.A.; Qi, J.; Behbahani, M.; Kambhampati, M.; Kelleher, N.L.; et al. Histone tail analysis reveals H3K36me2 and H4K16ac as epigenetic signatures of diffuse intrinsic pontine glioma. J. Exp. Clin. Cancer Res. 2020, 39, 261. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Fang, X.; Xue, B. Four methods to analyze H3K27M mutation in diffuse midline gliomas. Pathol. Res. Pract. 2020, 216, 153065. [Google Scholar] [CrossRef]
- Lebrun, L.; Allard-Demoustiez, S.; Gilis, N.; Van Campenhout, C.; Rodesch, M.; Roman, C.; Calò, P.; Lolli, V.; David, P.; Fricx, C.; et al. Clinicopathological and molecular characterization of a case classified by DNA-methylation profiling as “CNS embryonal tumor with BRD4–LEUTX fusion”. Acta Neuropathol. Commun. 2023, 11, 46. [Google Scholar] [CrossRef]
- Capper, D.; Stichel, D.; Sahm, F.; Jones, D.T.W.; Schrimpf, D.; Sill, M.; Schmid, S.; Hovestadt, V.; Reuss, D.E.; Koelsche, C.; et al. Practical implementation of DNA methylation and copy-number-based CNS tumor diagnostics: The Heidelberg experience. Acta Neuropathol. 2018, 136, 181–210. [Google Scholar] [CrossRef]
- Wiśniewski, K.; Ghaly, A.; Drummond, K.; Fahlstrӧm, A. H3 K27M-Altered Diffuse Midline Gliomas: A Review. Indian J. Neurosurg. 2023. [Google Scholar] [CrossRef]
- Wu, Z.; Abdullaev, Z.; Pratt, D.; Chung, H.-J.; Skarshaug, S.; Zgonc, V.; Perry, C.; Pack, S.; Saidkhodjaeva, L.; Nagaraj, S.; et al. Impact of the methylation classifier and ancillary methods on CNS tumor diagnostics. Neuro Oncol. 2022, 24, 571–581. [Google Scholar] [CrossRef]
- Capper, D.; Jones, D.T.W.; Sill, M.; Hovestadt, V.; Schrimpf, D.; Sturm, D.; Koelsche, C.; Sahm, F.; Chavez, L.; Reuss, D.E.; et al. DNA methylation-based classification of central nervous system tumours. Nature 2018, 555, 469–474. [Google Scholar] [CrossRef] [PubMed]
- Santana-Santos, L.; Kam, K.L.; Dittmann, D.; De Vito, S.; McCord, M.; Jamshidi, P.; Fowler, H.; Wang, X.; Aalsburg, A.M.; Brat, D.J.; et al. Validation of Whole Genome Methylation Profiling Classifier for Central Nervous System Tumors. J. Mol. Diagn. 2022, 24, 924–934. [Google Scholar] [CrossRef] [PubMed]
- Ceccarelli, M.; Barthel, F.P.; Malta, T.M.; Sabedot, T.S.; Salama, S.R.; Murray, B.A.; Morozova, O.; Newton, Y.; Radenbaugh, A.; Pagnotta, S.M.; et al. Molecular Profiling Reveals Biologically Discrete Subsets and Pathways of Progression in Diffuse Glioma. Cell 2016, 164, 550–563. [Google Scholar] [CrossRef] [PubMed]
- Vitanza, N.A.; Monje, M. Diffuse Intrinsic Pontine Glioma: From Diagnosis to Next-Generation Clinical Trials. Curr. Treat. Options Neurol. 2019, 21, 37. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Feng, L.; Ji, P.; Liu, J.; Guo, S.; Zhai, Y.; Sankey, E.W.; Wang, Y.; Xue, Y.; Wang, N.; et al. Clinical Features and Molecular Markers on Diffuse Midline Gliomas With H3K27M Mutations: A 43 Cases Retrospective Cohort Study. Front. Oncol. 2021, 10, 602553. [Google Scholar] [CrossRef] [PubMed]
- Louis, D.N.; Aldape, K.; Brat, D.J.; Capper, D.; Ellison, D.W.; Hawkins, C.; Paulus, W.; Perry, A.; Reifenberger, G.; Figarella-Branger, D.; et al. Announcing cIMPACT-NOW: The Consortium to Inform Molecular and Practical Approaches to CNS Tumor Taxonomy. Acta Neuropathol. 2017, 133, 1–3. [Google Scholar] [CrossRef]
- Louis, D.N.; Perry, A.; Reifenberger, G.; von Deimling, A.; Figarella-Branger, D.; Cavenee, W.K.; Ohgaki, H.; Wiestler, O.D.; Kleihues, P.; Ellison, D.W. The 2016 World Health Organization Classification of Tumors of the Central Nervous System: A summary. Acta Neuropathol. 2016, 131, 803–820. [Google Scholar] [CrossRef]
- Hauser, P. Classification and Treatment of Pediatric Gliomas in the Molecular Era. Children 2021, 8, 739. [Google Scholar] [CrossRef]
- Johung, T.B.; Monje, M. Diffuse Intrinsic Pontine Glioma: New Pathophysiological Insights and Emerging Therapeutic Targets. Curr. Neuropharmacol. 2017, 15, 88–97. [Google Scholar] [CrossRef]
- Gonzalez Castro, L.N.; Wesseling, P. The cIMPACT-NOW updates and their significance to current neuro-oncology practice. Neuro Oncology Pract. 2021, 8, 4–10. [Google Scholar] [CrossRef]
- Ortega, F.V.; García, A.A.; Pérez, T.M.; Ortega, F.V.; García, A.A.; Pérez, T.M. Tumores cerebrales en niños. Pediatr. Integr. 2016, 20, 401–411. [Google Scholar]
- Lapin, D.H.; Tsoli, M.; Ziegler, D.S. Genomic Insights into Diffuse Intrinsic Pontine Glioma. Front. Oncol. 2017, 7, 57. [Google Scholar] [CrossRef] [PubMed]
- Gallitto, M.; Lazarev, S.; Wasserman, I.; Stafford, J.M.; Wolden, S.L.; Terezakis, S.A.; Bindra, R.S.; Bakst, R.L. Role of Radiation Therapy in the Management of Diffuse Intrinsic Pontine Glioma: A Systematic Review. Adv. Radiat. Oncol. 2019, 4, 520–531. [Google Scholar] [CrossRef]
- Izzuddeen, Y.; Gupta, S.; Haresh, K.P.; Sharma, D.; Giridhar, P.; Rath, G.K. Hypofractionated radiotherapy with temozolomide in diffuse intrinsic pontine gliomas: A randomized controlled trial. J. Neurooncol. 2020, 146, 91–95. [Google Scholar] [CrossRef]
- Hu, X.; Fang, Y.; Hui, X.; Jv, Y.; You, C. Radiotherapy for diffuse brainstem glioma in children and young adults. Cochrane Database Syst. Rev. 2016, 6, CD010439. [Google Scholar] [CrossRef]
- Gwak, H.-S.; Park, H.J. Developing chemotherapy for diffuse pontine intrinsic gliomas (DIPG). Crit. Rev. Oncol. Hematol. 2017, 120, 111–119. [Google Scholar] [CrossRef] [PubMed]
- Bailey, S.; Howman, A.; Wheatley, K.; Wherton, D.; Boota, N.; Pizer, B.; Fisher, D.; Kearns, P.; Picton, S.; Saran, F.; et al. Diffuse intrinsic pontine glioma treated with prolonged temozolomide and radiotherapy—Results of a United Kingdom phase II trial (CNS 2007 04). Eur. J. Cancer 2013, 49, 3856–3862. [Google Scholar] [CrossRef] [PubMed]
- Baxter, P.A.; Su, J.M.; Onar-Thomas, A.; Billups, C.A.; Li, X.-N.N.; Poussaint, T.Y.; Smith, E.R.; Thompson, P.; Adesina, A.; Ansell, P.; et al. A phase I/II study of veliparib (ABT-888) with radiation and temozolomide in newly diagnosed diffuse pontine glioma: A Pediatric Brain Tumor Consortium study. Neuro Oncol. 2020, 22, 875–885. [Google Scholar] [CrossRef]
- Panandiker, A.S.P.; Wong, J.K.; Nedelka, M.A.; Wu, S.; Gajjar, A.; Broniscer, A. Effect of time from diagnosis to start of radiotherapy on children with diffuse intrinsic pontine glioma. Pediatr. Blood Cancer 2014, 61, 1180–1183. [Google Scholar] [CrossRef]
- Park, J.; Yea, J.W.; Park, J.W. Hypofractionated radiotherapy versus conventional radiotherapy for diffuse intrinsic pontine glioma. Medicine 2020, 99, e22721. [Google Scholar] [CrossRef]
- Zaghloul, M.S.; Nasr, A.; Tolba, M.; Refaat, A.; Youssef, A.; Mosaab, A.; Enayet, A.; Arafa, O.; Maher, E.; Eldebawy, E. Hypofractionated Radiation Therapy For Diffuse Intrinsic Pontine Glioma: A Noninferiority Randomized Study Including 253 Children. Int. J. Radiat. Oncol. 2022, 113, 360–368. [Google Scholar] [CrossRef]
- Muroi, A.; Mizumoto, M.; Ishikawa, E.; Ihara, S.; Fukushima, H.; Tsurubuchi, T.; Sakurai, H.; Matsumura, A. Proton therapy for newly diagnosed pediatric diffuse intrinsic pontine glioma. Child Nerv. Syst. 2020, 36, 507–512. [Google Scholar] [CrossRef]
- Navarria, P.; Minniti, G.; Clerici, E.; Tomatis, S.; Pinzi, V.; Ciammella, P.; Galaverni, M.; Amelio, D.; Scartoni, D.; Scoccianti, S.; et al. Re-irradiation for recurrent glioma: Outcome evaluation, toxicity and prognostic factors assessment. A multicenter study of the Radiation Oncology Italian Association (AIRO). J. Neurooncol. 2019, 142, 59–67. [Google Scholar] [CrossRef]
- Mizumoto, M.; Murayama, S.; Akimoto, T.; Demizu, Y.; Fukushima, T.; Ishida, Y.; Oshiro, Y.; Numajiri, H.; Fuji, H.; Okumura, T.; et al. Proton beam therapy for pediatric malignancies: A retrospective observational multicenter study in Japan. Cancer Med. 2016, 5, 1519–1525. [Google Scholar] [CrossRef]
- Mizumoto, M.; Murayama, S.; Akimoto, T.; Demizu, Y.; Fukushima, T.; Ishida, Y.; Oshiro, Y.; Numajiri, H.; Fuji, H.; Okumura, T.; et al. Long-term follow-up after proton beam therapy for pediatric tumors: A Japanese national survey. Cancer Sci. 2017, 108, 444–447. [Google Scholar] [CrossRef]
- Kilburn, L.B.; Kocak, M.; Baxter, P.; Poussaint, T.Y.; Paulino, A.C.; McIntyre, C.; Lemenuel-Diot, A.; Lopez-Diaz, C.; Kun, L.; Chintagumpala, M.; et al. A pediatric brain tumor consortium phase II trial of capecitabine rapidly disintegrating tablets with concomitant radiation therapy in children with newly diagnosed diffuse intrinsic pontine gliomas. Pediatr. Blood Cancer 2018, 65, e26832. [Google Scholar] [CrossRef]
- Veldhuijzen van Zanten, S.E.M.; El-Khouly, F.E.; Jansen, M.H.A.; Bakker, D.P.; Sanchez Aliaga, E.; Haasbeek, C.J.A.; Wolf, N.I.; Zwaan, C.M.; Vandertop, W.P.; van Vuurden, D.G.; et al. A phase I/II study of gemcitabine during radiotherapy in children with newly diagnosed diffuse intrinsic pontine glioma. J. Neurooncol. 2017, 135, 307–315. [Google Scholar] [CrossRef]
- Jakacki, R.I.; Cohen, K.J.; Buxton, A.; Krailo, M.D.; Burger, P.C.; Rosenblum, M.K.; Brat, D.J.; Hamilton, R.L.; Eckel, S.P.; Zhou, T.; et al. Phase 2 study of concurrent radiotherapy and temozolomide followed by temozolomide and lomustine in the treatment of children with high-grade glioma: A report of the Children’s Oncology Group ACNS0423 study. Neuro. Oncol. 2016, 18, 1442–1450. [Google Scholar] [CrossRef]
- Bellat, V.; Alcaina, Y.; Tung, C.-H.H.; Ting, R.; Michel, A.O.; Souweidane, M.; Law, B. A combined approach of convection-enhanced delivery of peptide nanofiber reservoir to prolong local DM1 retention for diffuse intrinsic pontine glioma treatment. Neuro. Oncol. 2020, 22, 1495–1504. [Google Scholar] [CrossRef]
- Siedek, F.; Yeo, S.Y.; Heijman, E.; Grinstein, O.; Bratke, G.; Heneweer, C.; Puesken, M.; Persigehl, T.; Maintz, D.; Grüll, H. Magnetic Resonance-Guided High-Intensity Focused Ultrasound (MR-HIFU): Technical Background and Overview of Current Clinical Applications (Part 1). RöFo 2019, 191, 522–530. [Google Scholar] [CrossRef]
- Palumbo, P.; Daffinà, J.; Bruno, F.; Arrigoni, F.; Splendiani, A.; Di Cesare, E.; Barile, A.; Masciocchi, C. Basics in Magnetic Resonance guided Focused Ultrasound: Technical basis and clinical application. A brief overview. Acta Biomed. 2021, 92, e2021403. [Google Scholar] [CrossRef]
- Zhang, X.; Ye, D.; Yang, L.; Yue, Y.; Sultan, D.; Pacia, C.P.; Pang, H.; Detering, L.; Heo, G.S.; Luehmann, H.; et al. Magnetic Resonance Imaging-Guided Focused Ultrasound-Based Delivery of Radiolabeled Copper Nanoclusters to Diffuse Intrinsic Pontine Glioma. ACS Appl. Nano Mater. 2020, 3, 11129–11134. [Google Scholar] [CrossRef]
- Conq, J.; Joudiou, N.; Ucakar, B.; Vanvarenberg, K.; Préat, V.; Gallez, B. Assessment of Hyperosmolar Blood–Brain Barrier Opening in Glioblastoma via Histology with Evans Blue and DCE-MRI. Biomedicines 2023, 11, 1957. [Google Scholar] [CrossRef]
- Ishida, J.; Alli, S.; Bondoc, A.; Golbourn, B.; Sabha, N.; Mikloska, K.; Krumholtz, S.; Srikanthan, D.; Fujita, N.; Luck, A.; et al. MRI-guided focused ultrasound enhances drug delivery in experimental diffuse intrinsic pontine glioma. J. Control. Release 2021, 330, 1034–1045. [Google Scholar] [CrossRef]
- Alli, S.; Figueiredo, C.A.; Golbourn, B.; Sabha, N.; Wu, M.Y.; Bondoc, A.; Luck, A.; Coluccia, D.; Maslink, C.; Smith, C.; et al. Brainstem blood brain barrier disruption using focused ultrasound: A demonstration of feasibility and enhanced doxorubicin delivery. J. Control. Release 2018, 281, 29–41. [Google Scholar] [CrossRef]
- Bailleul, Q.; Navarin, P.; Arcicasa, M.; Bal-Mahieu, C.; Carcaboso, A.M.; Le Bourhis, X.; Furlan, A.; Meignan, S.; Leblond, P. Evofosfamide Is Effective against Pediatric Aggressive Glioma Cell Lines in Hypoxic Conditions and Potentiates the Effect of Cytotoxic Chemotherapy and Ionizing Radiations. Cancers 2021, 13, 1804. [Google Scholar] [CrossRef]
- Wu, G.; Diaz, A.K.; Paugh, B.S.; Rankin, S.L.; Ju, B.; Li, Y.; Zhu, X.; Qu, C.; Chen, X.; Zhang, J.; et al. The genomic landscape of diffuse intrinsic pontine glioma and pediatric non-brainstem high-grade glioma. Nat. Genet. 2014, 46, 444–450. [Google Scholar] [CrossRef] [PubMed]
- Ryall, S.; Krishnatry, R.; Arnoldo, A.; Buczkowicz, P.; Mistry, M.; Siddaway, R.; Ling, C.; Pajovic, S.; Yu, M.; Rubin, J.B.; et al. Targeted detection of genetic alterations reveal the prognostic impact of H3K27M and MAPK pathway aberrations in paediatric thalamic glioma. Acta Neuropathol. Commun. 2016, 4, 93. [Google Scholar] [CrossRef] [PubMed]
- Filbin, M.G.; Tirosh, I.; Hovestadt, V.; Shaw, M.L.; Escalante, L.E.; Mathewson, N.D.; Neftel, C.; Frank, N.; Pelton, K.; Hebert, C.M.; et al. Developmental and oncogenic programs in H3K27M gliomas dissected by single-cell RNA-seq. Science 2018, 360, 331–335. [Google Scholar] [CrossRef] [PubMed]
- Karremann, M.; Gielen, G.H.; Hoffmann, M.; Wiese, M.; Colditz, N.; Warmuth-Metz, M.; Bison, B.; Claviez, A.; van Vuurden, D.G.; von Bueren, A.O.; et al. Diffuse high-grade gliomas with H3 K27M mutations carry a dismal prognosis independent of tumor location. Neuro. Oncol. 2018, 20, 123–131. [Google Scholar] [CrossRef]
- Solomon, D.A.; Wood, M.D.; Tihan, T.; Bollen, A.W.; Gupta, N.; Phillips, J.J.J.; Perry, A. Diffuse Midline Gliomas with Histone H3-K27M Mutation: A Series of 47 Cases Assessing the Spectrum of Morphologic Variation and Associated Genetic Alterations. Brain Pathol. 2016, 26, 569–580. [Google Scholar] [CrossRef] [PubMed]
- Pagès, M.; Beccaria, K.; Boddaert, N.; Saffroy, R.; Besnard, A.; Castel, D.; Fina, F.; Barets, D.; Barret, E.; Lacroix, L.; et al. Co-occurrence of histone H3 K27M and BRAF V600E mutations in paediatric midline grade I ganglioglioma. Brain Pathol. 2018, 28, 103–111. [Google Scholar] [CrossRef] [PubMed]
- Ryall, S.; Zapotocky, M.; Fukuoka, K.; Nobre, L.; Guerreiro Stucklin, A.; Bennett, J.; Siddaway, R.; Li, C.; Pajovic, S.; Arnoldo, A.; et al. Integrated Molecular and Clinical Analysis of 1000 Pediatric Low-Grade Gliomas. Cancer Cell 2020, 37, 569–583.e5. [Google Scholar] [CrossRef] [PubMed]
- Schüller, U.; Iglauer, P.; Dorostkar, M.M.; Mawrin, C.; Herms, J.; Giese, A.; Glatzel, M.; Neumann, J.E. Mutations within FGFR1 are associated with superior outcome in a series of 83 diffuse midline gliomas with H3F3A K27M mutations. Acta Neuropathol. 2021, 141, 323–325. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, A.T.; Colin, C.; Nanni-Metellus, I.; Padovani, L.; Maurage, C.-A.; Varlet, P.; Miquel, C.; Uro-Coste, E.; Godfraind, C.; Lechapt-Zalcman, E.; et al. Evidence for BRAF V600E and H3F3A K27M double mutations in paediatric glial and glioneuronal tumours. Neuropathol. Appl. Neurobiol. 2015, 41, 403–408. [Google Scholar] [CrossRef] [PubMed]
- FDA Approves Dabrafenib with Trametinib for Pediatric Patients with Low-Grade Glioma with a BRAF V600E Mutation. Available online: https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-dabrafenib-trametinib-pediatric-patients-low-grade-glioma-braf-v600e-mutation (accessed on 30 June 2023).
- Katagi, H.; Louis, N.; Unruh, D.; Sasaki, T.; He, X.; Zhang, A.; Ma, Q.; Piunti, A.; Shimazu, Y.; Lamano, J.B.; et al. Radiosensitization by histone H3 demethylase inhibition in diffuse intrinsic pontine glioma. Clin. Cancer Res. 2019, 25, 5572–5583. [Google Scholar] [CrossRef]
- Carvalho, D.M.; Richardson, P.J.; Olaciregui, N.; Stankunaite, R.; Lavarino, C.; Molinari, V.; Corley, E.A.; Smith, D.P.; Ruddle, R.; Donovan, A.; et al. Repurposing Vandetanib plus Everolimus for the Treatment of ACVR1-Mutant Diffuse Intrinsic Pontine Glioma. Cancer Discov. 2022, 12, 416–431. [Google Scholar] [CrossRef]
- Wei, L.; He, F.; Zhang, W.; Chen, W.; Yu, B. Bioinformatics analysis of microarray data to reveal the pathogenesis of diffuse intrinsic pontine glioma. Biol. Res. 2018, 51, 26. [Google Scholar] [CrossRef]
- Keyaerts, M.; Xavier, C.; Heemskerk, J.; Devoogdt, N.; Everaert, H.; Ackaert, C.; Vanhoeij, M.; Duhoux, F.P.; Gevaert, T.; Simon, P.; et al. Phase I Study of 68 Ga-HER2-Nanobody for PET/CT Assessment of HER2 Expression in Breast Carcinoma. J. Nucl. Med. 2016, 57, 27–33. [Google Scholar] [CrossRef]
- Allen, J.E.; Kline, C.L.B.; Prabhu, V.V.; Wagner, J.; Ishizawa, J.; Madhukar, N.; Lev, A.; Baumeister, M.; Zhou, L.; Lulla, A.; et al. Discovery and clinical introduction of first-in-class imipridone ONC201. Oncotarget 2016, 7, 74380–74392. [Google Scholar] [CrossRef]
- Allen, J.E.; Krigsfeld, G.; Mayes, P.A.; Patel, L.; Dicker, D.T.; Patel, A.S.; Dolloff, N.G.; Messaris, E.; Scata, K.A.; Wang, W.; et al. Dual Inactivation of Akt and ERK by TIC10 Signals Foxo3a Nuclear Translocation, TRAIL Gene Induction, and Potent Antitumor Effects. Sci. Transl. Med. 2013, 5, 171ra17. [Google Scholar] [CrossRef]
- Chi, A.S.; Tarapore, R.S.; Hall, M.D.; Shonka, N.; Gardner, S.; Umemura, Y.; Sumrall, A.; Khatib, Z.; Mueller, S.; Kline, C.; et al. Pediatric and adult H3 K27M-mutant diffuse midline glioma treated with the selective DRD2 antagonist ONC201. J. Neurooncol. 2019, 145, 97–105. [Google Scholar] [CrossRef]
- Gardner, S.L.; Tarapore, R.S.; Allen, J.; McGovern, S.L.; Zaky, W.; Odia, Y.; Daghistani, D.; Diaz, Z.; Hall, M.D.; Khatib, Z.; et al. Phase I dose escalation and expansion trial of single agent ONC201 in pediatric diffuse midline gliomas following radiotherapy. Neuro Oncol. Adv. 2022, 4, vdac143. [Google Scholar] [CrossRef] [PubMed]
- Odia, Y.; Koschmann, C.J.; Tarapore, R.; Allen, J.; Zaky, W.T.; Hall, M.D.; Daghistani, D.; Khatib, Z.; Aguilera, D.; MacDonald, T.J.; et al. Window-of-opportunity study of ONC201 in pediatric patients with diffuse intrinsic pontine glioma (DIPG) and thalamic glioma. J. Clin. Oncol. 2022, 40, TPS2082. [Google Scholar] [CrossRef]
- Pruss, M.; Dwucet, A.; Tanriover, M.; Hlavac, M.; Kast, R.E.; Debatin, K.-M.; Wirtz, C.R.; Halatsch, M.-E.; Siegelin, M.D.; Westhoff, M.-A.; et al. Dual metabolic reprogramming by ONC201/TIC10 and 2-Deoxyglucose induces energy depletion and synergistic anti-cancer activity in glioblastoma. Br. J. Cancer 2020, 122, 1146–1157. [Google Scholar] [CrossRef] [PubMed]
- Kawakibi, A.R.; Tarapore, R.S.; Gardner, S.; Chi, A.; Kurz, S.; Wen, P.Y.; Arrillaga-Romany, I.; Batchelor, T.T.; Butowski, N.A.; Sumrall, A.; et al. HGG-18. Clinical Efficacy of ONC201 in thalamic H3 K27M-mutant glioma. Neuro. Oncol. 2020, 22, iii347. [Google Scholar] [CrossRef]
- Duchatel, R.; Jackson, E.; Patabendige, A.; Cain, J.; Tsoli, M.; Monje, M.; Alvaro, F.; Ziegler, D.; Dun, M. DIPG-03. Targeting pi3k Using the Blood Brain Barrier Penetrable Inhibitor, gdc-0084, for the Treatment of Diffuse Intrinsic Pontine Glioma (DIPG). Neuro. Oncol. 2019, 21, ii68. [Google Scholar] [CrossRef]
- Parakh, S.; Nicolazzo, J.; Scott, A.M.; Gan, H.K. Antibody Drug Conjugates in Glioblastoma—Is There a Future for Them? Front. Oncol. 2021, 11, 718590. [Google Scholar] [CrossRef]
- Lian, X.; Kats, D.; Rasmussen, S.; Martin, L.R.; Karki, A.; Keller, C.; Berlow, N.E. Design considerations of an IL13Rα2 antibody–drug conjugate for diffuse intrinsic pontine glioma. Acta Neuropathol. Commun. 2021, 9, 88. [Google Scholar] [CrossRef]
- Lassman, A.B.; Pugh, S.L.; Wang, T.J.C.; Aldape, K.; Gan, H.K.; Preusser, M.; Vogelbaum, M.A.; Sulman, E.P.; Won, M.; Zhang, P.; et al. Depatuxizumab mafodotin in EGFR-amplified newly diagnosed glioblastoma: A phase III randomized clinical trial. Neuro. Oncol. 2023, 25, 339–350. [Google Scholar] [CrossRef]
- Weller, M.; Butowski, N.; Tran, D.D.; Recht, L.D.; Lim, M.; Hirte, H.; Ashby, L.; Mechtler, L.; Goldlust, S.A.; Iwamoto, F.; et al. Rindopepimut with temozolomide for patients with newly diagnosed, EGFRvIII-expressing glioblastoma (ACT IV): A randomised, double-blind, international phase 3 trial. Lancet Oncol. 2017, 18, 1373–1385. [Google Scholar] [CrossRef] [PubMed]
- Oller-Salvia, B.; Sánchez-Navarro, M.; Giralt, E.; Teixidó, M. Blood–brain barrier shuttle peptides: An emerging paradigm for brain delivery. Chem. Soc. Rev. 2016, 45, 4690–4707. [Google Scholar] [CrossRef] [PubMed]
- Panigrahy, A.; Jakacki, R.I.; Pollack, I.F.; Ceschin, R.; Okada, H.; Nelson, M.D.; Kohanbash, G.; Dhall, G.; Bluml, S. Magnetic Resonance Spectroscopy Metabolites as Biomarkers of Disease Status in Pediatric Diffuse Intrinsic Pontine Gliomas (DIPG) Treated with Glioma-Associated Antigen Peptide Vaccines. Cancers 2022, 14, 5995. [Google Scholar] [CrossRef] [PubMed]
- Katiyar, V.; De Jesus, O. Tumor Treatment Fields in Oncology. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2023. Available online: https://www.ncbi.nlm.nih.gov/books/NBK576401/ (accessed on 12 February 2023).
- Stupp, R.; Wong, E.T.; Kanner, A.A.; Steinberg, D.; Engelhard, H.; Heidecke, V.; Kirson, E.D.; Taillibert, S.; Liebermann, F.; Dbalý, V.; et al. NovoTTF-100A versus physician’s choice chemotherapy in recurrent glioblastoma: A randomised phase III trial of a novel treatment modality. Eur. J. Cancer 2012, 48, 2192–2202. [Google Scholar] [CrossRef] [PubMed]
- Ram, Z.; Kim, C.-Y.; Hottinger, A.F.; Idbaih, A.; Nicholas, G.; Zhu, J.-J. Efficacy and Safety of Tumor Treating Fields (TTFields) in Elderly Patients with Newly Diagnosed Glioblastoma: Subgroup Analysis of the Phase 3 EF-14 Clinical Trial. Front. Oncol. 2021, 11, 671972. [Google Scholar] [CrossRef]
- Korshoej, A.R.; Mikic, N.; Hansen, F.L.; Saturnino, G.B.; Thielscher, A.; Bomzon, Z. Enhancing Tumor Treating Fields Therapy with Skull-Remodeling Surgery. The Role of Finite Element Methods in Surgery Planning. Annu. Int. Conf. IEEE Eng. Med. Biol. Soc. 2019, 2019, 6995–6997. [Google Scholar]
- Korshoej, A.R.; Sørensen, J.C.H.; von Oettingen, G.; Poulsen, F.R.; Thielscher, A. Optimization of tumor treating fields using singular value decomposition and minimization of field anisotropy. Phys. Med. Biol. 2019, 64, 04NT03. [Google Scholar] [CrossRef]
- Ibn Essayed, W.; Jarvis, C.A.; Bernstock, J.D.; Slingerland, A.; Albanese, J.; Friedman, G.K.; Arnaout, O.; Baird, L. Positioning Transclival Tumor-Treating Fields for the Treatment of Diffuse Intrinsic Pontine Gliomas. Life 2023, 13, 601. [Google Scholar] [CrossRef]
- Mueller, S.; Taitt, J.M.; Villanueva-Meyer, J.E.; Bonner, E.R.; Nejo, T.; Lulla, R.R.; Goldman, S.; Banerjee, A.; Chi, S.N.; Whipple, N.S.; et al. Mass cytometry detects H3.3K27M-specific vaccine responses in diffuse midline glioma. J. Clin. Investig. 2020, 130, 6325–6337. [Google Scholar] [CrossRef]
- Gállego Pérez-Larraya, J.; Garcia-Moure, M.; Labiano, S.; Patiño-García, A.; Dobbs, J.; Gonzalez-Huarriz, M.; Zalacain, M.; Marrodan, L.; Martinez-Velez, N.; Puigdelloses, M.; et al. Oncolytic DNX-2401 Virus for Pediatric Diffuse Intrinsic Pontine Glioma. N. Engl. J. Med. 2022, 386, 2471–2481. [Google Scholar] [CrossRef]
- Kebudi, R.; Cakir, F.B.; Bay, S.B.; Gorgun, O.; Altınok, P.; Iribas, A.; Agaoglu, F.Y.; Darendeliler, E. Nimotuzumab-containing regimen for pediatric diffuse intrinsic pontine gliomas: A retrospective multicenter study and review of the literature. Child Nerv. Syst. 2019, 35, 83–89. [Google Scholar] [CrossRef] [PubMed]
- Fried, I.; Lossos, A.; Ben Ami, T.; Dvir, R.; Toledano, H.; Ben Arush, M.W.; Postovski, S.; Abu Kuidar, A.; Yalon, M.; Weintraub, M.; et al. Preliminary results of immune modulating antibody MDV9300 (pidilizumab) treatment in children with diffuse intrinsic pontine glioma. J. Neurooncol. 2018, 136, 189–195. [Google Scholar] [CrossRef]
- Chen, Y.; Zhao, C.; Li, S.; Wang, J.; Zhang, H. Immune Microenvironment and Immunotherapies for Diffuse Intrinsic Pontine Glioma. Cancers 2023, 15, 602. [Google Scholar] [CrossRef] [PubMed]
- Mishra, D.; Rajendran, S.; Zhu, X.; Nazzaro, M.; Kumar, S.S.; Mchugh, T.; Rajappa, P.; Drissi, R. DIPG-16. Evaluation of myeloid component of DIPG microenvironment. Neuro. Oncol. 2022, 24, i21. [Google Scholar] [CrossRef]
- Bausart, M.; Préat, V.; Malfanti, A. Immunotherapy for glioblastoma: The promise of combination strategies. J. Exp. Clin. Cancer Res. 2022, 41, 35. [Google Scholar] [CrossRef] [PubMed]
- Mount, C.W.; Majzner, R.G.; Sundaresh, S.; Arnold, E.P.; Kadapakkam, M.; Haile, S.; Labanieh, L.; Hulleman, E.; Woo, P.J.; Rietberg, S.P.; et al. Potent antitumor efficacy of anti-GD2 CAR T cells in H3-K27M+ diffuse midline gliomas letter. Nat. Med. 2018, 24, 572–579. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Qiu, Y.; Zhang, P.; Chen, Z.; Chen, S.; Huang, W.; Wang, B.; Yu, X.; Guo, D. Immunogenic Cell Death Enhances Immunotherapy of Diffuse Intrinsic Pontine Glioma: From Preclinical to Clinical Studies. Pharmaceutics 2022, 14, 1762. [Google Scholar] [CrossRef]
- Majzner, R.G.; Ramakrishna, S.; Yeom, K.W.; Patel, S.; Chinnasamy, H.; Schultz, L.M.; Richards, R.M.; Jiang, L.; Barsan, V.; Mancusi, R.; et al. GD2-CAR T cell therapy for H3K27M-mutated diffuse midline gliomas. Nature 2022, 603, 934–941. [Google Scholar] [CrossRef]
- Ruiz-López, E.; Schuhmacher, A.J. Transportation of Single-Domain Antibodies through the Blood-Brain Barrier. Biomolecules 2021, 11, 1131. [Google Scholar] [CrossRef]
- Ruiz-López, E.; Jovčevska, I.; González-Gómez, R.; Tejero, H.; Al-Shahrour, F.; Muyldermans, S.; Schuhmacher, A.J. Nanobodies targeting ABCC3 for immunotargeted applications in glioblastoma. Sci. Rep. 2022, 12, 22581. [Google Scholar] [CrossRef]
- Ruiz-López, E.; Calatayud-Pérez, J.; Castells-Yus, I.; Gimeno-Peribáñez, M.J.; Mendoza-Calvo, N.; Morcillo, M.Á.; Schuhmacher, A.J. Diagnosis of Glioblastoma by Immuno-Positron Emission Tomography. Cancers 2021, 14, 74. [Google Scholar] [CrossRef] [PubMed]
- Pothin, E.; Lesuisse, D.; Lafaye, P. Brain Delivery of Single-Domain Antibodies: A Focus on VHH and VNAR. Pharmaceutics 2020, 12, 937. [Google Scholar] [CrossRef] [PubMed]
| Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).