The Impact of Microbiota on the Gut–Brain Axis: Examining the Complex Interplay and Implications
Abstract
1. Introduction
2. Materials and Methods
2.1. Microbiota
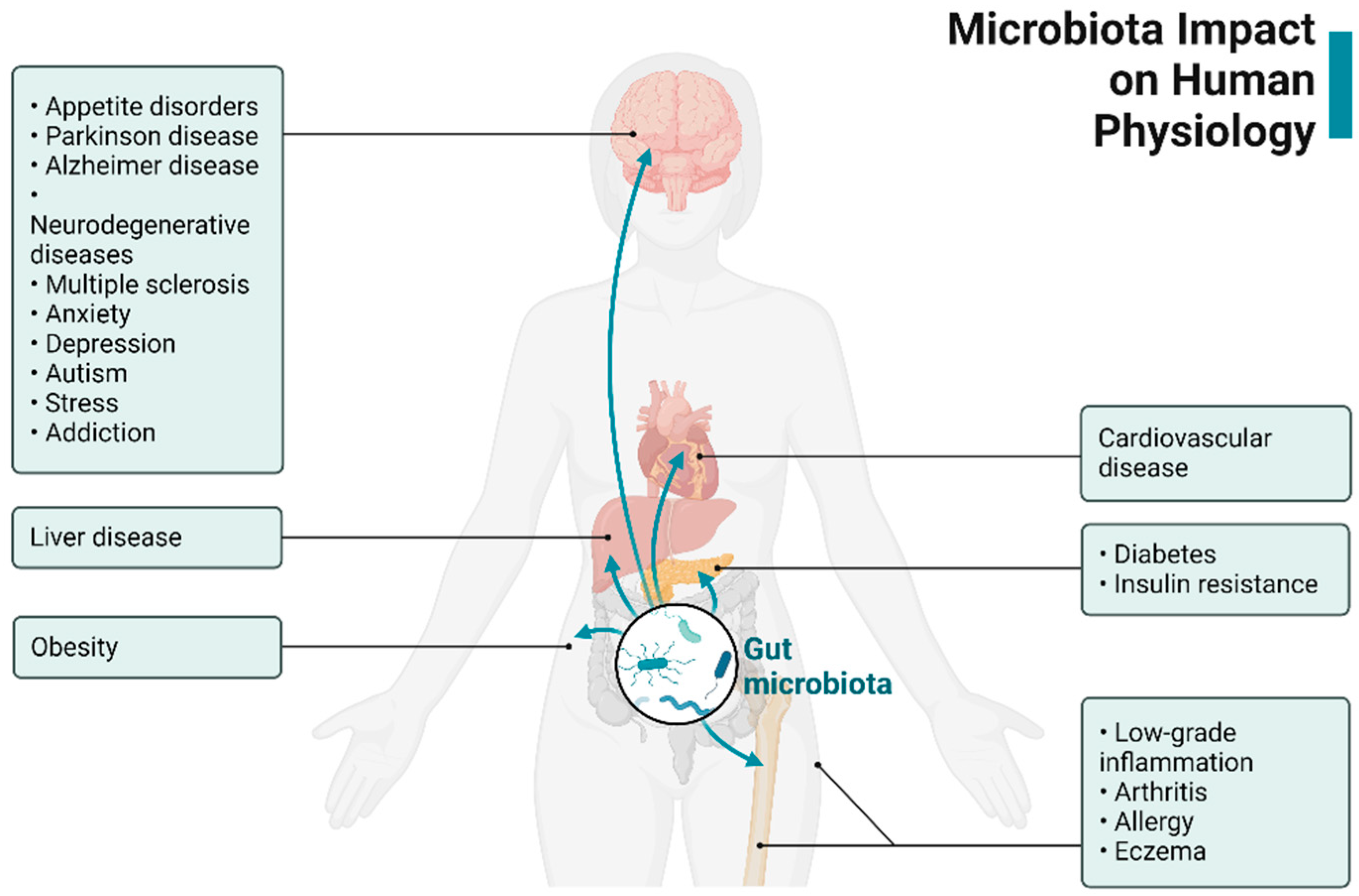
2.1.1. Microbiota and Its Influence on ENS and CNS
2.1.2. Microbiota and CNS
2.1.3. Microbiota and ENS
2.2. CNS Disorder and Their Gi Consequences
2.2.1. Neurodegenerative Disorders and Involvement of ENS
2.2.2. Serotonin
2.2.3. Autism and Serotonin
2.2.4. Nutrients and Cognition
2.3. GI Diseases and Their Associations with CNS
2.3.1. Inflammatory Bowel Disease and CNS Manifestations
2.3.2. Irritable Bowel Syndrome and ENS
2.3.3. Sirtuins-Interlinking ENS, CNS and Beyond
2.3.4. Gastrointestinal Functional Disorder and MOOD Disorders
2.3.5. Stress-Induced GI Disorders
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rhee, S.H.; Pothoulakis, C.; Mayer, E.A. Principles and clinical implications of the brain–gut–enteric microbiota axis. Nat. Rev. Gastroenterol. Hepatol. 2009, 6, 306–314. [Google Scholar] [CrossRef]
- Cryan, J.F.; O’Riordan, K.J.; Sandhu, K.; Peterson, V.; Dinan, T.G. The gut microbiome in neurological disorders. Lancet Neurol. 2020, 19, 179–194. [Google Scholar] [CrossRef]
- Strandwitz, P. Neurotransmitter modulation by the gut microbiota. Brain Res. 2018, 1693, 128–133. [Google Scholar] [CrossRef] [PubMed]
- Nezami, B.G.; Srinivasan, S. Enteric Nervous System in the Small Intestine: Pathophysiology and Clinical Implications. Curr. Gastroenterol. Rep. 2010, 12, 358–365. [Google Scholar] [CrossRef] [PubMed]
- Baetge, G.; Gershon, M.D. Transient catecholaminergic (TC) cells in the vagus nerves and bowel of fetal mice: Relationship to the development of enteric neurons. Dev. Biol. 1989, 132, 189–211. [Google Scholar] [CrossRef]
- Rao, M.; Gershon, M.D. The bowel and beyond: The enteric nervous system in neurological disorders. Nat. Rev. Gastroenterol. Hepatol. 2016, 13, 517–528. [Google Scholar] [CrossRef] [PubMed]
- Gianino, S.; Grider, J.R.; Cresswell, J.; Enomoto, H.; Heuckeroth, R.O. GDNF availability determines enteric neuron number by controlling precursor proliferation. Development 2003, 130, 2187–2198. [Google Scholar] [CrossRef]
- Stoddard, C.J. Current concepts of gastrointestinal motility and electrical activity. Br. J. Hosp. Med. 1978, 20, 426+428–434. [Google Scholar]
- Macia, L.; Tan, J.; Vieira, A.T.; Leach, K.; Stanley, D.; Luong, S.; Maruya, M.; McKenzie, C.L.; Hijikata, A.; Wong, C.; et al. Metabolite-sensing receptors GPR43 and GPR109A facilitate dietary fibre-induced gut homeostasis through regulation of the inflammasome. Nat. Commun. 2015, 6, 6734. [Google Scholar] [CrossRef] [PubMed]
- Carabotti, M.; Scirocco, A.; Maselli, M.A.; Severi, C. The gut-brain axis: Interactions between enteric microbiota, central and enteric nervous systems. Ann. Gastroenterol. 2015, 28, 203–209. [Google Scholar] [PubMed]
- International MNTM. Gut Microbiota: Definition, Importance, and Medical Uses. Available online: https://www.medicalnewstoday.com/articles/307998 (accessed on 8 March 2023).
- Heiss, C.N.; Olofsson, L.E. The role of the gut microbiota in development, function and disorders of the central nervous system and the enteric nervous system. J. Neuroendocr. 2019, 31, e12684. [Google Scholar] [CrossRef]
- Sharon, G.; Sampson, T.R.; Geschwind, D.H.; Mazmanian, S.K. The Central Nervous System and the Gut Microbiome. Cell 2016, 167, 915–932. [Google Scholar] [CrossRef]
- Calvo-Barreiro, L.; Eixarch, H.; Montalban, X.; Espejo, C. Combined therapies to treat complex diseases: The role of the gut microbiota in multiple sclerosis. Autoimmun. Rev. 2018, 17, 165–174. [Google Scholar] [CrossRef] [PubMed]
- Gong, X.; Liu, X.; Li, C.; Chen, C.; Lin, J.; Li, A.; An, D.; Zhou, D.; Hong, Z. Alterations in the human gut microbiome in anti-N-methyl-D-aspartate receptor encephalitis. Ann. Clin. Transl. Neurol. 2019, 6, 1771–1781. [Google Scholar] [CrossRef] [PubMed]
- Rutsch, A.; Kantsjö, J.B.; Ronchi, F. The Gut-Brain Axis: How Microbiota and Host Inflammasome Influence Brain Physiology and Pathology. Front. Immunol. 2020, 11, 604179. [Google Scholar] [CrossRef]
- Ma, Q.; Xing, C.; Long, W.; Wang, H.Y.; Liu, Q.; Wang, R.-F. Impact of microbiota on central nervous system and neurological diseases: The gut-brain axis. J. Neuroinflamm. 2019, 16, 53. [Google Scholar] [CrossRef]
- Boulangé, C.L.; Neves, A.L.; Chilloux, J.; Nicholson, J.K.; Dumas, M.-E. Impact of the gut microbiota on inflammation, obesity, and metabolic disease. Genome Med. 2016, 8, 42. [Google Scholar] [CrossRef]
- Morrison, D.J.; Preston, T. Formation of short chain fatty acids by the gut microbiota and their impact on human metabolism. Gut Microbes 2016, 7, 189–200. [Google Scholar] [CrossRef]
- Furness, J.B. The enteric nervous system and neurogastroenterology. Nat. Rev. Gastroenterol. Hepatol. 2012, 9, 286–294. [Google Scholar] [CrossRef]
- Furness, J.B.; Callaghan, B.P.; Rivera, L.R.; Cho, H.J. The enteric nervous system and gastrointestinal innervation: Integrated local and central control. Adv. Exp. Med. Biol. 2014, 817, 39–71. [Google Scholar] [PubMed]
- Grabauskas, G.; Owyang, C. Plasticity of vagal afferent signaling in the gut. Medicina 2017, 53, 73–84. [Google Scholar] [CrossRef]
- Bauer, P.V.; Hamr, S.C.; Duca, F.A. Regulation of energy balance by a gut–brain axis and involvement of the gut microbiota. Cell. Mol. Life Sci. 2016, 73, 737–755. [Google Scholar] [CrossRef]
- Dockray, G.J. Enteroendocrine cell signalling via the vagus nerve. Curr. Opin. Pharmacol. 2013, 13, 954–958. [Google Scholar] [CrossRef] [PubMed]
- Park, H.-J.; Lee, S.-E.; Kim, H.-B.; Isaacson, R.; Seo, K.-W.; Song, K.-H. Association of Obesity with Serum Leptin, Adiponectin, and Serotonin and Gut Microflora in Beagle Dogs. J. Vet. Intern. Med. 2015, 29, 43–50. [Google Scholar] [CrossRef]
- Ge, X.; Pan, J.; Liu, Y.; Wang, H.; Zhou, W.; Wang, X. Intestinal Crosstalk between Microbiota and Serotonin and its Impact on Gut Motility. Curr. Pharm. Biotechnol. 2018, 19, 190–195. [Google Scholar] [CrossRef]
- de La Serre, C.B.; de Lartigue, G.; Raybould, H.E. Chronic exposure to Low dose bacterial lipopolysaccharide inhibits leptin signaling in vagal afferent neurons. Physiol. Behav. 2015, 139, 188–194. [Google Scholar] [CrossRef] [PubMed]
- Saad, M.J.A.; Santos, A.; Prada, P.O. Linking Gut Microbiota and Inflammation to Obesity and Insulin Resistance. Physiology 2016, 31, 283–293. [Google Scholar] [CrossRef] [PubMed]
- Chelakkot, C.; Ghim, J.; Ryu, S.H. Mechanisms regulating intestinal barrier integrity and its pathological implications. Exp. Mol. Med. 2018, 50, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Logsdon, A.F.; Erickson, M.A.; Rhea, E.M.; Salameh, T.S.; Banks, W.A. Gut reactions: How the blood–brain barrier connects the microbiome and the brain. Exp. Biol. Med. 2018, 243, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Celifarco, A.; Gerard, G.; Faegenburg, D.; Burakoff, R. Dysphagia as the sole manifestation of bilateral strokes. Am. J. Gastroenterol. 1990, 85, 610–613. [Google Scholar] [PubMed]
- Buchholz, D.W. Dysphagia associated with neurological disorders. Acta Otorhinolaryngol. Belg. 1994, 48, 143–155. [Google Scholar]
- Panebianco, M.; Marchese-Ragona, R.; Masiero, S.; Restivo, D.A. Dysphagia in neurological diseases: A literature review. Neurol. Sci. 2020, 41, 3067–3073. [Google Scholar] [CrossRef] [PubMed]
- Wiles, C.M. Neurogenic dysphagia. J. Neurol. Neurosurg. Psychiatry 1991, 54, 1037–1039. [Google Scholar] [CrossRef]
- Emmanuel, A. Neurogenic bowel dysfunction. F1000Research 2019, 8, 1800. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Yuan, M.; Liu, Y.; Zhao, Y.; Wang, J.; Guo, W. Incidence of constipation in stroke patients: A systematic review and meta-analysis. Medicine 2017, 96, e7225. [Google Scholar] [CrossRef]
- Winge, K.; Rasmussen, D.; Werdelin, L.M. Constipation in neurological diseases. J. Neurol. Neurosurg. Psychiatry 2003, 74, 13–19. [Google Scholar] [CrossRef]
- de Arce, E.P.; Landskron, G.; Hirsch, S.; Defilippi, C.; Madrid, A.M. Chronic Intestinal Pseudo-obstruction: Clinical and Manometric Characteristics in the Chilean Population. J. Neurogastroenterol. Motil. 2017, 23, 273–280. [Google Scholar] [CrossRef][Green Version]
- Lim, J.; Ashmore, D.; Oommen, C. A Rare Case of Chronic Small Bowel Pseudo-Obstruction. Cureus 2020, 12, e8003. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.-J.; Na, I.-H.; Choi, E.-S.; Jung, S.-H.; Yoon, J.-S. Occurrence of Intestinal Pseudo-obstruction in a Brainstem Hemorrhage Patient. Ann. Rehabil. Med. 2012, 36, 278–281. [Google Scholar] [CrossRef]
- Ould-Ahmed, M.; de Saint-Martin, L.; Ansart, S.; Renault, A.; Boles, J.M. Acute colonic pseudo-obstruction (Ogilvie’s syndrome) in pneumococcal meningo-encephalitis treated with neostigmine. Ann. Fr. Anesth. Reanim. 2003, 22, 900–903. [Google Scholar] [CrossRef]
- Intestinal Pseudo-Obstruction: StatPearls. Available online: https://www.ncbi.nlm.nih.gov/books/NBK560669/ (accessed on 8 March 2023).
- Brunner, H.G.; Hamel, B.C.; Rieu, P.; Howeler, C.J.; Peters, F.T. Intestinal pseudo-obstruction in myotonic dystrophy. J. Med. Genet. 1992, 29, 791–793. [Google Scholar] [CrossRef] [PubMed]
- Antonucci, A.; Fronzoni, L.; Cogliandro, L.; Cogliandro, R.F.; Caputo, C.; De Giorgio, R.; Pallotti, F.; Barbara, G.; Corinaldesi, R.; Stanghellini, V. Chronic intestinal pseudo-obstruction. World J. Gastroenterol. 2008, 14, 2953–2961. [Google Scholar] [CrossRef]
- Grover, M.; Farrugia, G.; Stanghellini, V. Gastroparesis: A turning point in understanding and treatment. Gut 2019, 68, 2238–2250. [Google Scholar] [CrossRef]
- Bharucha, A.E.; Kudva, Y.C.; Prichard, D.O. Diabetic Gastroparesis. Endocr. Rev. 2019, 40, 1318–1352. [Google Scholar] [CrossRef] [PubMed]
- Usai-Satta, P.; Bellini, M.; Morelli, O.; Geri, F.; Lai, M.; Bassotti, G. Gastroparesis: New insights into an old disease. World J. Gastroenterol. 2020, 26, 2333–2348. [Google Scholar] [CrossRef]
- Natale, G.; Pasquali, L.; Ruggieri, S.; Paparelli, A.; Fornai, F. Parkinson’s disease and the gut: A well known clinical association in need of an effective cure and explanation. Neurogastroenterol. Motil. 2008, 20, 741–749. [Google Scholar] [CrossRef] [PubMed]
- Natale, G.; Pasquali, L.; Paparelli, A.; Fornai, F. Parallel manifestations of neuropathologies in the enteric and central nervous systems. Neurogastroenterol. Motil. 2011, 23, 1056–1065. [Google Scholar] [CrossRef]
- Chung, K.A.; Pfeiffer, R.F. Gastrointestinal dysfunction in the synucleinopathies. Clin. Auton. Res. 2021, 31, 77–99. [Google Scholar] [CrossRef]
- Schaeffer, E.; Kluge, A.; Böttner, M.; Zunke, F.; Cossais, F.; Berg, D.; Arnold, P. Alpha Synuclein Connects the Gut-Brain Axis in Parkinson’s Disease Patients—A View on Clinical Aspects, Cellular Pathology and Analytical Methodology. Front. Cell Dev. Biol. 2020, 8, 573696. [Google Scholar] [CrossRef]
- Qualman, S.J.; Haupt, H.M.; Yang, P.; Hamilton, S.R. Esophageal Lewy bodies associated with ganglion cell loss in achalasia. Similarity to Parkinson’s disease. Gastroenterology 1984, 87, 848–856. [Google Scholar] [CrossRef]
- Lebouvier, T.; Chaumette, T.; Damier, P.; Coron, E.; Touchefeu, Y.; Vrignaud, S.; Naveilhan, P.; Galmiche, J.-P.; des Varannes, S.B.; Derkinderen, P.; et al. Pathological lesions in colonic biopsies during Parkinson’s disease. Gut 2008, 57, 1741–1743. [Google Scholar] [CrossRef] [PubMed]
- Ohlsson, B.; Englund, E. Atrophic Myenteric and Submucosal Neurons Are Observed in Parkinson’s Disease. Park. Dis. 2019, 2019, 7935820. [Google Scholar] [CrossRef]
- Fenyi, A.; Duyckaerts, C.; Bousset, L.; Braak, H.; Tredici, K.; Melki, R.; The Brainbank Neuro-Ceb Neuropathology Network. Seeding Propensity and Characteristics of Pathogenic αSyn Assemblies in Formalin-Fixed Human Tissue from the Enteric Nervous System, Olfactory Bulb, and Brainstem in Cases Staged for Parkinson’s Disease. Cells 2021, 10, 139. [Google Scholar] [CrossRef]
- Killinger, B.; Labrie, V. The Appendix in Parkinson’s Disease: From Vestigial Remnant to Vital Organ? J. Park. Dis. 2019, 9 (Suppl. S2), S345–S358. [Google Scholar] [CrossRef] [PubMed]
- Phillips, R.J.; Walter, G.C.; Ringer, B.E.; Higgs, K.M.; Powley, T.L. Alpha-synuclein immunopositive aggregates in the myenteric plexus of the aging Fischer 344 rat. Exp. Neurol. 2009, 220, 109–119. [Google Scholar] [CrossRef]
- Clairembault, T.; Kamphuis, W.; Leclair-Visonneau, L.; Rolli-Derkinderen, M.; Coron, E.; Neunlist, M.; Hol, E.M.; Derkinderen, P. Enteric GFAP expression and phosphorylation in Parkinson’s disease. J. Neurochem. 2014, 130, 805–815. [Google Scholar] [CrossRef] [PubMed]
- Clairembault, T.; Leclair-Visonneau, L.; Neunlist, M.; Derkinderen, P. Enteric glial cells: New players in Parkinson’s disease? Mov. Disord. 2015, 30, 494–498. [Google Scholar] [CrossRef] [PubMed]
- Benvenuti, L.; D’antongiovanni, V.; Pellegrini, C.; Antonioli, L.; Bernardini, N.; Blandizzi, C.; Fornai, M. Enteric Glia at the Crossroads between Intestinal Immune System and Epithelial Barrier: Implications for Parkinson Disease. Int. J. Mol. Sci. 2020, 21, 9199. [Google Scholar] [CrossRef] [PubMed]
- Natale, G.; Biagioni, F.; Busceti, C.L.; Gambardella, S.; Limanaqi, F.; Fornai, F. TREM Receptors Connecting Bowel Inflammation to Neurodegenerative Disorders. Cells 2019, 8, 1124. [Google Scholar] [CrossRef]
- Caputi, V.; Giron, M.C. Microbiome-Gut-Brain Axis and Toll-Like Receptors in Parkinson’s Disease. Int. J. Mol. Sci. 2018, 19, 1689. [Google Scholar] [CrossRef]
- Dutta, S.K.; Verma, S.; Jain, V.; Surapaneni, B.K.; Vinayek, R.; Phillips, L.; Nair, P.P. Parkinson’s Disease: The Emerging Role of Gut Dysbiosis, Antibiotics, Probiotics, and Fecal Microbiota Transplantation. J. Neurogastroenterol. Motil. 2019, 25, 363–376. [Google Scholar] [CrossRef] [PubMed]
- Fleming, M.A., 2nd; Ehsan, L.; Moore, S.R.; Levin, D.E. The Enteric Nervous System and Its Emerging Role as a Therapeutic Target. Gastroenterol. Res. Pract. 2020, 2020, 8024171. [Google Scholar] [CrossRef] [PubMed]
- Roth, W.; Zadeh, K.; Vekariya, R.; Ge, Y.; Mohamadzadeh, M. Tryptophan Metabolism and Gut-Brain Homeostasis. Int. J. Mol. Sci. 2021, 22, 2973. [Google Scholar] [CrossRef] [PubMed]
- Young, S.N. How to increase serotonin in the human brain without drugs. J. Psychiatry Neurosci. 2007, 32, 394–399. [Google Scholar]
- Sadalla, S.; Milani, S. The Relationship between the Serotonin Metabolism, Gut-Microbiota and the Gut-Brain Axis. Curr. Drug Metab. 2019, 20, 646–655. [Google Scholar] [CrossRef]
- Berger, M.; Gray, J.A.; Roth, B.L. The Expanded Biology of Serotonin. Annu. Rev. Med. 2009, 60, 355–366. [Google Scholar] [CrossRef]
- Israelyan, N.; Del Colle, A.; Li, Z.; Park, Y.; Xing, A.; Jacobsen, J.P.R.; Luna, R.A.; Jensen, D.D.; Madra, M.; Saurman, V.; et al. Effects of Serotonin and Slow-Release 5-Hydroxytryptophan on Gastrointestinal Motility in a Mouse Model of Depression. Gastroenterology 2019, 157, 507–521.e4. [Google Scholar] [CrossRef] [PubMed]
- Pourhamzeh, M.; Moravej, F.G.; Arabi, M.; Shahriari, E.; Mehrabi, S.; Ward, R.; Ahadi, R.; Joghataei, M.T. The Roles of Serotonin in Neuropsychiatric Disorders. Cell. Mol. Neurobiol. 2022, 42, 1671–1692. [Google Scholar] [CrossRef] [PubMed]
- Chugani, D.C. Serotonin in autism and pediatric epilepsies. Ment. Retard. Dev. Disabil. Res. Rev. 2004, 10, 112–116. [Google Scholar] [CrossRef]
- Leshem, R.; Bar-Oz, B.; Diav-Citrin, O.; Gbaly, S.; Soliman, J.; Renoux, C.; Matok, I. Selective Serotonin Reuptake Inhibitors (SSRIs) and Serotonin Norepinephrine Reuptake Inhibitors (SNRIs) During Pregnancy and the Risk for Autism spectrum disorder (ASD) and Attention deficit hyperactivity disorder (ADHD) in the Offspring: A True Effect or a Bias? A Systematic Review & Meta-Analysis. Curr. Neuropharmacol. 2021, 19, 896–906. [Google Scholar] [CrossRef]
- Muller, C.; Anacker, A.; Veenstra-VanderWeele, J. The serotonin system in autism spectrum disorder: From biomarker to animal models. Neuroscience 2016, 321, 24–41. [Google Scholar] [CrossRef] [PubMed]
- Anderson, B.M.; Schnetz-Boutaud, N.C.; Bartlett, J.; Wotawa, A.M.; Wright, H.H.; Abramson, R.K.; Cuccaro, M.L.; Gilbert, J.R.; Pericak-Vance, M.A.; Haines, J.L. Examination of association of genes in the serotonin system to autism. Neurogenetics 2009, 10, 209–216. [Google Scholar] [CrossRef]
- Devlin, B.; Cook, E.H.; Coon, H.; Dawson, G.; Grigorenko, E.L.; McMahon, W.; Minshew, N.; Pauls, D.; Smith, M.; Spence, M.A.; et al. Autism and the serotonin transporter: The long and short of it. Mol. Psychiatry 2005, 10, 1110–1116. [Google Scholar] [CrossRef] [PubMed]
- Tordjman, S.; Gutknecht, L.; Carlier, M.; Spitz, E.; Antoine, C.; Slama, F.; Carsalade, V.; Cohen, D.J.; Ferrari, P.; Roubertoux, P.L.; et al. Role of the serotonin transporter gene in the behavioral expression of autism. Mol. Psychiatry 2001, 6, 434–439. [Google Scholar] [CrossRef]
- Polverino, A.; Sorrentino, P.; Pesoli, M.; Mandolesi, L. Nutrition and cognition across the lifetime: An overview on epigenetic mechanisms. AIMS Neurosci. 2021, 8, 448–476. [Google Scholar] [CrossRef] [PubMed]
- Jirout, J.; LoCasale-Crouch, J.; Turnbull, K.; Gu, Y.; Cubides, M.; Garzione, S.; Evans, T.M.; Weltman, A.L.; Kranz, S. How Lifestyle Factors Affect Cognitive and Executive Function and the Ability to Learn in Children. Nutrients 2019, 11, 1953. [Google Scholar] [CrossRef] [PubMed]
- Cutuli, D. Functional and Structural Benefits Induced by Omega-3 Polyunsaturated Fatty Acids During Aging. Curr. Neuropharmacol. 2017, 15, 534–542. [Google Scholar] [CrossRef]
- Murphy, T.; Dias, G.P.; Thuret, S. Effects of Diet on Brain Plasticity in Animal and Human Studies: Mind the Gap. Neural Plast. 2014, 2014, 563160. [Google Scholar] [CrossRef]
- Delgado-Morales, R.; Agís-Balboa, R.C.; Esteller, M.; Berdasco, M. Epigenetic mechanisms during ageing and neurogenesis as novel therapeutic avenues in human brain disorders. Clin. Epigenet. 2017, 9, 67. [Google Scholar] [CrossRef]
- Sasaki, H.; Matsuzaki, Y.; Meguro, K.; Ikarashi, Y.; Maruyama, Y.; Yamaguchi, S.; Sekizawa, K. Vitamin B12 improves cognitive disturbance in rodents fed a choline-deficient diet. Pharmacol. Biochem. Behav. 1992, 43, 635–639. [Google Scholar] [CrossRef]
- Bryan, J.; Calvaresi, E.; Hughes, D. Short-Term Folate, Vitamin B-12 or Vitamin B-6 Supplementation Slightly Affects Memory Performance But Not Mood in Women of Various Ages. J. Nutr. 2002, 132, 1345–1356. [Google Scholar] [CrossRef] [PubMed]
- Vogiatzoglou, A.; Refsum, H.; Johnston, C.; Smith, S.M.; Bradley, K.M.; de Jager, C.; Budge, M.M.; Smith, A.D. Vitamin B12 status and rate of brain volume loss in community-dwelling elderly. Neurology 2008, 71, 826–832. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.C.; Tyagi, A.K.; Deshmukh-Taskar, P.; Hinojosa, M.; Prasad, S.; Aggarwal, B.B. Downregulation of tumor necrosis factor and other proinflammatory biomarkers by polyphenols. Arch. Biochem. Biophys. 2014, 559, 91–99. [Google Scholar] [CrossRef]
- Soubry, A. POHaD: Why we should study future fathers. Environ. Epigenet. 2018, 4, dvy007. [Google Scholar] [CrossRef]
- Morís, G. Inflammatory bowel disease: An increased risk factor for neurologic complications. World J. Gastroenterol. 2014, 20, 1228–1237. [Google Scholar] [CrossRef]
- Nemati, R.; Mehdizadeh, S.; Salimipour, H.; Yaghoubi, E.; Alipour, Z.; Tabib, S.M.; Assadi, M. Neurological manifestations related to Crohn’s disease: A boon for the workforce. Gastroenterol. Rep. 2019, 7, 291–297. [Google Scholar] [CrossRef]
- Ferro, J.M. Neurologic Manifestations of Inflammatory Bowel Disease. Gastroenterol. Hepatol. 2014, 10, 599–600. [Google Scholar]
- Gershon, M.D. Nerves, reflexes, and the enteric nervous system: Pathogenesis of the irritable bowel syndrome. J. Clin. Gastroenterol. 2005, 39 (Suppl. S3), S184–S193. [Google Scholar] [CrossRef]
- Chandramowlishwaran, P.; Vijay, A.; Abraham, D.; Li, G.; Mwangi, S.M.; Srinivasan, S. Role of Sirtuins in Modulating Neurodegeneration of the Enteric Nervous System and Central Nervous System. Front. Neurosci. 2020, 14, 614331. [Google Scholar] [CrossRef]
- Khoury, N.; Koronowski, K.B.; Young, J.I.; Perez-Pinzon, M.A. The NAD+-Dependent Family of Sirtuins in Cerebral Ischemia and Preconditioning. Antioxid. Redox Signal. 2018, 28, 691–710. [Google Scholar] [CrossRef]
- Lakhan, S.E.; Kirchgessner, A. Gut microbiota and sirtuins in obesity-related inflammation and bowel dysfunction. J. Transl. Med. 2011, 9, 202. [Google Scholar] [CrossRef] [PubMed]
- Del Colle, A.; Israelyan, N.; Gross Margolis, K. Novel aspects of enteric serotonergic signaling in health and brain-gut disease. Am. J. Physiol. Gastrointest. Liver Physiol. 2020, 318, G130–G143. [Google Scholar] [CrossRef] [PubMed]
- Terry, N.; Margolis, K.G. Serotonergic Mechanisms Regulating the GI Tract: Experimental Evidence and Therapeutic Relevance. Handb. Exp. Pharmacol. 2017, 239, 319–342. [Google Scholar] [CrossRef] [PubMed]
- Gaspar, P.; Cases, O.; Maroteaux, L. The developmental role of serotonin: News from mouse molecular genetics. Nat. Rev. Neurosci. 2003, 4, 1002–1012. [Google Scholar] [CrossRef]
- Li, Z.; Chalazonitis, A.; Huang, Y.-Y.; Mann, J.J.; Margolis, K.G.; Yang, Q.M.; Kim, D.O.; Côté, F.; Mallet, J.; Gershon, M.D. Essential Roles of Enteric Neuronal Serotonin in Gastrointestinal Motility and the Development/Survival of Enteric Dopaminergic Neurons. J. Neurosci. 2011, 31, 8998–9009. [Google Scholar] [CrossRef]
- Suri, D.; Teixeira, C.M.; Cagliostro, M.K.C.; Mahadevia, D.; Ansorge, M.S. Monoamine-Sensitive Developmental Periods Impacting Adult Emotional and Cognitive Behaviors. Neuropsychopharmacology 2015, 40, 88–112. [Google Scholar] [CrossRef]
- McElhanon, B.O.; McCracken, C.; Karpen, S.; Sharp, W.G. Gastrointestinal Symptoms in Autism Spectrum Disorder: A Meta-analysis. Pediatrics 2014, 133, 872–883. [Google Scholar] [CrossRef]
- Margolis, K.G.; Buie, T.M.; Turner, J.B.; Silberman, A.E.; Feldman, J.F.; Murray, K.F.; McSwiggan-Hardin, M.; Levy, J.; Bauman, M.L.; Veenstra-VanderWeele, J.; et al. Development of a Brief Parent-Report Screen for Common Gastrointestinal Disorders in Autism Spectrum Disorder. J. Autism Dev. Disord. 2019, 49, 349–362. [Google Scholar] [CrossRef]
- Wigner, P.; Czarny, P.; Synowiec, E.; Bijak, M.; Białek, K.; Talarowska, M.; Galecki, P.; Szemraj, J.; Sliwinski, T. Association between single nucleotide polymorphisms of TPH1 and TPH2 genes, and depressive disorders. J. Cell. Mol. Med. 2018, 22, 1778–1791. [Google Scholar] [CrossRef]
- Tsai, S.-J.; Hong, C.-J.; Liou, Y.-J.; Yu, Y.W.-Y.; Chen, T.-J.; Hou, S.-J.; Yen, F.-C. Tryptophan hydroxylase 2 gene is associated with major depression and antidepressant treatment response. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2009, 33, 637–641. [Google Scholar] [CrossRef]
- Gutknecht, L.; Jacob, C.; Strobel, A.; Kriegebaum, C.; Müller, J.; Zeng, Y.; Markert, C.; Escher, A.; Wendland, J.; Reif, A.; et al. Tryptophan hydroxylase-2 gene variation influences personality traits and disorders related to emotional dysregulation. Int. J. Neuropsychopharmacol. 2007, 10, 309–320. [Google Scholar] [CrossRef] [PubMed]
- Kim, P.; Gadani, A.; Abdul-Baki, H.; Mitre, R.; Mitre, M. Fecal microbiota transplantation in recurrent Clostridium difficile infection: A retrospective single-center chart review. JGH Open 2019, 3, 4–9. [Google Scholar] [CrossRef]
- Hosseinzadeh, S.T.; Poorsaadati, S.; Radkani, B.; Forootan, M. Psychological disorders in patients with chronic constipation. Gastroenterol. Hepatol. Bed Bench 2011, 4, 159–163. [Google Scholar]
- Zhang, X.; Gainetdinov, R.R.; Beaulieu, J.-M.; Sotnikova, T.D.; Burch, L.H.; Williams, R.B.; Schwartz, D.A.; Krishnan, K.R.R.; Caron, M.G. Loss-of-Function Mutation in Tryptophan Hydroxylase-2 Identified in Unipolar Major Depression. Neuron 2005, 45, 11–16. [Google Scholar] [CrossRef] [PubMed]
- Huerta-Franco, M.R.; Vargas-Luna, M.; Tienda, P.; Delgadillo-Holtfort, I.; Balleza-Ordaz, M.; Flores-Hernandez, C. Effects of occupational stress on the gastrointestinal tract. World J. Gastrointest. Pathophysiol. 2013, 4, 108–118. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.P.; Sung, I.-K.; Kim, J.H.; Lee, S.-Y.; Park, H.S.; Shim, C.S. The Effect of Emotional Stress and Depression on the Prevalence of Digestive Diseases. J. Neurogastroenterol. Motil. 2015, 21, 273–282. [Google Scholar] [CrossRef] [PubMed]
- Yaribeygi, H.; Panahi, Y.; Sahraei, H.; Johnston, T.P.; Sahebkar, A. The impact of stress on body function: A review. EXCLI J. 2017, 16, 1057–1072. [Google Scholar] [CrossRef]
- Mayer, E.A. The neurobiology of stress and gastrointestinal disease. Gut 2000, 47, 861–869. [Google Scholar] [CrossRef]
- Konturek, P.C.; Brzozowski, T.; Konturek, S.J. Stress and the gut: Pathophysiology, clinical consequences, diagnostic approach and treatment options. J. Physiol. Pharmacol. 2011, 62, 591–599. [Google Scholar]
- Fikree, A.; Byrne, P. Management of functional gastrointestinal disorders. Clin. Med. 2021, 21, 44–52. [Google Scholar] [CrossRef]
- Meerveld, B.G.-V.; Johnson, A.C. Mechanisms of Stress-induced Visceral Pain. J. Neurogastroenterol. Motil. 2018, 24, 7–18. [Google Scholar] [CrossRef] [PubMed]
- Oshima, T.; Miwa, H. Epidemiology of Functional Gastrointestinal Disorders in Japan and in the World. J. Neurogastroenterol. Motil. 2015, 21, 320–329. [Google Scholar] [CrossRef] [PubMed]
- Lacy, B.E.; Patel, N.K. Rome Criteria and a Diagnostic Approach to Irritable Bowel Syndrome. J. Clin. Med. 2017, 6, 99. [Google Scholar] [CrossRef] [PubMed]
- Francis, P.; Zavala, S.R. Functional Dyspepsia: StatPearls. Available online: https://www.ncbi.nlm.nih.gov/books/NBK554563/ (accessed on 4 June 2023).
- Mawdsley, J.E.; Rampton, D.S. Psychological stress in IBD: New insights into pathogenic and therapeutic implications. Gut 2005, 54, 1481–1491. [Google Scholar] [CrossRef]
- Sun, Y.; Li, L.; Xie, R.; Wang, B.; Jiang, K.; Cao, H. Stress Triggers Flare of Inflammatory Bowel Disease in Children and Adults. Front. Pediatr. 2019, 7, 432. [Google Scholar] [CrossRef]
- Bednarikova, H.; Kascakova, N.; Furstova, J.; Zelinkova, Z.; Falt, P.; Hasto, J.; Tavel, P. Life Stressors in Patients with Inflammatory Bowel Disease: Comparison with a Population-Based Healthy Control Group in the Czech Republic. Int. J. Environ. Res. Public Health 2021, 18, 3237. [Google Scholar] [CrossRef]
- Shen, X.; Yang, H.; Wu, Y.; Zhang, D.; Jiang, H. Meta-analysis: Association of Helicobacter pylori infection with Parkinson’s diseases. Helicobacter 2017, 22, e12398. [Google Scholar]
- Fasano, A.; Bove, F.; Gabrielli, M.; Petracca, M.; Zocco, M.A.; Ragazzoni, E.; Barbaro, F.; Piano, C.; Fortuna, S.; Tortora, A.; et al. The role of small intestinal bacterial overgrowth in Parkinson’s disease. Mov. Disord. 2013, 28, 1241–1249. [Google Scholar] [CrossRef]
- Quigley, E.M.M. Microbiota-Brain-Gut Axis and Neurodegenerative Diseases. Curr. Neurol. Neurosci. Rep. 2017, 17, 94. [Google Scholar] [CrossRef]
- Sun, M.-F.; Shen, Y.-Q. Dysbiosis of gut microbiota and microbial metabolites in Parkinson’s Disease. Ageing Res. Rev. 2018, 45, 53–61. [Google Scholar] [CrossRef]
- Keane, R.W.; Dietrich, W.D.; de Rivero Vaccari, J.P. Inflammasome Proteins as Biomarkers of Multiple Sclerosis. Front. Neurol. 2018, 9, 135. [Google Scholar] [CrossRef]
- Inoue, M.; Shinohara, M.L. The role of interferon-β in the treatment of multiple sclerosis and experimental autoimmune encephalomyelitis—In the perspective of inflammasomes. Immunology 2013, 139, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Malaguarnera, L.; Motta, M.; Di Rosa, M.; Anzaldi, M.; Malaguarnera, M. Interleukin-18 and transforming growth factor-beta 1 plasma levels in Alzheimer’s disease and vascular dementia. Neuropathology 2006, 26, 307–312. [Google Scholar] [CrossRef] [PubMed]
- Saresella, M.; La Rosa, F.; Piancone, F.; Zoppis, M.; Marventano, I.; Calabrese, E.; Rainone, V.; Nemni, R.; Mancuso, R.; Clerici, M. The NLRP3 and NLRP1 inflammasomes are activated in Alzheimer’s disease. Mol. Neurodegener. 2016, 11, 23. [Google Scholar] [CrossRef] [PubMed]
- Mogi, M.; Harada, M.; Kondo, T.; Riederer, P.; Inagaki, H.; Minami, M.; Nagatsu, T. Interleukin-1 beta, interleukin-6, epidermal growth factor and transforming growth factor-alpha are elevated in the brain from parkinsonian patients. Neurosci. Lett. 1994, 180, 147–150. [Google Scholar] [CrossRef]
- Alcocer-Gómez, E.; Ulecia-Morón, C.; Marín-Aguilar, F.; Rybkina, T.; Casas-Barquero, N.; Ruiz-Cabello, J.; Ryffel, B.; Apetoh, L.; Ghiringhelli, F.; Bullón, P.; et al. Stress-Induced Depressive Behaviors Require a Functional NLRP3 Inflammasome. Mol. Neurobiol. 2016, 53, 4874–4882. [Google Scholar] [CrossRef]
- Baroja-Mazo, A.; Martín-Sánchez, F.; Gomez, A.I.; Martínez, C.M.; Amores-Iniesta, J.; Compan, V.; Barberà-Cremades, M.; Yagüe, J.; Ruiz-Ortiz, E.; Antón, J.; et al. The NLRP3 inflammasome is released as a particulate danger signal that amplifies the inflammatory response. Nat. Immunol. 2014, 15, 738–748. [Google Scholar] [CrossRef]
- Collins, S.M. A role for the gut microbiota in IBS. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 497–505. [Google Scholar] [CrossRef]
- El-Salhy, M.; Hatlebakk, J.G.; Hausken, T. Diet in Irritable Bowel Syndrome (IBS): Interaction with Gut Microbiota and Gut Hormones. Nutrients 2019, 11, 1824. [Google Scholar] [CrossRef]
- Ni, J.; Wu, G.D.; Albenberg, L.; Tomov, V.T. Gut microbiota and IBD: Causation or correlation? Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 573–584. [Google Scholar] [CrossRef]
- Koloski, N.A.; Jones, M.; Kalantar, J.; Weltman, M.; Zaguirre, J.; Talley, N.J. The brain–gut pathway in functional gastrointestinal disorders is bidirectional: A 12-year prospective population-based study. Gut 2012, 61, 1284–1290. [Google Scholar] [CrossRef] [PubMed]
- Koloski, N.A.; Jones, M.; Talley, N.J. Evidence that independent gut-to-brain and brain-to-gut pathways operate in the irritable bowel syndrome and functional dyspepsia: A 1-year population-based prospective study. Aliment. Pharmacol. Ther. 2016, 44, 592–600. [Google Scholar] [CrossRef] [PubMed]
- Panara, A.J.; Yarur, A.J.; Rieders, B.; Proksell, S.; Deshpande, A.R.; Abreu, M.T.; Sussman, D.A. The incidence and risk factors for developing depression after being diagnosed with inflammatory bowel disease: A cohort study. Aliment. Pharmacol. Ther. 2014, 39, 802–810. [Google Scholar] [CrossRef]
- Lix, L.M.; Graff, L.A.; Walker, J.R.; Clara, I.; Rawsthorne, P.; Rogala, L.; Miller, N.; Ediger, J.; Pretorius, T.; Bernstein, C.N. Longitudinal study of quality of life and psychological functioning for active, fluctuating, and inactive disease patterns in inflammatory bowel disease. Inflamm. Bowel Dis. 2008, 14, 1575–1584. [Google Scholar] [CrossRef]
- Anne-Sophie Ouellette, C.R.; Lemieux, S.; Tchernof, A.; Biertho, L.; Bégin, C. Diagnostic and Statistical Manual of Mental Disorders Fourth Edition Text Revision (DSM-IV-TR); American Psychiatric Association: Washington, DC, USA, 2000. [Google Scholar] [CrossRef]
- Johnson, J.; Campisi, J.; Sharkey, C.; Kennedy, S.; Nickerson, M.; Greenwood, B.; Fleshner, M. Catecholamines mediate stress-induced increases in peripheral and central inflammatory cytokines. Neuroscience 2005, 135, 1295–1307. [Google Scholar] [CrossRef] [PubMed]
- Farhadi, A.; Keshavarzian, A.; Van de Kar, L.D.; Jakate, S.; Domm, A.; Zhang, L.; Shaikh, M.; Banan, A.; Fields, J.Z. Heightened Responses to Stressors in Patients with Inflammatory Bowel Disease. Am. J. Gastroenterol. 2005, 100, 1796–1804. [Google Scholar] [CrossRef] [PubMed]
- Greenmyer, J.R.; Niaz, T.; Kohorst, M.A.; Stephens, E.H.; Anderson, J.H. Chromogenic Factor X Assay for Monitoring Warfarin Anticoagulation in a Child with a Prosthetic Mitral Valve. Mayo Clin. Proc. Innov. Qual. Outcomes 2021, 5, 811–816. [Google Scholar] [CrossRef]
- Rahimi, H.R.; Shiri, M.; Razmi, A. Antidepressants can treat inflammatory bowel disease through regulation of the nuclear factor-κB/nitric oxide pathway and inhibition of cytokine production: A hypothesis. World J. Gastrointest. Pharmacol. Ther. 2012, 3, 83–85. [Google Scholar] [CrossRef]
- Ford, A.C.; Lacy, B.E.; Harris, L.A.; Quigley, E.M.M.; Moayyedi, P. Effect of Antidepressants and Psychological Therapies in Irritable Bowel Syndrome: An Updated Systematic Review and Meta-Analysis. Am. J. Gastroenterol. 2019, 114, 21–39. [Google Scholar] [CrossRef]
- Gracie, D.J.; Hamlin, P.J.; Ford, A.C. The influence of the brain–gut axis in inflammatory bowel disease and possible implications for treatment. Lancet Gastroenterol. Hepatol. 2019, 4, 632–642. [Google Scholar] [CrossRef]
- Mohajeri, M.H.; La Fata, G.; Steinert, R.E.; Weber, P. Relationship between the gut microbiome and brain function. Nutr. Rev. 2018, 76, 481–496. [Google Scholar] [CrossRef] [PubMed]

| Si No | Disease | Findings |
|---|---|---|
| 1 | Multiple Sclerosis | a. Caspase-1 and ASC (apoptosis-associated speck-like protein) in serum are predictors for the severity of the disease [124]. b. IL-1β, Caspase-1 ↑, IL-18, in CSF and PBMC (Peripheral Blood Mononuclear Cells) [125]. |
| 2 | Alzheimer’s Disease | a. IL-18, Caspase-1, ASC, and IL-1β, in PBMCs. b. IL-1β, IL-18 in astrocytes, microglia, and neurons surrounding Aβ plaques [126,127]. |
| 3 | Parkinson’s disease | a. Caspase-1, IL-1β in the striatum and serum [128]. |
| 4 | Neuropsychiatric disorder | a. Increased activity of NLRP3 inflammasome [129,130,131]. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chaudhry, T.S.; Senapati, S.G.; Gadam, S.; Mannam, H.P.S.S.; Voruganti, H.V.; Abbasi, Z.; Abhinav, T.; Challa, A.B.; Pallipamu, N.; Bheemisetty, N.; et al. The Impact of Microbiota on the Gut–Brain Axis: Examining the Complex Interplay and Implications. J. Clin. Med. 2023, 12, 5231. https://doi.org/10.3390/jcm12165231
Chaudhry TS, Senapati SG, Gadam S, Mannam HPSS, Voruganti HV, Abbasi Z, Abhinav T, Challa AB, Pallipamu N, Bheemisetty N, et al. The Impact of Microbiota on the Gut–Brain Axis: Examining the Complex Interplay and Implications. Journal of Clinical Medicine. 2023; 12(16):5231. https://doi.org/10.3390/jcm12165231
Chicago/Turabian StyleChaudhry, Tuba Shahid, Sidhartha Gautam Senapati, Srikanth Gadam, Hari Priya Sri Sai Mannam, Hima Varsha Voruganti, Zainab Abbasi, Tushar Abhinav, Apurva Bhavana Challa, Namratha Pallipamu, Niharika Bheemisetty, and et al. 2023. "The Impact of Microbiota on the Gut–Brain Axis: Examining the Complex Interplay and Implications" Journal of Clinical Medicine 12, no. 16: 5231. https://doi.org/10.3390/jcm12165231
APA StyleChaudhry, T. S., Senapati, S. G., Gadam, S., Mannam, H. P. S. S., Voruganti, H. V., Abbasi, Z., Abhinav, T., Challa, A. B., Pallipamu, N., Bheemisetty, N., & Arunachalam, S. P. (2023). The Impact of Microbiota on the Gut–Brain Axis: Examining the Complex Interplay and Implications. Journal of Clinical Medicine, 12(16), 5231. https://doi.org/10.3390/jcm12165231






