Early Management of Severe Biliary Infection in the Era of the Tokyo Guidelines
Abstract
1. Introduction
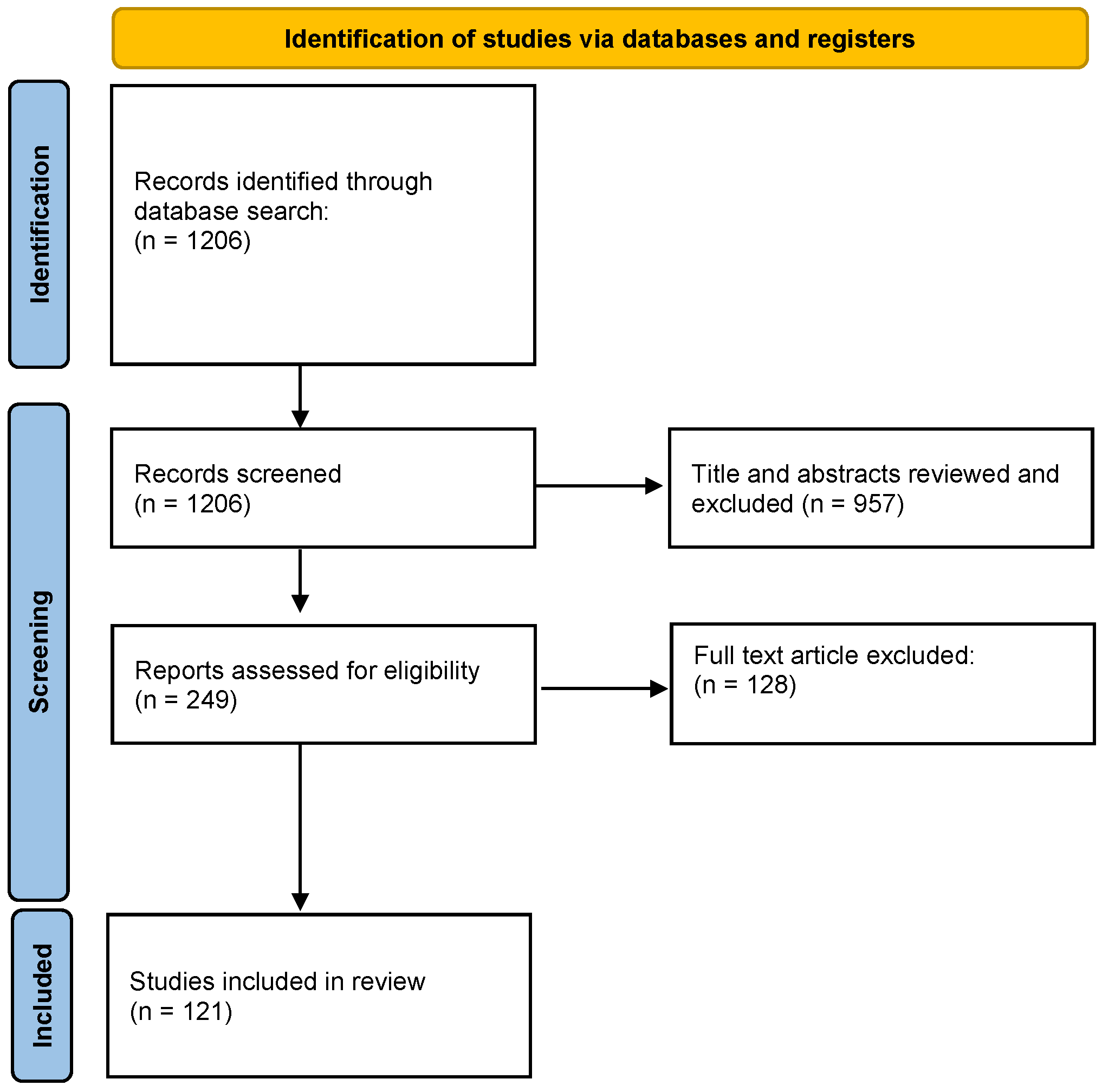
2. Materials and Methods
3. Results
3.1. Early Diagnosis
3.1.1. Acute Calculous Cholecystitis
3.1.2. Acute Cholangitis
3.2. Early Assessment of Severity
3.3. Early Administration of Antibiotics
3.3.1. Acute Cholecystitis
3.3.2. Acute Cholangitis
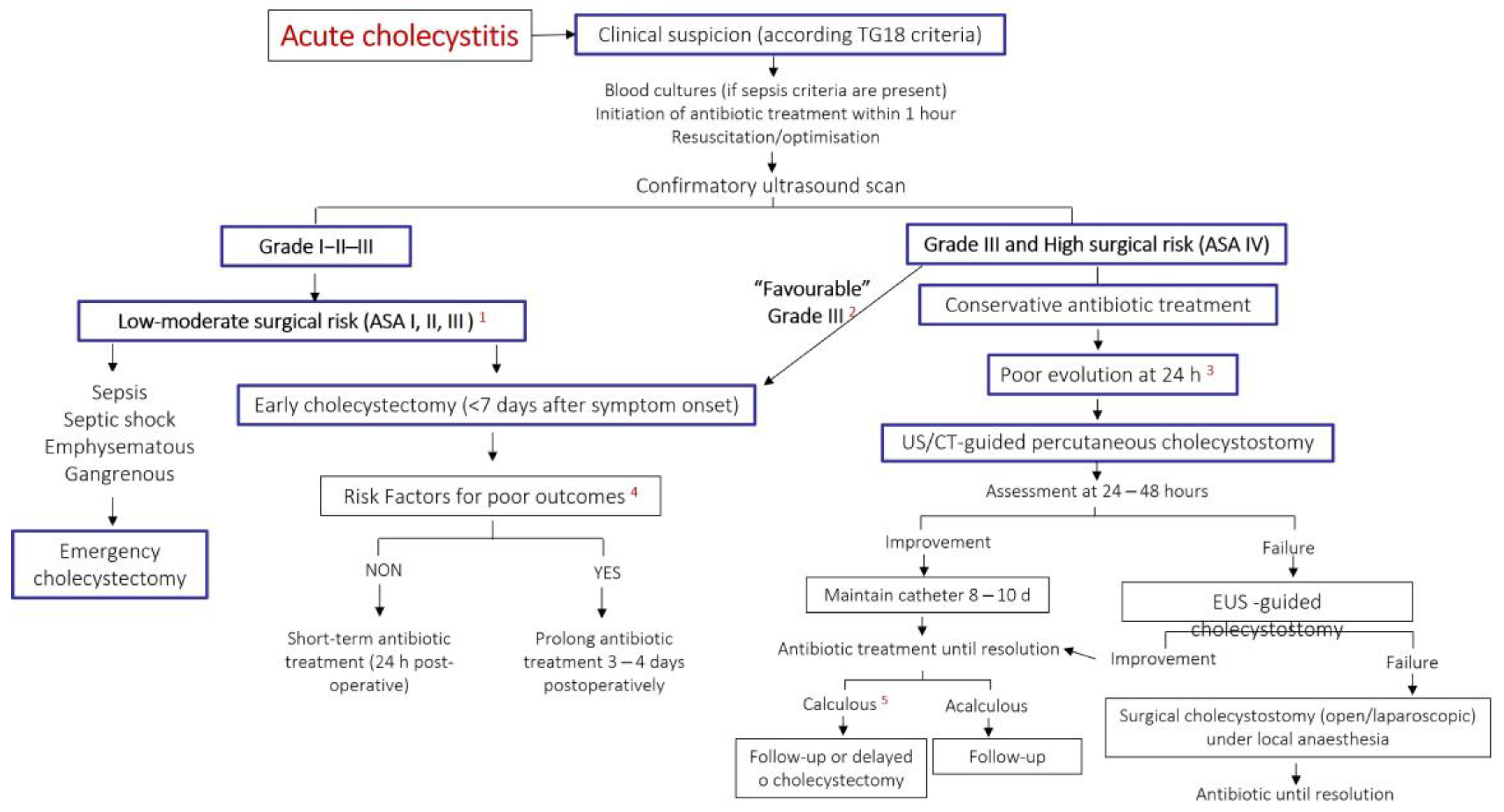
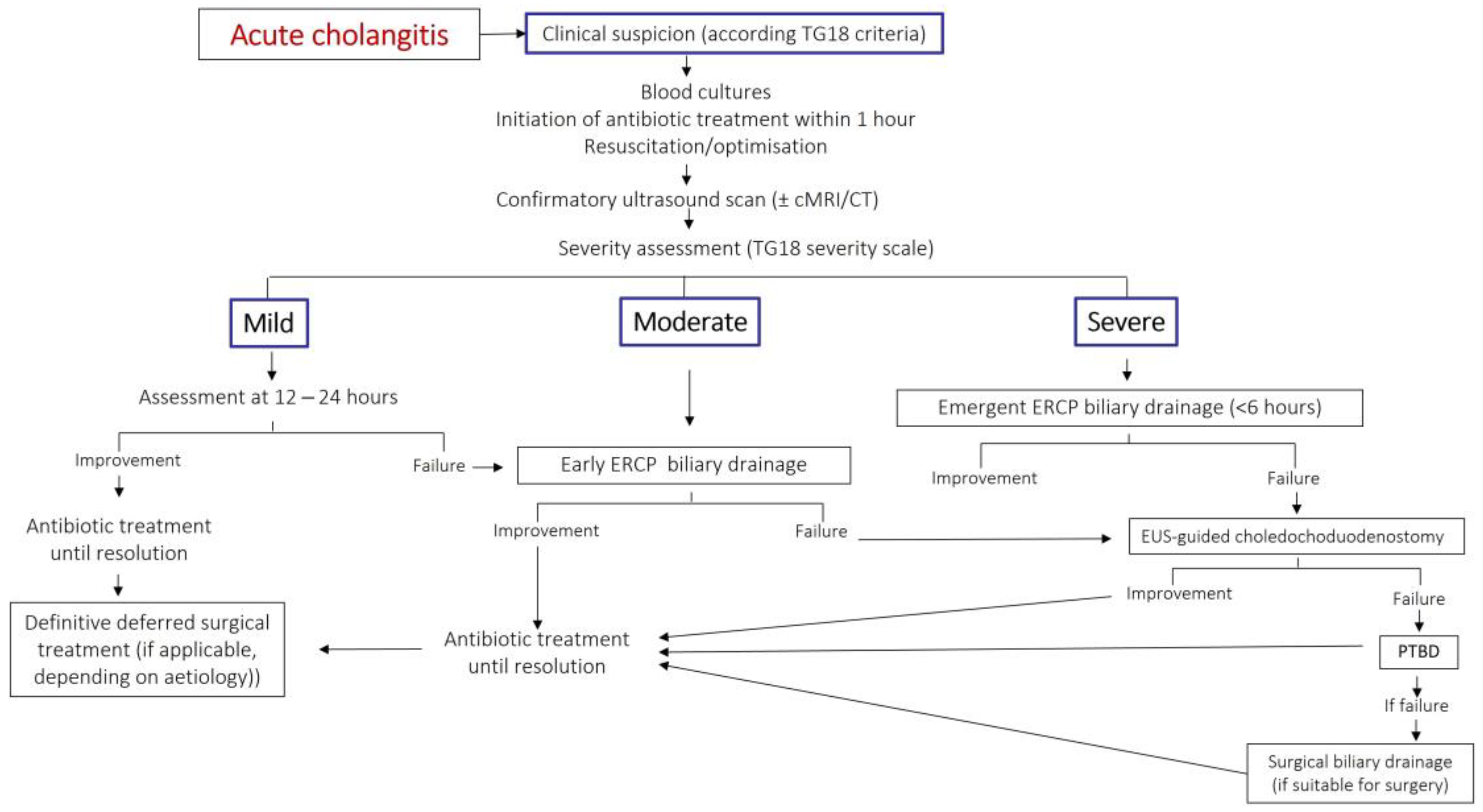
3.4. Timely Source Control
3.4.1. Early Cholecystectomy
3.4.2. Cholecystostomy
3.4.3. Biliary Drainage in Acute Cholangitis
3.5. Special Situations
3.5.1. Gangrenous Cholecystitis
3.5.2. Emphysematous Cholecystitis
3.5.3. Acute Acalculous Cholecystitis
| ORIGIN | Community-Acquired | Health Care-Associated Infections | |
|---|---|---|---|
| DIAGNOSE | Acute Calculous Cholecystitis | Acute Calculous Cholecystitis Acute Cholangitis (c) | Acalculous Cholecystitis in Critical Patient Cholangitis with Biliary Stent Cholangitis after ERCP or PTHC |
| SEVERITY | MILD-MODERATE | SEVERE | |
| WITHOUT Risk factors of poor evolution (a) | Amoxicillin-clavulanate± gentamicin (b) or Ertapenem or Cephalosporin 2nd + metronidazole Gentamicin or aztreonam + metronidazole * | Piperacilin-tazobactam or Meropenem, imipenem or doripenem (d) Tigecycline * ± Aztreonam or Amikacin (d) | Piperacilin-tazobactam ± amikacin (d) or Meropenem, imipenem or doripenem (d) ± Linezolid, daptomicine, or glycopeptide ± Fluconazole or candin (e) or Tigecyclin (d) + ceftazidime or amikacin o colistine ± Fluconazole or candin (e) Tigecyclin + Amikacin ± Fluconazole or echinocandin (e) * |
| WITH Risk factors of poor evolution (a) | Ertapenem Tigecycline * | Meropenem or imipenem(d) or Tigecycline + ceftazidime, cefepime or amikacin Tigecyclin + Aztreonam or Amikacin * | |
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Liang, L.; Moore, B.; Soni, A. National Inpatient Hospital Costs: The Most Expensive Conditions by Payer, 2017: Statistical Brief #261. Healthc. Cost Util. Proj. Stat. Briefs 2006, 204, 1–15. [Google Scholar]
- Cervellin, G.; Mora, R.; Ticinesi, A.; Meschi, T.; Comelli, I.; Catena, F.; Lippi, G. Epidemiology and outcomes of acute abdominal pain in a large urban Emergency Department: Retrospective analysis of 5,340 cases. Ann. Transl. Med. 2016, 4, 362. [Google Scholar] [CrossRef]
- Guirguis, R.N.; Nashaat, E.H.; Yassin, A.E.; Ibrahim, W.A.; Saleh, A.S.; Bahaa, M.; El-Meteini, M.; Fathy, M.; Dabbous, H.M.; Montasser, I.F.; et al. Biliary complications in recipients of living donor liver transplantation: A single-centre study. World J. Hepatol. 2021, 13, 2081–2103. [Google Scholar] [CrossRef] [PubMed]
- Kiriyama, S.; Kozaka, K.; Takada, T.; Strasberg, S.M.; Pitt, H.A.; Gabata, T.; Hata, J.; Liau, K.-H.; Miura, F.; Horiguchi, A.; et al. Tokyo Guidelines 2018: Diagnostic criteria and severity grading of acute cholangitis (with videos). J. Hepato-Biliary-Pancreat. Sci. 2018, 25, 17–30. [Google Scholar] [CrossRef] [PubMed]
- Kuo, C.H.; Changchien, C.S.; Chen, J.J.; Tai, D.I.; Chiou, S.S.; Lee, C.M. Septic Acute Cholecystitis. Scand. J. Gastroenterol. 1995, 30, 272–275. [Google Scholar] [CrossRef]
- Ortega, M.; Marco, F.; Soriano, A.; Almela, M.; Martinez, J.A.; Lopez, J.; Pitart, C.; Mensa, J. Epidemiology and prognostic determinants of bacteraemic biliary tract infection. J. Antimicrob. Chemother. 2012, 67, 1508–1513. [Google Scholar] [CrossRef]
- Sugiyama, M.; Atomi, Y. Treatment of Acute Cholangitis Due to Choledocholithiasis in Elderly and Younger Patients. Arch. Surg. 1997, 132, 1129–1133. [Google Scholar] [CrossRef]
- Esposito, A.L.; Gleckman, R.A.; Cram, S.; Crowley, M.; McCabe, F.; Drapkin, M.S. Community-Acquired Bacteremia in the Elderly: Analysis of One Hundred Consecutive Episodes. J. Am. Geriatr. Soc. 1980, 28, 315–319. [Google Scholar] [CrossRef] [PubMed]
- Auda, A.; Al Abdullah, R.; Khalid, M.O.; Alrasheed, W.Y.; Alsulaiman, S.A.; Almulhem, F.T.; Almaideni, M.F.; Alhikan, A. Acute Cholecystitis Presenting With Septic Shock as the First Presentation in an Elderly Patient. Cureus 2022, 14, 20981. [Google Scholar] [CrossRef]
- Takada, T.; Kawarada, Y.; Nimura, Y.; Yoshida, M.; Mayumi, T.; Sekimoto, M.; Miura, F.; Wada, K.; Hirota, M.; Yamashita, Y.; et al. Background: Tokyo Guidelines for the management of acute cholangitis and chole-cystitis. J. Hepato-Biliary-Pancreat. Sci. 2007, 14, 1–10. [Google Scholar] [CrossRef]
- Sartelli, M.; Catena, F.; Ansaloni, L.; Leppaniemi, A.; Taviloglu, K.; van Goor, H.; Viale, P.; Lazzareschi, D.V.; Coccolini, F.; Corbella, D.; et al. Complicated intra-abdominal infections in Europe: A comprehensive review of the CIAO study. World J. Emerg. Surg. 2012, 7, 36. [Google Scholar] [CrossRef] [PubMed]
- Ansaloni, L.; Pisano, M.; Coccolini, F.; Peitzmann, A.B.; Fingerhut, A.; Catena, F.; Agresta, F.; Allegri, A.; Bailey, I.; Balogh, Z.J.; et al. 2016 WSES guidelines on acute calculous cholecystitis. World J. Emerg. Surg. 2016, 11, 25. [Google Scholar] [CrossRef] [PubMed]
- Bass, G.A.; Gillis, A.E.; Cao, Y.; Mohseni, S.; Shamiyeh, A.; Rosetti, L.; Klimbacher, G.; Klugsberger, B.; Healy, P.; Moriarty, C.; et al. Self-reported and actual adherence to the Tokyo guidelines in the European snapshot audit of complicated calculous biliary disease. BJS Open 2020, 4, 622–629. [Google Scholar] [CrossRef]
- Lee, S.-W.; Yang, S.-S.; Chang, C.-S.; Yeh, H.-J. Impact of the Tokyo guidelines on the management of patients with acute calculous cholecystitis. J. Gastroenterol. Hepatol. 2009, 24, 1857–1861. [Google Scholar] [CrossRef]
- Takada, T.; Strasberg, S.M.; Solomkin, J.; Pitt, H.A.; Gomi, H.; Yoshida, M.; Mayumi, T.; Miura, F.; Gouma, D.J.; Garden, O.J.; et al. TG13: Updated Tokyo Guidelines for the management of acute cholangitis and cholecystitis. J. Hepato-Biliary-Pancreat. Sci. 2013, 20, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Takada, T. Tokyo Guidelines 2018: Updated Tokyo Guidelines for the management of acute cholangitis/acute cholecystitis. J. Hepatobiliary Pancreat. Sci. 2018, 25, 526. [Google Scholar] [CrossRef]
- Cumpston, M.; Li, T.; Page, M.; Chandler, J.; Welch, V.; Higgins, J.P.; Thomas, J. Updated guidance for trusted systematic reviews: A new edition of the Cochrane Handbook for Systematic Reviews of Interventions. Cochrane Database Syst. Rev. 2019, 10, ED000142. [Google Scholar] [CrossRef] [PubMed]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, 71. [Google Scholar] [CrossRef]
- Yokoe, M.; Hata, J.; Takada, T.; Strasberg, S.M.; Bun, T.A.Y.; Wakabayashi, G.; Kozaka, K.; Endo, I.; DeZiel, D.J.; Miura, F.; et al. Tokyo Guidelines 2018: Diagnostic criteria and severity grading of acute cholecystitis (with videos). J. Hepatobiliary Pancreat. Sci. 2018, 25, 41–54. [Google Scholar] [CrossRef]
- Yokoe, M.; Takada, T.; Mayumi, T.; Yoshida, M.; Hasegawa, H.; Norimizu, S.; Hayashi, K.; Umemura, S.; Orito, E. Accuracy of the Tokyo Guidelines for the diagnosis of acute cholangitis and cholecystitis taking into consideration the clinical practice pattern in Japan. J. Hepato-Biliary-Pancreat. Sci. 2011, 18, 250–257. [Google Scholar] [CrossRef] [PubMed]
- Indar, A.A.; Beckingham, I.J. Acute cholecystitis. Br. Med. J. 2002, 325, 639–643. [Google Scholar] [CrossRef]
- Kiewiet, J.J.S.; Leeuwenburgh, M.M.N.; Bipat, S.; Bossuyt, P.M.M.; Stoker, J.; Boermeester, M.A. A Systematic Review and Meta-Analysis of Diagnostic Performance of Imaging in Acute Cholecystitis. Radiology 2012, 264, 708–720. [Google Scholar] [CrossRef]
- Regimbeau, J.M.; Fuks, D.; Pautrat, K.; Mauvais, F.; Haccart, V.; Msika, S.; Mathonnet, M.; Scotté, M.; Paquet, J.C.; Vons, C.; et al. Effect of Postoperative Antibiotic Administration on Postoperative Infection Following Cholecystectomy for Acute Calculous Cholecystitis: A randomized clinical trial. JAMA 2014, 312, 145–154. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, M. Acute cholangitis—An update. World J. Gastrointest. Pathophysiol. 2018, 9, 1–7. [Google Scholar] [CrossRef]
- Melzer, M.; Toner, R.; Lacey, S.; Bettany, E.; Rait, G. Biliary tract infection and bacteraemia: Presentation, structural abnormalities, causative organisms and clinical outcomes. Postgrad. Med. J. 2007, 83, 773–776. [Google Scholar] [CrossRef]
- Sun, G.; Han, L.; Yang, Y.; Linghu, E.; Li, W.; Cai, F.; Kong, J.; Wang, X.; Meng, J.; Du, H.; et al. Comparison of two editions of Tokyo guidelines for the management of acute cholangitis. J. Hepato-Biliary-Pancreat. Sci. 2014, 21, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Kiriyama, S.; Takada, T.; Hwang, T.-L.; Akazawa, K.; Miura, F.; Gomi, H.; Mori, R.; Endo, I.; Itoi, T.; Yokoe, M.; et al. Clinical application and verification of the TG13 diagnostic and severity grading criteria for acute cholangitis: An international multicenter observational study. J. Hepato-Biliary-Pancreat. Sci. 2017, 24, 329–337. [Google Scholar] [CrossRef] [PubMed]
- Abboud, P.-A.C.; Malet, P.F.; Berlin, J.A.; Staroscik, R.; Cabana, M.D.; Clarke, J.R.; Shea, J.A.; Schwartz, J.; Williams, S.V. Predictors of common bile duct stones prior to cholecystectomy: A meta-analysis. Gastrointest. Endosc. 1996, 44, 450–457. [Google Scholar] [CrossRef]
- Singer, M.; Deutschman, C.S.; Seymour, C.W.; Shankar-Hari, M.; Annane, D.; Bauer, M.; Bellomo, R.; Bernard, G.R.; Chiche, J.-D.; Coopersmith, C.M.; et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA 2016, 315, 801–810. [Google Scholar] [CrossRef]
- Kaukonen, K.-M.; Bailey, M.; Pilcher, D.; Cooper, D.J.; Bellomo, R. Systemic Inflammatory Response Syndrome Criteria in Defining Severe Sepsis. New Engl. J. Med. 2015, 372, 1629–1638. [Google Scholar] [CrossRef]
- Mikkelsen, M.E.; Miltiades, A.N.; Gaieski, D.F.; Goyal, M.; Fuchs, B.D.; Shah, C.V.; Bellamy, S.L.; Christie, J.D. Serum lactate is associated with mortality in severe sepsis independent of organ failure and shock*. Crit. Care Med. 2009, 37, 1670–1677. [Google Scholar] [CrossRef]
- Dellinger, R.P.; Levy, M.M.; Rhodes, A.; Annane, D.; Gerlach, H.; Opal, S.M.; Sevransky, J.E.; Sprung, C.L.; Douglas, I.S.; Jaeschke, R.; et al. Surviving sepsis campaign: International guidelines for management of severe sepsis and septic shock: 2012. Crit. Care Med. 2013, 41, 580–637. [Google Scholar] [CrossRef]
- Vincent, J.L.; Moreno, R.; Takala, J.; Willatts, S.; De Mendonça, A.; Bruining, H.; Reinhart, C.K.; Suter, P.M.; Thijs, L.G. The SOFA (Sepsis-related Organ Failure Assessment) score to describe organ dysfunction/failure: On behalf of the Working Group on Sepsis-Related Problems of the European Society of Intensive Care Medicine. Intensive Care Med. 1996, 22, 707–710. [Google Scholar] [CrossRef] [PubMed]
- Seymour, C.W.; Liu, V.X.; Iwashyna, T.J.; Brunkhorst, F.M.; Rea, T.D.; Scherag, A.; Rubenfeld, G.; Kahn, J.M.; Shankar-Hari, M.; Singer, M.; et al. Assessment of clinical criteria for sepsis for the third international consensus def-initions for sepsis and septic shock (sepsis-3). J. Am. Med. Assoc 2016, 315, 762–774. [Google Scholar] [CrossRef]
- Levy, M.M.; Fink, M.P.; Marshall, J.C.; Abraham, E.; Angus, D.; Cook, D.; Cohen, J.; Opal, S.M.; Vincent, J.-L.; Ramsay, G. 2001 SCCM/ESICM/ACCP/ATS/SIS International Sepsis Definitions Conference. Crit. Care Med. 2003, 31, 1250–1256. [Google Scholar] [CrossRef]
- Tusgul, S.; Carron, P.-N.; Yersin, B.; Calandra, T.; Dami, F. Low sensitivity of qSOFA, SIRS criteria and sepsis definition to identify infected patients at risk of complication in the prehospital setting and at the emergency department triage. Scand. J. Trauma Resusc. Emerg. Med. 2017, 25, 108. [Google Scholar] [CrossRef]
- Haydar, S.; Spanier, M.; Weems, P.; Wood, S.; Strout, T. Comparison of QSOFA score and SIRS criteria as screening mechanisms for emergency department sepsis. Am. J. Emerg. Med. 2017, 35, 1730–1733. [Google Scholar] [CrossRef]
- Askim, Å.; Moser, F.; Gustad, L.T.; Stene, H.; Gundersen, M.; Åsvold, B.O.; Dale, J.; Bjørnsen, L.P.; Damås, J.K.; Solligård, E. Poor performance of quick-SOFA (qSOFA) score in predicting severe sepsis and mortality—A prospective study of patients admitted with infection to the emergency department. Scand. J. Trauma Resusc. Emerg. Med. 2017, 25, 56. [Google Scholar] [CrossRef] [PubMed]
- Williams, J.M.; Greenslade, J.H.; McKenzie, J.V.; Chu, K.; Brown, A.F.T.; Lipman, J. SIRS, qSOFA and organ dysfunction: Insights from a prospective database of emergency department patients with infection. Chest 2017, 151, 586–596. [Google Scholar] [CrossRef] [PubMed]
- Zheng, R.; Zhang, Y.; Rong, Z.; Huang, W.; Fu, X. Surviving Sepsis Campaign: International guidelines for management of sepsis and septic shock 2021, interpretation and expectation. Chin. Crit. Care Med. 2021, 33, 1159–1164. [Google Scholar]
- Levy, M.M.; Evans, L.E.; Rhodes, A. The Surviving Sepsis Campaign Bundle: 2018 update. Intensiv. Care Med. 2018, 44, 925–928. [Google Scholar] [CrossRef]
- Csendes, A.; Burdiles, P.; Maluenda, F.; Diaz, J.C.; Csendes, P.; Mitru, N. Simultaneous Bacteriologic Assessment of Bile From Gallbladder and Common Bile Duct in Control Subjects and Patients With Gallstones and Common Duct Stones. Arch. Surg. 1996, 131, 389–394. [Google Scholar] [CrossRef] [PubMed]
- Chang, W.-T.; Lee, K.-T.; Wang, S.-R.; Chuang, S.-C.; Kuo, K.-K.; Chen, J.-S.; Sheen, P.-C. Bacteriology and antimicrobial susceptibility in biliary tract disease: An audit of 10-year’s experience. Kaohsiung J. Med. Sci. 2002, 18, 221–228. [Google Scholar] [PubMed]
- Gomi, H.; Solomkin, J.S.; Schlossberg, D.; Okamoto, K.; Takada, T.; Strasberg, S.M.; Ukai, T.; Endo, I.; Iwashita, Y.; Hibi, T.; et al. Tokyo Guidelines 2018: Antimicrobial therapy for acute cholangitis and chole-cystitis. J. Hepato-Biliary-Pancreat. Sci. 2018, 25, 3–16. [Google Scholar] [CrossRef]
- Marne, C.; Pallarés, R.; Martín, R.; Sitges-Serra, A. Gangrenous cholecystitis and acute cholangitis associated with anaerobic bacteria in bile. Eur. J. Clin. Microbiol. Infect. Dis. 1986, 5, 35–39. [Google Scholar] [CrossRef]
- Weber, A.; Schneider, J.; Wagenpfeil, S.; Winkle, P.; Riedel, J.; Wantia, N.; Feihl, S.; Römmler, F.; Baur, D.M.; Schmid, R.M.; et al. Spectrum of pathogens in acute cholangitis in patients with and without biliary endoprosthesis. J. Infect. 2013, 67, 111–121. [Google Scholar] [CrossRef]
- Nomura, T.; Shirai, Y.; Hatakeyama, K. Enterococcal Bactibilia in Patients with Malignant Biliary Obstruction. Dig. Dis. Sci. 2000, 45, 2183–2186. [Google Scholar] [CrossRef]
- Martínez-Martínez, L.; Calvo, J. The growing problem of antibiotic resistance in clinically relevant Gram-negative bacteria: Current situation. Enferm. Infecc. Microbiol. Clin. 2010, 28 (Suppl. S2), 25–31. [Google Scholar] [CrossRef]
- Sokal, A.; Sauvanet, A.; Fantin, B.; de Lastours, V. Acute cholangitis: Diagnosis and management. J. Visc. Surg. 2019, 156, 515–525. [Google Scholar] [CrossRef] [PubMed]
- Rice, L.B.; Hutton-Thomas, R.; Lakticova, V.; Helfand, M.S.; Donskey, C.J. β-Lactam antibiotics and gastrointestinal colonization with vancomy-cin-resistant enterococci. J. Infect. Dis. 2004, 189, 1113–1118. [Google Scholar] [CrossRef] [PubMed]
- Ledro-Cano, D. Suspected choledocholithiasis: Endoscopic ultrasound or magnetic resonance cholangio-pancreatography? A systematic review. Eur. J. Gastroenterol. Hepatol. 2007, 19, 1007–1011. [Google Scholar] [CrossRef]
- Wintenberger, C.; Guery, B.; Bonnet, E.; Castan, B.; Cohen, R.; Diamantis, S.; Lesprit, P.; Maulin, L.; Péan, Y.; Peju, E.; et al. Proposal for shorter antibiotic therapies. Médecine Mal. Infect. 2017, 47, 92–141. [Google Scholar] [CrossRef] [PubMed]
- Van Lent, A.U.; Bartelsman, J.F.; Tytgat, G.N.; Speelman, P.; Prins, J.M. Duration of antibiotic therapy for cholangitis after successful endoscopic drainage of the biliary tract. Gastrointest. Endosc. 2002, 55, 518–522. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Cheng, Y.; Xiong, X.Z.; Lin, Y.X.; Wu, S.J.; Cheng, N.S. Two-stage vs single-stage management for concomitant gallstones and common bile duct stones. World J. Gastroenterol. 2012, 18, 3156–3166. [Google Scholar] [CrossRef]
- Okamoto, K.; Suzuki, K.; Takada, T.; Strasberg, S.M.; Asbun, H.J.; Endo, I.; Iwashita, Y.; Hibi, T.; Pitt, H.A.; Umezawa, A.; et al. Tokyo Guidelines 2018: Flowchart for the management of acute cholecystitis. J. Hepato-Biliary-Pancreat. Sci. 2018, 25, 55–72. [Google Scholar] [CrossRef]
- Papi, C.; Catarci, M.; D’Ambrosio, L.; Gili, L.; Koch, M.; Grassi, G.B.; Capurso, L. Timing of Cholecystectomy for Acute Calculous Cholecystitis: A Meta-Analysis. Am. J. Gastroenterol. 2004, 99, 147–155. [Google Scholar] [CrossRef] [PubMed]
- Gurusamy, K.S.; Koti, R.; Fusai, G.; Davidson, B.R. Early versus delayed laparoscopic cholecystectomy for uncomplicated biliary colic. Cochrane Database Syst. Rev. 2013, 6, CD007196. [Google Scholar] [CrossRef]
- Winbladh, A.; Gullstrand, P.; Svanvik, J.; Sandström, P. Systematic review of cholecystostomy as a treatment option in acute cholecystitis. HPB 2009, 11, 183–193. [Google Scholar] [CrossRef] [PubMed]
- Campanile, F.C.; Catena, F.; Coccolini, F.; Lotti, M.; Piazzalunga, D.; Pisano, M.; Ansaloni, L. The need for new “patient-related” guidelines for the treatment of acute cholecystitis. World J. Emerg. Surg. 2011, 6, 1–3. [Google Scholar] [CrossRef]
- Enochsson, L.; Swahn, F.; Arnelo, U.; Nilsson, M.; Löhr, M.; Persson, G. Data from 11.074 ERCP procedures from the Swedish register for gallstone surgery and ERCP (GallRiks). Pancreatology 2010, 72, 1175–1184. [Google Scholar]
- Serban, D.; Socea, B.; Balasescu, S.A.; Badiu, C.D.; Tudor, C.; Dascalu, A.M.; Vancea, G.; Spataru, R.I.; Sabau, A.D.; Sabau, D.; et al. Safety of Laparoscopic Cholecystectomy for acute cholecystitis in the elderly: A multivariate analysis of risk factors for intra and postoperative complications. Medicina 2021, 57, 230. [Google Scholar] [CrossRef]
- de Mestral, C.; Rotstein, O.D.; Laupacis, A.; Hoch, J.S.; Zagorski, B.; Alali, A.S.; Nathens, A.B. Comparative operative outcomes of early and delayed cholecystectomy for acute cholecystitis: A population-based propensity score analysis. Ann. Surg. 2014, 259, 10–15. [Google Scholar] [CrossRef]
- Cho, S.H.; Oh, D.; Song, T.J.; Park, D.H.; Seo, D.-W.; Lee, S.K.; Kim, M.-H.; Lee, Y.N.; Moon, J.H.; Lee, S.S. Comparison of the effectiveness and safety of lumen-apposing metal stents and anti-migrating tubular self-expandable metal stents for EUS-guided gallbladder drainage in high surgical risk patients with acute cholecystitis. Gastrointest. Endosc. 2020, 91, 543–550. [Google Scholar] [CrossRef] [PubMed]
- Hemerly, M.C.; de Moura, D.T.H.; Junior, E.S.D.M.; Proença, I.M.; Ribeiro, I.B.; Yvamoto, E.Y.; Ribas, P.H.B.V.; Sánchez-Luna, S.A.; Bernardo, W.M.; de Moura, E.G.H. Endoscopic ultrasound (EUS)-guided cholecystostomy versus percutaneous cholecystostomy (PTC) in the management of acute cholecystitis in patients unfit for surgery: A systematic review and meta-analysis. Surg. Endosc. 2022, 37, 2421–2438. [Google Scholar] [CrossRef] [PubMed]
- Lai, E.C.; Mok, F.P.; Tan, E.S.; Lo, C.-M.; Fan, S.-T.; You, K.-T.; Wong, J. Endoscopic biliary drainage for severe acute cholangitis. New Engl. J. Med. 1992, 326, 1582–1586. [Google Scholar] [CrossRef] [PubMed]
- Mori, Y.; Itoi, T.; Baron, T.H.; Takada, T.; Strasberg, S.M.; Pitt, H.A.; Ukai, T.; Shikata, S.; Noguchi, Y.; Teoh, A.Y.B.; et al. Tokyo Guidelines 2018: Management strategies for gallbladder drainage in patients with acute cholecystitis (with videos). J. Hepato-Biliary-Pancreat. Sci. 2018, 25, 87–95. [Google Scholar] [CrossRef]
- Gigot, J.F.M.; Leese, T.M.; Dereme, T.M.; Coutinho, J.M.; Castaing, D.M.; Bismuth, H.M. Acute Cholangitis. Ann. Surg. 1989, 209, 435–438. [Google Scholar] [CrossRef]
- Adler, D.G.; Baron, T.H.; Davila, R.E.; Egan, J.; Hirota, W.K.; Leighton, J.A.; Qureshi, W.; Rajan, E.; Zuckerman, M.J.; Fanelli, R.; et al. ASGE guideline: The role of ERCP in diseases of the biliary tract and the pancreas. Gastrointest. Endosc. 2005, 62, 1–8. [Google Scholar] [CrossRef]
- Du, L.; Cen, M.; Zheng, X.; Luo, L.; Siddiqui, A.; Kim, J.J. Timing of Performing Endoscopic Retrograde Cholangiopancreatography and Inpatient Mortality in Acute Cholangitis: A Systematic Review and Meta-Analysis. Clin. Transl. Gastroenterol. 2020, 11, e00158. [Google Scholar] [CrossRef] [PubMed]
- Doyle, J.B.; Sethi, A. Endoscopic Ultrasound-Guided Biliary Drainage. J. Clin. Med. 2023, 12, 2736. [Google Scholar] [CrossRef]
- Moole, H.; Bechtold, M.L.; Forcione, D.; Puli, S.R. A meta-analysis and systematic review: Success of endoscopic ultrasound guided biliary stenting in patients with inoperable malignant biliary strictures and a failed ERCP. Medicine 2017, 96, e5154. [Google Scholar] [CrossRef]
- Wang, K.; Zhu, J.; Xing, L.; Wang, Y.; Jin, Z.; Li, Z. Assessment of efficacy and safety of EUS-guided biliary drainage: A systematic review. Gastrointest. Endosc. 2016, 83, 1218–1227. [Google Scholar] [CrossRef] [PubMed]
- Teefey, S.A.; Dahiya, N.; Middleton, W.D.; Bajaj, S.; Dahiya, N.; Ylagan, L.; Hildebolt, C.F. Acute cholecystitis: Do sonographic findings and WBC count predict gangrenous changes? Am. J. Roentgenol. 2013, 200, 363–369. [Google Scholar] [CrossRef] [PubMed]
- Dhir, T.; Schiowitz, R. Old man gallbladder syndrome: Gangrenous cholecystitis in the unsuspected patient population. Int. J. Surg. Case Rep. 2015, 11, 46–49. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, A.; Shikata, K.; Uchida, H.A.; Shinoura, S.; Yokomichi, N.; Ogawa, D.; Sato-Horiguchi, C.; Yagi, T.; Wada, J.; Makino, H. Case of emphysematous cholecystitis in a patient with type 2 diabetes mellitus associated with schizophrenia. J. Diabetes Investig. 2012, 3, 534–535. [Google Scholar] [CrossRef]
- Sunnapwar, A.; Raut, A.A.; Nagar, A.M.; Katre, R. Emphysematous cholecystitis: Imaging findings in nine patients. Indian J. Radiol. Imaging 2011, 21, 142–146. [Google Scholar] [CrossRef]
- Yoshida, M.; Takada, T.; Kawarada, Y.; Tanaka, A.; Nimura, Y.; Gomi, H.; Hirota, M.; Miura, F.; Wada, K.; Mayumi, T.; et al. Antimicrobial therapy for acute cholecystitis: Tokyo Guidelines. J. Hepato-Biliary-Pancreat. Surg. 2007, 14, 83–90. [Google Scholar] [CrossRef]
- Ganpathi, I.S.; Diddapur, R.K.; Eugene, H.; Karim, M. Acute acalculous cholecystitis: Challenging the myths. HPB 2007, 9, 131–134. [Google Scholar] [CrossRef]
- Aledo, V.S.; Iñíguez, L.G.; Funes, D.F.; Prats, M.C.; Albasini, J.L.A. Is cholecystectomy the treatment of choice for acute acalculous cholecystitis? A systematic review of the literature. Rev. Española. Enferm. Dig. 2017, 109, 708–718. [Google Scholar] [CrossRef]
- Rehman, T.; Deboisblanc, B.P. Persistent Fever in the ICU. Chest 2014, 145, 158–165. [Google Scholar] [CrossRef]
- Huffman, J.L.; Schenker, S. Acute Acalculous Cholecystitis: A Review. Clin. Gastroenterol. Hepatol. 2010, 8, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Amillo-Zaragüeta, M.; Nve, E.; Casanova, D.; Garro, P.; Badia, J.M. The Importance of Early Management of Severe Biliary Infection: Current Concepts. Int. Surg. 2021, 105, 667–678. [Google Scholar] [CrossRef]
| DIAGNOSIS Suspicion: one criterion A + one criterion B Definitive diagnosis: one criterion A + one criterion B + one criterion C | A. Local inflammation A-1 Murphy’s sign A-2 Pain/mass/tenderness in upper right quadrant | B. Systemic inflammation B-1 Fever B-2 Elevated CPR B-3 Elevated leukocyte count | C. Image C-1 Characteristic findings of acute cholecystitis |
| SEVERITY | SEVERE (grade III) | MODERATE (grade II) | MILD (grade I) |
| It is associated with dysfunction in one of the following organs/systems 1. Cardiovascular: hypotension requiring dopamine > 5 µg/kg/min, or any dose of norepinephrine 2. Neurological: decrease in the level of consciousness 3. Respiratory: PaO2/FiO2 ratio <300 4. Renal: oliguria, creatinine > 2.0 mg/dL 5. Liver: PT-INR > 1.5 6. Haematological: platelet count < 100,000 mm3 | It is associated with one of the following: 1. Leukocytosis (>18,000/mm3) 2. Palpable mass with tenderness in upper right quadrant 3. Duration of symptoms > 72 h 4. Marked local inflammation (gangrenous cholecystitis, pericholecystic abscess, liver abscess, biliary peritonitis, emphysematous cholecystitis) | Does not meet criteria for severe or moderate cholecystitis. It can be defined as acute cholecystitis in a healthy patient without organic dysfunction and with mild inflammatory changes in the gallbladder |
| DIAGNOSIS Suspicion: one criterion A + one criterion B or C Definitive diagnosis: one criterion A + one criterion B + one criterion C | A. Systemic inflammation A-1 Fever and/or chills A-2 Inflammatory response (CPR elevation, elevated leukocyte count) | B. Cholestasis B-1 Jaundice (bilirubin ≥ 2 mg/dL) B-2 Altered liver function (elevated alkaline phosphatases, gamma-GT, and/or transaminases) | C. Image C-1 Bile duct dilation C-2 Evidence of etiology (stricture, lithiasis, stent) |
| SEVERITY | SEVERE (grade III) | MODERATE (grade II) | MILD (grade I) |
| Dysfunction of one of the following organs/systems: 1. Cardiovascular dysfunction: hypotension requiring dopamine ≥ 5 µg/kg per min, or any dose of norepinephrine 2. Neurological dysfunction: disturbance of consciousness 3. Respiratory dysfunction: PaO2/FiO2 ratio < 300 4. Renal dysfunction: oliguria, serum creatinine > 2.0 mg/dL 5. Hepatic dysfunction: PT-INR > 1.5 6. Haematological dysfunction: platelet count < 100,000/mm3 | Two of the following conditions: 1. abnormal WBC count (>12,000/mm3 or <4000/mm3) 2. High fever ≥ 39 °C 3. Age ≥ 75 years 4. Hyperbilirubinemia (total bilirubin ≥ 5 mg/dL) 5. Hypoalbuminemia | “Grade I” acute cholangitis does not meet the criteria of “Grade II” or “Grade III” acute cholangitis. |
| Microorganisms | Proportion of Isolates | ||
|---|---|---|---|
| Bile (%) | Blood Culture (%) | ||
| Community-Acquired | Nosocomial | ||
| Gram-negative | |||
| Escherichia coli | 31–44 | 35–62 | 23 |
| Klebsiella spp. | 9–20 | 12–28 | 16 |
| Pseudomonas spp. | 0.5–19 | 4–14 | 17 |
| Enterobacter spp. | 5–9 | 2–7 | 7 |
| Acinetobacter spp. | - | 3 | 7 |
| Citrobacter spp. | - | 2–6 | 5 |
| Gram-positive | |||
| Enterococcus spp. | 3–34 | 10–23 | 20 |
| Streptococcus spp. | 2–10 | 6–9 | 5 |
| Staphylococcus spp. | 0 | 2 | 4 |
| Anaerobes | 4–20 | 1 | 2 |
| Others | - | 17 | 11 |
| Related to the inadequacy of antibiotic treatment | Risk of infection by unusual organisms (Enterobacteria-ESBL, Pseudomonas spp.) |
| Hospitalisation > 5 days | |
| Antibiotic treatment > 3–5 days in the last 6 weeks | |
| Biliary stent | |
| Cholangitis after ERCP 1 | |
| Related to the severity of infection | Sepsis, septic shock |
| Related with comorbidities | Immunosuppression Malnutrition Diabetes Chronic renal failure Chronic obstructive pulmonary disease Liver cirrhosis |
| Age-related | >70 years old |
| Increasing isolation of Enterococcus spp., Enterococcus faecium, and Pseudomonas in acute cholangitis. |
| Increased resistance of E. coli and Klebsiella spp. to quinolones. |
| QuickSOFA (qSOFA) proposed as a new bedside score to identify sepsis (2016) but not recommended as an alternative to SIRS, NEWS, MEWS by the Surviving Sepsis Campaign 2021. |
Surviving Sepsis Campaign “Hour-1 bundle” (revised 2021):
|
Changes in Tokyo Guidelines 2007, 2013, and 2018 (TG07, TG13, TG18)
|
| Role of endoscopic ultrasound (EUS) in the diagnosis of choledocholithiasis. |
| Early laparoscopic cholecystectomy is recommended in acute cholecystitis. |
| Cholecystostomy is recommended in unstable patients with acute cholecystitis. |
| Early biliary drainage is recommended in acute cholangitis. |
| New endoscopic techniques combined with EUS are incorporated for drainage of the gallbladder or bile ducts. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nve, E.; Badia, J.M.; Amillo-Zaragüeta, M.; Juvany, M.; Mourelo-Fariña, M.; Jorba, R. Early Management of Severe Biliary Infection in the Era of the Tokyo Guidelines. J. Clin. Med. 2023, 12, 4711. https://doi.org/10.3390/jcm12144711
Nve E, Badia JM, Amillo-Zaragüeta M, Juvany M, Mourelo-Fariña M, Jorba R. Early Management of Severe Biliary Infection in the Era of the Tokyo Guidelines. Journal of Clinical Medicine. 2023; 12(14):4711. https://doi.org/10.3390/jcm12144711
Chicago/Turabian StyleNve, Esther, Josep M. Badia, Mireia Amillo-Zaragüeta, Montserrat Juvany, Mónica Mourelo-Fariña, and Rosa Jorba. 2023. "Early Management of Severe Biliary Infection in the Era of the Tokyo Guidelines" Journal of Clinical Medicine 12, no. 14: 4711. https://doi.org/10.3390/jcm12144711
APA StyleNve, E., Badia, J. M., Amillo-Zaragüeta, M., Juvany, M., Mourelo-Fariña, M., & Jorba, R. (2023). Early Management of Severe Biliary Infection in the Era of the Tokyo Guidelines. Journal of Clinical Medicine, 12(14), 4711. https://doi.org/10.3390/jcm12144711







