Mid- and Long-Term Atrio-Ventricular Functional Changes in Children after Recovery from COVID-19
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Population
2.2. Data Collection and Definitions
2.3. Control Group
2.4. Serological Assays
2.5. Cardiac Evaluation
2.6. Reproducibility
2.7. Statistical Analysis
3. Results
3.1. Baseline Characteristics
3.2. Standard Echocardiographic Measurements
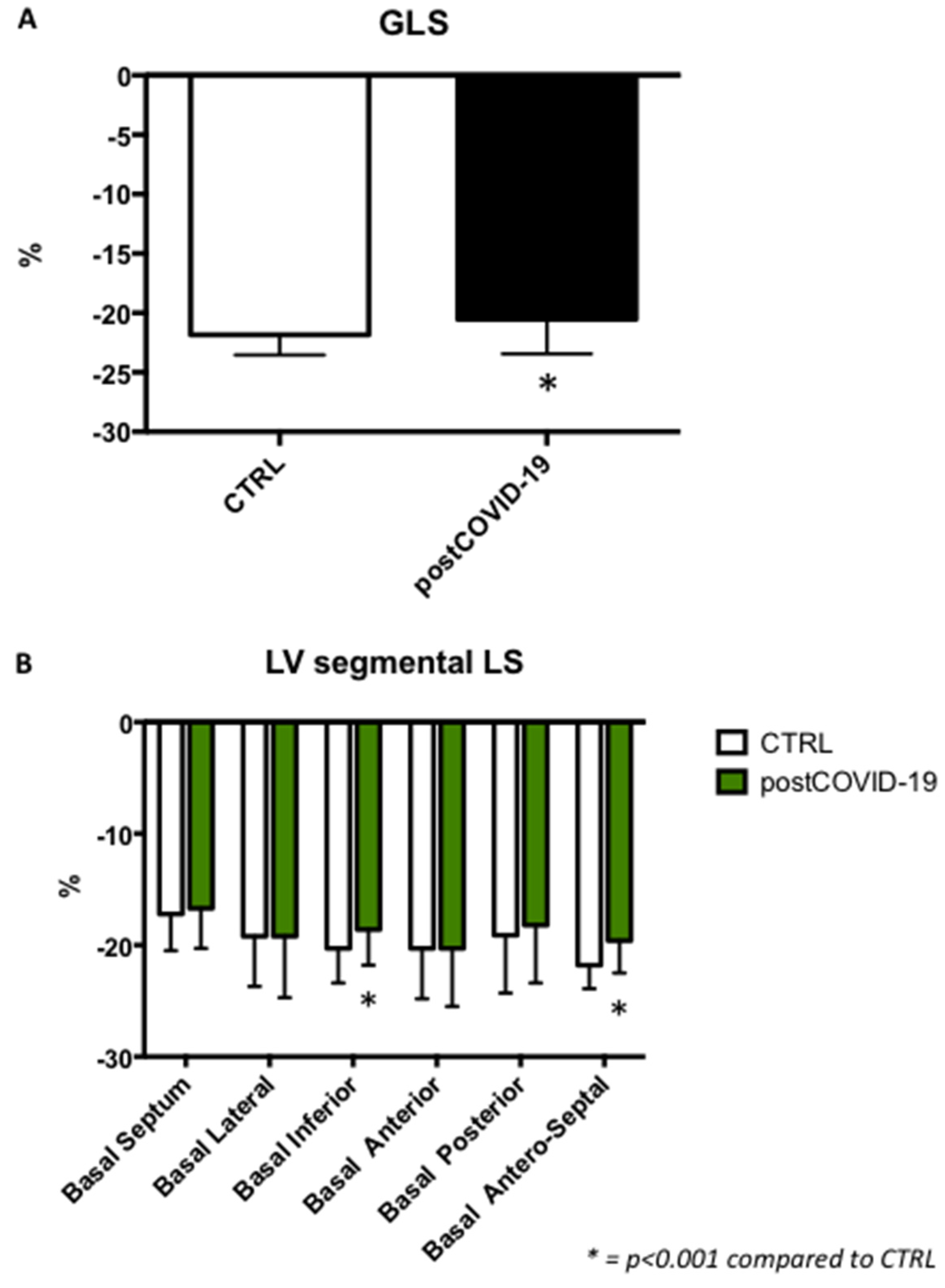
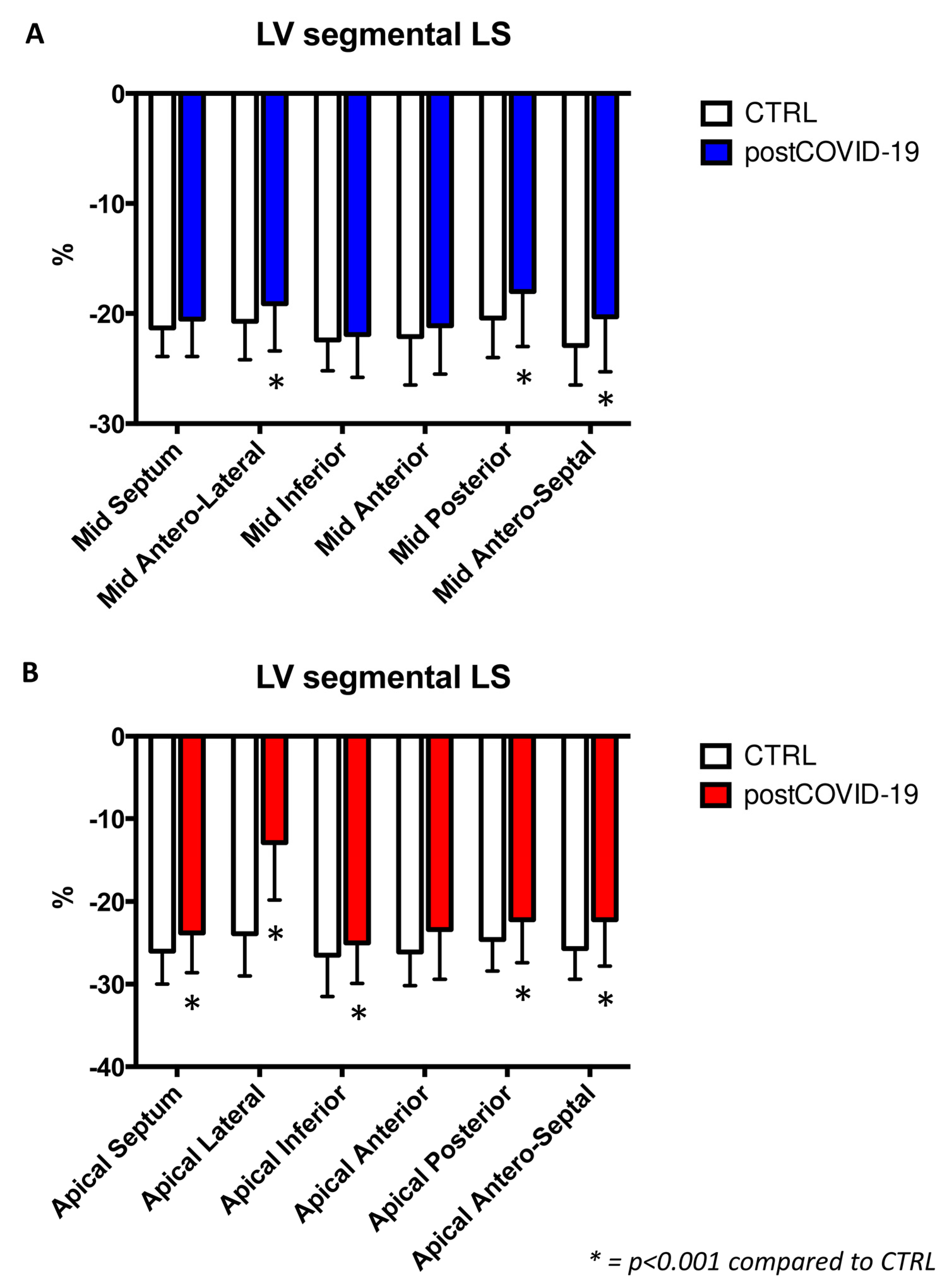
3.3. Left Ventricular Longitudinal Strain
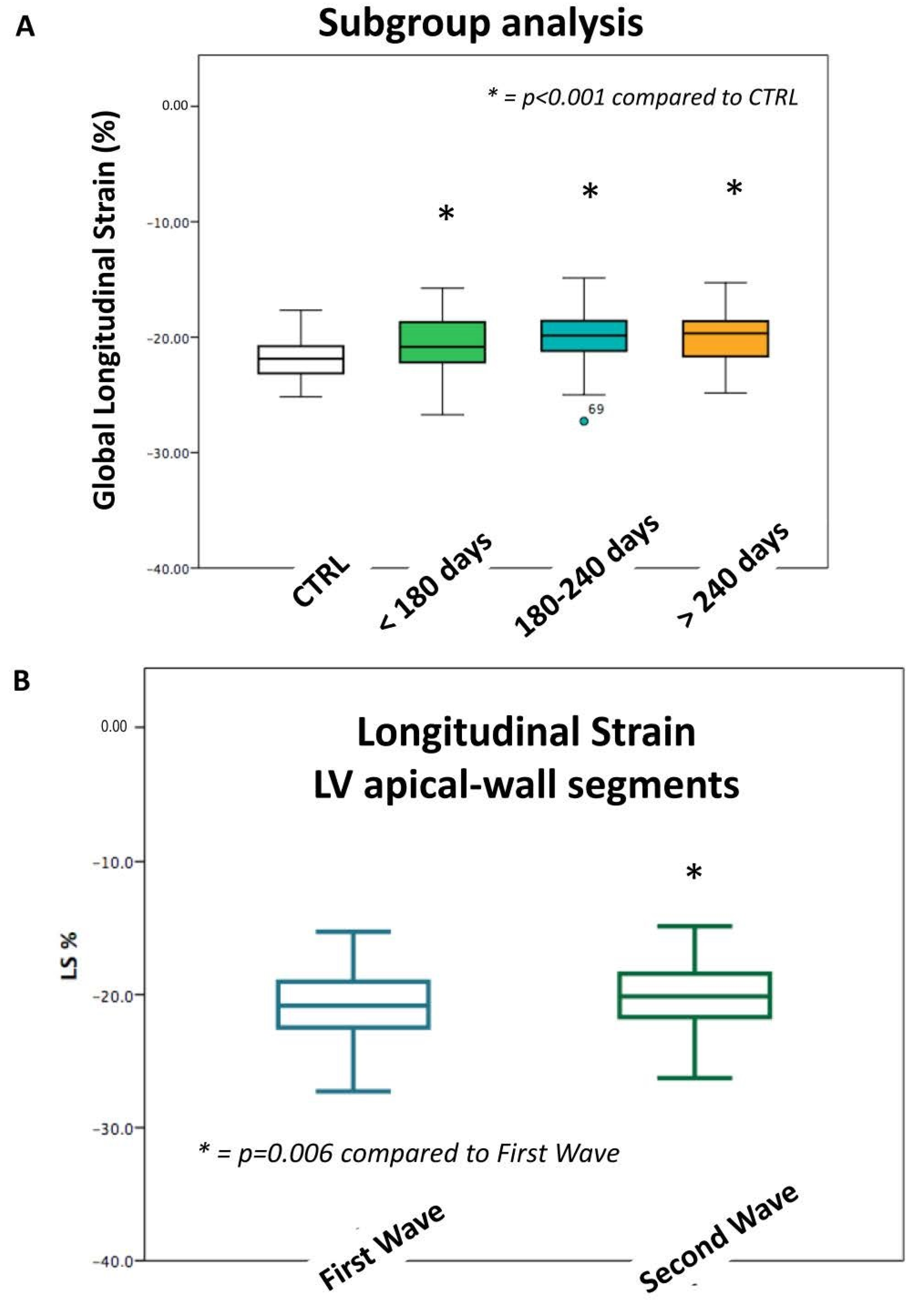
3.4. Left Ventricular Longitudinal Strain According to Different Pandemic Waves
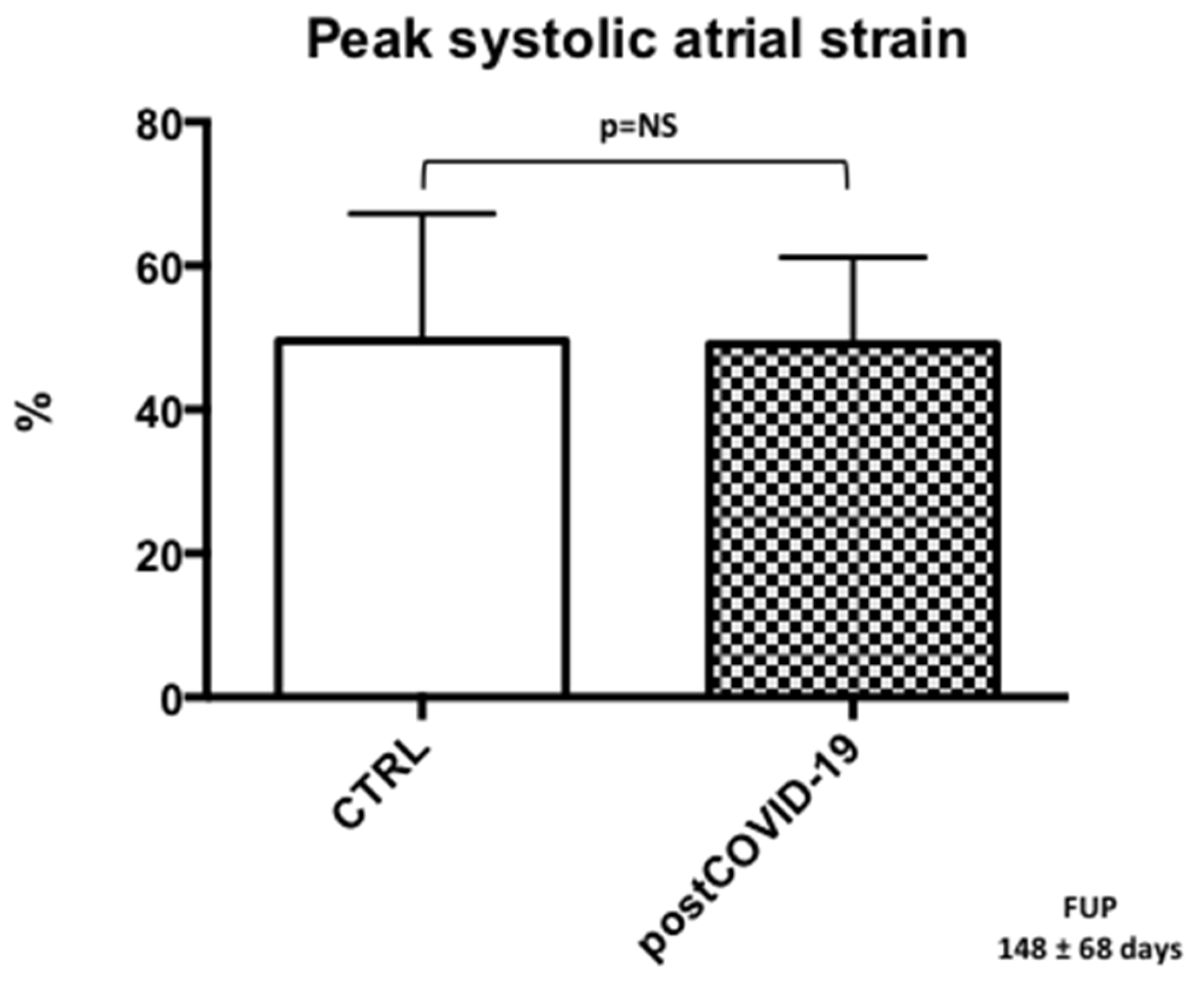
3.5. Left Atrial Reservoir Strain
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Feldstein, L.R.; Tenforde, M.W.; Friedman, K.G.; Newhams, M.; Rose, E.B.; Dapul, H.; Soma, V.L.; Maddux, A.B.; Mourani, P.M.; Bowens, C.; et al. Characteristics and Outcomes of US Children and Adolescents with Multisystem Inflammatory Syndrome in Children (MIS-C) Compared with Severe Acute COVID-19. JAMA 2021, 325, 1074–1087. [Google Scholar] [CrossRef]
- Valverde, I.; Singh, Y.; Sanchez-de-Toledo, J.; Theocharis, P.; Chikermane, A.; Di Filippo, S.; Kuciñska, B.; Mannarino, S.; Tamariz-Martel, A.; Gutierrez-Larraya, F.; et al. Acute Cardiovascular Manifestations in 286 Children with Multisystem Inflammatory Syndrome Associated with COVID-19 Infection in Europe. Circulation 2021, 143, 21–32. [Google Scholar] [CrossRef] [PubMed]
- Sirico, D.; Basso, A.; Reffo, E.; Cavaliere, A.; Castaldi, B.; Sabatino, J.; Meneghel, A.; Martini, G.; Da Dalt, L.; Zulian, F.; et al. Early Echocardiographic and Cardiac MRI Findings in Multisystem Inflammatory Syndrome in Children. J. Clin. Med. 2021, 10, 3360. [Google Scholar] [CrossRef] [PubMed]
- Sirico, D.; Di Chiara, C.; Costenaro, P.; Bonfante, F.; Cozzani, S.; Plebani, M.; Reffo, E.; Castaldi, B.; Donà, D.; Da Dalt, L.; et al. Left ventricular longitudinal strain alterations in asymptomatic or mildly symptomatic paediatric patients with SARS-CoV-2 infection. Eur. Hear. J. Cardiovasc. Imaging 2021, 23, 1083–1089. [Google Scholar] [CrossRef] [PubMed]
- Matsubara, D.; Kauffman, H.L.; Wang, Y.; Calderon-Anyosa, R.; Nadaraj, S.; Elias, M.D.; White, T.J.; Torowicz, D.L.; Yubbu, P.; Giglia, T.M.; et al. Echocardiographic Findings in Pediatric Multisystem Inflammatory Syndrome Associated with COVID-19 in the United States. J. Am. Coll. Cardiol. 2020, 76, 1947–1961. [Google Scholar] [CrossRef]
- Sirico, D.; Basso, A.; Sabatino, J.; Reffo, E.; Cavaliere, A.; Biffanti, R.; Cerutti, A.; Castaldi, B.; Zulian, F.; Da Dalt, L.; et al. Evolution of echocardiographic and cardiac magnetic resonance imaging abnormalities during follow-up in patients with multisystem inflammatory syndrome in children. Eur. Heart J. Cardiovasc. Imaging 2022, 23, 1066–1074. [Google Scholar] [CrossRef]
- Narayanan, A.; Aurigemma, G.P.; Chinali, M.; Hill, J.C.; Meyer, T.E.; Tighe, D.A. Cardiac mechanics in mild hypertensive heart disease: A speckle-strain imaging study. Circ. Cardiovasc. Imaging 2009, 2, 382–390. [Google Scholar] [CrossRef]
- Potter, E.; Marwick, T.H. Assessment of Left Ventricular Function by Echocardiography: The Case for Routinely Adding Global Longitudinal Strain to Ejection Fraction. JACC Cardiovasc. Imaging 2018, 11, 260–274. [Google Scholar] [CrossRef]
- Baruch, G.; Rothschild, E.; Sadon, S.; Szekely, Y.; Lichter, Y.; Kaplan, A.; Taieb, P.; Banai, A.; Hochstadt, A.; Merdler, I.; et al. Evolution of right and left ventricle routine and speckle-tracking echocardiography in patients recovering from coronavirus disease 2019: A longitudinal study. Eur. Heart J. Cardiovasc. Imaging 2021, 23, 1055–1065. [Google Scholar] [CrossRef]
- WHO. Clinical Management of COVID-19. Living Guidance for Clinical Management of COVID-19, 23 November 2021. 2021. Available online: https://www.who.int/publications/i/item/WHO-2019-nCoV-clinical-2021-2 (accessed on 23 March 2021).
- Padoan, A.; Bonfante, F.; Pagliari, M.; Bortolami, A.; Negrini, D.; Zuin, S.; Bozzato, D.; Cosma, C.; Sciacovelli, L.; Plebani, M. Analytical and clinical performances of five immunoassays for the detection of SARS-CoV-2 antibodies in comparison with neutralization activity. EBioMedicine 2020, 62, 103101. [Google Scholar] [CrossRef]
- Bonfante, F.; Costenaro, P.; Cantarutti, A.; Di Chiara, C.; Bortolami, A.; Petrara, M.R.; Carmona, F.; Pagliari, M.; Cosma, C.; Cozzani, S.; et al. Mild SARS-CoV-2 Infections and Neutralizing Antibody Titers. Pediatrics 2021, 148, e2021052173. [Google Scholar] [CrossRef] [PubMed]
- Di Salvo, G.; Siblini, G.; Issa, Z.; Mohammed, H.; Abu Hazeem, A.; Pergola, V.; Muhanna, N.; Al Qweai, N.; Galzerano, D.; Fadel, B.; et al. Left ventricular mechanics in patients with abnormal origin of the left main coronary artery from the pulmonary trunk late after successful repair. Cardiology 2017, 136, 71–76. [Google Scholar] [CrossRef] [PubMed]
- Di Salvo, G.; Al Bulbul, Z.; Issa, Z.; Fadel, B.; Al-Sehly, A.; Pergola, V.; Al Halees, Z.; Al Fayyadh, M. Left ventricular mechanics after arterial switch operation: A speckle-tracking echocardiography study. J. Cardiovasc. Med. 2016, 17, 217–224. [Google Scholar] [CrossRef] [PubMed]
- Sabatino, J.; Di Salvo, G.; Prota, C.; Bucciarelli, V.; Josen, M.; Paredes, J.; Borrelli, N.; Sirico, D.; Prasad, S.; Indolfi, C.; et al. Left atrial strain to identify diastolic dysfunction in children with cardiomyopathies. J. Clin. Med. 2019, 8, 1243. [Google Scholar] [CrossRef]
- Sabatino, J.; Di Salvo, G.; Krupickova, S.; Fraisse, A.; Prota, C.; Bucciarelli, V.; Josen, M.; Paredes, J.; Sirico, D.; Voges, I.; et al. Left Ventricular Twist Mechanics to Identify Left Ventricular Noncompaction in Childhood. Circ Cardiovasc. Imaging 2019, 12, e007805. [Google Scholar] [CrossRef] [PubMed]
- Molteni, E.; Sudre, C.H.; Canas, L.S.; Bhopal, S.S.; Hughes, R.C.; Antonelli, M.; Murray, B.; Kläser, K.; Kerfoot, E.; Chen, L.; et al. Illness duration and symptom profile in symptomatic UK school-aged children tested for SARS-CoV-2. Lancet Child Adolesc. Health 2021, 5, 708–718. [Google Scholar] [CrossRef]
- Funk, A.L.; Florin, T.A.; Kuppermann, N.; Tancredi, D.J.; Xie, J.; Kim, K.; Neuman, M.I.; Ambroggio, L.; Plint, A.C.; Mintegi, S.; et al. Pediatric Emergency Research Network-COVID-19 Study Team. Outcomes of SARS-CoV-2-Positive Youths Tested in Emergency Departments: The Global PERN-COVID-19 Study. JAMA Netw. Open 2022, 5, e2142322. [Google Scholar] [CrossRef]
- Daniels, C.J.; Rajpal, S.; Greenshields, J.T.; Rosenthal, G.L.; Chung, E.H.; Terrin, M.; Jeudy, J.; Mattson, S.E.; Law, I.H.; Borchers, J.; et al. Big Ten COVID-19 Cardiac Registry Investigators. Prevalence of Clinical and Subclinical Myocarditis in Competitive Athletes with Recent SARS-CoV-2 Infection: Results From the Big Ten COVID-19 Cardiac Registry. JAMA Cardiol. 2021, 6, 1078–1087. [Google Scholar] [CrossRef]
- Groff, D.; Sun, A.; Ssentongo, A.E.; Ba, D.M.; Parsons, N.; Poudel, G.R.; Lekoubou, A.; Oh, J.S.; Ericson, J.E.; Ssentongo, P.; et al. Short-term and Long-term Rates of Postacute Sequelae of SARS-CoV-2 Infection: A Systematic Review. JAMA Netw. Open 2021, 4, e2128568. [Google Scholar] [CrossRef]
- Petersen, S.E.; Friedrich, M.G.; Leiner, T.; Elias, M.D.; Ferreira, V.M.; Fenski, M.; Flamm, S.D.; Fogel, M.; Garg, R.; Halushka, M.K.; et al. Cardiovascular Magnetic Resonance for Patients With COVID-19. JACC Cardiovasc. Imaging 2021, 15, 685–699. [Google Scholar] [CrossRef]
- Munblit, D.; Nicholson, T.R.; Needham, D.M.; Seylanova, N.; Parr, C.; Chen, J.; Kokorina, A.; Sigfrid, L.; Buonsenso, D.; Bhatnagar, S.; et al. Studying the post-COVID-19 condition: Research challenges, strategies, and importance of Core Outcome Set development. BMC Med. 2022, 20, 50. [Google Scholar] [CrossRef] [PubMed]
- Davis, H.E.; Assaf, G.S.; McCorkell, L.; Wei, H.; Low, R.J.; Re’Em, Y.; Redfield, S.; Austin, J.P.; Akrami, A. Characterizing long COVID in an international cohort: 7 months of symptoms and their impact. Eclinicalmedicine 2021, 38, 101019. [Google Scholar] [CrossRef]
- Ziauddeen, N.; Gurdasani, D.; O’Hara, M.E.; Hastie, C.; Roderick, P.; Yao, G.; Alwan, N.A. Characteristics of long covid: Findings from a social media survey. medRxiv 2021, 75, A90. [Google Scholar] [CrossRef]
- Robinson, F.A.; Mihealsick, R.P.; Wagener, B.M.; Hanna, P.; Poston, M.D.; Efimov, I.R.; Shivkumar, K.; Hoover, D.B. Role of angiotensin-converting enzyme 2 and pericytes in cardiac complications of COVID-19 infection. Am. J. Physiol. Circ. Physiol. 2020, 319, H1059–H1068. [Google Scholar] [CrossRef]
- Nalbandian, A.; Sehgal, K.; Gupta, A.; Madhavan, M.V.; McGroder, C.; Stevens, J.S.; Cook, J.R.; Nordvig, A.S.; Shalev, D.; Sehrawat, T.S.; et al. Post-acute COVID-19 syndrome. Nat. Med. 2021, 27, 601–615. [Google Scholar] [CrossRef] [PubMed]
- Escher, F.; Pietsch, H.; Aleshcheva, G.; Bock, T.; Baumeier, C.; Elsaesser, A.; Wenzel, P.; Hamm, C.; Westenfeld, R.; Schultheiss, M.; et al. Detection of viral SARS-CoV-2 genomes and histopathological changes in endomyocardial biopsies. ESC Heart Fail. 2020, 7, 2440–2447. [Google Scholar] [CrossRef] [PubMed]
- Almamlouk, R.; Kashour, T.; Obeidat, S.; Bois, M.C.; Maleszewski, J.J.; Omrani, O.A.; Tleyjeh, R.; Berbari, E.; Chakhachiro, Z.; Zein-Sabatto, B.; et al. COVID-19–Associated cardiac pathology at the postmortem evaluation: A collaborative systematic review. Clin. Microbiol. Infect. 2022, 28, 1066–1075. [Google Scholar] [CrossRef] [PubMed]
- Biering-Sørensen, T.; Biering-Sørensen, S.R.; Olsen, F.J.; Sengeløv, M.; Jørgensen, P.G.; Mogelvang, R.; Shah, A.M.; Jensen, J.S. Global Longitudinal Strain by Echocardiography Predicts Long-Term Risk of Cardiovascular Morbidity and Mortality in a Low-Risk General Population: The Copenhagen City Heart Study. Circ. Cardiovasc. Imaging 2017, 10, e005521. [Google Scholar] [CrossRef]
- Verdonschot, J.A.J.; Henkens, M.T.H.M.; Wang, P.; Schummers, G.; Raafs, A.G.; Krapels, I.P.C.; van Empel, V.; Heymans, S.R.B.; Brunner-La Rocca, H.P.; Knackstedt, C. A global longitudinal strain cut-off value to predict adverse outcomes in individuals with a normal ejection fraction. ESC Heart Fail. 2021, 8, 4343–4345. [Google Scholar] [CrossRef]
| Clinical and Echocardiographic Variables | Post-COVID-19 (N = 157) | CTRL (N = 107) |
|---|---|---|
| Age (yrs) | 7.7 ± 4.5 | 11.0 ± 4.3 |
| Age range (yrs) | 0.3–18 | 0.3–18 |
| Female, n (%) | 62 (39) | 41 (38) |
| Body Surface Area (BSA) (m2) | 1.2 ± 0.4 | 1.3 ± 0.4 |
| LVEDD (mm) | 39.1 ± 6.7 | 40.7 ± 6.3 |
| LVEDD Z score | −0.33 ± 0.91 | −0.23 ± 0.75 |
| LVESD (mm) | 24.8 ± 4.7 | 26.2 ± 4.4 |
| LVESD Z score | −0.16 ± 1.06 | −0.15 ± 0.82 |
| LVEF (%) | 65.6 ± 4 | 65.0 ± 5 |
| LAVi (mL/m2) | 17.3 ± 6.4 | 17.7 ± 4.9 |
| TAPSE (mm) | 19.8 ±3.0 | 20.1 ±3.4 |
| E/A ratio | 1.9 ± 0.51 | 1.8 ± 0.51 |
| E/E’ avg, ratio | 5.9 ± 1.1 | 6.2 ± 1.3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sabatino, J.; Di Chiara, C.; Di Candia, A.; Sirico, D.; Donà, D.; Fumanelli, J.; Basso, A.; Pogacnik, P.; Cuppini, E.; Romano, L.R.; et al. Mid- and Long-Term Atrio-Ventricular Functional Changes in Children after Recovery from COVID-19. J. Clin. Med. 2023, 12, 186. https://doi.org/10.3390/jcm12010186
Sabatino J, Di Chiara C, Di Candia A, Sirico D, Donà D, Fumanelli J, Basso A, Pogacnik P, Cuppini E, Romano LR, et al. Mid- and Long-Term Atrio-Ventricular Functional Changes in Children after Recovery from COVID-19. Journal of Clinical Medicine. 2023; 12(1):186. https://doi.org/10.3390/jcm12010186
Chicago/Turabian StyleSabatino, Jolanda, Costanza Di Chiara, Angela Di Candia, Domenico Sirico, Daniele Donà, Jennifer Fumanelli, Alessia Basso, Pietro Pogacnik, Elena Cuppini, Letizia Rosa Romano, and et al. 2023. "Mid- and Long-Term Atrio-Ventricular Functional Changes in Children after Recovery from COVID-19" Journal of Clinical Medicine 12, no. 1: 186. https://doi.org/10.3390/jcm12010186
APA StyleSabatino, J., Di Chiara, C., Di Candia, A., Sirico, D., Donà, D., Fumanelli, J., Basso, A., Pogacnik, P., Cuppini, E., Romano, L. R., Castaldi, B., Reffo, E., Cerutti, A., Biffanti, R., Cozzani, S., Giaquinto, C., & Di Salvo, G. (2023). Mid- and Long-Term Atrio-Ventricular Functional Changes in Children after Recovery from COVID-19. Journal of Clinical Medicine, 12(1), 186. https://doi.org/10.3390/jcm12010186








