Improving Upper Extremity Bradykinesia in Parkinson’s Disease: A Randomized Clinical Trial on the Use of Gravity-Supporting Exoskeletons
Abstract
:1. Introduction
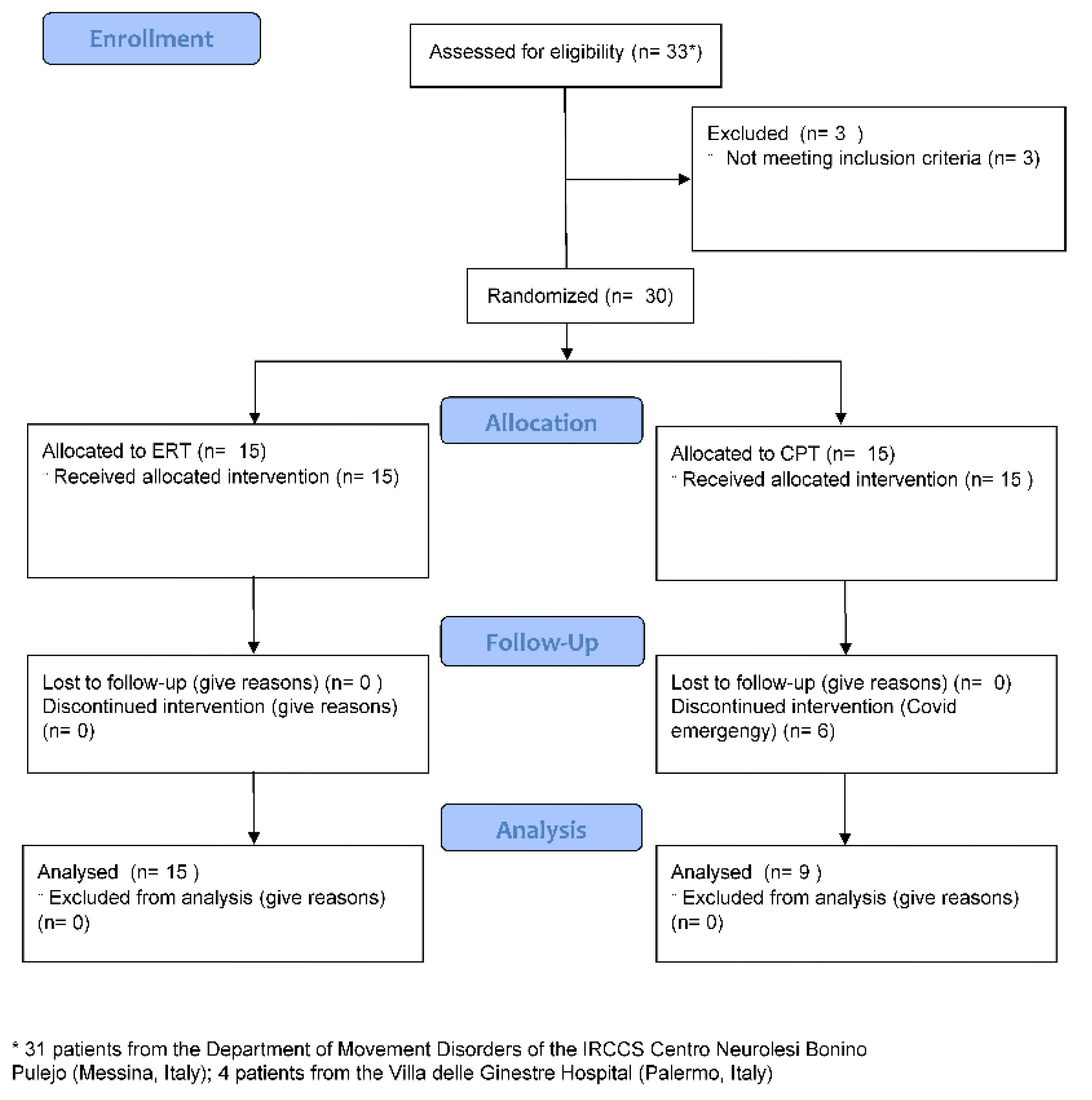
2. Materials and Methods
2.1. Study Design and Population
2.2. Randomization
2.3. Sample Size and Power Analysis
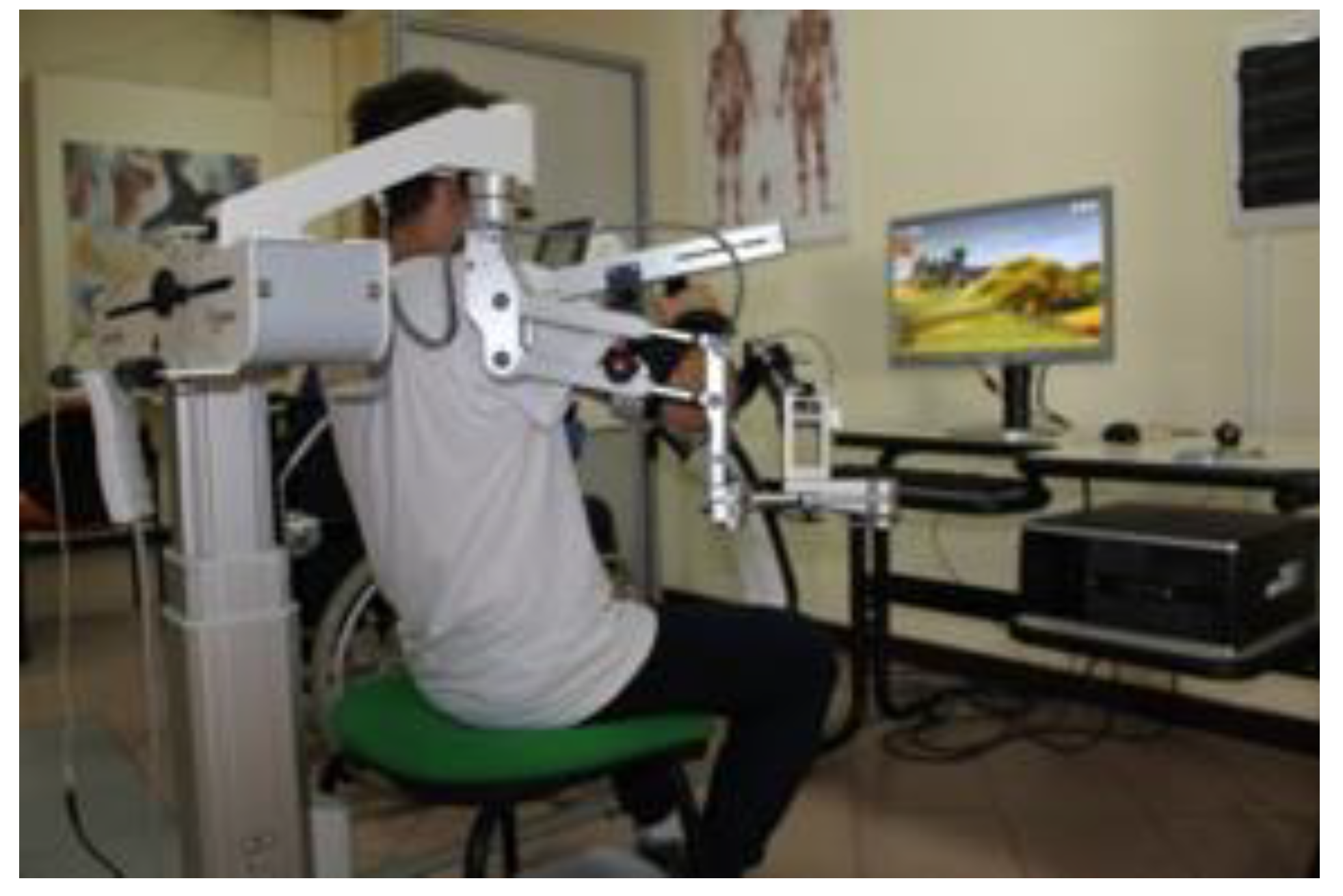
2.4. Experimental Robotic Therapy
2.5. Conventional Physical Therapy
2.6. Outcome Measures
2.6.1. Primary Outcome Measure
2.6.2. Secondary Outcome Measures
2.7. Statistical Analysis
3. Results
3.1. Baseline (T0)
3.2. Post-Treatment (T1)
4. Discussion
Strengths and Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ponsen, M.M.; Daffertshofer, A.; Wolters, E.C.; Beek, P.J.; Berendse, H.W. Impairment of complex upper limb motor function in de novo Parkinson’s disease. Parkinsonism Relat. Disord. 2008, 14, 199–204. [Google Scholar] [CrossRef] [PubMed]
- Fitts, P.M. The information capacity of the human motor system in controlling the amplitude of movement. J. Exp. Psychol. 1954, 47, 381–391. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sanes, J.N. Information processing deficits in Parkinson’s disease during movement. Neuropsychologia 1985, 23, 381–392. [Google Scholar] [CrossRef]
- Quinn, L.; Busse, M.; Dal Bello-Haas, V. Management of upper extremity dysfunction in people with Parkinson disease and Huntington disease: Facilitating outcomes across the disease lifespan. J. Hand Ther. 2013, 26, 148–155. [Google Scholar] [CrossRef] [PubMed]
- Rocchi, L.; Chiari, L.; Horak, F.B. Effects of deep brain stimulation and levodopa on postural sway in Parkinson’s disease. J. Neurol. Neurosurg. Psychiatry 2002, 73, 267–274. [Google Scholar] [CrossRef]
- Raggi, A.; Leonardi, M.; Ajovalasit, D.; Carella, F.; Soliveri, P.; Albanese, A.; Romito, L. Disability and profiles of functioning of patients with Parkinson’s disease described with ICF classification. Int. J. Rehabil. Res. 2011, 34, 141–150. [Google Scholar] [CrossRef]
- Diederich, N.J.; Moore, C.G.; Leurgans, S.E.; Chmura, T.A.; Goetz, C.G. Parkinson disease with old-age onset: A comparative study with subjects with middle-age onset. Arch. Neurol. 2003, 60, 529–533. [Google Scholar] [CrossRef]
- Soh, S.E.; McGinley, J.L.; Watts, J.J.; Iansek, R.; Murphy, A.T.; Menz, H.B.; Huxham, F.; Morris, M.E. Determinants of health-related quality of life in people with Parkinson’s disease: A path analysis. Qual. Life Res. 2013, 22, 1543–1553. [Google Scholar] [CrossRef]
- Berardelli, A.; Rothwell, J.C.; Thompson, P.D.; Hallett, M. Pathophysiology of bradykinesia in Parkinson’s disease. Brain 2001, 124, 2131–2146. [Google Scholar] [CrossRef] [Green Version]
- Espay, A.J.; Beaton, D.E.; Morgante, F.; Gunraj, C.A.; Lang, A.E.; Chen, R. Impairments of speed and amplitude of movement in Parkinson’s disease: A pilot study. Mov. Disord. 2009, 24, 1001–1008. [Google Scholar] [CrossRef]
- Hallett, M.; Khoshbin, S. A physiological mechanism of bradykinesia. Brain 1980, 103, 301–314. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, M.A.; Rodrigues, A.M.; Caballero, R.M.; Petersen, R.D.; Shim, J.K. Strength and isometric torque control in individuals with Parkinson’s disease. Exp. Brain Res. 2008, 184, 445–450. [Google Scholar] [CrossRef] [PubMed]
- Bologna, M.; Paparella, G.; Fasano, A.; Hallett, M.; Berardelli, A. Evolving concepts on bradykinesia. Brain 2020, 143, 727–750. [Google Scholar] [CrossRef] [PubMed]
- Vercruysse, S.; Gilat, M.; Shine, J.M.; Heremans, E.; Lewis, S.; Nieuwboer, A. Freezing beyond gait in Parkinson’s disease: A review of current neurobehavioral evidence. Neurosci. Biobehav. Rev. 2014, 43, 213–227. [Google Scholar] [CrossRef] [PubMed]
- Picelli, A.; Tamburin, S.; Passuello, M.; Waldner, A.; Smania, N. Robot-assisted arm training in patients with Parkinson’s disease: A pilot study. J. Neuroeng. Rehabil. 2014, 11, 28. [Google Scholar] [CrossRef] [Green Version]
- Calabrò, R.S.; Cacciola, A.; Bertè, F.; Manuli, A.; Leo, A.; Bramanti, A.; Naro, A.; Milardi, D.; Bramanti, P. Robotic gait rehabilitation and substitution devices in neurological disorders: Where are we now? Neurol. Sci. 2016, 37, 503–514. [Google Scholar] [CrossRef]
- Brognara, L.; Navarro-Flores, E.; Iachemet, L.; Serra-Catalá, N.; Cauli, O. Beneficial Effect of Foot Plantar Stimulation in Gait Parameters in Individuals with Parkinson’s Disease. Brain Sci. 2020, 10, 69. [Google Scholar] [CrossRef] [Green Version]
- Mehrholz, J.; Hädrich, A.; Platz, T.; Kugler, J.; Pohl, M. Electromechanical and robot-assisted arm training for improving generic activities of daily living, arm function, and arm muscle strength after stroke. Cochrane Database Syst. Rev. 2012, 6, CD006876. [Google Scholar] [CrossRef]
- Bonanno, L.; Russo, M.; Bramanti, A.; Calabrò, R.S.; Marino, S. Functional connectivity in multiple sclerosis after robotic rehabilitative treatment: A case report. Medicine 2019, 98, e15047. [Google Scholar] [CrossRef]
- Maggio, M.G.; Russo, M.; Cuzzola, M.F.; Destro, M.; Rosa, G.L.; Molonia, F.; Bramanti, P.; Lombardo, G.; Luca, R.D.; Calabrò, R.S. Virtual reality in multiple sclerosis rehabilitation: A review on cognitive and motor outcomes. J. Clin. Neurosci. 2019, 65, 106–111. [Google Scholar] [CrossRef]
- Clark, W.E.; Sivan, M.; O’Connor, R.J. Evaluating the use of robotic and virtual reality rehabilitation technologies to improve function in stroke survivors: A narrative review. J. Rehabil. Assist. Technol. Eng. 2019, 6, 2055668319863557. [Google Scholar] [CrossRef]
- Baur, K.; Schättin, A.; de Bruin, E.D.; Riener, R.; Duarte, J.E.; Wolf, P. Trends in robot-assisted and virtual reality-assisted neuromuscular therapy: A systematic review of health-related multiplayer games. J. Neuroeng. Rehabil. 2018, 15, 107. [Google Scholar] [CrossRef] [PubMed]
- Park, J.; Chung, Y. The effects of robot-assisted gait training using virtual reality and auditory stimulation on balance and gait abilities in persons with stroke. NeuroRehabilitation 2018, 43, 227–235. [Google Scholar] [CrossRef]
- Smania, N.; Picelli, A.; Geroin, C.; Munari, D.; Waldner, A.; Gandolfi, M. Robot-assisted gait training in patients with Parkinson’s disease. Neurodegener. Dis. Manag. 2013, 3, 321–330. [Google Scholar] [CrossRef]
- Capecci, M.; Pournajaf, S.; Galafate, D.; Sale, P.; Pera, D.L.; Goffredo, M.; Pandis, M.F.D.; Andrenelli, E.; Pennacchioni, M.; Ceravolo, M.G.; et al. Clinical effects of robot-assisted gait training and treadmill training for Parkinson’s disease. A randomized control. Trial. Ann. Phys. Rehabil. Med. 2019, 62, 303–312. [Google Scholar] [CrossRef]
- Cifuentes, C.A.; Frizera, A. Human-Robot Interaction for Assisting. In Human-Robot Interaction Strategies for Walker-Assisted Locomotion; Locomotion, H., Ed.; Springer: Berlin/Heidelberg, Germany, 2016; pp. 17–31. [Google Scholar]
- Scaletta, T.; Komada, S.; Oboe, R. Development of a human assistive robot to support hip joint movement during sit-to-stand using non-linear springs. IEEJ J. Ind. Appl. 2016, 5, 261–266. [Google Scholar] [CrossRef] [Green Version]
- Huen, D.; Liu, J.; Lo, B. An integrated wearable robot for tremor suppression with context aware sensing. In Proceedings of the 2016 IEEE 13th International Conference on Wearable and Implantable Body Sensor Networks (BSN), San Francisco, CA, USA, 14–17 June 2016. [Google Scholar]
- Voiculescu, I.; Cameron, S.; Zabarauskas, M.; Kozlowski, P. Towards Robot-Assisted Rehabilitation of Upper Limb dysfunction. In Advances in Robot Design and Intelligent Control: Advances in Intelligent Systems and Computing; Borangiu, T., Ed.; Springer: Berlin/Heidelberg, Germany, 2016; pp. 347–355. [Google Scholar]
- Asakawa, T.; Sugiyama, K.; Nozaki, T.; Sameshima, T.; Kobayashi, S.; Wang, L.; Hong, Z.; Chen, S.; Li, C.; Namba, H. Can the Latest Computerized Technologies Revolutionize Conventional Assessment Tools and Therapies for a Neurological Disease? The Example of Parkinson’s Disease. Neurol. Med.-Chir. 2019, 59, 69–78. [Google Scholar] [CrossRef] [Green Version]
- Hughes, A.J.; Daniel, S.E.; Kilford, L.; Lees, A.J. Accuracy of clinical diagnosis of idiopathic Parkinson’s disease: A clinico-pathological study of 100 cases. J. Neurol. Neurosurg. Psychiatry 1992, 55, 181–184. [Google Scholar] [CrossRef] [Green Version]
- Hoehn, M.M.; Yahr, M.D. Parkinsonism: Onset, progression and mortality. Neurology 1967, 17, 427–442. [Google Scholar] [CrossRef] [Green Version]
- Folstein, M.F.; Folstein, S.E.; McHugh, P.R. “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J. Psychiatr. Res. 1975, 12, 189–198. [Google Scholar] [CrossRef]
- Movement Disorder Society Task Force on Rating Scales for Parkinson’s Disease. The Unified Parkinson’s Disease Rating Scale (UPDRS): Status and recommendations. Mov. Disord. 2003, 18, 738–750. [Google Scholar] [CrossRef] [PubMed]
- Proud, E.L.; Bilney, B.; Miller, K.J.; Morris, M.E.; McGinley, J.L. Measuring Hand Dexterity in People with Parkinson’s Disease: Reliability of Pegboard Tests. Am. J. Occup. Ther. 2019, 73, p1–p7304205050. [Google Scholar] [CrossRef] [PubMed]
- Colomer, C.; Baldoví, A.; Torromé, S.; Navarro, M.D.; Moliner, B.; Ferri, J.; Noé, E. Efficacy of Armeo® Spring during the chronic phase of stroke. Study Mild Moderate Cases Hemiparesis Neurol. 2013, 28, 261–267. [Google Scholar] [CrossRef]
- Oxford Grice, K.; Vogel, K.A.; Le, V.; Mitchell, A.; Muniz, S.; Vollmer, M.A. Adult norms for a commercially available Nine Hole Peg Test for finger dexterity. Am. J. Occup. Ther. 2003, 57, 570–573. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Raciti, L.; Nicoletti, A.; Mostile, G.; Bonomo, R.; Contrafatto, D.; Dibilio, V.; Luca, A.; Sciacca, G.; Cicero, C.E.; Vasta, R.; et al. Validation of the UPDRS section IV for detection of motor fluctuations in Parkinson’s disease. Parkinsonism Relat. Disord. 2016, 27, 98–101. [Google Scholar] [CrossRef]
- Fugl-Meyer, A.R.; Jääskö, L.; Leyman, I.; Olsson, S.; Steglind, S. The post-stroke hemiplegic patient. 1. a method for evaluation of physical performance. Scand. J. Rehabil. Med. 1975, 7, 13–31. [Google Scholar] [PubMed]
- Opara, J.; Małecki, A.; Małecka, E.; Socha, T. Motor assessment in Parkinson`s disease. Ann. Agric. Environ. Med. 2017, 24, 411–415. [Google Scholar] [CrossRef]
- Lee, K.S.; Lee, W.H.; Hwang, S. Modified constraint-induced movement therapy improves fine and gross motor performance of the upper limb in Parkinson disease. Am. J. Phys. Med. Rehabil. 2011, 90, 380–386. [Google Scholar] [CrossRef]
- Keith, R.A.; Granger, C.V.; Hamilton, B.B.; Sherwin, F.S. The functional independence measure: A new tool for rehabilitation. Adv. Clin. Rehabil. 1987, 1, 6–18. [Google Scholar]
- Childs, J.D.; Piva, S.R.; Fritz, J.M. Responsiveness of the numeric pain rating scale in patients with low back pain. Spine 2005, 30, 1331–1334. [Google Scholar] [CrossRef]
- Demeurisse, G.; Demol, O.; Robaye, E. Motor evaluation in vascular hemiplegia. Eur. Neurol. 1980, 19, 382–389. [Google Scholar] [CrossRef] [PubMed]
- Calabrò, R.S.; Naro, A.; Russo, M.; Leo, A.; Luca, R.D.; Balletta, T.; Buda, A.; Rosa, G.L.; Bramanti, A.; Bramanti, P. The role of virtual reality in improving motor performance as revealed by EEG: A randomized clinical trial. J. Neuroeng. Rehabil. 2017, 14, 53. [Google Scholar] [CrossRef] [PubMed]
- Raciti, L.; Pizzurro, R.; Occhipinti, F.; Manuli, A.; Corallo, F.; Calabrò, R.S. A multidisciplinary advanced approach in central pontine myelinolysis recovery: Considerations about a case report. Disabil. Rehabil. Assist. Technol. 2020, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Maggio, M.G.; Torrisi, M.; Buda, A.; Luca, R.D.; Piazzitta, D.; Cannavò, A.; Leo, A.; Milardi, D.; Manuli, A.; Calabro, R.S. Effects of robotic neurorehabilitation through lokomat plus virtual reality on cognitive function in patients with traumatic brain injury: A retrospective case-control study. Int. J. Neurosci. 2020, 130, 117–123. [Google Scholar] [CrossRef]
- Calabrò, R.S.; Russo, M.; Naro, A.; Milardi, D.; Balletta, T.; Leo, A.; Filoni, S.; Bramanti, P. Who May Benefit from Armeo Power Treatment? A Neurophysiological Approach to Predict Neurorehabilitation Outcomes. PM R. 2016, 8, 971–978. [Google Scholar] [CrossRef]
- Bruni, M.F.; Melegari, C.; De Cola, M.C.; Bramanti, A.; Bramanti, P.; Calabrò, R.S. What does best evidence tell us about robotic gait rehabilitation in stroke patients: A systematic review and meta-analysis. J. Clin. Neurosci. 2018, 48, 11–17. [Google Scholar] [CrossRef]
- Moon, S.; Huang, C.K.; Sadeghi, M.; Akinwuntan, A.E.; Devos, H. Proof-of-Concept of the Virtual Reality Comprehensive Balance Assessment and Training for Sensory Organization of Dynamic Postural Control. Front. Bioeng. Biotechnol. 2021, 9, 678006. [Google Scholar] [CrossRef]
- Naro, A.; Pignolo, L.; Bruschetta, D.; Calabrò, R.S. What about the role of the cerebellum in music-associated functional recovery? A secondary EEG analysis of a randomized clinical trial in patients with Parkinson disease. Parkinsonism Relat. Disord. 2022, 96, 57–64. [Google Scholar] [CrossRef]
- Straudi, S.; Benedetti, M.G.; Venturini, E.; Manca, M.; Foti, C.; Basaglia, N. Does robot-assisted gait training ameliorate gait abnormalities in multiple sclerosis? A pilot randomized-control trial. NeuroRehabilitation 2013, 33, 555–563. [Google Scholar] [CrossRef]
- Masiero, S.; Armani, M.; Rosati, G. Upper-limb robot-assisted therapy in rehabilitation of acute stroke patients: Focused review and results of new randomized controlled trial. J. Rehabil. Res. Dev. 2011, 48, 355–366. [Google Scholar] [CrossRef]
- Maggio, M.G.; Luca, R.D.; Manuli, A.; Buda, A.; Cuzzola, M.F.; Leonardi, S.; D’Aleo, G.; Bramanti, P.; Russo, M.; Calabrò, R.S. Do patients with multiple sclerosis benefit from semi-immersive virtual reality? A randomized clinical trial on cognitive and motor outcomes. Appl. Neuropsychol. Adult 2022, 29, 59–65. [Google Scholar] [CrossRef]
- López-López, D.; Pérez-Ríos, M.; Ruano-Ravina, A.; Losa-Iglesias, M.E.; Becerro-de-Bengoa-Vallejo, R.; Romero-Morales, C.; Calvo-Lobo, C.; Navarro-Flores, E. Impact of quality of life related to foot problems: A case-control study. Sci. Rep. 2021, 11, 14515. [Google Scholar] [CrossRef]
- Mazzoni, P.; Shabbott, B.; Cortés, J.C. Motor control abnormalities in Parkinson’s disease. Cold Spring Harb. Perspect. Med. 2012, 2, a009282. [Google Scholar] [CrossRef] [Green Version]
- Intention to treat analysis and per protocol analysis: Complementary information. Prescrire Int. 2012, 21, 304–306.
- Tripepi, G.; Chesnaye, N.C.; Dekker, F.W.; Zoccali, C.; Jager, K.J. Intention to treat and per protocol analysis in clinical trials. Nephrology 2020, 25, 513–517. [Google Scholar] [CrossRef] [Green Version]
- Taveggia, G.; Borboni, A.; Salvi, L.; Mule, C.; Fogliaresi, S.; Villafañe, J.H.; Casale, R. Efficacy of robot-assisted rehabilitation for the functional recovery of the upper limb in post-stroke patients: A randomized controlled study. Eur. J. Phys. Rehabil. Med. 2016, 52, 767–773. [Google Scholar]
- Gijbels, D.; Lamers, I.; Kerkhofs, L.; Alders, G.; Knippenberg, E.; Feys, P. The Armeo Spring as training tool to improve upper limb functionality in multiple sclerosis: A pilot study. J. Neuroeng. Rehabil. 2011, 8, 5. [Google Scholar] [CrossRef] [Green Version]
- Ishikuro, K.; Dougu, N.; Nukui, T.; Yamamoto, M.; Nakatsuji, Y.; Kuroda, S.; Matsushita, I.; Nishimaru, H.; Araujo, M.F.P.; Nishijo, H. Effects of Transcranial Direct Current Stimulation (tDCS) Over the Frontal Polar Area on Motor and Executive Functions in Parkinson’s Disease; A Pilot Study. Front. Aging Neurosci. 2018, 10, 231. [Google Scholar] [CrossRef] [Green Version]
| ERT (n = 15) | CPT (n = 9) | p-Value | |
|---|---|---|---|
| Age (years) | 65.7 (7) | 62.7 (10.1) | 0.1 |
| Disease Duration (years) | 5.3 (3.4) | 6.2 (4.6) | 0.6 |
| LEDD (mg/day) | 544 (198) | 583 (191) | 0.7 |
| H&Y | 2 (2–3) | 2 (2–3) | 0.4 |
| T0 | T1 | Within-Group Comparison | Between-Group Comparison | ||
|---|---|---|---|---|---|
| 9HPT | ERT | 42.2 (17) | 34.1 (14) | 0.006 | 0.004 |
| CPT | 35.1 (6.8) | 31.4 (5.4) | 0.9 | ||
| UPDRS-III | ERT | 28 (23–33) | 21 (16–26) | 0.06 | 0.5 |
| CPT | 37 (31.5–41) | 32 (23.25;40) | 0.9 | ||
| P-NRS | ERT | 2.5 (0.5–3.5) | 1.1 (0.3–1.8) | 0.007 | 0.9 |
| CPT | 4 (3–5) | 1 (0–1.5) | 0.01 | ||
| MI-UE | ERT | 72 (65–80) * | 89 (83–94) * | 0.04 | 0.0001 |
| CPT | 77 (73.25–82) | 82 (79.25;88.5) | 0.8 | ||
| FIM | ERT | 104 (98–109) * | 110 (105–115) * | 0.6 | 0.6 |
| CPT | 100 (99–103) | 101 (100–106) | 0.9 | ||
| FMA-UE | ERT | 48 (45–52) | 53 (5–56) | 0.007 | 0.009 |
| CPT | 53 (51–55) | 56 (52.5–59.5) | 0.9 |
| T0 | T1 | Within-Group Comparison | Between-Group Comparison | ||
|---|---|---|---|---|---|
| 9HPT | ERT | 42.2 (17.3) * | 34.1 (13.9) | 0.001 | 0.7 |
| CPT | 35.1 (6.8) | 31.4 (5.4) | 0.003 | ||
| UPDRS-III | ERT | 28 (23.25–32) | 21 (15–22.5) | 0.07 | 0.7 |
| CPT | 28 (26.25–31) | 24 (23.25–26) | 0.1 | ||
| P-NRS | ERT | 2.1 (1.1–4.1) | 1.3 (1.2–1.4) | 0.01 | 0.001 |
| CPT | 1.7 (1.6–1.9) | 1.5 (1.4–1.6) | 0.3 | ||
| MI-UE | ERT | 69 (64.25–76) * | 87 (82.25–94) | 0.001 | 0.002 |
| CPT | 84 (78–91.5) | 82 (78.25–90.5) | 0.4 | ||
| FIM | ERT | 103 (100.25–106.5) * | 109 (104.25–112.5) | 0.0001 | 0.4 |
| CPT | 86 (82.25–90) | 122 (118.25–124) | 0.0001 | ||
| FMA-UE | ERT | 48 (45.25–51) | 52 (50.25–54) | 0.001 | 0.008 |
| CPT | 56 (50–57.5) | 57 (52.26–59) | 0.08 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Raciti, L.; Pignolo, L.; Perini, V.; Pullia, M.; Porcari, B.; Latella, D.; Isgrò, M.; Naro, A.; Calabrò, R.S. Improving Upper Extremity Bradykinesia in Parkinson’s Disease: A Randomized Clinical Trial on the Use of Gravity-Supporting Exoskeletons. J. Clin. Med. 2022, 11, 2543. https://doi.org/10.3390/jcm11092543
Raciti L, Pignolo L, Perini V, Pullia M, Porcari B, Latella D, Isgrò M, Naro A, Calabrò RS. Improving Upper Extremity Bradykinesia in Parkinson’s Disease: A Randomized Clinical Trial on the Use of Gravity-Supporting Exoskeletons. Journal of Clinical Medicine. 2022; 11(9):2543. https://doi.org/10.3390/jcm11092543
Chicago/Turabian StyleRaciti, Loredana, Loris Pignolo, Valentina Perini, Massimo Pullia, Bruno Porcari, Desiree Latella, Marco Isgrò, Antonino Naro, and Rocco Salvatore Calabrò. 2022. "Improving Upper Extremity Bradykinesia in Parkinson’s Disease: A Randomized Clinical Trial on the Use of Gravity-Supporting Exoskeletons" Journal of Clinical Medicine 11, no. 9: 2543. https://doi.org/10.3390/jcm11092543
APA StyleRaciti, L., Pignolo, L., Perini, V., Pullia, M., Porcari, B., Latella, D., Isgrò, M., Naro, A., & Calabrò, R. S. (2022). Improving Upper Extremity Bradykinesia in Parkinson’s Disease: A Randomized Clinical Trial on the Use of Gravity-Supporting Exoskeletons. Journal of Clinical Medicine, 11(9), 2543. https://doi.org/10.3390/jcm11092543







