Epigenetic Risks of Medically Assisted Reproduction
Abstract
1. Introduction
2. Epigenetics in Development and Imprinted Genes
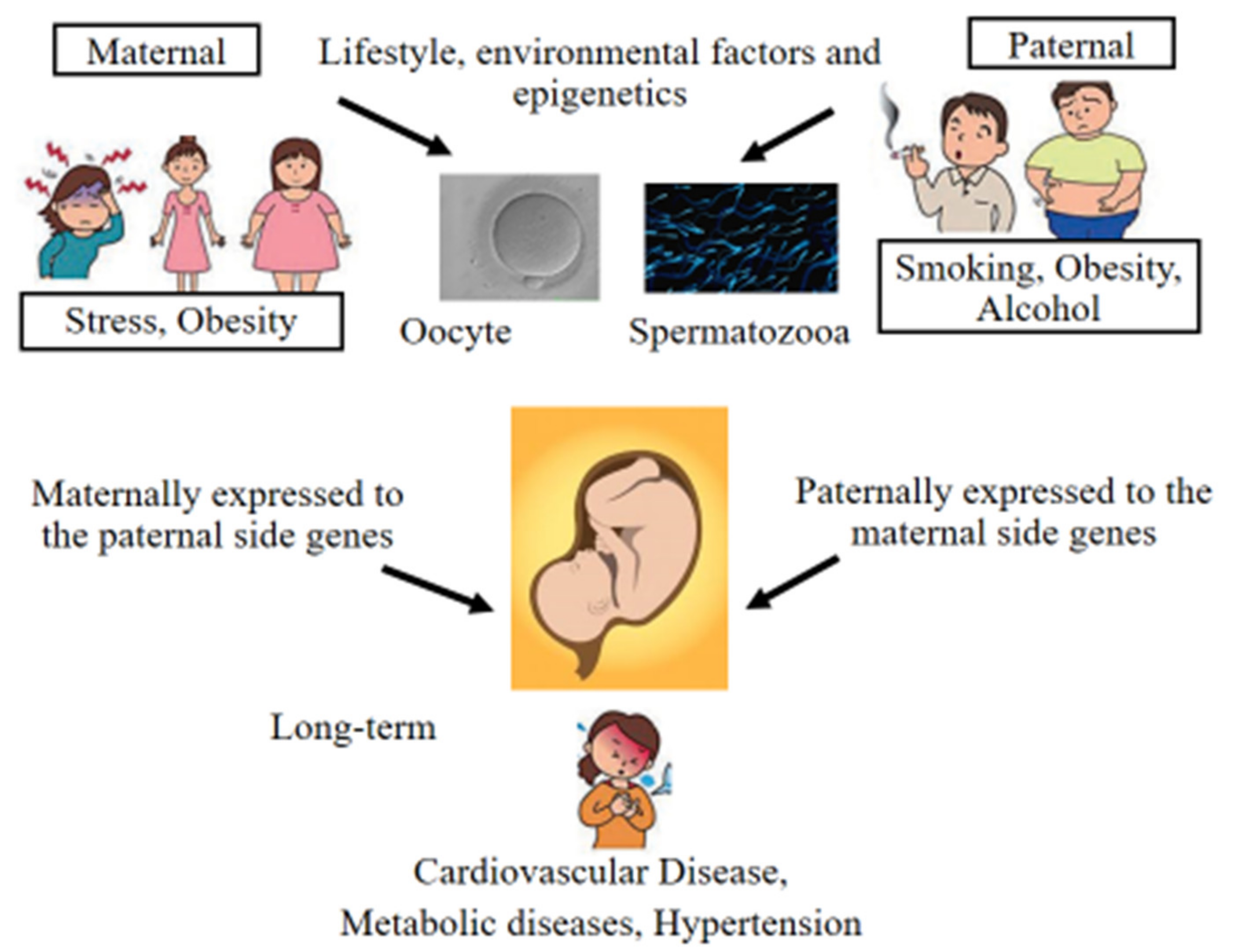
3. Epigenetic Alterations and Imprinting Disorders in ART
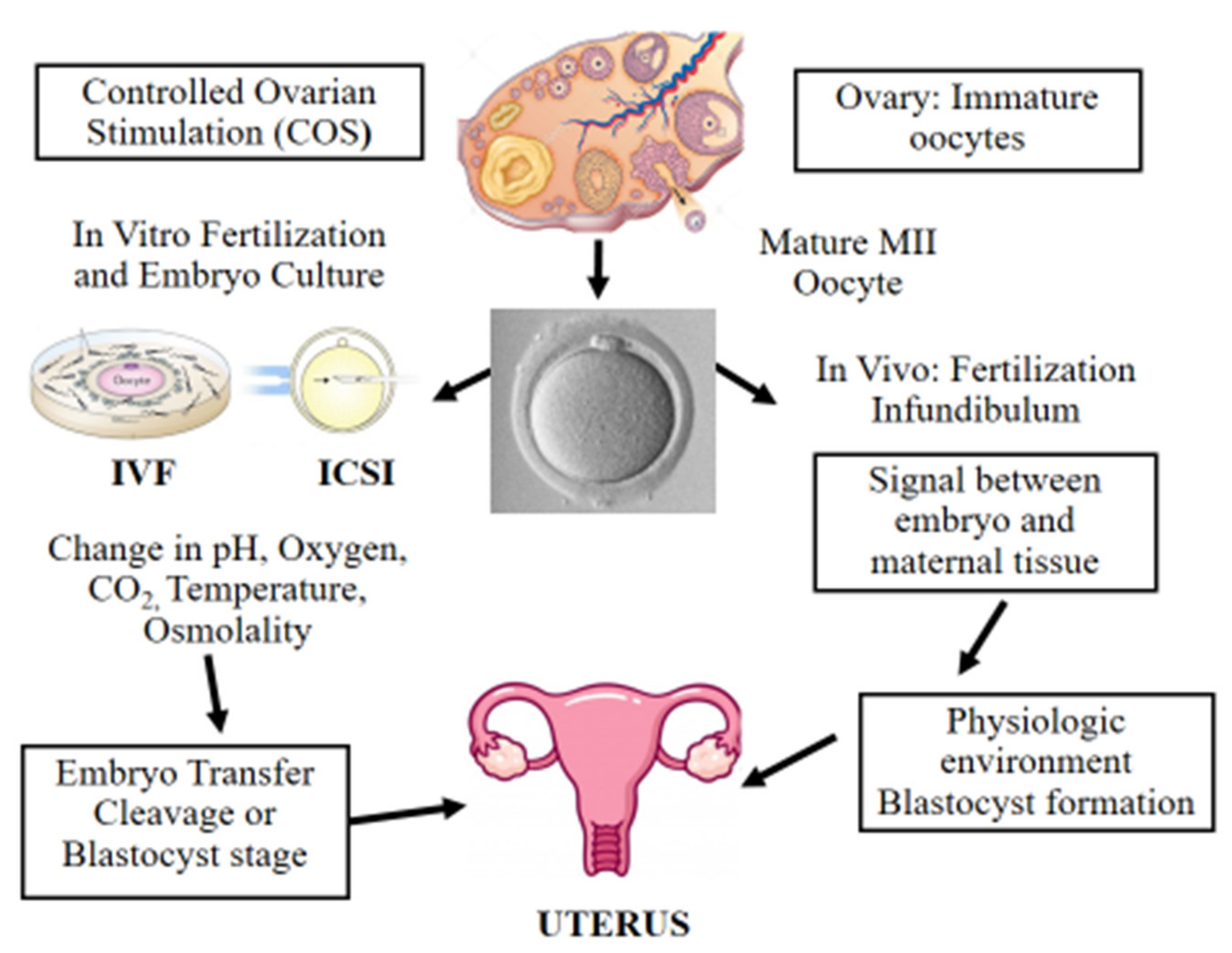
4. Controlled Ovarian Stimulation in ART
5. Fertilization Procedures: In Vitro Fertilization (IVF) and Intracytoplasmic Sperm Injection (ICSI)
6. Epigenetic Alterations Following In Vitro Culture
7. Oxygen Tension
8. In Vitro Culture and Human Birthweight
9. Cardiometabolic Complications in ART-Conceived Children
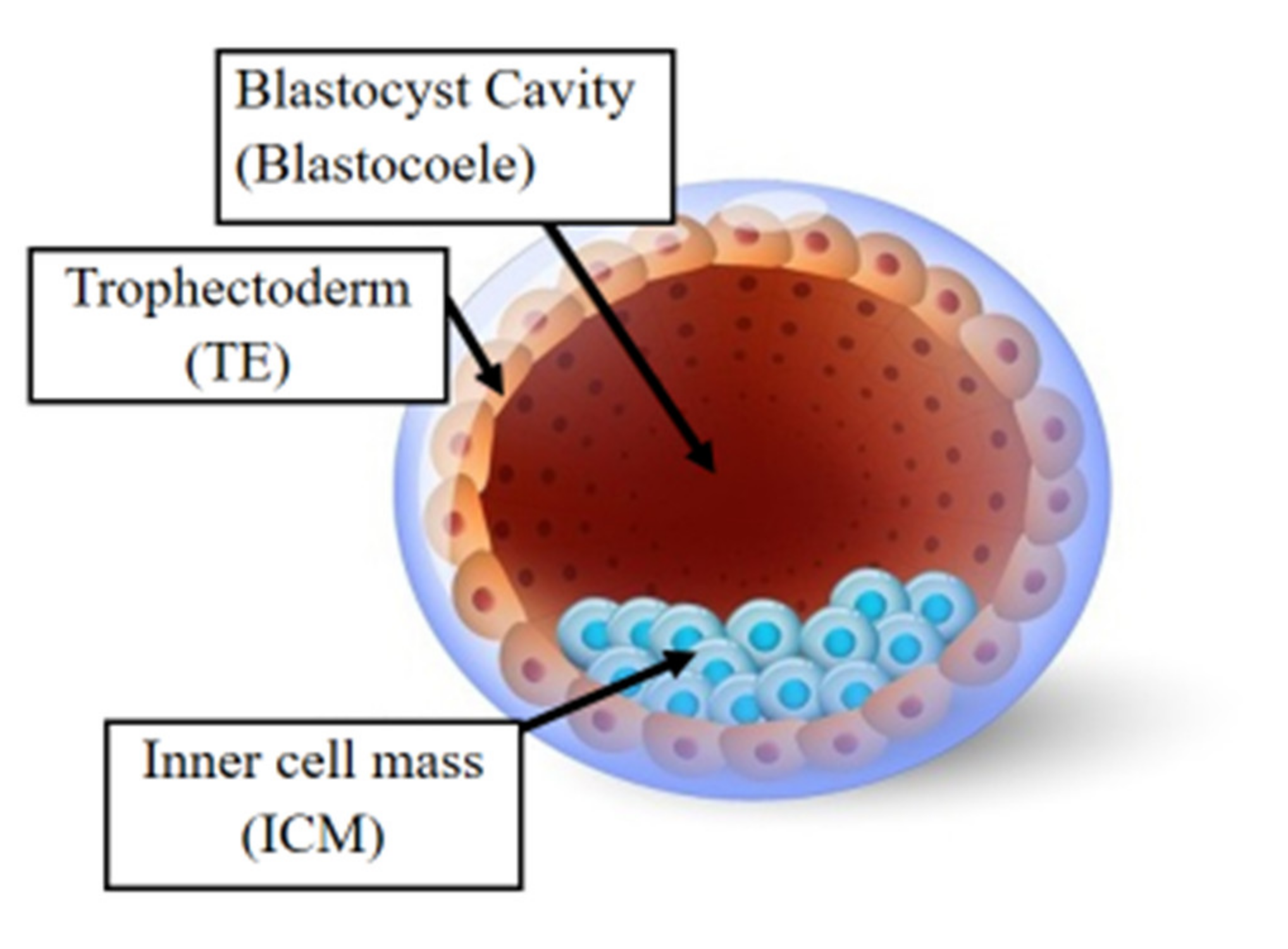
10. Epigenetic Alterations and Preimplantation Genetic Testing Following ART
11. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- De Geyter, C.; Wyns, C.; Calhaz-Jorge, C.; De Mouzon, J.; Ferraretti, A.P.; Kupka, M.; Andersen, A.N.; Nygren, K.G.; Goossens, V. 20 years of the European IVF-monitoring Consortium registry: What have we learned? A comparison with registries from two other regions. Hum. Reprod. 2020, 35, 2832–2849. [Google Scholar] [CrossRef]
- Steptoe, P.; Edwards, R. Birth after the reimplantation of a human embryo. Lancet 1978, 312, 366. [Google Scholar] [CrossRef]
- Thoma, M.E.; McLain, A.; Louis, J.F.; King, R.B.; Trumble, A.C.; Sundaram, R.; Louis, G.B. Prevalence of infertility in the United States as estimated by the current duration approach and a traditional constructed approach. Fertil. Steril. 2013, 99, 1324–1331. [Google Scholar] [CrossRef] [PubMed]
- Ventura-Juncá, P.; Irarrázaval, I.; Rolle, A.J.; Gutiérrez, J.I.; Moreno, R.D.; Santos, M.J. In vitro fertilization (IVF) in mammals: Epigenetic and developmental alterations. Scientific and bioethical implications for IVF in humans. Biol. Res. 2015, 48, 68. [Google Scholar] [CrossRef]
- Young, L.E.; Sinclair, K.D.; Wilmut, I. Large offspring syndrome in cattle and sheep. Rev. Reprod. 1998, 3, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Young, L.E.; Fernandes, K.; McEvoy, T.G.; Butterwith, S.C.; Gutierrez, C.G.; Carolan, C.; Broadbent, P.J.; Robinson, J.J.; Wilmut, I.; Sinclair, K. Epigenetic change in IGF2R is associated with fetal overgrowth after sheep embryo culture. Nat. Genet. 2001, 27, 153–154. [Google Scholar] [CrossRef] [PubMed]
- Doherty, A.S.; Mann, M.R.; Tremblay, K.D.; Bartolomei, M.S.; Schultz, R.M. Differential Effects of Culture on Imprinted H19 Expression in the Preimplantation Mouse Embryo1. Biol. Reprod. 2000, 62, 1526–1535. [Google Scholar] [CrossRef]
- Barberet, J.; Binquet, C.; Guilleman, M.; Doukani, A.; Choux, C.; Bruno, C.; Bourredjem, A.; Chapusot, C.; Bourc’his, D.; Duffourd, Y.; et al. Do assisted reproductive technologies and in vitro embryo culture influence the epigenetic control of imprinted genes and transposable elements in children? Hum. Reprod. 2021, 36, 479–492. [Google Scholar] [CrossRef]
- Schieve, L.A.; Meikle, S.F.; Ferre, C.; Peterson, H.B.; Jeng, G.; Wilcox, L.S. Low and Very Low Birth Weight in Infants Conceived with Use of Assisted Reproductive Technology. N. Engl. J. Med. 2002, 346, 731–737. [Google Scholar] [CrossRef]
- Sunkara, S.K.; Antonisamy, B.; Redla, A.C.; Kamath, M.S. Female causes of infertility are associated with higher risk of preterm birth and low birth weight: Analysis of 117 401 singleton live births following IVF. Hum. Reprod. 2020, 36, 676–682. [Google Scholar] [CrossRef]
- Terho, A.M.; Pelkonen, S.; Opdahl, S.; Romundstad, L.B.; Bergh, C.; Wennerholm, U.B.; Henningsen, A.A.; Pinborg, A.; Gissler, M.; Tiitinen, A. High birth weight and large-for-gestational-age in singletons born after frozen compared to fresh embryo transfer, by gestational week: A Nordic register study from the CoNARTaS group. Hum. Reprod. 2021, 36, 1083–1092. [Google Scholar] [CrossRef] [PubMed]
- Cox, G.F.; Bürger, J.; Lip, V.; Mau, U.A.; Sperling, K.; Wu, B.-L.; Horsthemke, B. Intracytoplasmic Sperm Injection May Increase the Risk of Imprinting Defects. Am. J. Hum. Genet. 2002, 71, 162–164. [Google Scholar] [CrossRef] [PubMed]
- DeBaun, M.R.; Niemitz, E.L.; Feinberg, A.P. Association of In Vitro Fertilization with Beckwith-Wiedemann Syndrome and Epigenetic Alterations of LIT1 and H19. Am. J. Hum. Genet. 2003, 72, 156–160. [Google Scholar] [CrossRef] [PubMed]
- Waddington, C.H. The epigenotype. Int. J. Epidemiol. 2012, 411, 10–13. [Google Scholar] [CrossRef] [PubMed]
- Russo, V.E.A.; Martienssen, R.A.; Riggs, A.D. Epigenetic Mechanisms of Gene Regulation; Cold Spring Harbor Laboratory Press: Plainview, NY, USA, 1996. [Google Scholar]
- Skinner, M.K. Environmental epigenomics and disease susceptibility. EMBO Rep. 2011, 12, 620–622. [Google Scholar] [CrossRef]
- Santos, F.; Hyslop, L.; Stojkovic, P.; Leary, C.; Murdoch, A.; Reik, W.; Stojkovic, M.; Herbert, M.; Dean, W. Evaluation of epigenetic marks in human embryos derived from IVF and ICSI. Hum. Reprod. 2010, 25, 2387–2395. [Google Scholar] [CrossRef]
- Klose, R.J.; Bird, A.P. Genomic DNA methylation: The mark and its mediators. Trends Biochem. Sci. 2006, 31, 89–97. [Google Scholar] [CrossRef]
- Bannister, A.J.; Kouzarides, T. Regulation of chromatin by histone modifications. Cell Res. 2011, 21, 381–395. [Google Scholar] [CrossRef]
- Rivera, C.M.; Ren, B. Mapping Human Epigenomes. Cell 2013, 155, 39–55. [Google Scholar] [CrossRef]
- Hirst, M.; Marra, M.A. Epigenetics and human disease. Int. J. Biochem. Cell Biol. 2009, 41, 136–146. [Google Scholar] [CrossRef]
- Weber, W. Cancer epigenetics. Prog. Mol. Biol. Transl. Sci. 2010, 95, 299–349. [Google Scholar]
- Skaar, D.A.; Li, Y.; Bernal, A.J.; Hoyo, C.; Murphy, S.; Jirtle, R.L. The Human Imprintome: Regulatory Mechanisms, Methods of Ascertainment, and Roles in Disease Susceptibility. ILAR J. 2012, 53, 341–358. [Google Scholar] [CrossRef]
- Allegrucci, C.; Thurston, A.; Lucas, E.; Young, L. Epigenetics and the germline. Reproduction 2005, 129, 137–149. [Google Scholar] [CrossRef]
- Glaser, R.L.; Ramsay, J.P.; Morison, I.M. The imprinted gene and parent-of-origin effect database now includes parental origin of de novo mutations. Nucleic Acids Res. 2006, 34, D29–D31. [Google Scholar] [CrossRef] [PubMed]
- Tremblay, K.D.; Duran, K.L.; Bartolomei, M.S. A 5’ 2-kilobase-pair region of the imprinted mouse H19 gene exhibits exclusive paternal methylation throughout development. Mol. Cell. Biol. 1997, 17, 4322–4329. [Google Scholar] [CrossRef] [PubMed]
- Thorvaldsen, J.L.; Duran, K.L.; Bartolomei, M.S. Deletion of the H19 differentially methylated domain results in loss of im-printed expression of H19 and Igf2. Genes Dev. 1998, 12, 3693–3702. [Google Scholar] [CrossRef] [PubMed]
- Koerner, M.V.; Pauler, F.M.; Huang, R.; Barlow, D.P. The function of non-coding RNAs in genomic imprinting. Development 2009, 136, 1771–1783. [Google Scholar] [CrossRef] [PubMed]
- Marcho, C.; Cui, W.; Mager, J. Epigenetic dynamics during preimplantation development. Reproduction 2015, 150, R109–R120. [Google Scholar] [CrossRef]
- Tunster, S.J.; Jensen, A.B.; John, R.M. Imprinted genes in mouse placental development and the regulation of fetal energy stores. Reproduction 2013, 145, R117–R137. [Google Scholar] [CrossRef]
- Eggermann, T.; de Nanclares, G.P.; Maher, E.R.; Temple, I.K.; Tümer, Z.; Monk, D.; Mackay, D.J.; Grønskov, K.; Riccio, A.; Linglart, A.; et al. Imprinting disorders: A group of congenital disorders with overlapping patterns of molecular changes affecting imprinted loci. Clin. Epigenet. 2015, 7, 123. [Google Scholar] [CrossRef]
- White, C.R.; Denomme, M.M.; Tekpetey, F.R.; Feyles, V.; Power, S.G.A.; Mann, M.R.W. High Frequency of Imprinted Methylation Errors in Human Preimplantation Embryos. Sci. Rep. 2015, 5, 17311. [Google Scholar] [CrossRef] [PubMed]
- Huntriss, J.D.; Hemmings, K.E.; Hinkins, M.; Rutherford, A.J.; Sturmey, R.G.; Elder, K.; Picton, H.M. Variable imprinting of the MEST gene in human preimplantation embryos. Eur. J. Hum. Genet. 2012, 21, 40–47. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Robbins, K.M.; Wells, K.D.; Rivera, R.M. Large offspring syndrome: A bovine model for the human loss-of-imprinting overgrowth syndrome Beckwith-Wiedemann. Epigenetics 2013, 8, 591–601. [Google Scholar] [CrossRef] [PubMed]
- Hiura, H.; Okae, H.; Chiba, H.; Miyauchi, N.; Sato, F.; Sato, A.; Arima, T. Imprinting methylation errors in ART. Reprod. Med. Biol. 2014, 13, 193–202. [Google Scholar] [CrossRef]
- Lazaraviciute, G.; Kauser, M.; Bhattacharya, S.; Haggarty, P.; Bhattacharya, S. A systematic review and meta-analysis of DNA methylation levels and imprinting disorders in children conceived by IVF/ICSI compared with children conceived spontane-ously. Hum. Reprod. Update 2014, 20, 840–852. [Google Scholar] [CrossRef]
- Vermeiden, J.P.; Bernardus, R.E. Are imprinting disorders more prevalent after human in vitro fertilization or intracytoplasmic sperm injection? Fertil. Steril. 2013, 99, 642–651. [Google Scholar] [CrossRef]
- Henningsen, A.A.; Gissler, M.; Rasmussen, S.; Opdahl, S.; Wennerholm, U.B.; Spangmose, A.L.; Tiitinen, A.; Bergh, C.; Romundstad, L.B.; Laivuori, H.; et al. Imprinting disorders in children born after ART: A Nordic study from the CoNARTaS group. Hum. Reprod. 2020, 35, 1178–1184. [Google Scholar] [CrossRef]
- Hattori, H.; Hiura, H.; Kitamura, A.; Miyauchi, N.; Kobayashi, N.; Takahashi, S.; Okae, H.; Kyono, K.; Kagami, M.; Ogata, T.; et al. Association of four imprinting disorders and ART. Clin. Epigenet. 2019, 11, 21. [Google Scholar] [CrossRef]
- Chen, Z.; Hagen, D.E.; Elsik, C.G.; Ji, T.; Morris, C.J.; Moon, L.E.; Rivera, R.M. Characterization of global loss of imprinting in fetal overgrowth syndrome induced by assisted reproduction. Proc. Natl. Acad. Sci. USA 2015, 112, 4618–4623. [Google Scholar] [CrossRef]
- Van der Auwera, I.; D’Hooghe, T. Superovulation of female mice delays embryonic and fetal development. Hum. Reprod. 2001, 16, 1237–1243. [Google Scholar] [CrossRef]
- Market-Velker, B.A.; Zhang, L.; Magri, L.S.; Bonvissuto, A.C.; Mann, M.R. Dual effects of superovulation: Loss of maternal and paternal imprinted methylation in a dose-dependent manner. Hum. Mol. Genet. 2009, 19, 36–51. [Google Scholar] [CrossRef] [PubMed]
- Chang, A.S.; Moley, K.H.; Wangler, M.; Feinberg, A.; DeBaun, M.R. Association between Beckwith-Wiedemann syndrome and assisted reproductive technology: A case series of 19 patients. Fertil. Steril. 2005, 83, 349–354. [Google Scholar] [CrossRef] [PubMed]
- Voronina, E.; Wessel, G.M. The Regulation of Oocyte Maturation. Curr. Top. Dev. Biol. 2003, 58, 53–110. [Google Scholar] [PubMed]
- Telfer, E. Progress and prospects for developing human immature oocytes in vitro. Reproduction 2019, 158, F45–F54. [Google Scholar] [CrossRef]
- Mak, W.; Weaver, J.R.; Bartolomei, M.S. Is ART changing the epigenetic landscape of imprinting? Anim. Reprod. 2010, 7, 168–176. [Google Scholar]
- Sato, A.; Otsu, E.; Negishi, H.; Utsunomiya, T.; Arima, T. Aberrant DNA methylation of imprinted loci in superovulated oo-cytes. Hum. Reprod. 2007, 22, 26–35. [Google Scholar] [CrossRef]
- Laprise, S.L. Implications of epigenetics and genomic imprinting in assisted reproductive technologies. Mol. Reprod. Dev. 2009, 76, 1006–1018. [Google Scholar] [CrossRef]
- Fauque, P.; Jouannet, P.; Lesaffre, C.; Ripoche, M.-A.; Dandolo, L.; Vaiman, D.; Jammes, H. Assisted Reproductive Technology affects developmental kinetics, H19 Imprinting Control Region methylation and H19 gene expression in individual mouse embryos. BMC Dev. Biol. 2007, 7, 116. [Google Scholar] [CrossRef]
- Huo, Y.; Yan, Z.Q.; Yuan, P.; Qin, M.; Kuo, Y.; Li, R.; Yan, L.Y.; Feng, H.L.; Qiao, J. Single-cell DNA methylation sequencing reveals epigenetic alterations in mouse oocytes superovulated with different dosages of gonadotropins. Clin. Epigenetics 2020, 12, 75. [Google Scholar] [CrossRef]
- Saenz-De-Juano, M.D.; Ivanova, E.; Billooye, K.; Herța, A.-C.; Smitz, J.; Kelsey, G.; Anckaert, E. Correction to: Genome-wide assessment of DNA methylation in mouse oocytes reveals effects associated with in vitro growth, superovulation, and sexual maturity. Clin. Epigenet. 2020, 12, 18. [Google Scholar] [CrossRef]
- Yu, B.; Smith, T.H.; Battle, S.L.; Ferrell, S.; Hawkins, R.D. Superovulation alters global DNA methylation in early mouse embryo development. Epigenetics 2019, 14, 780–790. [Google Scholar] [CrossRef] [PubMed]
- Kalthur, G.; Salian, S.R.; Nair, R.; Mathew, J.; Adiga, S.K.; Kalthur, S.G.; Zeegers, D.; Hande, M.P. Distribution pattern of cytoplasmic organelles, spindle integrity, oxidative stress, octamer-binding transcription factor 4 (Oct4) expression and de-velopmental potential of oocytes following multiple superovulation. Reprod. Fertil. Dev. 2016, 28, 2027–2038. [Google Scholar] [CrossRef] [PubMed]
- Tang, S.-B.; Yang, L.-L.; Zhang, T.-T.; Wang, Q.; Yin, S.; Luo, S.-M.; Shen, W.; Ge, Z.-J.; Sun, Q.-Y. Multiple superovulations alter histone modifications in mouse early embryos. Reproduction 2019, 157, 511–523. [Google Scholar] [CrossRef] [PubMed]
- Uysal, F.; Ozturk, S.; Akkoyunlu, G. Superovulation alters DNA methyltransferase protein expression in mouse oocytes and early em-bryos. J. Assist. Reprod. Genet. 2018, 35, 503–513. [Google Scholar] [CrossRef]
- Kindsfather, A.J.; Czekalski, M.A.; Pressimone, C.A.; Erisman, M.P.; Mann, M.R. Perturbations in imprinted methylation from assisted reproductive technologies but not advanced maternal age in mouse preimplantation embryos. Clin. Epigenet. 2019, 11, 162. [Google Scholar] [CrossRef]
- Chen, X.; Huang, Y.; Huang, H.; Guan, Y.; Li, M.; Jiang, X.; Yu, M.; Yang, X. Effects of superovulation, in vitro fertilization, and oocyte in vitro maturation on imprinted gene Grb10 in mouse blastocysts. Arch. Gynecol. Obstet. 2018, 298, 1219–1227. [Google Scholar] [CrossRef]
- Palermo, G.; Joris, H.; Devroey, P.; Van Steirteghem, A.C. Pregnancies after intracytoplasmic injection of single spermatozoon into an oocyte. Lancet 1992, 340, 17–18. [Google Scholar] [CrossRef]
- Hewitson, L.; Simerly, C.; Dominko, T.; Schatten, G. Cellular and molecular events after in vitro fertilization and intracyto-plasmic sperm injection. Theriogenology 2000, 53, 95–104. [Google Scholar] [CrossRef]
- Sullivan, E.A.; Zegers-Hochschild, F.; Mansour, R.; Ishihara, O.; De Mouzon, J.; Nygren, K.G.; Adamson, G.D. International Committee for Monitoring Assisted Reproductive Technologies (ICMART) world report: Assisted reproductive technology 2004. Hum. Reprod. 2013, 28, 1375–1390. [Google Scholar] [CrossRef]
- Minor, A.; Chow, V.; Ma, S. Aberrant DNA methylation at imprinted genes in testicular sperm retrieved from men with ob-structive azoospermia and undergoing vasectomy reversal. Reproduction 2011, 141, 749–757. [Google Scholar] [CrossRef]
- Kobayashi, H.; Sato, A.; Otsu, E.; Hiura, H.; Tomatsu, C.; Utsunomiya, T.; Sasaki, H.; Yaegashi, N.; Arima, T. Aberrant DNA methylation of imprinted loci in sperm from oligospermic patients. Hum. Mol. Genet. 2007, 16, 2542–2551. [Google Scholar] [CrossRef] [PubMed]
- Marques, C.J.; Francisco, T.; Sousa, S.; Carvalho, F.; Barros, A.; Sousa, M. Methylation defects of imprinted genes in human testicular spermatozoa. Fertil. Steril. 2010, 94, 585–594. [Google Scholar] [CrossRef] [PubMed]
- Whitelaw, N.; Bhattacharya, S.; Hoad, G.; Horgan, G.W.; Hamilton, M.; Haggarty, P. Epigenetic status in the offspring of spontaneous and assisted conception. Hum. Reprod. 2014, 29, 1452–1458. [Google Scholar] [CrossRef] [PubMed]
- Rancourt, R.; Harris, H.; Michels, K. Methylation levels at imprinting control regions are not altered with ovulation induction or in vitro fertilization in a birth cohort. Hum. Reprod. 2012, 27, 2208–2216. [Google Scholar] [CrossRef][Green Version]
- Xu, N.; Barlow, G.M.; Cui, J.; Wang, E.T.; Lee, B.; Akhlaghpour, M.; Kroener, L.; Williams, J.; Rotter, J.I.; Chen, Y.-D.I.; et al. Comparison of Genome-Wide and Gene-Specific DNA Methylation Profiling in First-Trimester Chorionic Villi From Pregnancies Conceived With Infertility Treatments. Reprod. Sci. 2016, 24, 996–1004. [Google Scholar] [CrossRef]
- Choufani, S.; Turinsky, A.L.; Melamed, N.; Greenblatt, E.; Brudno, M.; Bérard, A.; Fraser, W.D.; Weksberg, R.; Trasler, J.; Monnier, P.; et al. Impact of assisted reproduction, infertility, sex and paternal factors on the placental DNA methylome. Hum. Mol. Genet. 2018, 28, 372–385. [Google Scholar] [CrossRef]
- Tierling, S.; Souren, N.Y.; Gries, J.; LoPorto, C.; Groth, M.; Lutsik, P.; Neitzel, H.; Utz-Billing, I.; Gillessen-Kaesbach, G.; Kentenich, H.; et al. Assisted reproductive technologies do not enhance the variability of DNA methylation imprints in human. J. Med. Genet. 2009, 47, 371–376. [Google Scholar] [CrossRef]
- El Hajj, N.; Haertle, L.; Dittrich, M.; Denk, S.; Lehnen, H.; Hahn, T.; Schorsch, M.; Haaf, T. DNA methylation signatures in cord blood of ICSI children. Hum. Reprod. 2017, 32, 1761–1769. [Google Scholar] [CrossRef]
- Choux, C.; Binquet, C.; Carmignac, V.; Bruno, C.; Chapusot, C.; Barberet, J.; LaMotte, M.; Sagot, P.; Bourc’His, D.; Fauque, P. The epigenetic control of transposable elements and imprinted genes in newborns is affected by the mode of conception: ART versus spontaneous conception without underlying infertility. Hum. Reprod. 2017, 33, 331–340. [Google Scholar] [CrossRef]
- Rivera, R.M.; Stein, P.; Weaver, J.R.; Mager, J.; Schultz, R.M.; Bartolomei, M.S. Manipulations of mouse embryos prior to implantation result in aberrant expression of imprinted genes on day 9.5 of development. Hum. Mol. Genet. 2007, 17, 1–14. [Google Scholar] [CrossRef]
- Nelissen, E.C.; Dumoulin, J.C.; Daunay, A.; Evers, J.L.; Tost, J.; van Montfoort, A.P. Placentas from pregnancies conceived by IVF/ICSI have a reduced DNA methylation level at the H19 and MEST differentially methylated regions. Hum. Reprod. 2013, 28, 1117–1126. [Google Scholar] [CrossRef] [PubMed]
- Fortier, A.L.; Lopes, F.L.; Darricarrère, N.; Martel, J.; Trasler, J.M. Superovulation alters the expression of imprinted genes in the midgestation mouse placenta. Hum. Mol. Genet. 2008, 17, 1653–1665. [Google Scholar] [CrossRef] [PubMed]
- De Waal, E.; Mak, W.; Calhoun, S.; Stein, P.; Ord, T.; Krapp, C.; Coutifaris, C.; Schultz, R.M.; Bartolomei, M. In Vitro Culture Increases the Frequency of Stochastic Epigenetic Errors at Imprinted Genes in Placental Tissues from Mouse Concepti Produced Through Assisted Reproductive Technologies1. Biol. Reprod. 2014, 90, 22. [Google Scholar] [CrossRef]
- Yang, H.; Ma, Z.; Peng, L.; Kuhn, C.; Rahmeh, M.; Mahner, S.; Jeschke, U.; von Schönfeldt, V. Comparison of Histone H3K4me3 between IVF and ICSI Technologies and between Boy and Girl Offspring. Int. J. Mol. Sci. 2021, 22, 8574. [Google Scholar] [CrossRef]
- Choux, C.; Petazzi, P.; Sánchez, A.M.; Mora, J.R.H.; Monteagudo, A.; Sagot, P.; Monk, D.; Fauque, P. The hypomethylation of imprinted genes in IVF/ICSI placenta samples is associated with concomitant changes in histone modifications. Epigenetics 2020, 15, 1386–1395. [Google Scholar] [CrossRef]
- Potabattula, R.; Zacchini, F.; Ptak, G.E.; Dittrich, M.; Müller, T.; El Hajj, N.; Hahn, T.; Drummer, C.; Behr, R.; Lucas-Hahn, A.; et al. Increasing methylation of sperm rDNA and other repetitive elements in the aging male mammalian germline. Aging Cell 2020, 19, e13181. [Google Scholar] [CrossRef]
- Potabattula, R.; Dittrich, M.; Böck, J.; Haertle, L.; Müller, T.; Hahn, T.; Schorsch, M.; Hajj, N.E.; Haaf, T. Allele-specific methylation of imprinted genes in fetal cord blood is influenced by cis-acting genetic variants and parental factors. Epigenomics 2018, 10, 1315–1326. [Google Scholar] [CrossRef]
- Mitchell, M.; Strick, R.; Strissel, P.L.; Dittrich, R.; McPherson, N.O.; Lane, M.; Pliushch, G.; Potabattula, R.; Haaf, T.; El Hajj, N. Gene expression and epigenetic aberrations in F1-placentas fathered by obese males. Mol Reprod Dev. 2017, 84, 316–328. [Google Scholar] [CrossRef]
- Potabattula, R.; Dittrich, M.; Schorsch, M.; Hahn, T.; Haaf, T.; El Hajj, N. Male obesity effects on sperm and next-generation cord blood DNA methylation. PLoS ONE 2019, 14, e0218615. [Google Scholar] [CrossRef]
- Bernhardt, L.; Dittrich, M.; El-Merahbi, R.; Saliba, A.E.; Müller, T.; Sumara, G.; Vogel, J.; Nichols-Burns, S.; Mitchell, M.; Haaf, T.; et al. A genome-wide transcriptomic analysis of embryos fathered by obese males in a murine model of diet-induced obesity. Sci. Rep. 2021, 11, 1979. [Google Scholar] [CrossRef]
- Atsem, S.; Reichenbach, J.; Potabattula, R.; Dittrich, M.; Nava, C.; Depienne, C.; Böhm, L.; Rost, S.; Hahn, T.; Schorsch, M.; et al. Paternal age effects on sperm FOXK1 and KCNA7 methylation and transmission into the next generation. Hum. Mol. Genet. 2016, 25, 4996–5005. [Google Scholar] [CrossRef] [PubMed]
- Vieweg, M.; Dvorakova-Hortova, K.; Dudkova, B.; Waliszewski, P.; Otte, M.; Oels, B.; Hajimohammad, A.; Turley, H.; Schorsch, M.; Schuppe, H.-C.; et al. Methylation analysis of histone H4K12ac-associated promoters in sperm of healthy donors and subfertile patients. Clin. Epigenet. 2015, 7, 31. [Google Scholar] [CrossRef] [PubMed]
- El Hajj, N.; Zechner, U.; Schneider, E.; Tresch, A.; Gromoll, J.; Hahn, T.; Schorsch, M.; Haaf, T. Methylation status of imprinted genes and repetitive elements in sperm DNA from infertile males. Sex. Dev. 2011, 5, 60–69. [Google Scholar] [CrossRef]
- Schon, S.B.; Luense, L.J.; Wang, X.; Bartolomei, M.S.; Coutifaris, C.; Garcia, B.A.; Berger, S.J. Histone modification sig-natures in human sperm distinguish clinical abnormalities. J. Assist. Reprod. Genet. 2019, 36, 267–275. [Google Scholar] [CrossRef] [PubMed]
- Dang, V.Q.; Vuong, L.N.; Luu, T.M.; Pham, T.D.; Ho, T.M.; Ha, A.N.; Truong, B.T.; Phan, A.K.; Nguyen, D.P.; Pham, T.N.; et al. Intracytoplasmic sperm injection versus conventional in-vitro fertilisation in couples with infertility in whom the male partner has normal total sperm count and motility: An open-label, randomised controlled trial. Lancet 2021, 397, 1554–1563. [Google Scholar] [CrossRef]
- Practice Committees of American Society for Reproductive Medicine and Society for Assisted Reproduction Technology. Intracytoplasmic sperm injection (ICSI) for non- male factor indications: A committee opinion. Fertil. Steril. 2020, 114, 239–245. [Google Scholar] [CrossRef]
- Huntriss, J.; Picton, H.M. Epigenetic consequences of assisted reproduction and infertility on the human preimplantation embryo. Hum. Fertil. 2008, 11, 85–94. [Google Scholar] [CrossRef]
- Osman, E.; Franasiak, J.; Scott, R. Oocyte and Embryo Manipulation and Epigenetics. Semin. Reprod. Med. 2018, 36, e1–e9. [Google Scholar] [CrossRef]
- Siqueira, L.G.; Silva, M.V.G.; Panetto, J.; Viana, J. Consequences of assisted reproductive technologies for offspring function in cattle. Reprod. Fertil. Dev. 2020, 32, 82–97. [Google Scholar] [CrossRef]
- El Hajj, N.; Haaf, T. Epigenetic disturbances in in vitro cultured gametes and embryos: Implications for human assisted reproduction. Fertil. Steril. 2013, 99, 632–641. [Google Scholar] [CrossRef]
- Market-Velker, B.; Fernandes, A.; Mann, M. Side-by-Side Comparison of Five Commercial Media Systems in a Mouse Model: Suboptimal In Vitro Culture Interferes with Imprint Maintenance1. Biol. Reprod. 2010, 83, 938–950. [Google Scholar] [CrossRef]
- Estill, M.S.; Bolnick, J.M.; Waterland, R.A.; Bolnick, A.D.; Diamond, M.P.; Krawetz, S.A. Assisted reproductive technology alters deoxyribonucleic acid methylation profiles in bloodspots of newborn infants. Fertil. Steril. 2016, 106, 629–639. [Google Scholar] [CrossRef] [PubMed]
- Katari, S.; Turan, N.; Bibikova, M.; Erinle, O.; Chalian, R.; Foster, M.; Gaughan, J.P.; Coutifaris, C.; Sapienza, C. DNA methylation and gene expression differences in children conceived in vitro or in vivo. Hum. Mol. Genet. 2009, 18, 3769–3778. [Google Scholar] [CrossRef] [PubMed]
- Castillo-Fernandez, J.; Loke, Y.J.; Bass-Stringer, S.; Gao, F.; Xia, Y.; Wu, H.; Lu, H.; Liu, Y.; Wang, J.; Spector, T.D.; et al. DNA methylation changes at infertility genes in newborn twins conceived by in vitro fertilisation. Genome Med. 2017, 9, 28. [Google Scholar] [CrossRef] [PubMed]
- Schwarzer, C.; Esteves, T.C.; Araúzo-Bravo, M.J.; Le Gac, S.; Nordhoff, V.; Schlatt, S.; Boiani, M. ART culture conditions change the probability of mouse embryo gestation through defined cellular and molecular responses. Hum. Reprod. 2012, 27, 2627–2640. [Google Scholar] [CrossRef] [PubMed]
- Gad, A.; Schellander, K.; Hoelker, M.; Tesfaye, D. Transcriptome profile of early mammalian embryos in response to culture environment. Anim. Reprod. Sci. 2012, 134, 76–83. [Google Scholar] [CrossRef]
- Kleijkers, S.H.; Eijssen, L.M.; Coonen, E.; Derhaag, J.G.; Mantikou, E.; Jonker, M.J.; Mastenbroek, S.; Repping, S.; Evers, J.L.; Dumoulin, J.C.; et al. Differences in gene expression profiles between human preimplantation embryos cultured in two different IVF culture media. Hum. Reprod. 2015, 30, 2303–2311. [Google Scholar] [CrossRef]
- Mantikou, E.; Jonker, M.J.; Wong, K.M.; van Montfoort, A.P.; De Jong, M.; Breit, T.M.; Repping, S.; Mastenbroek, S. Factors affecting the gene ex-pression of in vitro cultured human preimplantation embryos. Hum. Reprod. 2016, 31, 298–311. [Google Scholar]
- Mulder, C.L.; Wattimury, T.M.; Jongejan, A.; de Winter-Korver, C.M.; van Daalen, S.K.M.; Struijk, R.B.; Borgman, S.C.M.; Wurth, Y.; Consten, D.; van Echten-Arends, J.; et al. Comparison of DNA methylation patterns of parentally imprinted genes in placenta derived from IVF conceptions in two different culture media. Hum. Reprod. 2020, 35, 516–528. [Google Scholar] [CrossRef]
- Fischer, B.; Bavister, B.D. Oxygen tension in the oviduct and uterus of rhesus monkeys, hamsters and rabbits. J. Reprod. Fertil. 1993, 99, 673–679. [Google Scholar] [CrossRef]
- Ng, K.Y.B.; Mingels, R.; Morgan, H.; Macklon, N.; Cheong, Y. In vivo oxygen, temperature and pH dynamics in the female reproductive tract and their importance in human conception: A systematic review. Hum. Reprod. Update 2017, 24, 15–34. [Google Scholar] [CrossRef] [PubMed]
- Gardner, D.K.; Lane, M. Ex vivo early embryo development and effects on gene expression and imprinting. Reprod. Fertil. Dev. 2005, 17, 361–370. [Google Scholar] [CrossRef]
- Sciorio, R.; Smith, G. Embryo culture at a reduced oxygen concentration of 5%: A mini review. Zygote 2019, 27, 355–361. [Google Scholar] [CrossRef]
- Katz-Jaffe, M.G.; Linck, D.W.; Schoolcraft, W.B.; Gardner, D.K. A proteomic analysis of mammalian preimplantation embryonic development. Reproduction 2005, 130, 899–905. [Google Scholar] [CrossRef]
- Rinaudo, P.F.; Giritharan, G.; Talbi, S.; Dobson, A.T.; Schultz, R.M. Effects of oxygen tension on gene expression in pre-implantation mouse embryos. Fertil. Steril. 2006, 86 (Suppl. 4), 1252–1265. [Google Scholar] [CrossRef] [PubMed]
- Meintjes, M.; Chantilis, S.J.; Douglas, J.D.; Rodriguez, A.J.; Guerami, A.R.; Bookout, D.M.; Barnett, B.D.; Madden, J.D. A controlled randomized trial evaluating the effect of lowered incubator oxygen tension on live births in a predominantly blas-tocyst transfer program. Hum. Reprod. 2009, 24, 300–307. [Google Scholar] [CrossRef] [PubMed]
- Gardner, D.K.; Kuramoto, T.; Tanaka, M.; Mitzumoto, S.; Montag, M.; Yoshida, A. Prospec-tive randomized multicentre comparison on sibling oocytes comparing G-Series media system with antioxidants versus standard G-Series media system. Reprod. Biomed. Online 2020, 40, 637–644. [Google Scholar] [CrossRef]
- Bontekoe, S.; Mantikou, E.; van Wely, M.; Seshadri, S.; Repping, S.; Mastenbroek, S. Low oxygen concentrations for embryo culture in assisted reproductive technologies. Cochrane Database Syst. Rev. 2012, CD008950. [Google Scholar] [CrossRef]
- Painter, R.C.; de Rooij, S.R.; Bossuyt, P.M.; Simmers, T.A.; Osmond, C.; Barker, D.J.; Bleker, O.P.; Roseboom, T.J. Early onset of coronary artery disease after prenatal exposure to the Dutch famine. Am. J. Clin. Nutr. 2006, 84, 322–327. [Google Scholar] [CrossRef]
- Dumoulin, J.C.; Land, J.A.; Van Montfoort, A.P.; Nelissen, E.C.; Coonen, E.; Derhaag, J.G.; Schreurs, I.L.; Dunselman, G.A.; Kester, A.D.; Geraedts, J.P.; et al. Effect of in vitro culture of human embryos on birthweight of newborns. Hum. Reprod. 2010, 25, 605–612. [Google Scholar] [CrossRef]
- Nelissen, E.C.; van Montfoort, A.; Coonen, E.; Derhaag, J.G.; Geraedts, J.P.; Smits, L.J.; Land, J.A.; Evers, J.; Dumoulin, J.C. Further evidence that culture media affect perinatal outcome: Findings after transfer of fresh and cryopreserved embryos. Hum. Reprod. 2012, 27, 1966–1976. [Google Scholar] [CrossRef] [PubMed]
- Vergouw, C.G.; Kostelijk, E.H.; Doejaaren, E.; Hompes, P.G.; Lambalk, C.B.; Schats, R. The influence of the type of embryo culture medium on neonatal birthweight after single embryo transfer in IVF. Hum. Reprod. 2012, 27, 2619–2626. [Google Scholar] [CrossRef] [PubMed]
- Roberts, S.A.; Vail, A. On the appropriate interpretation of evidence: The example of culture media and birth weight. Hum. Reprod. 2017, 32, 1151–1154. [Google Scholar] [CrossRef] [PubMed]
- Kleijkers, S.H.; Mantikou, E.; Slappendel, E.; Consten, D.; Van Echten-Arends, J.; Wetzels, A.M.; van Wely, M.; Smits, L.J.; van Montfoort, A.; Repping, S.; et al. Influence of embryo culture medium (G5 and HTF) on pregnancy and perinatal outcome after IVF: A multicenter RCT. Hum. Reprod. 2016, 31, 2219–2230. [Google Scholar] [CrossRef]
- Kleijkers, S.H.; van Montfoort, A.; Smits, L.J.M.; Viechtbauer, W.; Roseboom, T.J.; Nelissen, E.C.; Coonen, E.; Derhaag, J.G.; Bastings, L.; Schreurs, I.E.; et al. IVF culture medium affects post-natal weight in humans during the first 2 years of life. Hum. Reprod. 2014, 29, 661–669. [Google Scholar] [CrossRef]
- Lin, S.; Li, M.; Lian, Y.; Chen, L.; Liu, P. No effect of embryo culture media on birthweight and length of newborns. Hum. Reprod. 2013, 28, 1762–1767. [Google Scholar] [CrossRef]
- De Vos, A.; Janssens, R.; Van De Velde, H.; Haentjens, P.; Bonduelle, M.; Tournaye, H.; Verheyen, G. The type of culture medium and the duration of in vitro culture do not influence birthweight of ART singletons. Hum. Reprod. 2015, 30, 20–27. [Google Scholar] [CrossRef]
- Eskild, A.; Monkerud, L.; Tanbo, T. Birthweight and placental weight; do changes in culture media used for IVF matter? Comparisons with spontaneous pregnancies in the corresponding time periods. Hum. Reprod. 2013, 28, 3207–3214. [Google Scholar] [CrossRef]
- Kleijkers, S.H.; van Montfoort, A.P.; Smits, L.J.M.; Coonen, E.; Derhaag, J.G.; Evers, J.L.; Dumoulin, J.C. Age of G-1 PLUS v5 embryo culture medium is inversely associated with birthweight of the newborn. Hum. Reprod. 2015, 30, 1352–1357. [Google Scholar] [CrossRef]
- Zhu, J.; Li, M.; Chen, L.; Liu, P.; Qiao, J. The protein source in embryo culture media influences birthweight: A comparative study between G1 v5 and G1-PLUS v5. Hum. Reprod. 2014, 29, 1387–1392. [Google Scholar] [CrossRef]
- Zhu, J.; Lin, S.; Li, M.; Chen, L.; Lian, Y.; Liu, P.; Qiao, J. Effect of in vitro culture period on birthweight of singleton newborns. Hum. Reprod. 2014, 29, 448–454. [Google Scholar] [CrossRef] [PubMed]
- Zandstra, H.; Van Montfoort, A.P.; Dumoulin, J.C. Does the type of culture medium used influence birthweight of children born after IVF? Hum. Reprod. 2015, 30, 530–542. [Google Scholar] [CrossRef] [PubMed]
- Litzky, J.F.; Boulet, S.; Esfandiari, N.; Zhang, Y.; Kissin, D.M.; Theiler, R.; Marsit, C.J. Effect of frozen/thawed embryo transfer on birthweight, macrosomia, and low birthweight rates in US singleton infants. Am. J. Obstet. Gynecol. 2017, 218, 433.e1–433.e10. [Google Scholar] [CrossRef] [PubMed]
- Cetin, I.; Cozzi, V.; Antonazzo, P. Fetal development after assisted reproduction–a review. Placenta 2003, 24, S104–S113. [Google Scholar] [CrossRef]
- Källén, B.; Finnström, O.; Lindam, A.; Nilsson, E.; Nygren, K.G.; Otterblad, P.O. Congenital malformations in infants born after in vitro fertilization in Sweden. Birth Defects Res. A Clin. Mol. Teratol. 2010, 88, 137–143. [Google Scholar]
- Hart, R.; Norman, R.J. The longer-term health outcomes for children born as a result of IVF treatment: Part I–General health outcomes. Hum. Reprod. Update 2013, 19, 232–243. [Google Scholar] [CrossRef]
- Scherrer, U.; Rimoldi, S.F.; Rexhaj, E.; Stuber, T.; Duplain, H.; Garcin, S.; de Marchi, S.F.; Nicod, P.; Germond, M.; Allemann, Y.; et al. Systemic and pulmonary vascular dys-function in children conceived by assisted reproductive technologies. Circulation 2012, 125, 1890–1896. [Google Scholar] [CrossRef]
- Von Arx, R.; Allemann, Y.; Sartori, C.; Rexhaj, E.; Cerny, D.; De Marchi, S.F.; Soria, R.; Germond, M.; Scherrer, U.; Rimoldi, S.F. Right ventricular dysfunction in children and adolescents conceived by assisted reproductive technologies. J. Appl. Physiol. 2015, 118, 1200–1206. [Google Scholar] [CrossRef]
- Valenzuela-Alcaraz, B.; Cruz-Lemini, M.; Rodriguez-Lopez, M.; Goncé, A.; García-Otero, L.; Ayuso, H.; Sitges, M.; Bijnens, B.; Balasch, J.; Gratacós, E.; et al. Fetal cardiac remodeling in twin pregnancy conceived by assisted reproductive technology. Ultrasound Obstet. Gynecol. 2017, 51, 94–100. [Google Scholar] [CrossRef]
- Sakka, S.D.; Loutradis, D.; Kanaka-Gantenbein, C.; Margeli, A.; Papastamataki, M.; Papassotiriou, I.; Chrousos, G.P. Absence of insulin resistance and low-grade inflammation despite early metabolic syndrome manifestations in children born after in vitro fertilization. Fertil. Steril. 2010, 94, 1693–1699. [Google Scholar] [CrossRef]
- Ceelen, M.; van Weissenbruch, M.M.; Roos, J.C.; Vermeiden, J.P.; van Leeuwen, F.E.; Delemarre-van de Waal, H.A. Body composition in children and adolescents born after in vitro fertilization or spontaneous conception. J. Clin. Endocrinol. Metab. 2007, 92, 3417–3423. [Google Scholar] [CrossRef] [PubMed]
- Ceelen, M.; van Weissenbruch, M.M.; Vermeiden, J.P.; van Leeuwen, F.E.; Delemarre-van de Waal, H.A. Cardiometabolic differences in children born after in vitro fertilization: Follow-up study. J. Clin. Endocrinol. Metab. 2008, 93, 1682–1688. [Google Scholar] [CrossRef] [PubMed]
- Bi, W.; Xiao, Y.; Wang, X.; Cui, L.; Song, G.; Yang, Z.; Zhang, Y.; Ren, W. The association between assisted reproductive technology and cardiac remodeling in fetuses and early infants: A prospective cohort study. BMC Med. 2022, 20, 104. [Google Scholar] [CrossRef]
- Huang, J.Y.; Cai, S.; Huang, Z.; Tint, M.T.; Yuan, W.L.; Aris, I.M.; Godfrey, K.M.; Karnani, N.; Lee, Y.S.; Chan, J.K.Y.; et al. Analyses of child cardiometabolic phenotype following assisted reproductive technologies using a pragmatic trial emulation approach. Nat. Commun. 2021, 12, 5613. [Google Scholar] [CrossRef]
- Halliday, J.; Lewis, S.; Kennedy, J.; Burgner, D.P.; Juonala, M.; Hammarberg, K.; Amor, D.J.; Doyle, L.W.; Saffery, R.; Ranganathan, S.; et al. Health of adults aged 22 to 35 years con-ceived by assisted reproductive technology. Fertil. Steril. 2019, 112, 130–139. [Google Scholar] [CrossRef]
- Novakovic, B.; Lewis, S.; Halliday, J.; Kennedy, J.; Burgner, D.P.; Czajko, A.; Kim, B.; Sexton-Oates, A.; Juonala, M.; Hammarberg, K.; et al. Assisted reproductive technologies are associated with limited epigenetic variation at birth that largely resolves by adulthood. Nat. Commun. 2019, 10, 3922. [Google Scholar] [CrossRef] [PubMed]
- Denomme, M.M.; Mann, M.R. Genomic imprints as a model for the analysis of epigenetic stability during ARTs. Reproduction 2012, 144, 393–409. [Google Scholar] [CrossRef]
- Zechner, U.; Pliushch, G.; Schneider, E.; El Hajj, N.; Tresch, A.; Shufaro, Y.; Seidmann, L.; Coerdt, W.; Müller, A.M.; Haaf, T. Quantitative methylation analysis of developmentally important genes in human pregnancy losses after ART and spontaneous conception. Mol. Hum. Reprod. 2009, 16, 704–713. [Google Scholar] [CrossRef]
- Sciorio, R.; Dattilo, M. PGT-A preimplantation genetic testing for aneuploidies and embryo selection in routine ART cycles: Time to step back? Clin. Genet. 2020, 98, 107–115. [Google Scholar] [CrossRef]
- Dokras, A.; Sargent, I.; Ross, C.; Gardner, R.; Barlow, D. Trophectoderm biopsy in human blastocysts. Hum. Reprod. 1990, 5, 821–825. [Google Scholar] [CrossRef]
- Honguntikar, S.D.; Salian, S.R.; D’Souza, F.; Uppangala, S.; Kalthur, G.; Adiga, S.K. Epigenetic changes in preimplan-tation embryos subjected to laser manipulation. Lasers Med. Sci. 2017, 32, 2081–2087. [Google Scholar] [CrossRef] [PubMed]
- Glujovsky, D.; Farquhar, C.; Quinteiro Retamar, A.M.; Alvarez Sedo, C.R.; Blake, D. Cleavage stage versus blastocyst stage embryo transfer in assisted reproductive technology (Review). Cochrane Database Syst. Rev. 2016, 30, CD002118. [Google Scholar]
- Salilew-Wondim, D.; Saeed-Zidane, M.; Hoelker, M.; Gebremedhn, S.; Poirier, M.; Pandey, H.O.; Tholen, E.; Neuhoff, C.; Held, E.; Besenfelder, U.; et al. Genome-wide DNA methylation patterns of bovine blastocysts derived from in vivo embryos subjected to in vitro culture before, during or after embryonic genome activation. BMC Genom. 2018, 19, 424. [Google Scholar] [CrossRef] [PubMed]
- Beyer, C.; Osianlis, T.; Boekel, K.; Osborne, E.; Rombauts, L.; Catt, J.; Kralevski, V.; Aali, B.; Gras, L. Preimplantation genetic screening outcomes are associated with culture conditions. Hum. Reprod. 2009, 24, 1212–1220. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Picton, H.M.; Elder, K.; Houghton, F.D.; Hawkhead, J.A.; Rutherford, A.J.; Hogg, J.E.; Leese, H.J.; Harris, S.E. Association between amino acid turnover and chromosome aneuploidy during human preimplantation embryo development in vitro. Mol. Hum. Reprod. 2010, 16, 557–569. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sciorio, R.; El Hajj, N. Epigenetic Risks of Medically Assisted Reproduction. J. Clin. Med. 2022, 11, 2151. https://doi.org/10.3390/jcm11082151
Sciorio R, El Hajj N. Epigenetic Risks of Medically Assisted Reproduction. Journal of Clinical Medicine. 2022; 11(8):2151. https://doi.org/10.3390/jcm11082151
Chicago/Turabian StyleSciorio, Romualdo, and Nady El Hajj. 2022. "Epigenetic Risks of Medically Assisted Reproduction" Journal of Clinical Medicine 11, no. 8: 2151. https://doi.org/10.3390/jcm11082151
APA StyleSciorio, R., & El Hajj, N. (2022). Epigenetic Risks of Medically Assisted Reproduction. Journal of Clinical Medicine, 11(8), 2151. https://doi.org/10.3390/jcm11082151






