Advances and Challenges in Biomarkers Use for Coronary Microvascular Dysfunction: From Bench to Clinical Practice
Abstract
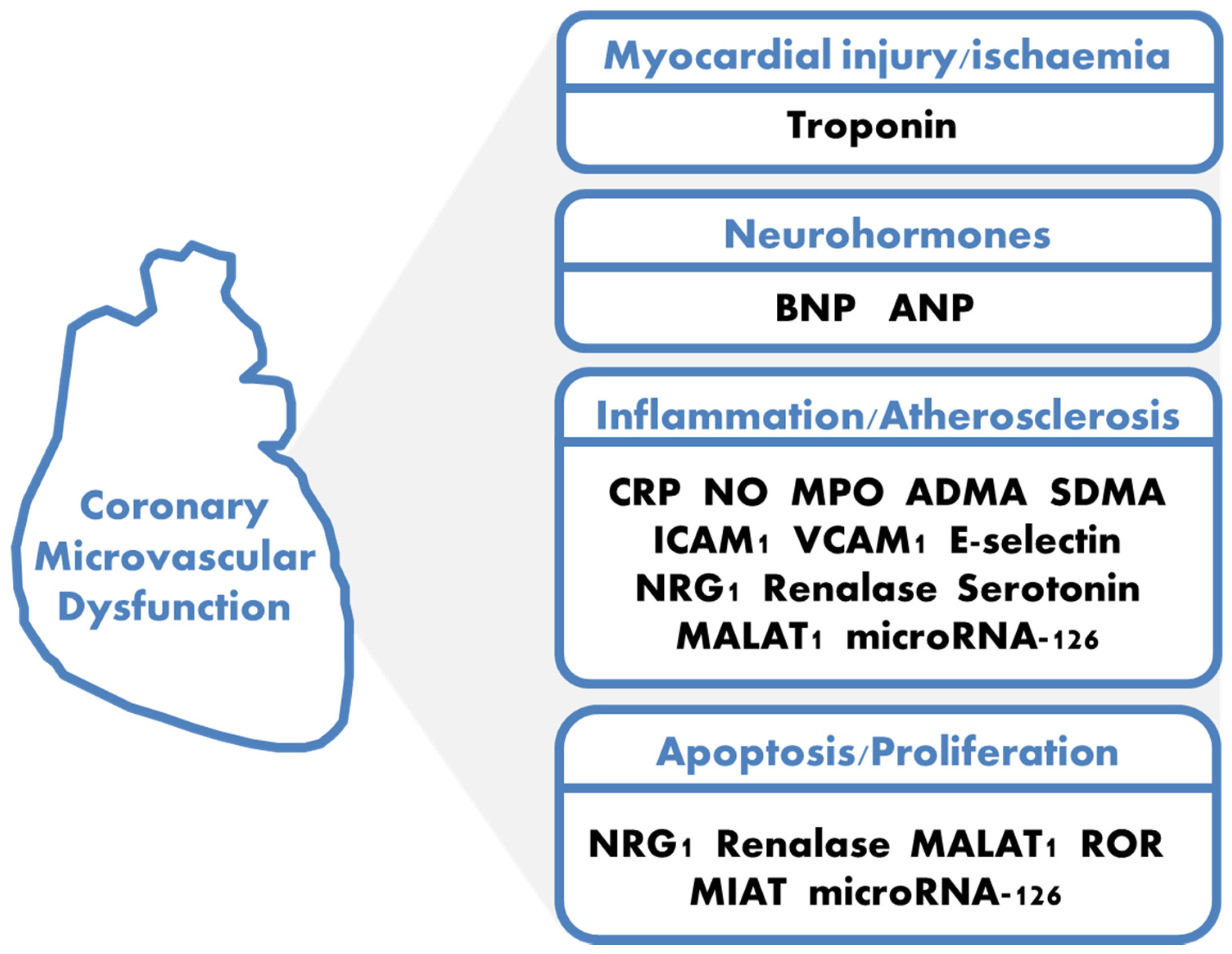
1. Definition and Pathophysiology
2. The Current State of the Art for the Diagnosis of CMD
3. Traditional Biomarkers: Troponin and Natriuretic Peptides
4. Endothelial Microvascular Inflammation: Nitric Oxide (NO), Myeloperoxidase (MPO), Asymmetric Dimethylarginine (ADMA), Symmetric Dimethylarginine (SDMA), Calprotectin, C-Reactive Protein (CRP)
5. Cell Adhesion Molecules: ICAM-1, VCAM-1, E-Selectin
6. Neuregulin-1
7. Renalase
8. Serotonin
9. Noncoding RNAs End Extracellular Vessels
10. Conclusions and Future Perspectives
11. Limitations and Methodology
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Padro, T.; Manfrini, O.; Bugiardini, R.; Canty, J.; Cenko, E.; De Luca, G.; Duncker, D.J.; Eringa, E.C.; Koller, A.; Tousoulis, D.; et al. ESC Working Group on Coronary Pathophysiology and Microcirculation position paper on ‘coronary microvascular dysfunction in cardiovascular disease’. Cardiovasc. Res. 2020, 116, 741–755. [Google Scholar] [CrossRef]
- Camici, P.G.; Crea, F. Coronary microvascular dysfunction. N. Engl. J. Med. 2007, 356, 830–840. [Google Scholar] [CrossRef]
- Li, X.; Sun, X.; Carmeliet, P. Hallmarks of Endothelial Cell Metabolism in Health and Disease. Cell Metab. 2019, 30, 414–433. [Google Scholar] [CrossRef]
- Dikalov, S.; Itani, H.A.; Richmond, B.; Arslanbaeva, L.; Vergeade, A.; Rahman, S.M.J.; Boutaud, O.; Blackwell, T.; Massion, P.P.; Harrison, D.G.; et al. Tobacco smoking induces cardiovascular mitochondrial oxidative stress, promotes endothelial dysfunction, and enhances hypertension. Am. J. Physiol. Heart Circ. Physiol. 2019, 316, H639–H646. [Google Scholar] [CrossRef]
- Miao, H.; Hu, Y.-L.; Shiu, Y.-T.; Yuan, S.; Zhao, Y.; Kaunas, R.; Wang, Y.; Jin, G.; Usami, S.; Chien, S. Effects of Flow Patterns on the Localization and Expression of VE-Cadherin at Vascular Endothelial Cell Junctions: In vivo and in vitro Investigations. J. Vasc. Res. 2005, 42, 77–89. [Google Scholar] [CrossRef]
- Souilhol, C.; Serbanovic-Canic, J.; Fragiadaki, M.; Chico, T.J.; Ridger, V.; Roddie, H.; Evans, P.C. Endothelial responses to shear stress in atherosclerosis: A novel role for developmental genes. Nat. Rev. Cardiol. 2019, 17, 52–63. [Google Scholar] [CrossRef] [PubMed]
- Thoumine, O.; Nerem, R.M.; Girard, F.R. Oscillatory shear stress and hydrostatic pressure modulate cell-matrix attachment proteins in cultured endothelial cells. In Vitro Cell Dev. Biol. Anim. 1995, 31, 45–54. [Google Scholar] [CrossRef] [PubMed]
- Hunt, B.J.; Jurd, K.M. Endothelial cell activation. A central pathophysiological process. BMJ 1998, 316, 1328–1329. [Google Scholar] [CrossRef] [PubMed]
- Rubinshtein, R.; Yang, E.H.; Rihal, C.S.; Prasad, A.; Lennon, R.J.; Best, P.J.; Lerman, L.O.; Lerman, A. Coronary microcirculatory vasodilator function in relation to risk factors among patients without obstructive coronary disease and low to intermediate Framingham score. Eur. Heart J. 2009, 31, 936–942. [Google Scholar] [CrossRef] [PubMed]
- Granger, D.N.; Rodrigues, S.F.; Yildirim, A.; Senchenkova, E.Y. Microvascular Responses to Cardiovascular Risk Factors. Microcirculation 2010, 17, 192–205. [Google Scholar] [CrossRef]
- Mundi, S.; Massaro, M.; Scoditti, E.; Carluccio, M.A.; van Hinsbergh, V.W.M.; Iruela-Arispe, M.L.; De Caterina, R. Endothelial permeability, LDL deposition, and cardiovascular risk factors—A review. Cardiovasc. Res. 2018, 114, 35–52. [Google Scholar] [CrossRef] [PubMed]
- Stokes, K.Y.; Granger, D.N. Platelets: A critical link between inflammation and microvascular dysfunction. J. Physiol. 2012, 590, 1023–1034. [Google Scholar] [CrossRef] [PubMed]
- Gavins, F.N.E.; Li, G.; Russell, J.; Perretti, M.; Granger, D.N. Microvascular thrombosis and CD40/CD40L signaling. J. Thromb. Haemost. 2011, 9, 574–581. [Google Scholar] [CrossRef] [PubMed]
- Szekeres, M.; Nádasy, G.L.; Dörnyei, G.; Szénási, A.; Koller, A. Remodeling of Wall Mechanics and the Myogenic Mechanism of Rat Intramural Coronary Arterioles in Response to a Short-Term Daily Exercise Program: Role of Endothelial Factors. J. Vasc. Res. 2018, 55, 87–97. [Google Scholar] [CrossRef] [PubMed]
- Yun, J.-S.; Park, Y.-M.; Cha, S.-A.; Ahn, Y.-B.; Ko, S.-H. Progression of cardiovascular autonomic neuropathy and cardio-vascular disease in type 2 diabetes. Cardiovasc. Diabetol. 2018, 17, 109. [Google Scholar] [CrossRef]
- Ong, P.; Camici, P.G.; Beltrame, J.F.; Crea, F.; Shimokawa, H.; Sechtem, U.; Kaski, J.C.; Merz, C.N.B.; Coronary Vasomotion Disorders International Study Group (COVADIS). International standardization of diagnostic criteria for microvascular angina. Int. J. Cardiol. 2018, 250, 16–20. [Google Scholar] [CrossRef]
- Ong, P.; Safdar, B.; Seitz, A.; Hubert, A.; Beltrame, J.F.; Prescott, E. Diagnosis of coronary microvascular dysfunction in the clinic. Cardiovasc. Res. 2020, 116, 841–855. [Google Scholar] [CrossRef]
- Knuuti, J.; Wijns, W.; Saraste, A.; Capodanno, D.; Barbato, E.; Funck-Brentano, C.; Bax, J.J. 2019 ESC Guidelines for the diagnosis and management of chronic coronary syndromes. Eur. Heart J. 2020, 41, 407–477. [Google Scholar] [CrossRef]
- Radico, F.; Cicchitti, V.; Zimarino, M.; De Caterina, R. Angina Pectoris and Myocardial Ischemia in the Absence of Obstructive Coronary Artery Disease: Practical Considerations for Diagnostic Tests. JACC Cardiovasc. Interv. 2014, 7, 453–463. [Google Scholar] [CrossRef]
- Echavarria-Pinto, M.; Escaned, J.; Macías, E.; Medina, M.; Gonzalo, N.; Petraco, R.; Macaya, C. Disturbed coronary hemodynamics in vessels with intermediate stenoses evaluated with fractional flow reserve: A combined analysis of epicardial and microcirculatory involvement in ischemic heart disease. Circulation 2013, 128, 2557–2566. [Google Scholar] [CrossRef]
- Mejía-Rentería, H.; Van Der Hoeven, N.; Van De Hoef, T.P.; Heemelaar, J.; Ryan, N.; Lerman, A.; Van Royen, N.; Escaned, J. Targeting the dominant mechanism of coronary microvascular dysfunction with intracoronary physiology tests. Int. J. Cardiovasc. Imaging 2017, 33, 1041–1059. [Google Scholar] [CrossRef] [PubMed]
- Melikian, N.; Vercauteren, S.; Fearon, W.; Cuisset, T.; MacCarthy, P.; Davidavičius, G.; Aarnoudse, W.; Bartunek, J.; Vanderheyden, M.; Wyffels, E.; et al. Quantitative assessment of coronary microvascular function in patients with and without epicardial atherosclerosis. J. Eur. Collab. Work. Gr. Interv. Cardiol. Eur. Soc. Cardiol. 2010, 5, 939–945. [Google Scholar] [CrossRef] [PubMed]
- Layland, J.J.; Whitbourn, R.J.; Burns, A.T.; Somaratne, J.; Leitl, G.; MacIsaac, A.I.; Wilson, A. The index of microvascular resistance identifies patients with periprocedural myocardial infarction in elective percutaneous coronary intervention. Heart 2012, 98, 1492–1497. [Google Scholar] [CrossRef] [PubMed]
- Alexander, Y.; Osto, E.; Schmidt-Trucksäss, A.; Shechter, M.; Trifunovic, D.; Duncker, D.J.; Aboyans, V.; Bäck, M.; Badimon, L.; Cosentino, F.; et al. Endothelial function in cardiovascular medicine: A consensus paper of the European Society of Cardi-ology Working Groups on Atherosclerosis and Vascular Biology, Aorta and Peripheral Vascular Diseases, Coronary Pathophysiology and Microcirculation, and thrombosis. Cardiovasc. Res. 2021, 117, 29–42. [Google Scholar]
- Thygesen, K.; Alpert, J.S.; Jaffe, A.S.; Chaitman, B.R.; Bax, J.J.; Morrow, D.A.; White, H.D.; Executive Group on behalf of the Joint European Society of Cardiology (ESC)/American College of Cardiology (ACC)/American Heart Association (AHA)/World Heart Federation (WHF) Task Force for the Universal Definition of Myocardial Infarction. Fourth Universal Definition of Myocardial Infarction (2018). J. Am. Coll. Cardiol. 2018, 72, 2231–2264. [Google Scholar] [CrossRef] [PubMed]
- Bugiardini, R.; Manfrini, O.; De Ferrari, G.M. Unanswered questions for management of acute coronary syndrome: Risk stratification of patients with minimal disease or normal findings on coronary angiography. Arch. Intern. Med. 2006, 166, 1391–1395. [Google Scholar] [CrossRef] [PubMed]
- Maino, A.; Di Stasio, E.; Grimaldi, M.C.; Cappannoli, L.; Rocco, E.; Vergallo, R.; Biscetti, F.; Baroni, S.; Urbani, A.; Landolfi, R.; et al. Prevalence and characteristics of myocardial injury during COVID-19 pandemic: A new role for high-sensitive troponin. Int. J. Cardiol. 2021, 338, 278–285. [Google Scholar] [CrossRef] [PubMed]
- Takashio, S.; Yamamuro, M.; Izumiya, Y.; Sugiyama, S.; Kojima, S.; Yamamoto, E.; Tsujita, K.; Tanaka, T.; Tayama, S.; Kaikita, K.; et al. Coronary Microvascular Dysfunction and Diastolic Load Correlate With Cardiac Troponin T Release Measured by a Highly Sensitive Assay in Patients With Nonischemic Heart Failure. J. Am. Coll. Cardiol. 2013, 62, 632–640. [Google Scholar] [CrossRef]
- Fujii, K.; Kawasaki, D.; Oka, K.; Akahori, H.; Iwasaku, T.; Fukunaga, M.; Eguchi, A.; Sawada, H.; Masutani, M.; Lee-Kawabata, M.; et al. The Impact of Pravastatin Pre-Treatment on Periprocedural Microcirculatory Damage in Patients Undergoing Percutaneous Coronary Intervention. JACC Cardiovasc. Interv. 2011, 4, 513–520. [Google Scholar] [CrossRef][Green Version]
- Kitabata, H.; Kubo, T.; Ishibashi, K.; Komukai, K.; Tanimoto, T.; Ino, Y.; Kashiwagi, M.; Ozaki, Y.; Shiono, Y.; Shimamura, K.; et al. Prognostic Value of Microvascular Resistance Index Immediately After Primary Percutaneous Coronary Intervention on Left Ventricular Remodeling in Patients With Reperfused Anterior Acute ST-Segment Elevation Myocardial Infarction. JACC Cardiovasc. Interv. 2013, 6, 1046–1054. [Google Scholar] [CrossRef]
- Herrmann, J.; Haude, M.; Lerman, A.; Schulz, R.; Volbracht, L.; Ge, J.; Schmermund, A.; Wieneke, H.; von Birgelen, C.; Eggebrecht, H.; et al. Abnormal Coronary Flow Velocity Reserve After Coronary Intervention Is Associated with Cardiac Marker Elevation. Circulation 2001, 103, 2339–2345. [Google Scholar] [CrossRef] [PubMed]
- Park, K.; Kim, M.; Cho, Y.-R.; Park, J.-S.; Park, T.-H.; Kim, M.H.; Kim, Y.-D. Association between Cardiac Troponin Level and Coronary Flow Reserve in Patients without Coronary Artery Disease: Insight from a Thermodilution Technique Using an Intracoronary Pressure Wire. Korean Circ. J. 2014, 44, 141–147. [Google Scholar] [CrossRef] [PubMed]
- Taqueti, V.R.; Everett, B.M.; Murthy, V.; Gaber, M.; Foster, C.R.; Hainer, J.; Blankstein, R.; Dorbala, S.; Di Carli, M.F. Interaction of Impaired Coronary Flow Reserve and Cardiomyocyte Injury on Adverse Cardiovascular Outcomes in Patients Without Overt Coronary Artery Disease. Circulation 2015, 131, 528–535. [Google Scholar] [CrossRef] [PubMed]
- McDonagh, T.A.; Metra, M.; Adamo, M.; Gardner, R.S.; Baumbach, A.; Böhm, M.; Burri, H.; Butler, J.; Čelutkienė, J.; Chioncel, O.; et al. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: Developed by the Task Force for the Diagnosis and Treatment of Acute and Chronic Heart Failure of the European Society of Cardiology (ESC) with the Special Contribution of the Heart Failure Association (HFA) of the ESC. Eur. Heart J. 2021, 42, 3599–3726. [Google Scholar] [CrossRef] [PubMed]
- Aspromonte, N.; Cappannoli, L.; Scicchitano, P.; Massari, F.; Pantano, I.; Massetti, M.; Crea, F.; Valle, R. Stay Home! Stay Safe! First Post-Discharge Cardiologic Evaluation of Low-Risk–Low-BNP Heart Failure Patients in COVID-19 Era. J. Clin. Med. 2021, 10, 2126. [Google Scholar] [CrossRef]
- Houben, A.J.; Krekels, M.M.; Schaper, N.; Fuss-Lejeune, M.J.; Rodriguez, S.A.; De Leeuw, P.W. Microvascular effects of atrial natriuretic peptide (ANP) in man: Studies during high and low salt diet. Cardiovasc. Res. 1998, 39, 442–450. [Google Scholar] [CrossRef][Green Version]
- Dudek, D.; Rzeszutko, L.; Dimitrow, P.P.; Bartus, S.; Sorysz, D.; Chyrchel, M.; Rakowski, T.; Zdzienicka, A.; Guevara, I.; Dembinska-Kiec, A.; et al. Circulating N-terminal brain natriuretic peptide precursor and endothelin levels in patients with syn-drome X and left bundle branch block with preserved systolic function. Int. J. Cardiol. 2001, 79, 25–30. [Google Scholar] [CrossRef]
- Knaapen, P.; Germans, T.; Camici, P.G.; Rimoldi, O.E.; Cate, F.J.T.; Berg, J.M.T.; Dijkmans, P.A.; Boellaard, R.; Van Dockum, W.G.; Götte, M.J.W.; et al. Determinants of coronary microvascular dysfunction in symptomatic hypertrophic cardiomyopathy. Am. J. Physiol. Heart Circ. Physiol. 2008, 294, H986–H993. [Google Scholar] [CrossRef]
- Mitchell, A.; Misialek, J.R.; Folsom, A.R.; Duprez, D.; Alonso, A.; Jerosch-Herold, M.; Sanchez, O.A.; Watson, K.E.; Sallam, T.; Konety, S.H. Usefulness of N-terminal Pro–brain Natriuretic Peptide and Myocardial Perfusion in Asymptomatic Adults (from the Multi-Ethnic Study of Atherosclerosis). Am. J. Cardiol. 2015, 115, 1341–1345. [Google Scholar] [CrossRef][Green Version]
- Liuzzo, G.; Biasucci, L.M.; Gallimore, J.R.; Grillo, R.L.; Rebuzzi, A.G.; Pepys, M.B.; Maseri, A. The prognostic value of C-reactive protein and serum amyloid a protein in severe unstable angina. N. Engl. J. Med. 1994, 331, 417–424. [Google Scholar] [CrossRef]
- Crea, F.; Liuzzo, G. Pathogenesis of acute coronary syndromes. J. Am. Coll. Cardiol. 2013, 61, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Pedicino, D.; Giglio, A.F.; Galiffa, V.A.; Cialdella, P.; Trotta, F.; Graziani, F.; Liuzzo, G. Infections, immunity and atherosclerosis: Pathogenic mechanisms and unsolved questions. Int. J. Cardiol. 2013, 166, 572–583. [Google Scholar] [CrossRef] [PubMed]
- Vinci, R.; Pedicino, D.; Bonanni, A.; D’Aiello, A.; Severino, A.; Pisano, E.; Ponzo, M.; Canonico, F.; Ciampi, P.; Russo, G.; et al. A Novel Monocyte Subset as a Unique Signature of Atherosclerotic Plaque Rupture. Front. Cell Dev. Biol. 2021, 9, 2666. [Google Scholar] [CrossRef] [PubMed]
- Basile, U.; La Rosa, G.; Napodano, C.; Pocino, K.; Cappannoli, L.; Gulli, F.; Cianfrocca, C.; Di Stasio, E.; Biasucci, L.M. Free light chains a novel biomarker of cardiovascular disease. A pilot study. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 2563–2569. [Google Scholar] [PubMed]
- D’Amario, D.; Cappetta, D.; Cappannoli, L.; Princi, G.; Migliaro, S.; Diana, G.; Chouchane, K.; Borovac, J.A.; Restivo, A.; Arcudi, A.; et al. Colchicine in ischemic heart disease: The good, the bad and the ugly. Clin. Res. Cardiol. 2021, 110, 1531–1542. [Google Scholar] [CrossRef]
- Taqueti, V.R.; Di Carli, M.F. Coronary Microvascular Disease Pathogenic Mechanisms and Therapeutic Options: JACC State-of-the-Art Review. J. Am. Coll. Cardiol. 2018, 72, 2625–2641. [Google Scholar] [CrossRef]
- Ohanyan, V.; Sisakian, H.; Peketi, P.; Parikh, A.; Chilian, W. A chicken and egg conundrum: Coronary microvascular dysfunction and heart failure with preserved ejection fraction. Am. J. Physiol. Circ. Physiol. 2018, 314, H1262–H1263. [Google Scholar] [CrossRef]
- D’Amario, D.; Migliaro, S.; Borovac, J.A.; Restivo, A.; Vergallo, R.; Galli, M.; Leone, A.M.; Montone, R.A.; Niccoli, G.; Aspromonte, N.; et al. Microvascular Dysfunction in Heart Failure with Preserved Ejection Fraction. Front. Physiol. 2019, 10, 1347. [Google Scholar] [CrossRef]
- von Leitner, E.-C.; Klinke, A.; Atzler, D.; Slocum, J.L.; Lund, N.; Kielstein, J.T.; Maas, R.; Schmidt-Haupt, R.; Pekarova, M.; Hellwinkel, O.; et al. Pathogenic Cycle between the Endogenous Nitric Oxide Synthase Inhibitor Asymmetrical Dimethylarginine and the Leukocyte-Derived Hemoprotein Myeloperoxidase. Circulation 2011, 124, 2735–2745. [Google Scholar] [CrossRef]
- Biasucci, L.M.; Maino, A.; Grimaldi, M.C.; Cappannoli, L.; Aspromonte, N. Novel Biomarkers in Heart Failure: New In-sight in Pathophysiology and Clinical Perspective. J. Clin. Med. 2021, 10, 2771. [Google Scholar] [CrossRef]
- Ky, B.; Putt, M.; Sawaya, H.; French, B.; Januzzi, J.L.; Sebag, I.A.; Plana, J.C.; Cohen, V.; Banchs, J.; Carver, J.R.; et al. Early Increases in Multiple Biomarkers Predict Subsequent Cardiotoxicity in Patients With Breast Cancer Treated With Doxorubicin, Taxanes, and Trastuzumab. J. Am. Coll. Cardiol. 2014, 63, 809–816. [Google Scholar] [CrossRef] [PubMed]
- Eiserich, J.P.; Baldus, S.; Brennan, M.-L.; Hoffman, S.L.; Zhang, C.; Tousson, A.; Castro, L.; Lusis, A.J.; Nauseef, W.M.; White, C.R.; et al. Myeloperoxidase, a Leukocyte-Derived Vascular NO Oxidase. Science 2002, 296, 2391–2394. [Google Scholar] [CrossRef] [PubMed]
- Schnabel, R.; Blankenberg, S.; Lubos, E.; Lackner, K.J.; Rupprecht, H.J.; Espinola-Klein, C.; Münzel, T. Asymmetric dimethylarginine and the risk of cardiovascular events and death in patients with coronary artery disease: Results from the AtheroGene Study. Circ. Res. 2005, 97, e53–e59. [Google Scholar] [CrossRef] [PubMed]
- Böger, R.H.; Sullivan, L.M.; Schwedhelm, E.; Wang, T.J.; Maas, R.; Benjamin, E.J.; Schulze, F.; Xanthakis, V.; Benndorf, R.A.; Vasan, R.S. Plasma Asymmetric Dimethylarginine and Incidence of Cardiovascular Disease and Death in the Community. Circulation 2009, 119, 1592–1600. [Google Scholar] [CrossRef]
- Böger, R.H.; Bode-Böger, S.M.; Szuba, A.; Tsao, P.S.; Chan, J.R.; Tangphao, O.; Cooke, J.P. Asymmetric dimethylarginine (ADMA): A novel risk factor for endothelial dysfunction: Its role in hypercholesterolemia. Circulation 1998, 98, 1842–1847. [Google Scholar] [CrossRef]
- Wang, Z.; Tang, W.H.W.; Cho, L.; Brennan, D.M.; Hazen, S.L. Targeted metabolomic evaluation of arginine methylation and cardiovascular risks: Potential mechanisms beyond nitric oxide synthase inhibition. Arterioscler. Thromb. Vasc. Biol. 2009, 29, 1383–1391. [Google Scholar] [CrossRef]
- Hage, C.; Michaëlsson, E.; Kull, B.; Miliotis, T.; Svedlund, S.; Linde, C.; Donal, E.; Daubert, J.; Gan, L.; Lund, L.H. Myeloperoxidase and related biomarkers are suggestive footprints of endothelial microvascular inflammation in HFpEF patients. ESC Heart Fail. 2020, 7, 1534–1546. [Google Scholar] [CrossRef]
- Ridker, P.M.; Cook, N. Clinical Usefulness of Very High and Very Low Levels of C-Reactive Protein Across the Full Range of Framingham Risk Scores. Circulation 2004, 109, 1955–1959. [Google Scholar] [CrossRef]
- Cook, N.R.; Paynter, N.P.; Eaton, C.B.; Manson, J.E.; Martin, L.W.; Robinson, J.G.; Rossouw, J.E.; Wassertheil-Smoller, S.; Ridker, P.M. Comparison of the Framingham and Reynolds Risk Scores for Global Cardiovascular Risk Prediction in the Multiethnic Women’s Health Initiative. Circulation 2012, 125, 1748–1756. [Google Scholar] [CrossRef]
- Cosín-Sales, J.; Pizzi, C.; Brown, S.; Kaski, J.C. C-reactive protein, clinical presentation, and ischemic activity in patients with chest pain and normal coronary angiograms. J. Am. Coll. Cardiol. 2003, 41, 1468–1474. [Google Scholar] [CrossRef]
- Recio-Mayoral, A.; Rimoldi, O.E.; Camici, P.G.; Kaski, J.C. Inflammation and microvascular dysfunction in cardiac syn-drome X patients without conventional risk factors for coronary artery disease. JACC. Cardiovasc. Imaging 2013, 6, 660–667. [Google Scholar] [CrossRef] [PubMed]
- Qamirani, E.; Ren, Y.; Kuo, L.; Hein, T.W. C-Reactive Protein Inhibits Endothelium-Dependent NO-Mediated Dilation in Coronary Arterioles by Activating p38 Kinase and NAD(P)H Oxidase. Arterioscler. Thromb. Vasc. Biol. 2005, 25, 995–1001. [Google Scholar] [CrossRef] [PubMed]
- Godo, S.; Takahashi, J.; Yasuda, S.; Shimokawa, H. Role of Inflammation in Coronary Epicardial and Microvascular Dysfunction. Eur. Cardiol. Rev. 2021, 16, e13. [Google Scholar] [CrossRef] [PubMed]
- Springer, T.A. Adhesion receptors of the immune system. Nature 1990, 346, 425–434. [Google Scholar] [CrossRef]
- Carlos, T.M.; Harlan, J.M. Leukocyte-endothelial adhesion molecules. Blood 1994, 84, 2068–2101. [Google Scholar] [CrossRef]
- Turhan, H.; Saydam, G.S.; Erbay, A.R.; Ayaz, S.; Yasar, A.S.; Aksoy, Y.; Basar, N.; Yetkin, E. Increased plasma soluble adhesion molecules; ICAM-1, VCAM-1, and E-selectin levels in patients with slow coronary flow. Int. J. Cardiol. 2006, 108, 224–230. [Google Scholar] [CrossRef]
- Tousoulis, D.; Davies, G.J.; Asimakopoulos, G.; Homaei, H.; Zouridakis, E.; Ahmed, N.; Kaski, J.C. Vascular cell adhesion molecule-1 and intercellular adhesion molecule-1 serum level in patients with chest pain and normal coronary arteries (syndrome X). Clin. Cardiol. 2001, 24, 301–304. [Google Scholar] [CrossRef]
- Lupattelli, G.; Lombardini, R.; Schillaci, G.; Ciuffetti, G.; Marchesi, S.; Siepi, D.; Mannarino, E. Flow-mediated vasoactivity and circulating adhesion molecules in hypertriglyceridemia: Association with small, dense LDL cholesterol particles. Am. Heart J. 2000, 140, 521–526. [Google Scholar] [CrossRef]
- Miwa, K.; Igawa, A.; Inoue, H. Soluble E-selectin, ICAM-1 and VCAM-1 levels in systemic and coronary circulation in patients with variant angina. Cardiovasc. Res. 1997, 36, 37–44. [Google Scholar] [CrossRef]
- Vaccarino, V.; Khan, D.; Votaw, J.; Faber, T.; Veledar, E.; Jones, D.P.; Goldberg, J.; Raggi, P.; Quyyumi, A.A.; Bremner, J.D. Inflammation is Related to Coronary Flow Reserve Detected by Positron Emission Tomography in Asymptomatic Male Twins. J. Am. Coll. Cardiol. 2011, 57, 1271–1279. [Google Scholar] [CrossRef]
- Siminiak, T.; Śmielecki, J.; Rzeźniczak, J.; Kaźmierczak, M.; Kalawski, R.; Wysocki, H. The effects of dipyridamole stress test on plasma levels of soluble adhesion molecules intracellular adhesion molecule-1, vascular cell adhesion molecule-1, E-selectin and L-selectin in patients with ischemic heart disease and patients with syndrome X. Coron. Artery Dis. 1999, 10, 235–254. [Google Scholar] [CrossRef] [PubMed]
- Britsch, S. The neuregulin-I/ErbB signaling system in development and disease. Adv. Anat. Embryol. Cell Biol. 2007, 190, 1–65. [Google Scholar] [PubMed]
- Hedhli, N.; Huang, Q.; Kalinowski, A.; Palmeri, M.; Hu, X.; Russell, R.R.; Russell, K.S. Endothelium-Derived Neuregulin Protects the Heart Against Ischemic Injury. Circulation 2011, 123, 2254–2262. [Google Scholar] [CrossRef] [PubMed]
- Kang, W.; Cheng, Y.; Zhou, F.; Wang, L.; Zhong, L.; Li, H.T.; Wang, X.; Dang, S.; Wang, X. Neuregulin-1 protects cardiac function in septic rats through multiple targets based on endothelial cells. Int. J. Mol. Med. 2019, 44, 1255–1266. [Google Scholar] [CrossRef] [PubMed]
- Kalinowski, A.; Plowes, N.J.R.; Huang, Q.; Berdejo-Izquierdo, C.; Russell, R.R.; Russell, K.S. Metalloproteinase-dependent cleavage of neuregulin and autocrine stimulation of vascular endothelial cells. FASEB J. 2010, 24, 2567–2575. [Google Scholar] [CrossRef] [PubMed]
- Timolati, F.; Ott, D.; Pentassuglia, L.; Giraud, M.-N.; Perriard, J.-C.; Suter, T.M.; Zuppinger, C. Neuregulin-1 beta attenuates doxorubicin-induced alterations of excitation–contraction coupling and reduces oxidative stress in adult rat cardiomyocytes. J. Mol. Cell. Cardiol. 2006, 41, 845–854. [Google Scholar] [CrossRef]
- Brero, A.; Ramella, R.; Fitou, A.; Dati, C.; Alloatti, G.; Gallo, M.P.; Levi, R. Neuregulin-1beta1 rapidly modulates nitric oxide syn-thesis and calcium handling in rat cardiomyocytes. Cardiovasc. Res. 2010, 88, 443–452. [Google Scholar] [CrossRef]
- Rupert, C.E.; Coulombe, K.L.K. The Roles of Neuregulin-1 in Cardiac Development, Homeostasis, and Disease. Biomark. Insights 2015, 10 (Suppl. 1), 1–9. [Google Scholar] [CrossRef]
- Hedhli, N.; Kalinowski, A.; Russell, K.S. Cardiovascular effects of neuregulin-1/ErbB signaling: Role in vascular signaling and angiogenesis. Curr. Pharm. Des. 2014, 20, 4899–4905. [Google Scholar] [CrossRef]
- Russell, K.S.; Stern, D.F.; Polverini, P.J.; Bender, J.R. Neuregulin activation of ErbB receptors in vascular endothelium leads to angiogenesis. Am. J. Physiol. Content 1999, 277, H2205–H2211. [Google Scholar] [CrossRef]
- Wu, C.; Gui, C.; Li, L.; Pang, Y.; Tang, Z.; Wei, J. Expression and secretion of neuregulin-1 in cardiac microvascular endothelial cells treated with angiogenic factors. Exp. Ther. Med. 2018, 15, 3577–3581. [Google Scholar] [CrossRef] [PubMed]
- Rohrbach, S.; Niemann, B.; Silber, R.E.; Holtz, J. Neuregulin receptors erbB2 and erbB4 in failing human myocardium -- de-pressed expression and attenuated activation. Basic Res. Cardiol. 2005, 100, 240–249. [Google Scholar] [CrossRef] [PubMed]
- Ky, B.; Kimmel, S.E.; Safa, R.N.; Putt, M.E.; Sweitzer, N.K.; Fang, J.C.; Sawyer, D.B.; Cappola, T.P. Neuregulin-1β Is Associated with Disease Severity and Adverse Outcomes in Chronic Heart Failure. Circulation 2009, 120, 310–317. [Google Scholar] [CrossRef] [PubMed]
- Miao, J.; Huang, S.; Su, Y.R.; Lenneman, C.A.; Wright, M.; Harrell, F.E.; Sawyer, D.B.; Lenihan, D.J. Effects of endogenous serum neuregulin-1β on morbidity and mortality in patients with heart failure and left ventricular systolic dysfunction. Biomarkers 2018, 23, 704–708. [Google Scholar] [CrossRef] [PubMed]
- Lemmens, K.; Doggen, K.; De Keulenaer, G.W. Role of neuregulin-1/ErbB signaling in cardiovascular physiology and disease: Implications for therapy of heart failure. Circulation 2007, 116, 954–960. [Google Scholar] [CrossRef] [PubMed]
- Hage, C.; Wärdell, E.; Linde, C.; Donal, E.; Lam, C.S.; Daubert, C.; Lund, L.H.; Månsson-Broberg, A. Circulating neuregulin1-β in heart failure with preserved and reduced left ventricular ejection fraction. ESC Heart Fail. 2020, 7, 445–455. [Google Scholar] [CrossRef] [PubMed]
- Kuramochi, Y.; Cote, G.M.; Guo, X.; Lebrasseur, N.K.; Cui, L.; Liao, R.; Sawyer, D.B. Cardiac endothelial cells regulate reactive oxy-gen species-induced cardiomyocyte apoptosis through neuregulin-1beta/erbB4 signaling. J. Biol. Chem. 2004, 279, 51141–51147. [Google Scholar] [CrossRef] [PubMed]
- Munk, M.; Memon, A.A.; Goetze, J.P.; Nielsen, L.B.; Nexo, E.; Sorensen, B.S. Hypoxia Changes the Expression of the Epidermal Growth Factor (EGF) System in Human Hearts and Cultured Cardiomyocytes. PLoS ONE 2012, 7, e40243. [Google Scholar] [CrossRef]
- Geisberg, C.A.; Wang, G.; Safa, R.N.; Smith, H.M.; Anderson, B.; Peng, X.-Y.; Veerkamp, B.; Zhao, D.X.; Blakemore, D.; Yu, C.; et al. Circulating neuregulin-1β levels vary according to the angiographic severity of coronary artery disease and ischemia. Coron. Artery Dis. 2011, 22, 577–582. [Google Scholar] [CrossRef]
- Paulus, W.J.; Tschöpe, C. A novel paradigm for heart failure with preserved ejection fraction: Comorbidities drive myocardial dysfunction and remodeling through coronary microvascular endothelial inflammation. J. Am. Coll. Cardiol. 2013, 62, 263–271. [Google Scholar] [CrossRef]
- Shah, S.J.; Lam, C.S.P.; Svedlund, S.; Saraste, A.; Hage, C.; Tan, R.S.; Beussink-Nelson, L.; Ljung Faxén, U.; Fermer, M.L.; Broberg, M.A.; et al. Prevalence and correlates of coronary microvascular dysfunction in heart failure with preserved ejection fraction: PROMIS-HFpEF. Eur. Heart J. 2018, 39, 3439–3450. [Google Scholar] [CrossRef] [PubMed]
- Mendes-Ferreira, P.; Maia-Rocha, C.; Adão, R.; Mendes, M.J.; Santos-Ribeiro, D.; Alves, B.S.; Cerqueira, R.J.; Castro-Chaves, P.; Lourenço, A.P.; De Keulenaer, G.W.; et al. Neuregulin-1 improves right ventricular function and attenuates experimental pulmonary arterial hypertension. Cardiovasc. Res. 2016, 109, 44–54. [Google Scholar] [CrossRef] [PubMed]
- Vermeulen, Z.; Hervent, A.S.; Dugaucquier, L.; Vandekerckhove, L.; Rombouts, M.; Beyens, M.; Schrijvers, D.M.; De Meyer, G.R.Y.; Maudsley, S.; De Keulenaer, G.W.; et al. Inhibitory actions of the NRG-1/ErbB4 pathway in macrophages during tissue fibrosis in the heart, skin, and lung. Am. J. Physiol. Heart Circ. Physiol. 2017, 313, H934–H945. [Google Scholar] [CrossRef] [PubMed]
- Galindo, C.L.; Kasasbeh, E.; Murphy, A.; Ryzhov, S.; Lenihan, S.; Ahmad, F.A.; Williams, P.; Nunnally, A.; Adcock, J.; Song, Y.; et al. An-ti-remodeling and anti-fibrotic effects of the neuregulin-1β glial growth factor 2 in a large animal model of heart failure. J. Am. Heart Assoc. 2014, 3, e000773. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Li, G.; Wang, P.; Velazquez, H.; Yao, X.; Li, Y.; Wu, Y.; Peixoto, A.; Crowley, S.; Desir, G.V. Renalase is a novel, soluble mono-amine oxidase that regulates cardiac function and blood pressure. J. Clin. Investig. 2005, 115, 1275–1280. [Google Scholar] [CrossRef]
- Zhou, M.; Ma, C.; Liu, W.; Liu, H.; Wang, N.; Kang, Q.; Li, P. Valsartan Promoting Atherosclerotic Plaque Stabilization by Upregu-lating Renalase: A Potential-Related Gene of Atherosclerosis. J. Cardiovasc. Pharmacol. Ther. 2015, 20, 509–519. [Google Scholar] [CrossRef]
- Tokinoya, K.; Shiromoto, J.; Sugasawa, T.; Yoshida, Y.; Aoki, K.; Nakagawa, Y.; Ohmori, H.; Takekoshi, K. Influence of acute exercise on renalase and its regulatory mechanism. Life Sci. 2018, 210, 235–242. [Google Scholar] [CrossRef]
- Yılmaz, Z.V.; Akkaş, E.; Yıldırım, T.; Yılmaz, R.; Erdem, Y. A novel marker in pregnant with preeclampsia: Renalase. J. Matern. Fetal. Neonatal Med. 2017, 30, 808–813. [Google Scholar] [CrossRef]
- Lienhart, W.D.; Gudipati, V.; Macheroux, P. The human flavoproteome. Arch Biochem Biophys. 2013, 535, 150–162. [Google Scholar] [CrossRef]
- Yin, J.; Lu, Z.; Wang, F.; Jiang, Z.; Lu, L.; Miao, N.; Wang, N. Renalase attenuates hypertension, renal injury and cardiac remodelling in rats with subtotal nephrectomy. J. Cell. Mol. Med. 2016, 20, 1106–1117. [Google Scholar] [CrossRef]
- Pries, A.R.; Badimon, L.; Bugiardini, R.; Camici, P.G.; Dorobantu, M.; Duncker, D.J.; Escaned, J.; Koller, A.; Piek, J.J.; de Wit, C. Coronary vascular regulation, remodelling, and collateralization: Mechanisms and clinical implications on behalf of the working group on coronary pathophysiology and microcirculation. Eur. Heart J. 2015, 36, 3134–3146. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Xie, Z.; Lin, M.; Huang, R.; Liang, Z.; Huang, W.; Jiang, W. Renalase Protects the Cardiomyocytes of Sprague-Dawley Rats Against Ischemia and Reperfusion Injury by Reducing Myocardial Cell Necrosis and Apoptosis. Kidney Blood Press. Res. 2015, 40, 215–222. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Wang, L.; Velazquez, H.; Safirstein, R.; Desir, G.V. Renalase: Its role as a cytokine, and an update on its association with type 1 diabetes and ischemic stroke. Curr. Opin. Nephrol. Hypertens. 2014, 23, 513–518. [Google Scholar] [CrossRef] [PubMed]
- Przybylowski, P.; Koc-Zorawska, E.; Malyszko, J.S.; Mysliwiec, M.; Malyszko, J. Renalase and Endothelial Dysfunction in Heart Transplant Recipients. Transplant. Proc. 2013, 45, 394–396. [Google Scholar] [CrossRef]
- Safdar, B.; Guo, X.; Johnson, C.; D’Onofrio, G.; Dziura, J.; Sinusas, A.J.; Testani, J.; Rao, V.; Desir, G. Elevated renalase levels in patients with acute coronary microvascular dysfunction—A possible biomarker for ischemia. Int. J. Cardiol. 2019, 279, 155–161. [Google Scholar] [CrossRef]
- Medvedev, A.; Kopylov, A.; Fedchenko, V.; Buneeva, O. Is renalase ready to become a biomarker of ischemia? Int. J. Cardiol. 2020, 307, 179. [Google Scholar] [CrossRef]
- Vanhoutte, P.M. Platelet-derived serotonin, the endothelium, and cardiovascular disease. J. Cardiovasc. Pharmacol. 1991, 17 (Suppl. 5), S6–S12. [Google Scholar] [CrossRef]
- Lanza, G.A.; Giordano, A.; Pristipino, C.; Calcagni, M.L.; Meduri, G.; Trani, C.; Franceschini, R.; Crea, F.; Troncone, L.; Maseri, A. Abnormal cardiac adrenergic nerve function in patients with syndrome X detected by [123I] metaiodobenzylguanidine myocardial scintigraphy. Circulation 1997, 96, 821–826. [Google Scholar] [CrossRef]
- Kaski, J.C.; Rosano, G.; Gavrielides, S.; Chen, L. Effects of angiotensin-converting enzyme inhibition on exercise-induced angina and ST segment depression in patients with microvascular angina. J. Am. Coll. Cardiol. 1994, 23, 652–657. [Google Scholar] [CrossRef]
- McFadden, E.P.; Clarke, J.G.; Davies, G.J.; Kaski, J.C.; Haider, A.W.; Maseri, A. Effect of Intracoronary Serotonin on Coronary Vessels in Patients with Stable Angina and Patients with Variant Angina. N. Engl. J. Med. 1991, 324, 648–654. [Google Scholar] [CrossRef]
- Nemecek, G.M.; Coughlin, S.R.; Handley, D.A.; Moskowitz, M.A. Stimulation of aortic smooth muscle cell mitogenesis by serotonin. Proc. Natl. Acad. Sci. USA 1986, 83, 674–678. [Google Scholar] [CrossRef] [PubMed]
- Vikenes, K.; Farstad, M.; Nordrehaug, J.E. Serotonin Is Associated with Coronary Artery Disease and Cardiac Events. Circulation 1999, 100, 483–489. [Google Scholar] [CrossRef] [PubMed]
- Figueras, J.; Domingo, E.; Cortadellas, J.; Padilla, F.; Dorado, D.G.; Segura, R.; Galard, R.; Soler, J.S. Comparison of Plasma Serotonin Levels in Patients with Variant Angina Pectoris Versus Healed Myocardial Infarction. Am. J. Cardiol. 2005, 96, 204–207. [Google Scholar] [CrossRef]
- Odaka, Y.; Takahashi, J.; Tsuburaya, R.; Nishimiya, K.; Hao, K.; Matsumoto, Y.; Ito, K.; Sakata, Y.; Miyata, S.; Manita, D.; et al. Plasma concentration of serotonin is a novel biomarker for coronary microvascular dysfunction in patients with suspected angina and unobstructive coronary arteries. Eur. Heart J. 2016, 38, 489–496. [Google Scholar] [CrossRef] [PubMed]
- Pizzi, C.; Manzoli, L.; Mancini, S.; Costa, G.M. Analysis of potential predictors of depression among coronary heart disease risk factors including heart rate variability, markers of inflammation, and endothelial function. Eur. Heart J. 2008, 29, 1110–1117. [Google Scholar] [CrossRef]
- Verma, S.; Wang, C.H.; Li, S.H.; Dumont, A.S.; Fedak, P.W.; Badiwala, M.V.; Dhillon, B.; Weisel, R.D.; Li, R.K.; Mickle, D.A.; et al. A self-fulfilling prophecy: C-reactive protein attenuates nitric oxide production and inhibits angiogenesis. Circulation 2002, 106, 913–919. [Google Scholar] [CrossRef] [PubMed]
- Himmerich, H.; Fulda, S.; Linseisen, J.; Seiler, H.; Wolfram, G.; Himmerich, S.; Gedrich, K.; Kloiber, S.; Lucae, S.; Ising, M.; et al. Depression, comorbidities and the TNF-alpha system. Eur. Psychiatry 2008, 23, 421–429. [Google Scholar] [CrossRef] [PubMed]
- Ungvari, Z.; Pacher, P.; Koller, A. Serotonin Reuptake Inhibitor Fluoxetine Decreases Arteriolar Myogenic Tone by Reducing Smooth Muscle [Ca2+]i. J. Cardiovasc. Pharmacol. 2000, 35, 849–854. [Google Scholar] [CrossRef]
- Lara, N.; Archer, S.L.; Baker, G.B.; Le Mellédo, J.M. Paroxetine-induced increase in metabolic endproducts of nitric oxide. J. Clin. Psychopharmacol. 2003, 23, 408–412. [Google Scholar] [CrossRef]
- Pizzi, C.; Mancini, S.; Angeloni, L.; Fontana, F.; Manzoli, L.; Costa, G.M. Effects of Selective Serotonin Reuptake Inhibitor Therapy on Endothelial Function and Inflammatory Markers in Patients with Coronary Heart Disease. Clin. Pharmacol. Ther. 2009, 86, 527–532. [Google Scholar] [CrossRef]
- Lekakis, J.; Ikonomidis, I.; Papoutsi, Z.; Moutsatsou, P.; Nikolaou, M.; Parissis, J.; Kremastinos, D.T. Selective serotonin re-uptake inhibitors decrease the cytokine-induced endothelial adhesion molecule expression, the endothelial adhesiveness to monocytes and the circulating levels of vascular adhesion molecules. Int. J. Cardiol. 2010, 139, 150–158. [Google Scholar] [CrossRef] [PubMed]
- Ma, S.X.; Bai, Z.F.; Wang, W.; Wu, H.Y. Effects of Microrna-93 on mouse cardiac microvascular endothelial cells injury and inflammatory response by mediating SPP1 through the NF-KappaB pathway. J. Cell. Biochem. 2017, 120, 2847–2858. [Google Scholar] [CrossRef] [PubMed]
- Qi, X.-F.; Li, Y.-J.; Chen, Z.-Y.; Kim, S.-K.; Lee, K.-J.; Cai, D.-Q. Involvement of the FoxO3a pathway in the ischemia/reperfusion injury of cardiac microvascular endothelial cells. Exp. Mol. Pathol. 2013, 95, 242–247. [Google Scholar] [CrossRef]
- Boos, C.J.; Jaumdally, R.J.; MacFadyen, R.J.; Varma, C.; Lip, G.Y.H. Circulating endothelial cells and von Willebrand factor as indices of endothelial damage/dysfunction in coronary artery disease: A comparison of central vs. peripheral levels and effects of coronary angioplasty. J. Thromb. Haemost. 2007, 5, 630–632. [Google Scholar] [CrossRef] [PubMed]
- Bonetti, P.O.; Lerman, L.O.; Lerman, A. Endothelial dysfunction: A marker of atherosclerotic risk. Arterioscler. Thromb. Vasc. Biol. 2003, 23, 168–175. [Google Scholar] [CrossRef]
- Widmer, R.J.; Chung, W.-Y.; Herrmann, J.; Jordan, K.L.; Lerman, L.O.; Lerman, A. The Association between Circulating MicroRNA Levels and Coronary Endothelial Function. PLoS ONE 2014, 9, e109650. [Google Scholar] [CrossRef]
- Poller, W.; Dimmeler, S.; Heymans, S.; Zeller, T.; Haas, J.; Karakas, M.; Leistner, D.M.; Jakob, P.; Nakagawa, S.; Blankenberg, S.; et al. Non-coding RNAs in cardiovascular diseases: Diagnostic and therapeutic perspectives. Eur. Heart J. 2018, 39, 2704–2716. [Google Scholar] [CrossRef]
- Schober, A.; Maleki, S.S.; Nazari-Jahantigh, M. Regulatory Non-coding RNAs in Atherosclerosis. Handb. Exp. Pharmacol. 2020, 270, 463–492. [Google Scholar] [CrossRef]
- Michalik, K.M.; You, X.; Manavski, Y.; Doddaballapur, A.; Zörnig, M.; Braun, T.; John, D.; Ponomareva, Y.; Chen, W.; Uchida, S.; et al. Long Noncoding RNA MALAT1 Regulates Endothelial Cell Function and Vessel Growth. Circ. Res. 2014, 114, 1389–1397. [Google Scholar] [CrossRef]
- Kluge, M.A.; Fetterman, J.L.; Vita, J.A. Mitochondria and Endothelial Function. Circ. Res. 2013, 112, 1171–1188. [Google Scholar] [CrossRef]
- Rosca, M.G.; Hoppel, C.L. Mitochondria in heart failure. Cardiovasc. Res. 2010, 88, 40–50. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Li, S.; Zhang, Y.; Wang, M.; Li, X.; Liu, S.; Xu, D.; Bao, Y.; Jia, P.; Wu, N.; et al. The lncRNA Malat1 regulates microvascular-lar function after myocardial infarction in mice via miR-26b-5p/Mfn1 axis-mediated mitochondrial dynamics. Redox Biol. 2021, 41, 101910. [Google Scholar] [CrossRef] [PubMed]
- Lugus, J.J.; Ngoh, G.A.; Bachschmid, M.M.; Walsh, K. Mitofusins are required for angiogenic function and modulate different signaling pathways in cultured endothelial cells. J. Mol. Cell. Cardiol. 2011, 51, 885–893. [Google Scholar] [CrossRef] [PubMed]
- Qin, W.-W.; Xin, Z.-L.; Wang, H.-Q.; Wang, K.-P.; Li, X.-Y.; Wang, X. Inhibiting lncRNA ROR suppresses growth, migration and angiogenesis in microvascular endothelial cells by up-regulating miR-26. Eur. Rev. Med. Pharmacol. Sci. 2018, 22, 7985–7993. [Google Scholar] [PubMed]
- Liao, J.; He, Q.; Li, M.; Chen, Y.; Liu, Y.; Wang, J. LncRNA MIAT: Myocardial infarction associated and more. Gene 2016, 578, 158–161. [Google Scholar] [CrossRef]
- Yan, B.; Yao, J.; Liu, J.Y.; Li, X.M.; Wang, X.Q.; Li, Y.J.; Tao, Z.F.; Song, Y.C.; Chen, Q.; Jiang, Q. lncRNA-MIAT regulates microvascular dysfunction by functioning as a competing endogenous RNA. Circ. Res. 2015, 116, 1143–1156. [Google Scholar]
- Zachary, I. VEGF signalling: Integration and multi-tasking in endothelial cell biology. Biochem. Soc. Trans. 2003, 31, 1171–1177. [Google Scholar] [CrossRef]
- Fish, J.E.; Santoro, M.M.; Morton, S.U.; Yu, S.; Yeh, R.-F.; Wythe, J.D.; Ivey, K.N.; Bruneau, B.G.; Stainier, D.Y.R.; Srivastava, D. miR-126 Regulates Angiogenic Signaling and Vascular Integrity. Dev. Cell 2008, 15, 272–284. [Google Scholar] [CrossRef]
- Nicoli, S.; Standley, C.; Walker, P.; Hurlstone, A.; Fogarty, K.E.; Lawson, N.D. MicroRNA-mediated integration of haemodynamics and VEGF signalling during angiogenesis. Nature 2010, 464, 1196–1200. [Google Scholar] [CrossRef]
- Yang, H.H.; Chen, Y.; Gao, C.Y.; Cui, Z.T.; Yao, J.M. Protective Effects of MicroRNA-126 on Human Cardiac Microvascular Endothelial Cells Against Hypoxia/Reoxygenation-Induced Injury and Inflammatory Response by Activating PI3K/Akt/eNOS Signaling Pathway. Cell. Physiol. Biochem. 2017, 42, 506–518. [Google Scholar] [CrossRef]
- Chen, L.; Wang, J.; Wang, B.; Yang, J.; Gong, Z.; Zhao, X.; Zhang, C.; Du, K. MiR-126 inhibits vascular endothelial cell apoptosis through targeting PI3K/Akt signaling. Ann. Hematol. 2016, 95, 365–374. [Google Scholar] [CrossRef] [PubMed]
- Li, H.-Y.; Zhao, X.; Liu, Y.-Z.; Meng, Z.; Wang, D.; Yang, F.; Shi, Q.-W. Plasma MicroRNA-126-5p is associated with the Complexity and Severity of Coronary Artery Disease in Patients with Stable Angina Pectoris. Cell. Physiol. Biochem. 2016, 39, 837–846. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Yang, H.; Li, W.-J.; Liu, Y.-H. LncRNA MALAT1 Promotes OGD-Induced Apoptosis of Brain Microvascular Endothelial Cells by sponging miR-126 to Repress PI3K/Akt Signaling Pathway. Neurochem. Res. 2020, 45, 2091–2099. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, M.F.; Zhu, H.; Millard, R.W.; Fan, G.-C. Exosomes Function in Pro- and Anti-Angiogenesis. Curr. Angiogenes 2013, 2, 54–59. [Google Scholar] [CrossRef]
- Beheshti, E.H.; Grau, G.E.R. Extracellular vesicles and microvascular pathology: Decoding the active dialogue. Microcirculation 2019, 26, e12485. [Google Scholar] [CrossRef]
- Shu, Z.; Tan, J.; Miao, Y.; Zhang, Q. The role of microvesicles containing microRNAs in vascular endothelial dysfunction. J. Cell. Mol. Med. 2019, 23, 7933–7945. [Google Scholar] [CrossRef]
- Zernecke, A.; Bidzhekov, K.; Noels, H.; Shagdarsuren, E.; Gan, L.; Denecke, B.; Hristov, M.; Köppel, T.; Jahantigh, M.N.; Lutgens, E.; et al. Delivery of MicroRNA-126 by Apoptotic Bodies Induces CXCL12-Dependent Vascular Protection. Sci. Signal. 2009, 2, ra81. [Google Scholar] [CrossRef]
- Givertz, M.M.; Slawsky, M.T.; Moraes, D.L.; McIntyre, K.M.; Colucci, W.S. Noninvasive determination of pulmonary artery wedge pressure in patients with chronic heart failure. Am. J. Cardiol. 2001, 87, 1213–1215. [Google Scholar] [CrossRef]
- Graziani, F.; Varone, F.; Crea, F.; Richeldi, L. Treating heart failure with preserved ejection fraction: Learning from pulmonary fibrosis. Eur. J. Heart Fail. 2018, 20, 1385–1391. [Google Scholar] [CrossRef]
| Modality | Agent | Pros | Cons |
|---|---|---|---|
| Transthoracic Doppler echocardiography | Adenosine/Dipyridamole | Easily accessible. No radiation exposure. | Need previous rule-out of obstructive CAD. Operator-dependent. |
| Myocardial contrast echocardiography | Echocardiographic contrast substance | No radiation exposure. Assessment of global perfusion. | Lacking availability of standardized commercial software. Operator-dependent. |
| Positron emission tomography (PET) | Adenosine tracer (15O-H2O, 13 Nammonia, 82 R(b) | Reference standard of non-invasive methods. Assessment of global perfusion at the same time. | Difficult availability. Expensive. Radiation exposure. Need previous rule-out of obstructive CAD. |
| Cardiac Magnetic Resonance (CMR) | Adenosine/Regadenoson Gadolinium-based substances | No radiation exposure. Assessment of global perfusion. Used in the setting of obstructive CAD and structural heart disease. | Difficult availability. Expensive. Nonlinear relationship of tissue contrast concentration and MR signal intensity. Need of specific protocol. |
| Computed Tomography (CT) | Adenosine/Regadenoson Iodine-based contrast agent | Assessment of global perfusion at the same time. Used in the setting of obstructive CAD. | Need of further validation. Radiation exposure. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rocco, E.; Grimaldi, M.C.; Maino, A.; Cappannoli, L.; Pedicino, D.; Liuzzo, G.; Biasucci, L.M. Advances and Challenges in Biomarkers Use for Coronary Microvascular Dysfunction: From Bench to Clinical Practice. J. Clin. Med. 2022, 11, 2055. https://doi.org/10.3390/jcm11072055
Rocco E, Grimaldi MC, Maino A, Cappannoli L, Pedicino D, Liuzzo G, Biasucci LM. Advances and Challenges in Biomarkers Use for Coronary Microvascular Dysfunction: From Bench to Clinical Practice. Journal of Clinical Medicine. 2022; 11(7):2055. https://doi.org/10.3390/jcm11072055
Chicago/Turabian StyleRocco, Erica, Maria Chiara Grimaldi, Alessandro Maino, Luigi Cappannoli, Daniela Pedicino, Giovanna Liuzzo, and Luigi Marzio Biasucci. 2022. "Advances and Challenges in Biomarkers Use for Coronary Microvascular Dysfunction: From Bench to Clinical Practice" Journal of Clinical Medicine 11, no. 7: 2055. https://doi.org/10.3390/jcm11072055
APA StyleRocco, E., Grimaldi, M. C., Maino, A., Cappannoli, L., Pedicino, D., Liuzzo, G., & Biasucci, L. M. (2022). Advances and Challenges in Biomarkers Use for Coronary Microvascular Dysfunction: From Bench to Clinical Practice. Journal of Clinical Medicine, 11(7), 2055. https://doi.org/10.3390/jcm11072055






