The Fate of Antibiotic Impregnated Cement Space in Treatment for Forefoot Osteomyelitis
Abstract
:1. Introduction
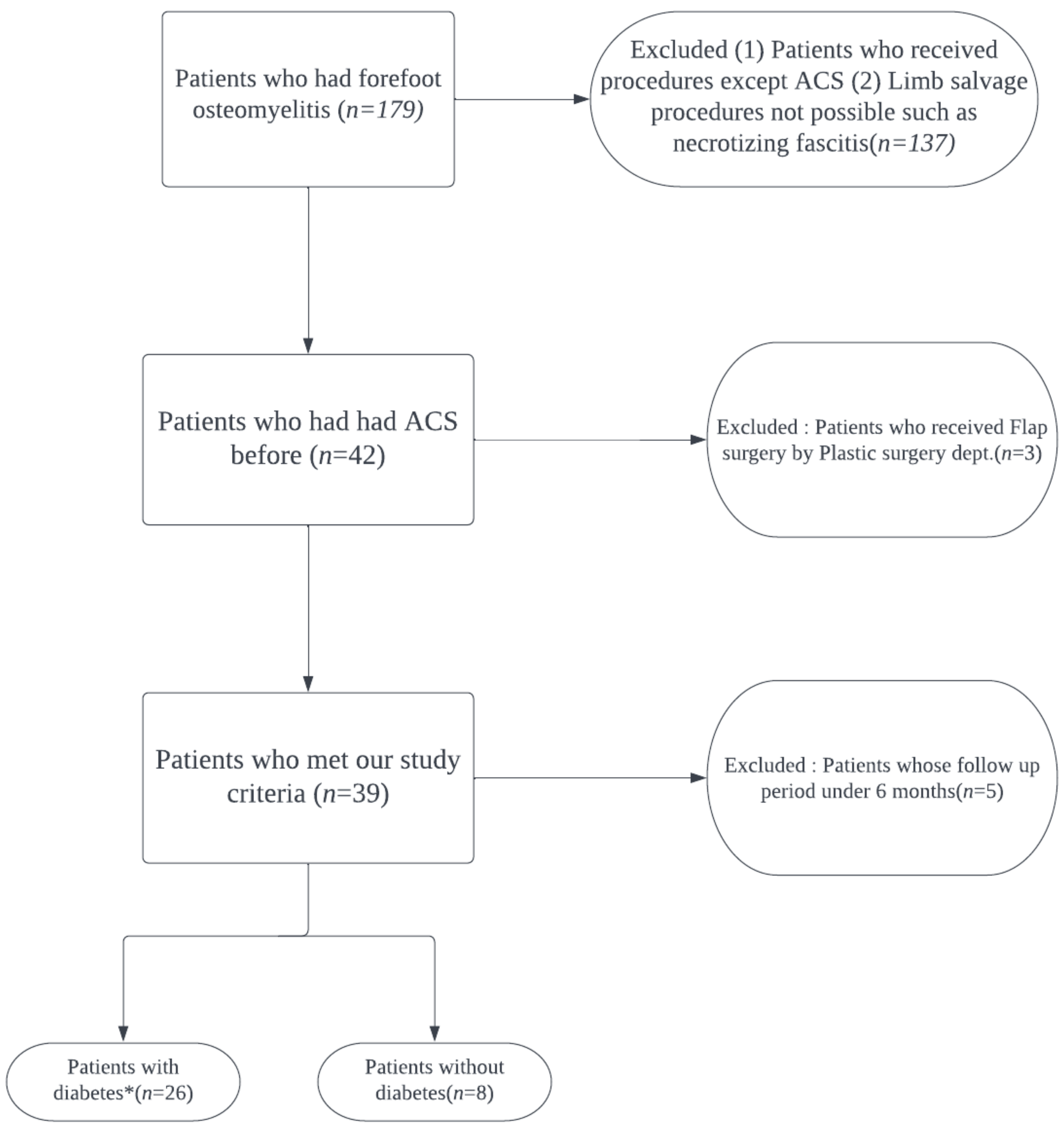
2. Materials and Methods
2.1. Study Subjects
2.2. Preoperative Evaluations
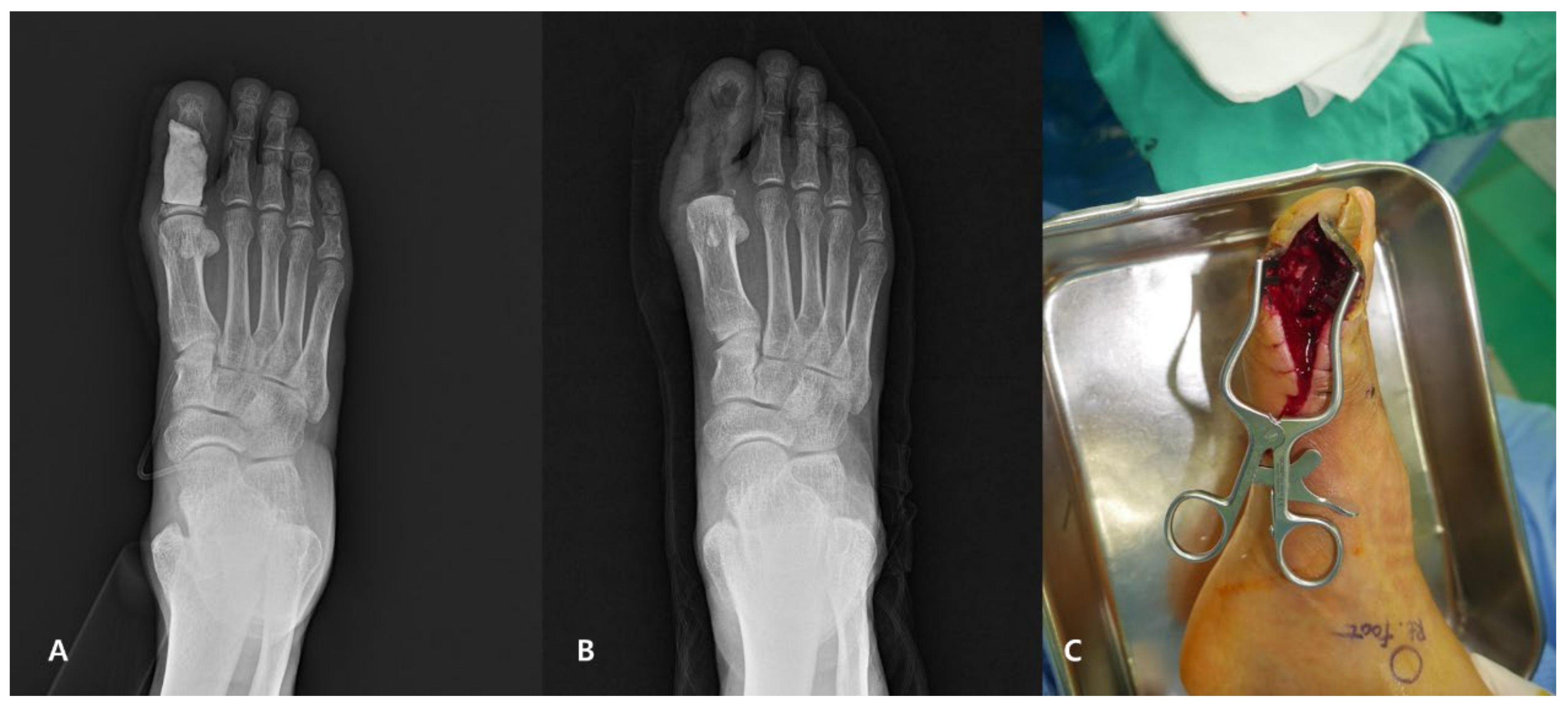
2.3. Surgical Technique
2.4. Outcome Assessments
2.5. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kim, Y.K.; Lee, H.S.; Seo, S.G.; Park, S.-H.; Boedijono, D. Comments on the Article “Results of Simple Conservative Treatment of Midfoot Charcot Arthropathy”: In Reply. Clin. Orthop. Surg. 2020, 12, 269–270. [Google Scholar] [CrossRef] [PubMed]
- Lipsky, B.A. Medical treatment of diabetic foot infections. Clin. Infect. Dis. 2004, 39, S104–S114. [Google Scholar] [CrossRef] [PubMed]
- Giurato, L.; Meloni, M.; Izzo, V.; Uccioli, L. Osteomyelitis in diabetic foot: A comprehensive overview. World J. Diabetes 2017, 8, 135. [Google Scholar] [CrossRef] [PubMed]
- Aragón-Sánchez, J.; Lázaro-Martínez, J.L.; Alvaro-Afonso, F.J.; Molinés-Barroso, R. Conservative Surgery of Diabetic Forefoot Osteomyelitis: How Can I Operate on This Patient without Amputation? Int. J. Low Extrem. Wounds 2015, 14, 108–131. [Google Scholar] [CrossRef]
- Kandemir, Ö.; Akbay, E.; Şahin, E.; Milcan, A.; Gen, R. Risk factors for infection of the diabetic foot with multi-antibiotic resistant microorganisms. J. Infect. 2007, 54, 439–445. [Google Scholar] [CrossRef]
- Berendt, A.; Peters, E.; Bakker, K.; Embil, J.; Eneroth, M.; Hinchliffe, R.; Jeffcoate, W.; Lipsky, B.; Senneville, E.; Teh, J. Diabetic foot osteomyelitis: A progress report on diagnosis and a systematic review of treatment. Diabetes/Metab. Res. Rev. 2008, 24, S145–S161. [Google Scholar] [CrossRef]
- Jeong, J.J.; Lee, H.S.; Choi, Y.R.; Kim, S.W.; Seo, J.H. Surgical treatment of non-diabetic chronic osteomyelitis involving the foot and ankle. Foot & Ankle Int. 2012, 33, 128–132. [Google Scholar]
- Dalla Paola, L.; Carone, A.; Morisi, C.; Cardillo, S.; Pattavina, M. Conservative surgical treatment of infected ulceration of the first metatarsophalangeal joint with osteomyelitis in diabetic patients. J. Foot Ankle Surg. 2015, 54, 536–540. [Google Scholar] [CrossRef]
- Aragón-Sánchez, J.; Lázaro-Martínez, J.L.; Campillo-Vilorio, N.; Quintana-Marrero, Y.; Hernández-Herrero, M.J. Controversies regarding radiological changes and variables predicting amputation in a surgical series of diabetic foot osteomyelitis. Foot Ankle Surg. 2012, 18, 233–236. [Google Scholar] [CrossRef]
- Lázaro-Martínez, J.L.; García-Madrid, M.; García-Álvarez, Y.; Álvaro-Afonso, F.J.; Sanz-Corbalán, I.; García-Morales, E. Conservative surgery for chronic diabetic foot osteomyelitis: Procedures and recommendations. J. Clin. Orthop. Trauma 2021, 16, 86–98. [Google Scholar] [CrossRef]
- Schmitt, D.R.; Killen, C.; Murphy, M.; Perry, M.; Romano, J.; Brown, N. The Impact of Antibiotic-Loaded Bone Cement on Antibiotic Resistance in Periprosthetic Knee Infections. Clin. Orthop. Surg. 2020, 12, 318–323. [Google Scholar] [CrossRef] [PubMed]
- Park, S.-J.; Cho, Y.; Lee, S.W.; Woo, H.-Y.; Lim, S.E. In Vitro Study Evaluating the Antimicrobial Activity of Vancomycin-Impregnated Cement Stored at Room Temperature in Methicillin-Resistant Staphylococcus aureus. J. Foot Ankle Surg. 2018, 22, 38–43. [Google Scholar]
- Schade, V.L.; Roukis, T.S. The role of polymethylmethacrylate antibiotic–loaded cement in addition to debridement for the treatment of soft tissue and osseous infections of the foot and ankle. J. Foot Ankle Surg. 2010, 49, 55–62. [Google Scholar] [CrossRef] [PubMed]
- York, B.; Cha, J.; Dao, A.; Gane, S.; Policinski, I.; Rahman, M. Diagnosis: Chronic osteomyelitis. Eplasty 2014, 14, ic8. [Google Scholar] [PubMed]
- Marais, L.; Ferreira, N.; Aldous, C.; Le Roux, T.L. The classification of chronic osteomyelitis. SA Orthop. J. 2014, 13, 22–28. [Google Scholar]
- Cierny, G., III. A clinical staging system for adult osteomyelitis. Contemp. Orthop. 1985, 10, 17–37. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Álvaro-Afonso, F.J.; Lázaro-Martínez, J.L.; García-Morales, E.; García-Álvarez, Y.; Sanz-Corbalán, I.; Molines-Barroso, R.J. Cortical disruption is the most reliable and accurate plain radiographic sign in the diagnosis of diabetic foot osteomyelitis. Diabet Med. 2019, 36, 258–259. [Google Scholar] [CrossRef]
- Malone, M.; Erasmus, A.; Schwarzer, S.; Lau, N.S.; Ahmad, M.; Dickson, H.G. Utilisation of the 2019 IWGDF diabetic foot infection guidelines to benchmark practice and improve the delivery of care in persons with diabetic foot infections. J. Foot Ankle Res. 2021, 14, 10. [Google Scholar] [CrossRef]
- Mantey, I.; Hill, R.; Foster, A.; Wilson, S.; Wade, J.; Edmonds, M. Infection of foot ulcers with Staphylococcus aureus associated with increased mortality in diabetic patients. Commun. Dis. Public Health 2000, 3, 288–290. [Google Scholar]
- Senneville, E.; Lombart, A.; Beltrand, E.; Valette, M.; Legout, L.; Cazaubiel, M.; Yazdanpanah, Y.; Fontaine, P. Outcome of diabetic foot osteomyelitis treated nonsurgically: A retrospective cohort study. Diabetes Care 2008, 31, 637–642. [Google Scholar] [CrossRef] [Green Version]
- Elmarsafi, T.; Oliver, N.G.; Steinberg, J.S.; Evans, K.K.; Attinger, C.E.; Kim, P.J. Long-term outcomes of permanent cement spacers in the infected foot. J. Foot Ankle Surg. 2017, 56, 287–290. [Google Scholar] [CrossRef] [PubMed]
- Krause, F.G.; deVries, G.; Meakin, C.; Kalla, T.P.; Younger, A.S. Outcome of transmetatarsal amputations in diabetics using antibiotic beads. Foot Ankle Int. 2009, 30, 486–493. [Google Scholar] [CrossRef] [PubMed]
- Niazi, N.S.; Drampalos, E.; Morrissey, N.; Jahangir, N.; Wee, A.; Pillai, A. Adjuvant antibiotic loaded bio composite in the management of diabetic foot osteomyelitis—A multicentre study. Foot 2019, 39, 22–27. [Google Scholar] [CrossRef] [PubMed]
- Melamed, E.A.; Peled, E. Antibiotic impregnated cement spacer for salvage of diabetic osteomyelitis. Foot Ankle Int. 2012, 33, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Fraimow, H.S. Systemic antimicrobial therapy in osteomyelitis. Semin. Plast. Surg. 2009, 23, 90–99. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wong, M.W.; Hui, M. Development of gentamicin resistance after gentamicin-PMMA beads for treatment of foot osteomyelitis: Report of two cases. Foot Ankle Int. 2005, 26, 1093–1095. [Google Scholar] [CrossRef]
- Marson, B.; Deshmukh, S.; Grindlay, D.; Ollivere, B.; Scammell, B. A systematic review of local antibiotic devices used to improve wound healing following the surgical management of foot infections in diabetics. Bone Joint J. 2018, 100, 1409–1415. [Google Scholar] [CrossRef]
| Demographic Data (n = 35) | |||
|---|---|---|---|
| Variable | Patients with DM (n = 27) | Patients without DM (n = 8) | p-Value |
| Age (yr) | 0.83 | ||
| Median | 64 | 60 | |
| IQR | 60.0–75.3 | 51–67 | |
| Sex | 1 | ||
| Male | 21 | 6 | |
| Female | 6 | 2 | |
| Follow-up (months) | 0.28 | ||
| Median | 16.7 | 34 | |
| IQR | 15.6–39.8 | 17–48.6 | |
| BMI (kg/m2) | 0.529 | ||
| Median | 22.7 | 22.7 | |
| IQR | 21.4–24 | 21.3–27.3 | |
| Comorbidity | |||
| Hypertension | 14 | 2 | 0.244 |
| Renal disease which requires hemodialysis | 6 | 0 | 0.299 |
| Cardiac diseases | 7 | 0 | 0.16 |
| Location | 0.313 | ||
| Phalanx (joint not involved) | 15 | 7 | |
| joint involvement | 10 | 1 | |
| Metatarsals (joint not involved) | 2 | 0 | |
| Maintenance of ACS | 0.858 | ||
| Remained | 15 | 6 | |
| Removed | 12 | 2 |
| The Microbiological Findings from Cultures at Operation Room | |
|---|---|
| Organisms | Number |
| Staphylococcus epidermidis | 2 |
| Staphylococcus aureus | 8 |
| MRSA | 4 |
| Enterococcus faecalis | 4 |
| Enterobacter cleacae | 1 |
| Pseudomonas aeruginosa | 4 |
| Proteus mirabilis | 2 |
| Streptococcus species | 3 |
| Serratia marcescens | 1 |
| Klebsiella pneumoniae | 1 |
| Morganella morganii | 2 |
| no growth | 3 |
| multiple organisms | 0 |
| Total | 35 |
| Comparing between ACS Retension Group and ACS Removed Group | |||
|---|---|---|---|
| Variable | ACS Retension Group (N = 21) | ACS Removed Group (N = 14) | p-Value |
| Age (yr) | 60.00 (52.5–68) | 58.00 (53.50–73.25) | 0.96 |
| mean Ankle brachial index (ABI) | 1.16 (1.08–1.22) | 1.096 (1.13–1.17) | 0.503 |
| mean Toe brachial index (TBI) | 0.77 (0.48–0.89) | 1.01 (0.93) | 0.03 |
| Numbers of previous surgeries before ACS insertion | 0 (0–2.5) | 0 (0.5–2) | 0.594 |
| Features of Why and Where ACS Were Failed, Time to Failure (%) | |
|---|---|
| Variables | |
| Retained ACS cases | 21 (60%) |
| ACS removed cases | 14 (40%) |
| Additional amputation needed | 1 (7.1%) |
| Reasons why ACS were removed | |
| infection not controlled | 8 (57.1%) |
| wound problem (such as skin penetraion, not healed op scar) | 5 (35.7%) |
| difficulty in walking | 1 (7.1%) |
| Location of ACS being failed (total number of each group) | |
| phalanx | 6 (out of 22, 27%) |
| MPJ (joint involved) | 5 (out of 11, 45%) |
| metatarsals | 2 (out of 2, 100%) |
| Other * | 1 (out of 1, 100%) |
| Time to failure in ACS removed groups (days) | |
| mean | 348 |
| Range | 2 to 2332 |
| Category of Antibiotics Prescribed to the Failure Group | |
|---|---|
| Variable | Number |
| Cephalosporins (first to third generation) | 5 |
| Piperacillin/Tazobactam | 1 |
| Floroquinolones | 4 |
| Vancomycin | 1 |
| Tigecycline | 1 |
| Carbapenem | 2 |
| Total | 14 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Woo, I.; Park, J.; Seok, H.; Kim, T.-g.; Moon, J.S.; Chung, S.M.; Park, C.H. The Fate of Antibiotic Impregnated Cement Space in Treatment for Forefoot Osteomyelitis. J. Clin. Med. 2022, 11, 1976. https://doi.org/10.3390/jcm11071976
Woo I, Park J, Seok H, Kim T-g, Moon JS, Chung SM, Park CH. The Fate of Antibiotic Impregnated Cement Space in Treatment for Forefoot Osteomyelitis. Journal of Clinical Medicine. 2022; 11(7):1976. https://doi.org/10.3390/jcm11071976
Chicago/Turabian StyleWoo, Inha, Jeongjin Park, Hyungyu Seok, Tae-gon Kim, Jun Sung Moon, Seung Min Chung, and Chul Hyun Park. 2022. "The Fate of Antibiotic Impregnated Cement Space in Treatment for Forefoot Osteomyelitis" Journal of Clinical Medicine 11, no. 7: 1976. https://doi.org/10.3390/jcm11071976
APA StyleWoo, I., Park, J., Seok, H., Kim, T.-g., Moon, J. S., Chung, S. M., & Park, C. H. (2022). The Fate of Antibiotic Impregnated Cement Space in Treatment for Forefoot Osteomyelitis. Journal of Clinical Medicine, 11(7), 1976. https://doi.org/10.3390/jcm11071976







