Prognostic Utility of a New Risk Stratification Protocol for Secondary Prevention in Patients Attending Cardiac Rehabilitation
Abstract
1. Introduction
2. Materials and Methods
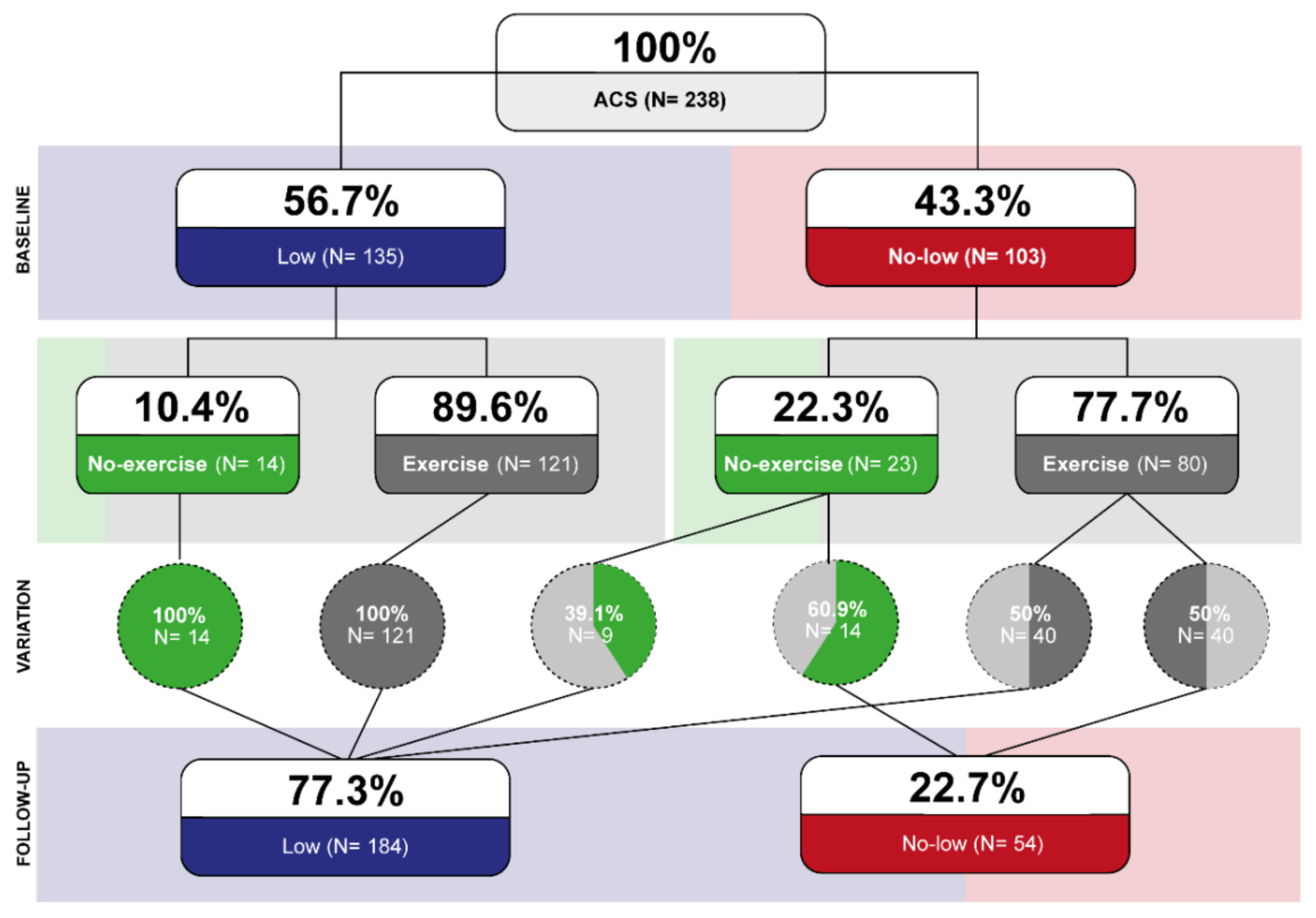
2.1. Study Design, Population and Study Variables
2.2. Cardiac Rehabilitation Program
2.3. Cardiac Risk Stratification Process
2.4. Aims and Endpoint
2.5. Ethics
2.6. Statistics
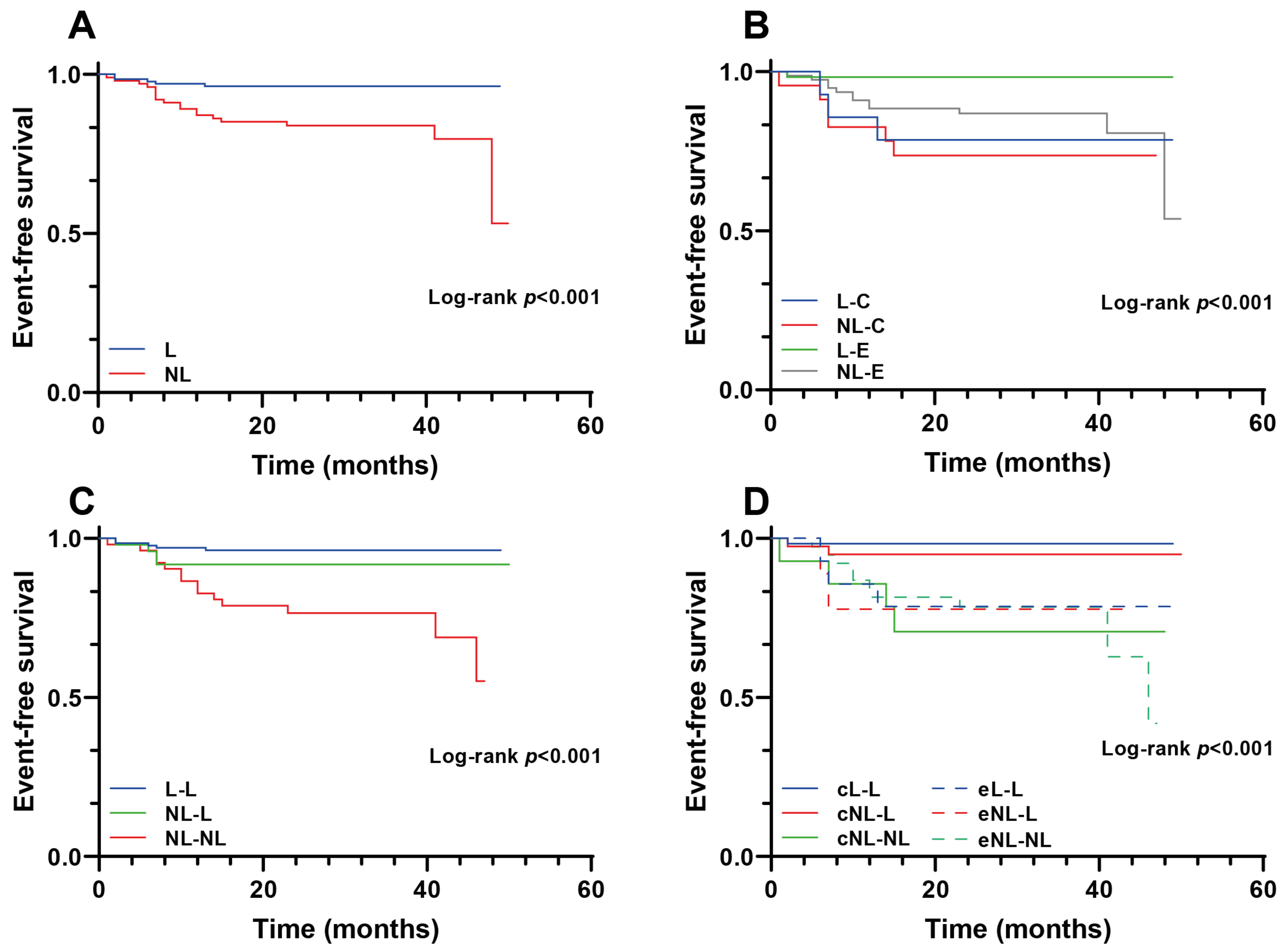
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ab Khan, M.; Hashim, M.J.; Mustafa, H.; Baniyas, M.Y.; Al Suwaidi, S.K.B.M.; AlKatheeri, R.; Alblooshi, F.M.K.; Almatrooshi, M.E.A.H.; Alzaabi, M.E.H.; Al Darmaki, R.S.; et al. Global Epidemiology of Ischemic Heart Disease: Results from the Global Burden of Disease Study. Cureus 2020, 12, e9349. [Google Scholar]
- Collet, J.P.; Thiele, H.; Barbato, E.; Barthélémy, O.; Bauersachs, J.; Bhatt, D.L.; Karia, N. 2020 ESC Guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation. Eur. Heart J. 2020, 42, 289–1367. [Google Scholar] [CrossRef] [PubMed]
- D’Ascenzo, F.; Biondi-Zoccai, G.; Moretti, C.; Bollati, M.; Omedè, P.; Sciuto, F.; Presutti, D.G.; Modena, M.G.; Gasparini, M.; Reed, M.; et al. TIMI, GRACE and alternative risk scores in Acute Coronary Syndromes: A meta-analysis of 40 derivation studies on 216,552 patients and of 42 validation studies on 31,625 patients. Contemp. Clin. Trials 2012, 33, 507–514. [Google Scholar] [CrossRef] [PubMed]
- Timóteo, A.T.; Rosa, S.A.; Nogueira, M.A.; Belo, A.; Ferreira, R.C. Validação externa do score de risco ProACS para estratificação de risco de doentes com síndrome coronária aguda. Rev. Port. Cardiol. 2016, 35, 323–328. [Google Scholar] [CrossRef]
- Tang, W.H.W.; Topol, E.J.; Fan, Y.; Wu, Y.; Cho, L.; Stevenson, C.; Ellis, S.G.; Hazen, S.L. Prognostic value of estimated functional capacity incremental to cardiac biomarkers in stable cardiac patients. J. Am. Heart Assoc. 2014, 3, e000960. [Google Scholar] [CrossRef]
- McNeer, J.F.; Margolis, J.R.; Lee, K.L.; Kisslo, A.J.; Peter, R.H.; Kong, Y.; Behar, V.S.; Wallace, A.G.; McCants, C.B.; Rosati, A.R. The role of the exercise test in the evaluation of patients for ischemic heart disease. Circulation 1978, 57, 64–70. [Google Scholar] [CrossRef]
- Winnige, P.; Filakova, K.; Hnatiak, J.; Dosbaba, F.; Bocek, O.; Pepera, G.; Papathanasiou, J.; Batalik, L.; Grace, S.L. Validity and Reliability of the Cardiac Rehabilitation Barriers Scale in the Czech Republic (CRBS-CZE): Determination of Key Barriers in East-Central Europe. Int. J. Environ. Res. Public Health 2021, 18, 13113. [Google Scholar] [CrossRef]
- Velasco, J.A.; Cosín, J.; Maroto, J.M.; Muñiz, J.; Casasnovas, J.A.; Plaza, I.; Abadal, L.T. Guías de práctica clínica de la Sociedad Española de Cardiología en prevención cardiovascular y rehabilitación cardíaca. Rev. Española Cardiol. 2000, 53, 1095–1120. [Google Scholar] [CrossRef]
- Procedimiento Rehabilitación Cardiaca—Sociedad Española de Cardiología. Available online: https://secardiologia.es/institucional/reuniones-institucionales/sec-calidad/sec-excelente/procedimientos/8722-procedimiento-rehabilitacion-cardiaca (accessed on 24 March 2022).
- American Association of Cardiovascular & Pulmonary Rehabilitation. Guidelines for Cardiac Rehabilitation Programs; American Association of Cardiovascular & Pulmonary Rehabilitation: West Palm Beach, FL, USA, 2016; p. 359. [Google Scholar]
- Da Silva, A.K.F.; da Costa de Rezende Barbosa, M.P.; Bernardo, A.F.B.; Vanderlei, F.M.; Pacagnelli, F.L.; Vanderlei, L.C.M. Cardiac risk stratification in cardiac rehabilitation programs: A review of protocols. Rev. Bras. Cir. Cardiovasc. 2014, 29, 255. [Google Scholar] [CrossRef]
- López-Aguilera, J.; Casado-Adam, P.; Heredia-Torres, M.A.; Mazuelos-Bellido, F. Effectiveness of Cardiac Rehabilitation in Increased Left Ventricle Ejection Fraction and Cardiovascular Secondary Prevention. Int. J. Clin. Cardiol. 2015, 2, 065. [Google Scholar] [CrossRef]
- Wang, Y.; Chien, C.W.; Xu, Y.; Tung, T.H. Effect of Exercise-Based Cardiac Rehabilitation on Left Ventricular Function in Asian Patients with Acute Myocardial Infarction after Percutaneous Coronary Intervention: A Meta-Analysis of Randomized Controlled Trials. Healthcare 2021, 9, 774. [Google Scholar] [CrossRef] [PubMed]
- Ibanez, B.; James, S.; Agewall, S.; Antunes, M.J.; Bucciarelli-Ducci, C.; Bueno, H.; Caforio, A.L.P.; Crea, F.; Goudevenos, A.J.; Halvorsen, S.; et al. 2017 ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation. Eur. Heart J. 2018, 39, 119–177. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Yan, B.P.; Nichol, M.B.; Tomlinson, B.; Lee, V.W. 2015 ESC Guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevationTask Force for the Management of Acute Coronary Syndromes in Patients Presenting without Persistent ST-Segment Elevation of the European Society of Cardiology (ESC). Eur. Heart J. 2016, 37, 267–315. [Google Scholar]
- Cabrera-Aguilera, I.; Ivern, C.; Badosa, N.; Marco, E.; Salas-Medina, L.; Mojón, D.; Vicente, M.; Llagostera, M.; Farré, N.; Ruiz-Bustillo, S. Impact of and Reasons for Not Performing Exercise Training After an Acute Coronary Syndrome in the Setting of an Interdisciplinary Cardiac Rehabilitation Program: Results From a Risk-Op- Acute Coronary Syndrome Ambispective Registry. Front. Physiol. 2021, 12, 2109. [Google Scholar] [CrossRef]
- Bellet, R.N.; Francis, R.L.; Jacob, J.S.; Healy, K.M.; Bartlett, H.J.; Adams, L.; Morris, N.R. Fast-track equivalent to traditional cardiac rehabilitation? Pilot study outcome. Eur. J. Physioterapy 2016, 18, 126–136. [Google Scholar] [CrossRef][Green Version]
- Vittinghoff, E.; McCulloch, C.E. Relaxing the rule of ten events per variable in logistic and Cox regression. Am. J. Epidemiol. 2007, 165, 710–718. [Google Scholar] [CrossRef]
- Avezum, A.; Makdisse, M.; Spencer, F.; Gore, J.M.; Fox, K.A.; Montalescot, G.; Grace Investigators. Impact of age on management and outcome of acute coronary syndrome: Observations from the global registry of acute coronary events (GRACE). Am. Heart J. 2005, 149, 67–73. [Google Scholar] [CrossRef]
- Antman, E.M.; Cohen, M.; Bernink, P.J.L.M. The TIMI risk score for unstable angina/non-ST elevation MI: A method for prognostication and therapeutic decision making. JAMA 2000, 284, 835–842. [Google Scholar] [CrossRef]
- Morrow, D.A.; Antman, E.M.; Charlesworth, A.; Cairns, R.; Murphy, S.A.; de Lemos, J.A.; Giugliano, R.P.; McCabe, C.H.; Braunwald, E. TIMI Risk Score for ST-Elevation Myocardial Infarction: A Convenient, Bedside, Clinical Score for Risk Assessment at Presentation. Circulation 2000, 102, 2031–2037. [Google Scholar] [CrossRef]
- Fox, A.A.K.; Dabbous, O.H.; Goldberg, R.J.; Pieper, K.S.; Eagle, A.K.; Van de Werf, F.; Avezum, A.; Goodman, S.G.; Flather, M.D.; Anderson, F.A., Jr.; et al. Prediction of risk of death and myocardial infarction in the six months after presentation with acute coronary syndrome: Prospective multinational observational study (GRACE). BMJ 2006, 333, 1091–1094. [Google Scholar] [CrossRef]
- Timmer, J.R.; Hoekstra, M.; Nijsten, M.W.; van der Horst, I.C.; Ottervanger, J.P.; Slingerland, R.J.; Dambrink, J.-H.E.; Bilo, H.J.; Zijlstra, F.; Hof, A.W.V. Prognostic value of admission glycosylated hemoglobin and glucose in nondiabetic patients with ST-segment-elevation myocardial infarction treated with percutaneous coronary intervention. Circulation 2011, 124, 704–711. [Google Scholar] [CrossRef] [PubMed]
- Waters, D.D.; Arsenault, B.J. Predicting Prognosis in Acute Coronary Syndromes: Makeover Time for TIMI and GRACE? Can. J. Cardiol. 2016, 32, 1290–1293. [Google Scholar] [CrossRef][Green Version]
- Pardaens, S.; Willems, A.-M.; Clays, E.; Baert, A.; Vanderheyden, M.; Verstreken, S.; Du Bois, I.; Vervloet, D.; De Sutter, J. The impact of drop-out in cardiac rehabilitation on outcome among coronary artery disease patients. Eur. J. Prev. Cardiol. 2017, 24, 1490–1497. [Google Scholar] [CrossRef] [PubMed]
- Kokkinos, P.; Faselis, C.; Myers, J.; Sui, X.; Zhang, J.; Blair, S.N. Age-specific exercise capacity threshold for mortality risk assessment in male veterans. Circulation 2014, 130, 653–658. [Google Scholar] [CrossRef] [PubMed]
- Morris, C.K.; Myers, J.; Froelicher, V.F.; Kawaguchi, T.; Ueshima, K.; Hideg, A. Nomogram based on metabolic equivalents and age for assessing aerobic exercise capacity in men. J. Am. Coll. Cardiol. 1993, 22, 175–182. [Google Scholar] [CrossRef]
- Sandercock, G.R.H.; Cardoso, F.; Almodhy, M.; Pepera, G. Cardiorespiratory fitness changes in patients receiving comprehensive outpatient cardiac rehabilitation in the UK: A multicentre study. Heart 2013, 99, 785–790. [Google Scholar] [CrossRef]
- Zhao, X.; Li, J.; Xian, Y.; Chen, J.; Gao, Z.; Qiao, S.; Yang, Y.; Gao, R.; Xu, B.; Yuan, J. Prognostic value of the GRACE discharge score for predicting the mortality of patients with stable coronary artery disease who underwent percutaneous coronary intervention. Catheter. Cardiovasc. Interv. 2020, 95, 550–557. [Google Scholar] [CrossRef]
- Smart, N.A.; King, N.; Lambert, J.D.; Pearson, M.J.; Campbell, J.; Risom, S.S.; Taylor, R.S. Exercise-based cardiac rehabilitation improves exercise capacity and health-related quality of life in people with atrial fibrillation: A systematic review and meta-analysis of randomised and non-randomised trials. Open Heart 2018, 5, e000880. [Google Scholar] [CrossRef]
- Budts, W.; Pieles, G.E.; Roos-Hesselink, J.W.; Sanz de la Garza, M.; D’Ascenzi, F.; Giannakoulas, G.; Papadakis, M. 2020 ESC Guidelines on sports cardiology and exercise in patients with cardiovascular disease. Eur. Heart J. 2021, 42, 17–96. [Google Scholar]
- McGregor, G.; Powell, R.; Kimani, P.; Underwood, M. Exercise-based cardiac rehabilitation for coronary heart disease. Cochrane Database Syst. Rev. 2016, 2016, CD001800. [Google Scholar]
- Niu, S.; Wang, F.; Yang, S.; Jin, Z.; Han, X.; Zou, S.; Guo, D.; Guo, C. Predictive value of cardiopulmonary fitness parameters in the prognosis of patients with acute coronary syndrome after percutaneous coronary intervention. J. Int. Med. Res. 2020, 48. [Google Scholar] [CrossRef] [PubMed]
| Characteristic/Variable | L (n = 135) | NL (n = 103) | p-Value |
|---|---|---|---|
| Anthropometric | |||
| Age (years) | 59.3 ± 10.2 | 63.3 ± 10.6 | 0.006 |
| Women | 19 (14.1) | 17 (16.5) | 0.604 |
| BMI (kg/m2) | 27.3 (24.7–29.3) | 26.9 (24.9–30.5) | 0.631 |
| Risk factors and comorbidities | |||
| Hypertension | 64 (47.4) | 56 (54.4) | 0.287 |
| Hyperlipidemia | 92 (68.1) | 65 (63.1) | 0.416 |
| Diabetes mellitus | 21 (15.6) | 28 (27.2) | 0.028 |
| Current smoker | 59 (43.7) | 33 (32.0) | 0.288 |
| Previous smoker > 1 year | 42 (31.1) | 35 (34.0) | |
| Previous smoker < 1 year | 5 (3.7) | 5 (4.9) | |
| COPD | 5 (3.7) | 10 (9.7) | 0.059 |
| Cerebrovascular disease | 3 (2.2) | 7 (6.8) | 0.081 |
| Peripheral vascular disease | 6 (4.4) | 3 (2.9) | 0.539 |
| Anemia | 13 (9.3) | 13 (12.6) | 0.464 |
| Chronic kidney disease | 3 (2.2) | 5 (4.9) | 0.264 |
| Diagnostics | |||
| STEMI | 60 (40.4) | 46 (44.7) | 0.684 |
| NSTEMI | 54 (40.0) | 37 (35.9) | |
| Unstable angina | 21 (15.6) | 20 (19.4) | |
| Previous ACS-MI | 17 (12.6) | 18 (17.5) | 0.292 |
| One vessel disease | 84 (62.2) | 51 (49.5) | 0.133 |
| Two vessels disease | 29 (21.5) | 26 (25.2) | |
| Three vessels disease | 17 (16.2) | 24 (23.3) | |
| Ejection fraction (%) | 60 (55.0–63.5) | 51 (43.5–60.0) | <0.001 |
| Exercise testing | |||
| METs | 10.3 (9.1–12.4) | 8.3 (6.6–9.8) | <0.001 |
| Maximum predicted heart rate (%) | 81 (73.0–89.0) | 79 (67.0–87.0) | 0.073 |
| Peak systolic blood pressure (mmHg) | 154 (142–173) | 148 (132–165) | 0.055 |
| Blood test | |||
| Glucose, mg/dL | 104 (94–124) | 116 (99–151) | 0.005 |
| Glycated hemoglobin, % | 5.6 (5.3–5.8) | 5.7 (5.4–6.7) | 0.007 |
| LDLc, mg/dL | 117 (90–136) | 107(81–136) | 0.323 |
| Outcomes | L (n = 135) | NL (n = 103) | p-Value |
|---|---|---|---|
| Follow-up METs | 11.8 (9.8–13.3) | 9.8 (7.9–11.4) | <0.001 |
| Relative increase in METs (%) | 7.6 (0.0–17.4) | 12.3 (2.6–27.4) | 0.019 |
| Absolute increase in METs | 0.8 (0.0–1.9) | 1.1 (0.2–2.1) | 0.173 |
| All causes re-admission | 27 (20.0) | 39 (37.9) | 0.002 |
| All causes death | 0 (0.0) | 2 (1.9) | 0.104 |
| Cardiac rehospitalization | 6 (4.4) | 19 (18.4) | <0.001 |
| Heart failure | 1 (0.7) | 3 (2.9) | 0.197 |
| Arrythmias | 2 (1.5) | 10 (9.7) | 0.004 |
| Revascularization | 4 (3.0) | 9 (8.7) | 0.052 |
| New ACS | 0 (0.0) | 7 (6.8) | 0.002 |
| Univariable HR (95% CI) | p-Value | Adjusted HR (95% CI) | p-Value | |
|---|---|---|---|---|
| No-low risk group | 4.32 (1.73–10.82) | 0.002 | 3.83 (1.51–9.68) | 0.005 |
| No-exercise group | 3.15 (1.39–7.15) | 0.006 | 2.52 (1.10–5.78) | 0.021 |
| Age (years) | 1.03 (0.99–1.07) | 0.103 | - | |
| Men | 1.22 (0.36–4.10) | 0.745 | - | |
| Mellitus diabetes | 1.40 (0.47–4.02) | 0.556 | - |
| Characteristic/Variable | L-C (n = 14) | NL-C (n = 23) | L-E (n = 121) | NL-E (n = 80) | p-Value |
|---|---|---|---|---|---|
| Anthropometric | |||||
| Age (years) | 61.4 ± 9.3 | 65.5 ± 12.1 | 59.8 ± 9.7 | 62.1 ± 10.1 | 0.005 |
| Women | 3 (21.4) | 5 (21.7) | 16 (13.2) | 12 (15.0) | 0.669 |
| BMI (kg/m2) | 26.7 (24.0–27.7) | 28.4 (23.5–31.2) | 27.3 (24.7–29.3) | 26.7 (24.8–30.5) | 0.830 |
| Risk factors and comorbidities | |||||
| Hypertension | 6 (42.9) | 16 (69.6) | 58 (47.9) | 40 (50.0) | 0.262 |
| Hyperlipidemia | 8 (57.1) | 13 (56.5) | 84(69.4) | 52 (65.0) | 0.557 |
| Diabetes mellitus | 1 (7.1) | 8 (34.8) | 20 (16.5) | 20 (25.0) | 0.088 |
| Current smoker | 9 (64.3) | 7 (30.4) | 50 (41.3) | 26 (32.5) | 0.478 |
| Previous smoker > 1 year | 3 (21.4) | 6 (26.1) | 39 (32.2) | 29 (36.3) | |
| Previous smoker < 1 year | 0 (0.0) | 1 (4.3) | 5 (4.1) | 4 (5.0) | |
| COPD | 1 (7.1) | 2 (8.7) | 4 (3.3) | 8 (10.0) | 0.269 |
| Cerebrovascular disease | 0 (0.0) | 2 (8.7) | 3 (2.5) | 5 (6.3) | 0.322 |
| Peripheral vascular disease | 1 (7.1) | 0 (0.0) | 5 (4.1) | 3 (3.8) | 0.710 |
| Anemia | 3 (21.4) | 4 (17.4) | 10 (8.3) | 9 (11.3) | 0.325 |
| Chronic kidney disease | 0 (0.0) | 2 (8.7) | 3 (2.5) | 3 (3.8) | 0.419 |
| CV family history | 3 (21.4) | 4 (17.4) | 26 (21.5) | 12 (15.0) | 0.702 |
| Sudden death family history | 0 (0.0) | 1 (4.3) | 6 (5.0) | 1 (1.3) | 0.457 |
| Diagnostics | |||||
| STEMI | 4 (28.6) | 9 (39.1) | 56 (46.3) | 37 (46.3) | 0.765 |
| NSTEMI | 7 (50.0) | 8 (34.8) | 47 (38.8) | 29 (36.3) | |
| Unstable Angina | 3 (21.4) | 6 (26.1) | 18 (14.9) | 14 (17.5) | |
| Previous ACS-MI | 3 (21.4) | 7 (30.4) | 14 (11.6) | 11 (13.8) | 0.109 |
| One vessel disease | 8 (57.1) | 7 (30.4) | 76 (62.8) | 44 (55.0) | 0.228 |
| Two vessels disease | 4 (28.6) | 7 (30.4) | 25 (20.7) | 19 (23.8) | |
| Three vessels disease | 2 (14.3) | 9 (39.1) | 15 (12.4) | 15 (18.8) | |
| Ejection fraction (%) | 62 (55.5–63.5) | 58 (51.0–60.0) | 60 (56.0–64.0) | 51 (41.0–60.0) | <0.001 |
| Exercise testing | |||||
| METs | 9.8 (8.5–11.8) | 6.8 (6.2–7.8) | 10.3 (9.3–12.6) | 8.5 (6.7–10.0) | <0.001 |
| Maximum predicted heart rate (%) | 80 (72.0–83.5) | 82 (71.0–87.0) | 81 (74.0–89.0) | 76 (65.5–85.5) | 0.088 |
| Peak systolic blood pressure (mmHg) | 155 (139–180) | 160 (133–183) | 154 (142–172) | 147 (132–161.5) | 0.103 |
| Blood test | |||||
| Glucose, mg/dL | 101 (87–112) | 126 (106–190) | 105 (95–124) | 116 (97–151) | 0.032 |
| Glycated hemoglobin, % | 5.6 (5.3–5.7) | 6.7 (5.7–8.3) | 5.6 (5.4–5.9) | 5.6 (5.4–6.5) | 0.016 |
| LDL, mg/dL | 115 (89–129) | 102 (69–110) | 118 (93–140) | 111 (84–139) | 0.127 |
| Outcomes | |||||
| Follow-up METs | 10.8 (8.3–12.2) | 8.1 (6.0–10.8) | 12.0 (9.8–13.3) | 9.8 (8.3–11.5) | <0.001 |
| Relative increase in METs (%) | 4.5 (−4.3–16.7) | 6.5 (−3.2–27.9) | 7.7 (0.8–17.6) | 14.3 (3.2–26.8) | 0.039 |
| Absolute increase in METs | 0.3 (−0.4–1.0) | 0.5 (−0.2–1.2) | 0.9 (0.0–1.9) | 1.3 (0.3–2.3) | 0.119 |
| Cardiac rehospitalization | 3 (21.4) | 6 (26.1) | 3 (2.5) | 13 (16.3) | <0.001 |
| All causes readmission | 7 (50.0) | 12 (52.2) | 20 (16.5) | 27 (33.8) | <0.001 |
| All causes death | 0 (0.0) | 1 (4.3) | 0 (0.0) | 1 (1.3) | 0.195 |
| Revascularization | 3 (21.4) | 3 (13.0) | 1 (0.8) | 6 (7.5) | 0.002 |
| New ACS | 0 (0.0) | 2 (8.7) | 0 (0.0) | 5 (6.3) | 0.020 |
| Outcomes | L-C (n = 14) | NL-C (n = 23) | L-E (n = 121) | NL-E (n = 80) | p-Value |
|---|---|---|---|---|---|
| Follow-up METs | 10.8 (8.3–12.2) | 8.1 (6.0–10.8) | 12.0 (9.8–13.3) | 9.8 (8.3–11.5) | <0.001 |
| Relative increase in METs (%) | 4.5 (−4.3–16.7) | 6.5 (−3.2–27.9) | 7.7 (0.8–17.6) | 14.3 (3.2–26.8) | 0.039 |
| Absolute increase in METs | 0.3 (−0.4–1.0) | 0.5 (−0.2–1.2) | 0.9 (0.0–1.9) | 1.3 (0.3–2.3) | 0.119 |
| All causes readmission | 7 (50.0) | 12 (52.2) | 20 (16.5) | 27 (33.8) | <0.001 |
| All causes death | 0 (0.0) | 1 (4.3) | 0 (0.0) | 1 (1.3) | 0.195 |
| Cardiac rehospitalization | 3 (21.4) | 6 (26.1) | 3 (2.5) | 13 (16.3) | <0.001 |
| Heart failure | 1 (7.1) | 1 (4.3) | 0 (0.0) | 2 (2.5) | 0.116 |
| Arrythmias | 0 (0.0) | 3 (13.0) | 2 (1.7) | 7 (8.8) | 0.029 |
| Revascularization | 3 (21.4) | 3 (13.0) | 1 (0.8) | 6 (7.5) | 0.002 |
| New ACS | 0 (0.0) | 2 (8.7) | 0 (0.0) | 5 (6.3) | 0.020 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cabrera-Aguilera, I.; Ivern, C.; Badosa, N.; Marco, E.; Duran, X.; Mojón, D.; Vicente, M.; Llagostera, M.; Farré, N.; Ruíz-Bustillo, S. Prognostic Utility of a New Risk Stratification Protocol for Secondary Prevention in Patients Attending Cardiac Rehabilitation. J. Clin. Med. 2022, 11, 1910. https://doi.org/10.3390/jcm11071910
Cabrera-Aguilera I, Ivern C, Badosa N, Marco E, Duran X, Mojón D, Vicente M, Llagostera M, Farré N, Ruíz-Bustillo S. Prognostic Utility of a New Risk Stratification Protocol for Secondary Prevention in Patients Attending Cardiac Rehabilitation. Journal of Clinical Medicine. 2022; 11(7):1910. https://doi.org/10.3390/jcm11071910
Chicago/Turabian StyleCabrera-Aguilera, Ignacio, Consolació Ivern, Neus Badosa, Ester Marco, Xavier Duran, Diana Mojón, Miren Vicente, Marc Llagostera, Nuria Farré, and Sonia Ruíz-Bustillo. 2022. "Prognostic Utility of a New Risk Stratification Protocol for Secondary Prevention in Patients Attending Cardiac Rehabilitation" Journal of Clinical Medicine 11, no. 7: 1910. https://doi.org/10.3390/jcm11071910
APA StyleCabrera-Aguilera, I., Ivern, C., Badosa, N., Marco, E., Duran, X., Mojón, D., Vicente, M., Llagostera, M., Farré, N., & Ruíz-Bustillo, S. (2022). Prognostic Utility of a New Risk Stratification Protocol for Secondary Prevention in Patients Attending Cardiac Rehabilitation. Journal of Clinical Medicine, 11(7), 1910. https://doi.org/10.3390/jcm11071910






