Obstructive Sleep Apnea and Atrial Fibrillation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Search Strategy
2.2. Selection Process
2.3. Quality Assessment
2.4. Definitions
2.5. Statistical Analysis
3. Results
3.1. Search Results and Characteristics of the Studies
3.2. Quality of the Studies
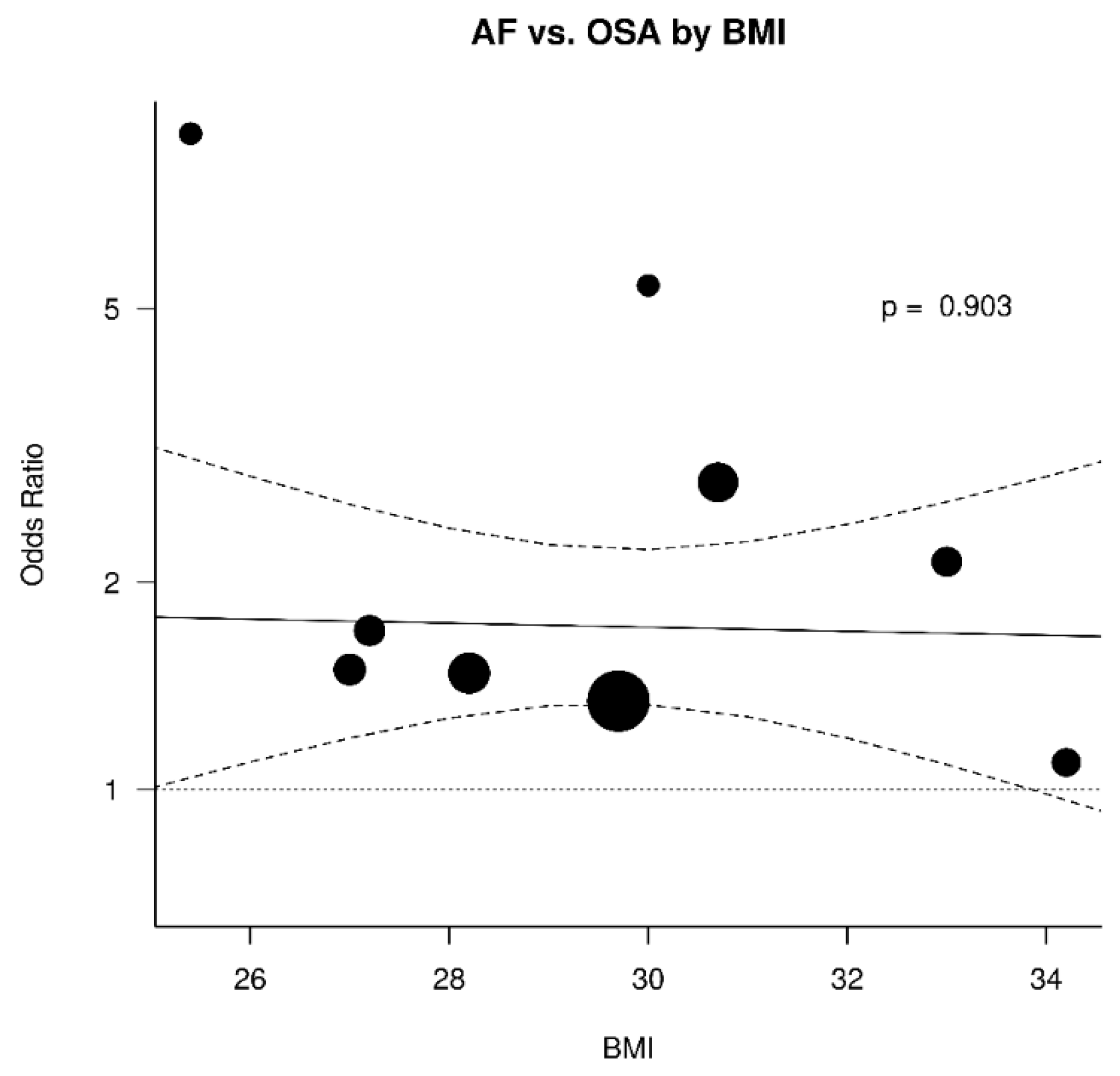
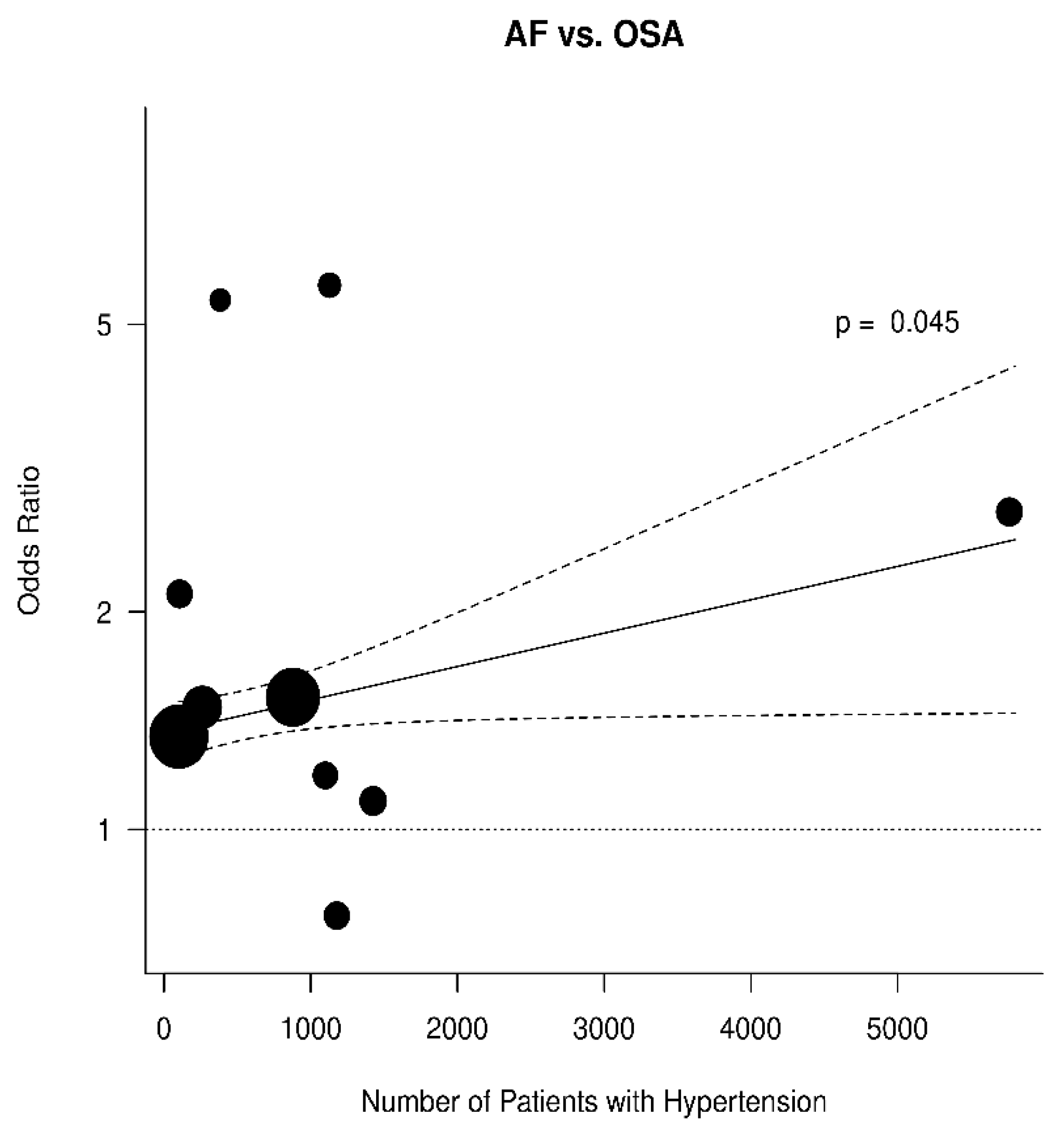
3.3. Relation between OSA and AF
3.4. Discussion
3.5. Limitations
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Conflicts of Interest
Appendix A
| Item | M | SD | |
|---|---|---|---|
| 1 | Study hypothesis/aim/objective described? | 0.91 | 0.10 |
| 2 | Main outcomes described in the introduction or methods? | 0.77 | 0.24 |
| 3 | Participant characteristics described? | 0.87 | 0.14 |
| 4 | Contacted participants representative? | 0.61 | 0.20 |
| 5 | Prepared participants representative? | 0.55 | 0.29 |
| 6 | Participants recruited from the same population? | 0.68 | 0.25 |
| 7 | Participants recruited over the same time? | 0.83 | 0.11 |
| 8 | Measures and experimental tasks described? | 0.89 | 0.11 |
| 9 | Main outcome measures valid and reliable? | 1.0 | 0.0 |
| 10 | Task engagement assessed? | 0.80 | 0.14 |
| 11 | Confounders described and controlled for? | 1.6 | 0.40 |
| 12 | Statistical tests appropriate? | 0.94 | 0.1 |
| 13 | Main findings described? | 1.0 | 0.0 |
| 14 | Estimates of the random variability in data main outcomes? | 0.44 | 0.36 |
| 15 | Probability values reported? | 0.56 | 0.31 |
| 16 | Withdrawals and drop-outs reported? | 0.55 | 0.30 |
| 17 | Data dredging made clear? | 0.46 | 0.36 |
| 18 | Sufficient power analysis provided? | 0.62 | 0.44 |
References
- Calvo, D.; Filgueiras-Rama, D.; Jalife, J. Mechanisms and drug development in atrial Fibrillation. Pharmacol. Rev. 2018, 70, 505–525. [Google Scholar] [CrossRef] [Green Version]
- January, C.T.; Wann, L.S.; Alpert, J.S.; Calkins, H.; Cigarroa, J.E.; Cleveland, J.C., Jr.; Conti, J.B.; Ellinor, P.T.; Ezekowitz, M.D.; Field, M.E.; et al. 2014 AHA/ACC/HRS guideline for the management of patients with atrial fibrillation: A report of the American College of Cardiology/American heart Association task force on practice guidelines and the heart rhythm society. Circulation 2014, 130, e199–e267. [Google Scholar] [CrossRef]
- Shamloo, A.S.; Dagres, N.; Arya, A.; Hindricks, G. Atrial fibrillation: A review of modifiable risk factors and preventive strategies. Rom. J. Intern. Med. 2019, 57, 99–109. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Drugs for atrial fibrillation. Med. Lett. Drugs Ther. 2019, 61, 137–144.
- Bennaghmouch, N.; de Veer, A.; Mahmoodi, B.K.; Jofre-Bonet, M.; Lip, G.Y.H.; Bode, K.; Ten Berg, J. Economic evaluation of the use of non-vitamin K oral anticoagulants in patients with atrial fibrillation on antiplatelet therapy: A modelling analysis using the healthcare system in the Netherlands. Eur. Heart J. Qual. Care Clin. Outcomes 2019, 5, 127–135. [Google Scholar] [CrossRef]
- Karnik, A.A.; Gopal, D.M.; Ko, D.; Benjamin, E.J.; Helm, R.H. Epidemiology of atrial fibrillation and heart failure: A growing and important problem. Cardiol. Clin. 2019, 37, 119–129. [Google Scholar] [CrossRef]
- Westerman, S.; Wenger, N. Gender differences in atrial fibrillation: A review of epidemiology, management, and outcomes. Curr. Cardiol. Rev. 2019, 15, 136–144. [Google Scholar] [CrossRef]
- Albrecht, M.; Koolhaas, C.M.; Schoufour, J.D.; van Rooij, F.J.; Kavousi, M.; Ikram, M.A.; Franco, O.H. Physical activity types and atrial fibrillation risk in the middle-aged and elderly: The Rotterdam study. Eur. J. Prev. Cardiol. 2018, 25, 1316–1323. [Google Scholar] [CrossRef] [Green Version]
- Alreshidan, M.; Roberts, H.G.; Rankin, J.S.; Wei, L.M.; Badhwar, V. Current approach to surgical ablation for atrial fibrillation. Semin. Thorac. Cardiovasc. Surg. 2019, 31, 141–145. [Google Scholar] [CrossRef]
- Bell, D.S.H.; Goncalves, E. Atrial fibrillation and type 2 diabetes: Prevalence, etiology, pathophysiology and effect of anti-diabetic therapies. Diabetes Obes. Metab. 2019, 21, 210–217. [Google Scholar] [CrossRef]
- De Sensi, F.; Costantino, S.; Limbruno, U.; Paneni, F. Atrial fibrillation in the cardiometabolic patient. Minerva Med. 2019, 110, 157–167. [Google Scholar] [CrossRef] [PubMed]
- Dobrev, D.; Aguilar, M.; Heijman, J.; Guichard, J.B.; Nattel, S. Postoperative atrial fibrillation: Mechanisms, manifestations and management. Nat. Rev. Cardiol. 2019, 16, 417–436. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Lin, X.; Ma, X.; Tao, J.; Zou, R.; Yang, S.; Liu, H.; Hua, P. Biatrial versus isolated left atrial ablation in atrial fibrillation: A systematic review and meta-analysis. BioMed Res. Int. 2018, 2018, 3651212. [Google Scholar] [CrossRef] [PubMed]
- Panchal, G.; Mahmood, M.; Lip, G.Y.H. Revisiting the risks of incident atrial fibrillation: A narrative review. Part 2. Kardiol. Pol. 2019, 77, 515–524. [Google Scholar] [CrossRef] [Green Version]
- Serban, R.C.; Scridon, A. Data linking diabetes mellitus and atrial fibrillation—How strong is the evidence? From epidemiology and pathophysiology to therapeutic implications. Can. J. Cardiol. 2018, 34, 1492–1502. [Google Scholar] [CrossRef] [PubMed]
- Sugumar, H.; Nanayakkara, S.; Prabhu, S.; Voskoboinik, A.; Kaye, D.M.; Ling, L.-H.; Kistler, P.M. Pathophysiology of atrial fibrillation and heart failure: Dangerous interactions. Cardiol. Clin. 2019, 37, 131–138. [Google Scholar] [CrossRef]
- Thomas, D.; Christ, T.; Fabritz, L.; Goette, A.; Hammwohner, M.; Heijman, J.; Kockskaemper, J.; Linz, D.; Odening, K.E.; Schweizer, P.A.; et al. German cardiac society working group on cellular electrophysiology state-of-the-art paper: Impact of molecular mechanisms on clinical arrhythmia management. Clin. Res. Cardiol. 2019, 108, 577–599. [Google Scholar] [CrossRef] [Green Version]
- Zimetbaum, P. Atrial fibrillation. Ann. Intern. Med. 2017, 166, ITC33–ITC48. [Google Scholar] [CrossRef]
- Delesie, M.; Knaepen, L.; Verbraecken, J.; Weytjens, K.; Dendale, P.; Heidbuchel, H.; Desteghe, L. Cardiorespiratory polygraphy for detection of obstructive sleep apnea in patients with atrial fibrillation. Front. Cardiovasc. Med. 2021, 8, 758548. [Google Scholar] [CrossRef]
- Young, T.; Evans, L.; Finn, L.; Palta, M. Estimation of the clinically diagnosed proportion of sleep apnea syndrome in middle-aged men and women. Sleep 1997, 20, 705–706. [Google Scholar] [CrossRef]
- Gami, A.S.; Pressman, G.; Caples, S.M.; Kanagala, R.; Gard, J.J.; Davison, D.E.; Malouf, J.F.; Ammash, N.M.; Friedman, P.A.; Somers, V.K. Association of atrial fibrillation and obstructive sleep apnea. Circulation 2004, 110, 364–367. [Google Scholar] [CrossRef] [PubMed]
- Sin, D.D.; Fitzgerald, F.; Parker, J.D.; Newton, G.; Floras, J.S.; Bradley, T.D. Risk factors for central and obstructive sleep apnea in 450 men and women with congestive heart failure. Am. J. Respir. Crit. Care Med. 1999, 160, 1101–1106. [Google Scholar] [CrossRef] [PubMed]
- Huang, B.; Liu, H.; Scherlag, B.J.; Sun, L.; Xing, S.; Xu, J.; Luo, M.; Guo, Y.; Cao, G.; Jiang, H. Atrial fibrillation in obstructive sleep apnea: Neural mechanisms and emerging therapies. Trends Cardiovasc. Med. 2020, 31, 127–132. [Google Scholar] [CrossRef] [PubMed]
- Linz, D.; Baumert, M.; Desteghe, L.; Kadhim, K.; Vernooy, K.; Kalman, J.M.; Dobrev, D.; Arzt, M.; Sastry, M.; Crijns, H.J.G.M.; et al. Nightly sleep apnea severity in patients with atrial fibrillation: Potential applications of long-term sleep apnea monitoring. Int. J. Cardiol. Heart Vasc. 2019, 24, 100424. [Google Scholar] [CrossRef]
- Perger, E.; Pengo, M.F.; Lombardi, C. Hypertension and atrial fibrillation in obstructive sleep apnea: Is it a menopause issue? Maturitas 2019, 124, 32–34. [Google Scholar] [CrossRef] [PubMed]
- Shantha, G.; Pelosi, F.; Morady, F. Relationship between obstructive sleep apnoea and AF. Arrhythm Electrophysiol. Rev. 2019, 8, 180–183. [Google Scholar] [CrossRef]
- Khalil, M.; Power, N.; Graham, E.; Deschenes, S.S.; Schmitz, N. The association between sleep and diabetes outcomes—A systematic review. Diabetes Res. Clin. Pract. 2020, 161, 108035. [Google Scholar] [CrossRef]
- Rundo, J.V. Obstructive sleep apnea basics. Clev. Clin. J. Med. 2019, 86 (Suppl. 1), 2–9. [Google Scholar] [CrossRef] [Green Version]
- Tetta, C.; Moula, A.I.; Matteucci, F.; Parise, O.; Maesen, B.; Johnson, D.; La Meir, M.; Gelsomino, S. Association between atrial fibrillation and Helicobacter pylori. Clin. Res. Cardiol. 2019, 108, 730–740. [Google Scholar] [CrossRef] [Green Version]
- Almeneessier, A.S.; Alasousi, N.; Sharif, M.M.; Pandi-Perumal, S.R.; Hersi, A.S.; BaHammam, A.S. Prevalence and Predictors of Arrhythmia in Patients with Obstructive Sleep Apnea. Sleep Sci. 2017, 10, 142–146. [Google Scholar] [CrossRef]
- Shamseer, L.; Moher, D.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M.; Shekelle, P.; Stewart, L.A. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015: Elaboration and explanation. BMJ 2015, 350, g7647. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hutton, B.; Salanti, G.; Caldwell, D.M.; Chaimani, A.; Schmid, C.H.; Cameron, C.; Ioannidis, J.P.A.; Straus, S.; Thorlund, K.; Jansen, J.P.; et al. The PRISMA extension statement for reporting of systematic reviews incorporating network meta-analyses of health care interventions: Checklist and explanations. Ann. Intern. Med. 2015, 162, 777–784. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sterne, J.A.; Hernan, M.A.; Reeves, B.C.; Savovic, J.; Berkman, N.D.; Viswanathan, M.; Henry, D.; Altman, D.G.; Ansari, M.T.; Boutron, I.; et al. ROBINS-I: A tool for assessing risk of bias in non-randomised studies of interventions. BMJ 2016, 355, i4919. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cochrane Handbook for Systematic Reviews of Interventions Version 6.0 Cochrane. 2019. Available online: www.training.cochrane.org/handbook (accessed on 13 August 2021).
- Downs, S.H.; Black, N. The feasibility of creating a checklist for the assessment of the methodological quality both of randomised and non-randomised studies of health care interventions. J. Epidemiol. Community Health 1998, 52, 377–384. [Google Scholar] [CrossRef] [Green Version]
- McHugh, M.L. Interrater reliability: The kappa statistic. Biochem. Med. 2012, 22, 276–282. [Google Scholar] [CrossRef]
- Park, J.G.; Ramar, K.; Olson, E.J. Updates on definition, consequences, and management of obstructive sleep apnea. Mayo Clin Proc. 2011, 86, 549–554, quiz 54–55. [Google Scholar] [CrossRef] [Green Version]
- Atrial Fibrillation Follow-up Investigation of Rhythm Management (AFFIRM) Investigators. A comparison of rate control and rhythm control in patients with atrial fibrillation. N. Eng. J. Med. 2002, 347, 1825–1833. [Google Scholar] [CrossRef]
- Adderley, N.J.; Subramanian, A.; Toulis, K.; Gokhale, K.; Taverner, T.; Hanif, W.; Haroon, S.; Thomas, G.N.; Sainsbury, C.; Tahrani, A.b.d.A.; et al. Obstructive sleep apnea, a risk factor for cardiovascular and microvascular disease in patients with type 2 diabetes: Findings from a population-based cohort study. Diabetes Care 2020, 43, 1868–1877. [Google Scholar] [CrossRef]
- Anzai, T.; Grandinetti, A.; Katz, A.R.; Hurwitz, E.L.; Wu, Y.Y.; Masaki, K. Association between central sleep apnea and atrial fibrillation/flutter in Japanese-American men: The Kuakini Honolulu Heart Program (HHP) and Honolulu-Asia Aging Study (HAAS). J. Electrocardiol. 2020, 61, 10–17. [Google Scholar] [CrossRef]
- Cadby, G.; McArdle, N.; Briffa, T.; Hillman, D.R.; Simpson, L.; Knuiman, M.; Hung, J. Severity of OSA is an independent predictor of incident atrial fibrillation hospitalization in a large sleep-clinic cohort. Chest 2015, 148, 945–952. [Google Scholar] [CrossRef] [Green Version]
- Gali, B.; Glasgow, A.E.; Greason, K.L.; Johnson, R.L.; Albright, R.C.; Habermann, E.B. Postoperative outcomes of patients with obstructive sleep apnea undergoing cardiac surgery. Ann. Thorac. Surg. 2020, 110, 1324–1332. [Google Scholar] [CrossRef] [PubMed]
- Gami, A.S.; Hodge, D.O.; Herges, R.M.; Olson, E.J.; Nykodym, J.; Kara, T.; Somers, V.K. Obstructive sleep apnea, obesity, and the risk of incident atrial fibrillation. J. Am. Coll. Cardiol. 2007, 49, 565–571. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ganga, H.V.; Nair, S.U.; Puppala, V.K.; Miller, W.L. Risk of new-onset atrial fibrillation in elderly patients with the overlap syndrome: A retrospective cohort study. J. Geriatr. Cardiol. 2013, 10, 129–134. [Google Scholar] [PubMed]
- Hendrikx, T.; Sundqvist, M.; Sandstrom, H.; Sahlin, C.; Rohani, M.; Al-Khalili, F.; Hornsten, R.; Blomberg, A.; Wester, P.; Rosenqvist, M.; et al. Atrial fibrillation among patients under investigation for suspected obstructive sleep apnea. PLoS ONE. 2017, 12, e0171575. [Google Scholar] [CrossRef] [Green Version]
- Korostovtseva, L.S.; Zvartau, N.E.; Rotar, O.P.; Sviryaev, Y.V.; Konradi, A.O. Predictors of heart rhythm disturbances in hypertensive obese patients with obstructive sleep apnea. J. Geriatr. Cardiol. 2017, 14, 553–562. [Google Scholar] [PubMed]
- Kwon, Y.; Gadi, S.; Shah, N.R.; Stout, C.; Blackwell, J.N.; Cho, Y.; Koene, R.J.; Mehta, N.; Mazimba, S.; Darby, A.E.; et al. Atrial fibrillation and objective sleep quality by slow wave sleep. J. Atr. Fibrillat. 2018, 11, 2031. [Google Scholar] [CrossRef]
- May, A.M.; Blackwell, T.; Stone, P.H.; Stone, K.L.; Cawthon, P.M.; Sauer, W.H.; Varosy, P.D.; Redline, S.; Mehra, R. Central sleep-disordered breathing predicts incident atrial fibrillation in older men. Am. J. Respir. Crit. Care Med. 2016, 193, 783–791. [Google Scholar] [CrossRef] [Green Version]
- Mehra, R.; Benjamin, E.J.; Shahar, E.; Gottlieb, D.J.; Nawabit, R.; Kirchner, H.L.; Sahadevan, J.; Redline, S. Association of nocturnal arrhythmias with sleep-disordered breathing: The Sleep Heart Health Study. Am. J. Respir. Crit. Care Med. 2006, 173, 910–916. [Google Scholar] [CrossRef]
- Mehra, R.; Stone, K.L.; Varosy, P.D.; Hoffman, A.R.; Marcus, G.M.; Blackwell, T.; Ibrahim, O.A.; Salem, R.; Redline, S. Nocturnal arrhythmias across a spectrum of obstructive and central sleep-disordered breathing in older men: Outcomes of sleep disorders in older men (MrOS sleep) study. Arch. Intern. Med. 2009, 169, 1147–1155. [Google Scholar] [CrossRef] [Green Version]
- Porthan, K.M.; Melin, J.H.; Kupila, J.T.; Venho, K.K.; Partinen, M.M. Prevalence of sleep apnea syndrome in lone atrial fibrillation: A case-control study. Chest 2004, 125, 879–885. [Google Scholar] [CrossRef]
- Selim, B.J.; Koo, B.B.; Qin, L.; Jeon, S.; Won, C.; Redeker, N.S.; Lampert, R.J.; Concato, J.P.; Bravata, D.M.; Ferguson, J.; et al. The association between nocturnal cardiac arrhythmias and sleep-disordered breathing: The DREAM study. J. Clin. Sleep Med. 2016, 12, 829–837. [Google Scholar] [CrossRef]
- Tanigawa, T.; Yamagishi, K.; Sakurai, S.; Muraki, I.; Noda, H.; Shimamoto, T.; Iso, H. Arterial oxygen desaturation during sleep and atrial fibrillation. Heart 2006, 92, 1854–1855. [Google Scholar] [CrossRef] [PubMed]
- Tung, P.; Levitzky, Y.S.; Wang, R.; Weng, J.; Quan, S.F.; Gottlieb, D.J.; Rueschman, M.; Punjabi, N.M.; Mehra, R.; Bertisch, S.; et al. Obstructive and central sleep apnea and the risk of incident atrial fibrillation in a community cohort of men and women. J. Am. Heart Assoc. 2017, 6, e004500. [Google Scholar] [CrossRef] [Green Version]
- Wang, S.; Cui, H.; Meng, L.; Wu, R.; Tang, B.; Zhu, C.; Yu, Q.; Huang, X.; Wang, S. Effect of septal myectomy on obstructive sleep apnoea syndrome in patients with hypertrophic obstructive cardiomyopathy. Heart Lung Circ. 2020, 29, 1366–1374. [Google Scholar] [CrossRef] [PubMed]
- Wong, J.K.; Maxwell, B.G.; Kushida, C.A.; Sainani, K.L.; Lobato, R.L.; Woo, Y.J.; Pearl, R.G. Obstructive sleep apnea is an independent predictor of postoperative atrial fibrillation in cardiac surgery. J. Cardiothorac. Vasc. Anesth. 2015, 29, 1140–1147. [Google Scholar] [CrossRef] [Green Version]
- Xu, H.; Wang, J.; Yuan, J.; Hu, F.; Yang, W.; Guo, C.; Luo, X.; Liu, R.; Cui, J.; Gao, X.; et al. Implication of apnea-hypopnea index, a measure of obstructive sleep apnea severity, for atrial fibrillation in patients with hypertrophic cardiomyopathy. J. Am. Heart Assoc. 2020, 9, e015013. [Google Scholar] [CrossRef]
- Zhang, L.; Hou, Y.; Po, S.S. Obstructive sleep apnoea and atrial fibrillation. Arrhythmia Electrophysiol. Rev. 2015, 4, 14–18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yeghiazarians, Y.; Jneid, H.; Tietjens, J.R.; Redline, S.; Brown, D.L.; El-Sherif, N.; Mehra, R.; Bozkurt, B.; Ndumele, C.E.; Somers, V.K.; et al. Obstructive Sleep Apnea and cardiovascular disease: A scientific statement from the American heart association. Circulation 2021, 144, e56–e67. [Google Scholar] [CrossRef] [PubMed]
- Lavergne, F.; Morin, L.; Armitstead, J.; Benjafield, A.; Richards, G.; Woehrle, H. Atrial fibrillation and sleep-disordered breathing. J. Thorac. Dis. 2015, 7, E575–E584. [Google Scholar] [PubMed]
- Marulanda-Londono, E.; Chaturvedi, S. The interplay between obstructive sleep apnea and atrial fibrillation. Front. Neurol. 2017, 8, 668. [Google Scholar] [CrossRef] [Green Version]
- Chao, T.F.; Liu, C.J.; Chen, S.J.; Wang, K.L.; Lin, Y.J.; Chang, S.L.; Lo, L.-W.; Hu, Y.-F.; Tuan, T.-C.; Chen, T.-J.; et al. Incidence and risk of atrial fibrillation in sleep-disordered breathing without coexistent systemic disease. Circ. J. 2014, 78, 2182–2187. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bangash, A.; Wajid, F.; Poolacherla, R.; Mim, F.K.; Rutkofsky, I.H. Obstructive sleep apnea and hypertension: A review of the relationship and pathogenic association. Cureus 2020, 12, e8241. [Google Scholar] [CrossRef] [PubMed]
- Gumprecht, J.; Domek, M.; Lip, G.Y.H.; Shantsila, A. Invited review: Hypertension and atrial fibrillation: Epidemiology, pathophysiology, and implications for management. J. Hum. Hypertens. 2019, 33, 824–836. [Google Scholar] [CrossRef] [PubMed]
- Wingerter, R.; Steiger, N.; Burrows, A.; Estes, N.A.M., III. Impact of lifestyle modification on atrial fibrillation. Am. J. Cardiol. 2020, 125, 289–297. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z. Aging, arterial stiffness, and hypertension. Hypertension 2015, 65, 252–256. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bseikri, M.; Lo, L.; Guilleminault, C. Obstructive sleep apnea: A syndrome from childhood to old-age. Pulmon. Ther. 2015, 1, 31–42. [Google Scholar] [CrossRef]
- Zathar, Z.; Karunatilleke, A.; Fawzy, A.M.; Lip, G.Y.H. Atrial fibrillation in older people: Concepts and controversies. Front. Med. 2019, 6, 175. [Google Scholar] [CrossRef]
- Coccina, F.; Pierdomenico, A.M.; De Rosa, M.; Cuccurullo, C.; Pierdomenico, S.D. Association of clinic and ambulatory blood pressure with new-onset atrial fibrillation: A meta-analysis of observational studies. J. Clin. Hypertens. 2021, 23, 1104–1111. [Google Scholar] [CrossRef]
- Lau, Y.F.; Yiu, K.H.; Siu, C.W.; Tse, H.F. Hypertension and atrial fibrillation: Epidemiology, pathophysiology and therapeutic implications. J. Hum. Hypertens. 2011, 26, 563–569. [Google Scholar] [CrossRef] [Green Version]
- Tietjens, J.R.; Claman, D.; Kezirian, E.J.; De Marco, T.; Mirzayan, A.; Sadroonri, B.; Goldberg, A.N.; Long, C.; Gerstenfeld, E.P.; Yeghiazarians, Y. Obstructive sleep apnea in cardiovascular disease: A review of the literature and proposed multidisciplinary clinical management strategy. J. Am. Heart Assoc. 2019, 8, e010440. [Google Scholar] [CrossRef] [Green Version]
- Linz, B.; Hohl, M.; Lang, L.; Wong, D.W.L.; Nickel, A.G.; De La Torre, C.; Sticht, C.; Wirth, K.; Boor, P.; Maack, C.; et al. Repeated exposure to transient obstructive sleep apnea-related conditions causes an atrial fibrillation substrate in a chronic rat model. Heart Rhythm. 2021, 18, 455–464. [Google Scholar] [CrossRef] [PubMed]
- Hohl, M.; Linz, B.; Böhm, M.; Linz, D. Obstructive sleep apnea and atrial arrhythmogenesis. Curr. Cardiol. Rev. 2014, 10, 362–368. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Christou, K.; Moulas, A.N.; Pastaka, C.; Gourgoulianis, K.I. Antioxidant capacity in obstructive sleep apnea patients. Sleep Med. 2003, 4, 225–228. [Google Scholar] [CrossRef]
- Haissaguerre, M.; Hocini, M.; Denis, A.; Shah, A.J.; Komatsu, Y.; Yamashita, S.; Daly, M.; Amraoui, S.; Zellerhoff, S.; Picat, M.-Q.; et al. Driver domains in persistent atrial fibrillation. Circulation 2014, 130, 530–538. [Google Scholar] [CrossRef] [Green Version]
- Sanchez, A.M.; Germany, R.; Lozier, M.R.; Schweitzer, M.D.; Kosseifi, S.; Anand, R. Central sleep apnea and atrial fibrillation: A review on pathophysiological mechanisms and therapeutic implications. IJC Heart Vasculat. 2020, 30, 100527. [Google Scholar] [CrossRef]
- Li, X.; Zhou, X.; Xu, X.; Dai, J.; Chen, C.; Ma, L.; Li, J.; Mao, W.; Zhu, M. Effects of continuous positive airway pressure treatment in obstructive sleep apnea patients with atrial fibrillation: A meta-analysis. Medicine 2021, 100, e25438. [Google Scholar] [CrossRef] [PubMed]
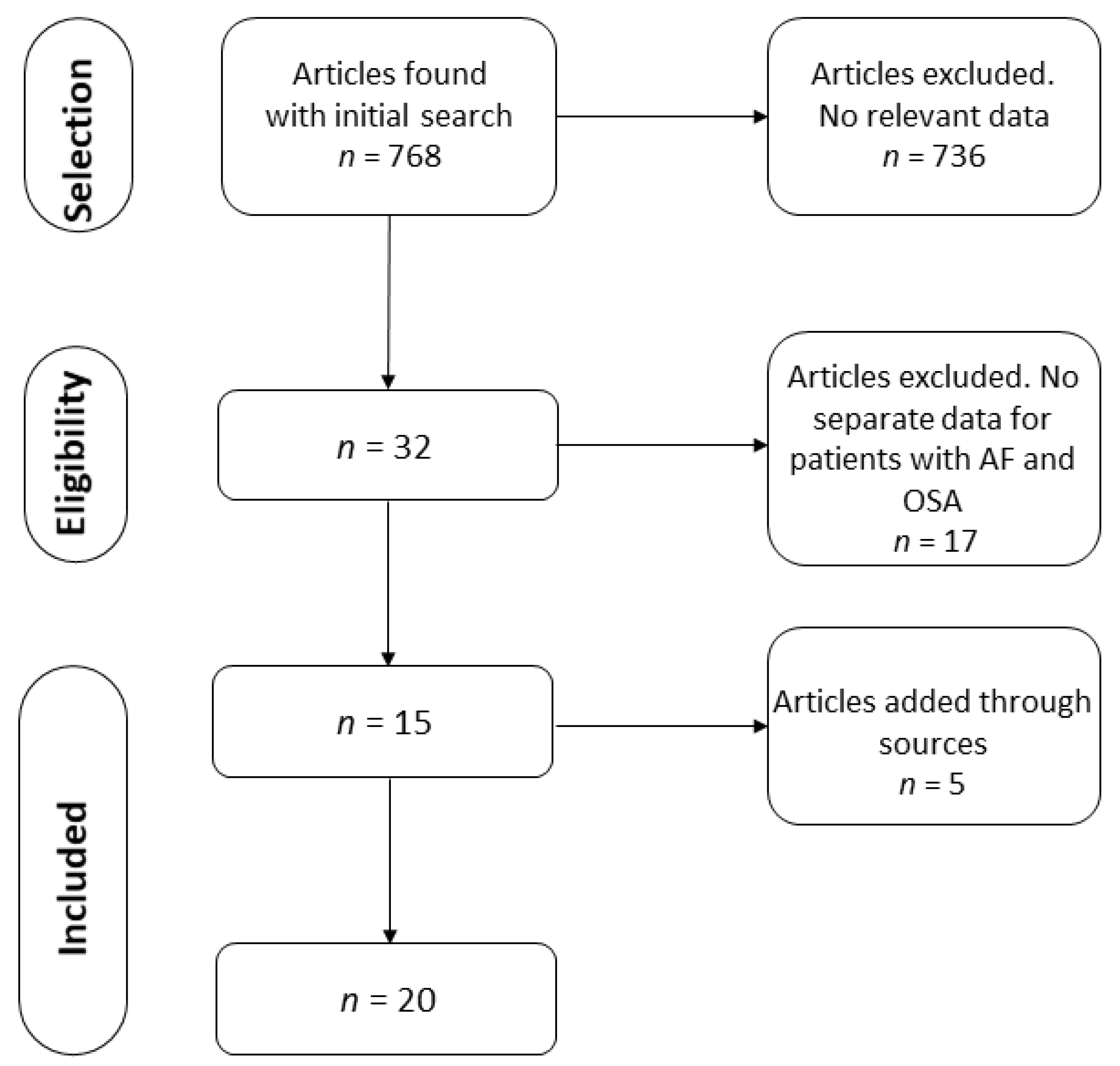
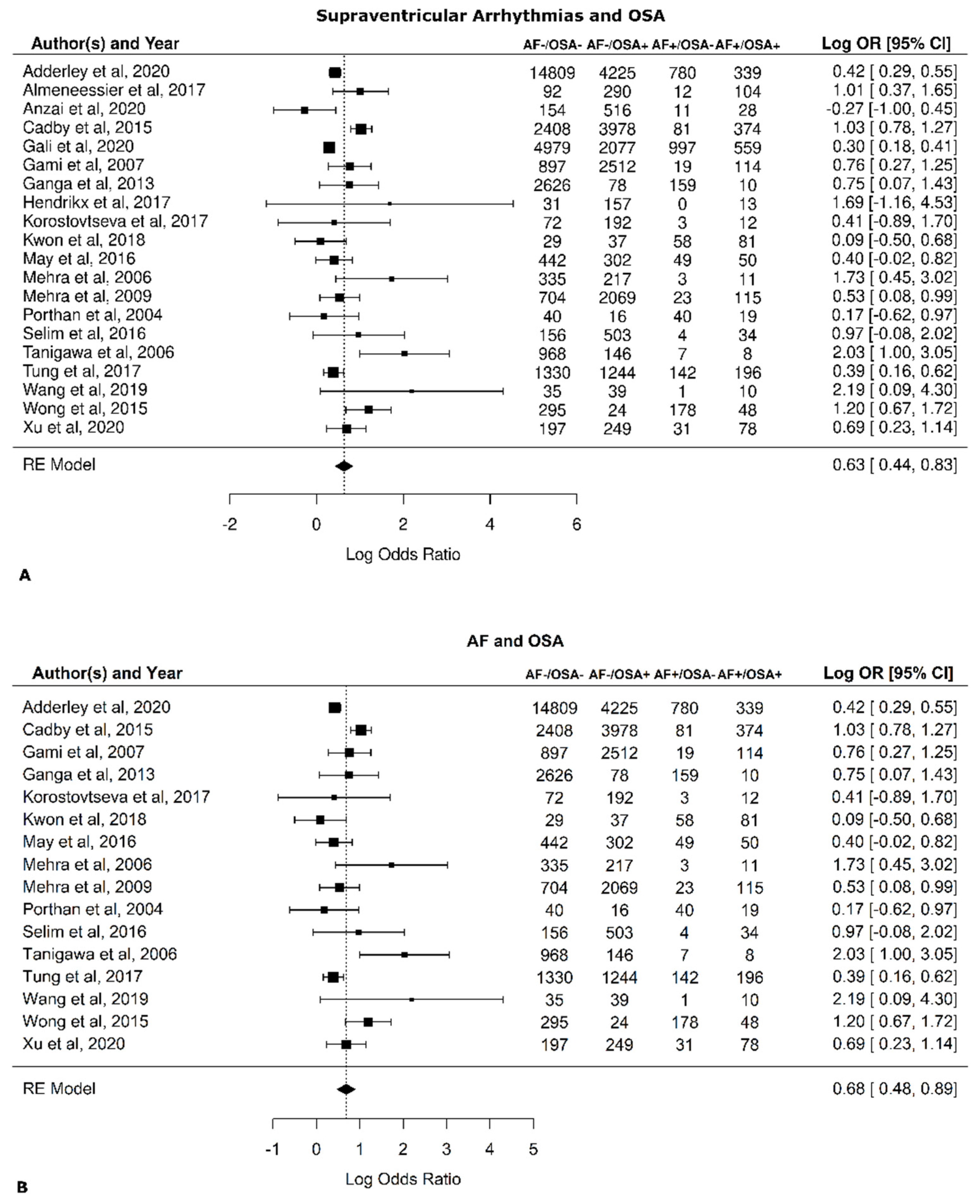

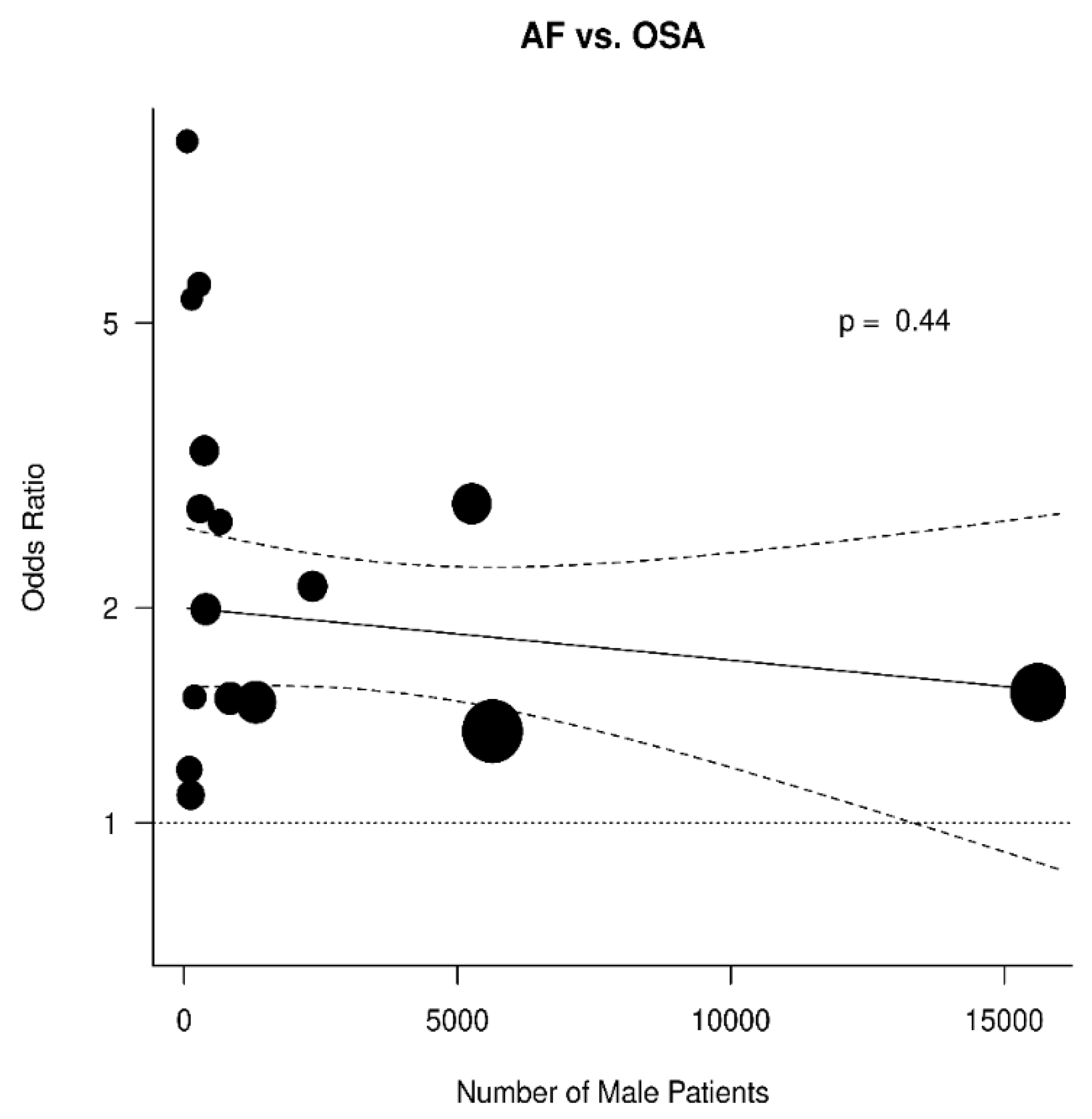
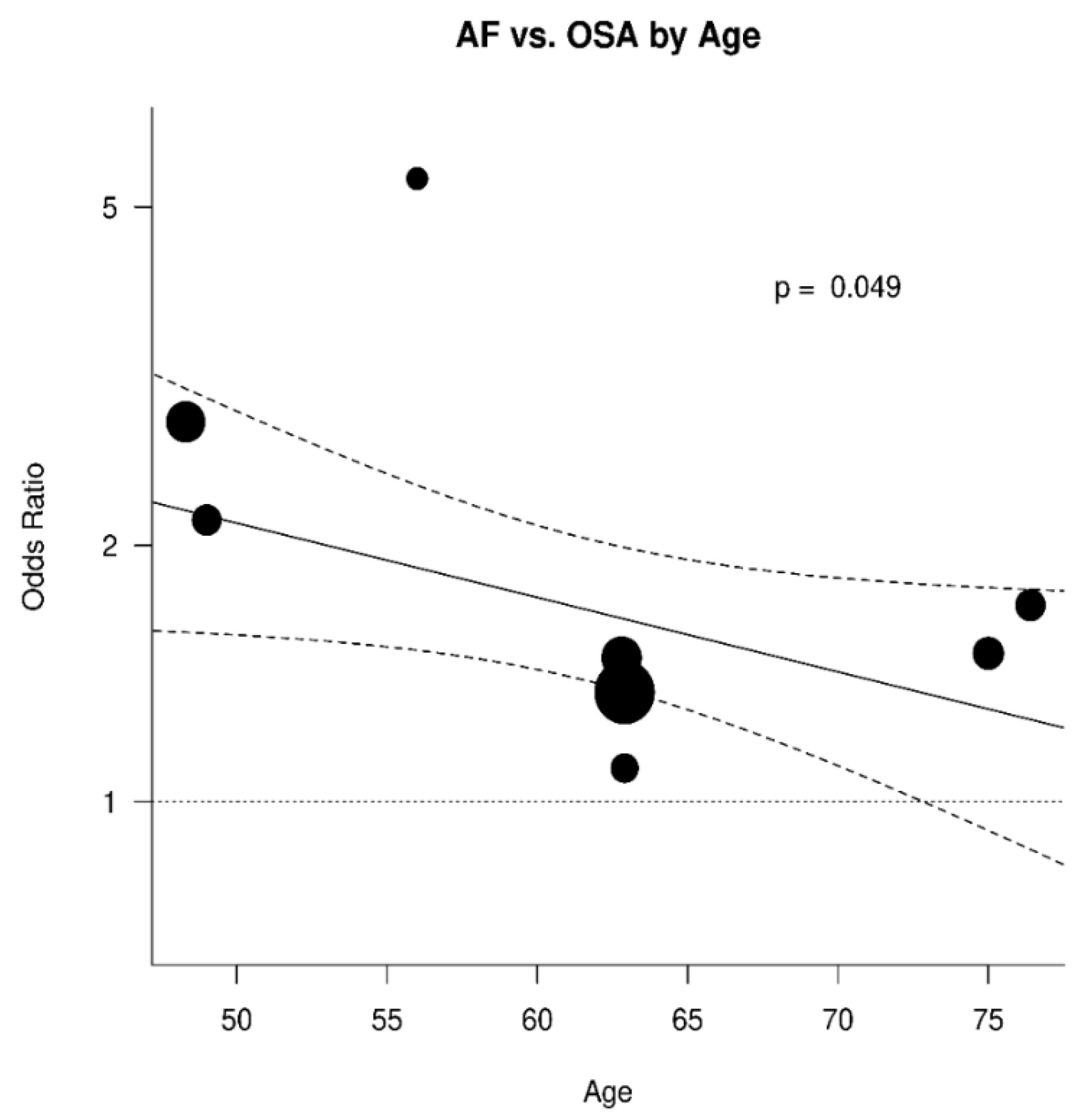

| Paper # | Paper | Total Patients | Total AF+ | AF+ OSA+ | AF+ OSA− | Total AF− | AF− OSA+ | AF− OSA− | Total OSA+ | Total OSA− |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Adderley et al., 2020 [39] | 20,153 | 1119 | 339 | 780 | 19,034 | 4225 | 14,809 | 4564 | 15,589 |
| 2 | Almeneessier et al., 2017 [30] | 498 | 116 | 104 | 12 | 382 | 290 | 92 | 394 | 104 |
| 3 | Anzai et al., 2020 [40] | 709 | 39 | 28 | 11 | 670 | 516 | 154 | 544 | 165 |
| 4 | Cadby et al., 2015 [41] | 6841 | 455 | 374 | 81 | 6386 | 3978 | 2408 | 4352 | 2489 |
| 5 | Gali et al., 2020 [42] | 8612 | 1556 | 559 | 997 | 7056 | 2077 | 4979 | 2636 | 5976 |
| 6 | Gami et al., 2007 [43] | 3542 | 133 | 114 | 19 | 3409 | 2512 | 897 | 2626 | 916 |
| 7 | Ganga et al., 2013 [44] | 2873 | 169 | 10 | 159 | 2704 | 78 | 2626 | 88 | 2785 |
| 8 | Hendrikx et al., 2017 [45] | 201 | 13 | 13 | 0 | 188 | 157 | 31 | 170 | 31 |
| 9 | Korostovtseva et al., 2017 [46] | 279 | 15 | 12 | 3 | 264 | 192 | 72 | 204 | 75 |
| 10 | Kwon et al., 2018 [47] | 205 | 139 | 81 | 58 | 66 | 37 | 29 | 118 | 87 |
| 11 | May et al., 2016 [48] | 843 | 99 | 50 | 49 | 744 | 302 | 442 | 352 | 491 |
| 12 | Mehra et al., 2006 [49] | 566 | 14 | 11 | 3 | 552 | 217 | 335 | 228 | 338 |
| 13 | Mehra et al., 2009 [50] | 2911 | 138 | 115 | 23 | 2773 | 2069 | 704 | 2184 | 727 |
| 14 | Porthan et al., 2004 [51] | 115 | 59 | 19 | 40 | 56 | 16 | 40 | 35 | 80 |
| 15 | Selim et al., 2016 [52] | 697 | 38 | 34 | 4 | 659 | 503 | 156 | 537 | 160 |
| 16 | Tanigawa et al., 2006 [53] | 1129 | 15 | 8 | 7 | 1114 | 146 | 968 | 154 | 975 |
| 17 | Tung et al., 2017 [54] | 2912 | 338 | 196 | 142 | 2574 | 1244 | 1330 | 1440 | 1472 |
| 18 | Wang et al., 2019 [55] | 85 | 11 | 10 | 1 | 74 | 39 | 35 | 49 | 36 |
| 19 | Wong et al., 2015 [56] | 545 | 226 | 48 | 178 | 319 | 24 | 295 | 72 | 473 |
| 20 | Xu et al. 2020 [57] | 555 | 109 | 78 | 31 | 446 | 249 | 197 | 327 | 228 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moula, A.I.; Parrini, I.; Tetta, C.; Lucà, F.; Parise, G.; Rao, C.M.; Mauro, E.; Parise, O.; Matteucci, F.; Gulizia, M.M.; et al. Obstructive Sleep Apnea and Atrial Fibrillation. J. Clin. Med. 2022, 11, 1242. https://doi.org/10.3390/jcm11051242
Moula AI, Parrini I, Tetta C, Lucà F, Parise G, Rao CM, Mauro E, Parise O, Matteucci F, Gulizia MM, et al. Obstructive Sleep Apnea and Atrial Fibrillation. Journal of Clinical Medicine. 2022; 11(5):1242. https://doi.org/10.3390/jcm11051242
Chicago/Turabian StyleMoula, Amalia Ioanna, Iris Parrini, Cecilia Tetta, Fabiana Lucà, Gianmarco Parise, Carmelo Massimiliano Rao, Emanuela Mauro, Orlando Parise, Francesco Matteucci, Michele Massimo Gulizia, and et al. 2022. "Obstructive Sleep Apnea and Atrial Fibrillation" Journal of Clinical Medicine 11, no. 5: 1242. https://doi.org/10.3390/jcm11051242
APA StyleMoula, A. I., Parrini, I., Tetta, C., Lucà, F., Parise, G., Rao, C. M., Mauro, E., Parise, O., Matteucci, F., Gulizia, M. M., La Meir, M., & Gelsomino, S. (2022). Obstructive Sleep Apnea and Atrial Fibrillation. Journal of Clinical Medicine, 11(5), 1242. https://doi.org/10.3390/jcm11051242







